Abstract
Purpose
The aim of this study was to predict key genes and their relationships for anxiety and nociceptive sensitivity related to Comt1 genetype.
Methods
The raw data of E-GEOD-20160 related to anxiety and nociceptive sensitivity were obtained. Pearson correlation coefficient of interaction in protein–protein interaction was calculated. Topological analysis was processed for protein–protein interaction network, and genes in this network were ranked based on their degrees. Ego genes were identified, and models were searched and refined. A total of 1000 randomized tests were processed for ego networks. The classification accuracy of each ego network was obtained in this process.
Results
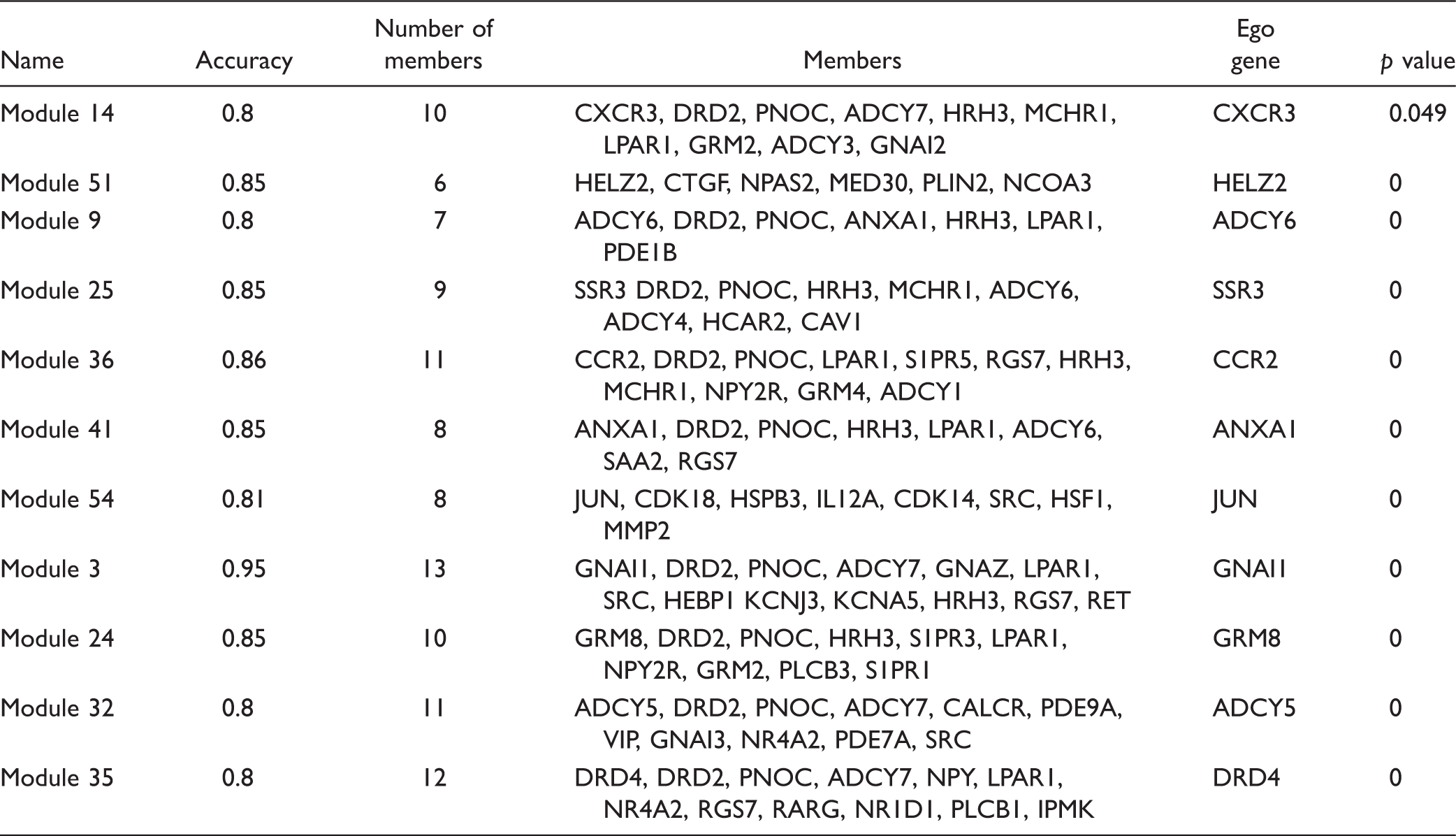
The interactions with genes in gene expression profiles were extracted, and protein–protein interaction was constructed. The protein–protein interaction included 4639 genes and 43,837 relationships. Differential co-expression network was constructed, and 74 ego genes were obtained. Thereinto, top five ego genes were ADCY2, GRM8, S1PR3, ADCY6, and ANXA1. After module searching and refinement, a total of 11 candidate modules were obtained, including module 14, module 51, and module 9. In addition, these 11 modules were confirmed to be with significance. Module 14 contained 10 genes, such as HRH3, DRD2, and CXCR3. Similarly, module 51 included six genes, such as HELZ2, NCOA3, and MED30.
Conclusions
Ego network analysis was a useful and comprehensive method for biomarkers screening. Several modules such as module 3 and module 36 were important subnetworks. Potential genes in these modules including ADCYs, GNAI1, DRD2, PNOC, CCR2, DRD2, and LPAR1 might be important genes in the research of anxiety and nociceptive sensitivity.
Keywords
Introduction
Anxiety and nociceptive sensitivity was a complex physiological process, which was induced by various factors. 1 Waszczuk et al. 2 found that genes, environments, and cognition commonly influenced the depression and anxiety symptoms of humans. In a rat model with post-traumatic stress disorder, nociception/orphanin FQ was found to be closely related with the process of allodynia and hyperalgesia. 3 Interestingly, anxiety was also a key factor of nociceptive sensitivity by participating in nociceptive response. 4
In the past decades, a number of studies have been carried out to explore the molecular mechanism of anxiety and nociceptive sensitivity and to identify key genes in different Comt1 genetypes. Catechol-O-methyltransferase (COMT) is confirmed to be a critical enzyme for human psychiatric disorders, which is important for dopamine catabolism. 5 Moreover, a short interspersed element (SINE) insertion can induce overexpression of Comt1. In mice models, decreased COMT was found to be related with pain sensitivity, and polymorphic variance of COMT was also associated with psychiatric disorders. 6 In patients with fibromyalgia, the frequency of genetic variations of COMT was significantly higher than the normal, and fibromyalgia with Val128Met SNP also showed higher sensitivity to pressure. 7 These data are certainly valued, but differently expressed genes based on expression data were still without higher reliability.
Normal bioinformatics always focus on individual genetic index, which could not comprehensively describe the character of network. In this study, a novel method of ego network analysis was used to identify significant networks associated with anxiety and nociceptive sensitivity, and accuracy was applied to predict key genes and their relationships for anxiety and nociceptive sensitivity related to Comt1 genetype.
Materials and methods
Data source and pretreatment
The raw data of E-GEOD-20160 were obtained from ArrayExpress database, which was related to anxiety and nociceptive sensitivity. 6 The platform of this profile was GPL1261 [Mouse430_2] Affymetrix Mouse Genome 430 2.0 Array. The raw data included 56 samples, including 24 samples with SINE and 32 samples without SINE. The raw data were preprocessed with three steps: background correction, repeated probes removing, and data normalization. After pretreatment, all probes were mapped to genes, and gene expression profiles were obtained.
Screening data of PPI
The protein–protein interaction (PPI) network of mice was downloaded, including 16,053 genes and 3,701,743 relationships. 8 PPI relationships with boundary value more than 0.8 were screened and regarded as background PPI. The interactions with genes in gene expression profiles were extracted, and PPI was constructed.
Ego genes identification and ego networks construction
Calculation of weight for interactions
Pearson correlation coefficient of interaction in PPI was calculated, and PPI with absolute value more than δ (δ = 0.8) was chosen as differently expressed interaction network. One-side t test was used to calculate the p value of each gene between samples with and without SINE. Then, the weight of each interaction in PPI was calculated by the following formula:
Ego genes identification
Topological analysis was processed for PPI network, and genes in this network were ranked based on their degrees. Every network (Gk = (V,Ek) (1 ≤ k ≤ M)) had an adjacency matrix (AK = (aijk) n × n). The importance of each genes for the network was calculated by the following formula which is shown by g(i) value (g(i) = z-score):
Thereinto, Nk(i) was the adjacency node of Gk, and k was the degree normalized weighted adjacency matrix. In addition, k = D−1/2AkD1/2, D was the diagonal matrix of Ak.
Then, nodes in PPI were ranked based on the z-score. The top 5% genes with higher degree and genes with degree more than 1 were chosen as ego genes. 9
Module searching for ego genes and refinement
A known ego gene v ∈ V was used as a differently expressed module. Then, gene U which was adjacent to v was added in this model, and new model C′ was obtained. The variation of classification accuracy between module V and C′ was calculated by formula of ΔS(C′,C) = S(C′)−S(C). Gene U increased the degree of module C when ΔS(C′,C) was more than 0. Following, all genes with ΔS(C′,C) > 0 was added in module C, until ΔS was not increased.
All modules were refined with the threshold of classification accuracy not less than 0.8. The selected modules were obtained as candidate ego modules.
Statistical analysis of ego networks
A total of 1000 randomized tests were processed for ego networks. The classification accuracy of each ego network was obtained in this process. The p value of each ego network was calculated by the following formula:
p = SUM possibility (randomized classification accuracy > candidate classification accuracy)/number of randomized tests. Ego network with p ≤ 0.05 was regarded with significance.
Results
PPI identification
The interactions with genes in gene expression profiles were extracted, and PPI was constructed. The PPI included 4639 genes and 43,837 relationships.
Ego genes identification
Differential co-expression network was constructed (Figure 1). Based on the threshold of top 5% and degree >1, a total of 74 ego genes were obtained (Supplement Table 1). Thereinto, top five ego genes were adenylate cyclase 2 (ADCY2, z-score = 216.4743), glutamate metabotropic receptor 8 (GRM8, z-score = 211.4734), sphingosine-1-phosphate receptor 3 (S1PR3, z-score = 195.16), adenylate cyclase 6 (ADCY6, z-score = 190.5954), and annexin A1 (ANXA1, z-score = 189.7538).
Differential co-expression network. Note: The nodes represent important genes for anxiety and nociceptive sensitivity, and edges represent their relationships between these genes. The blue and yellow genes represent normal and ego genes, respectively.
Ego networks construction and statistical analysis
Modules and their information.
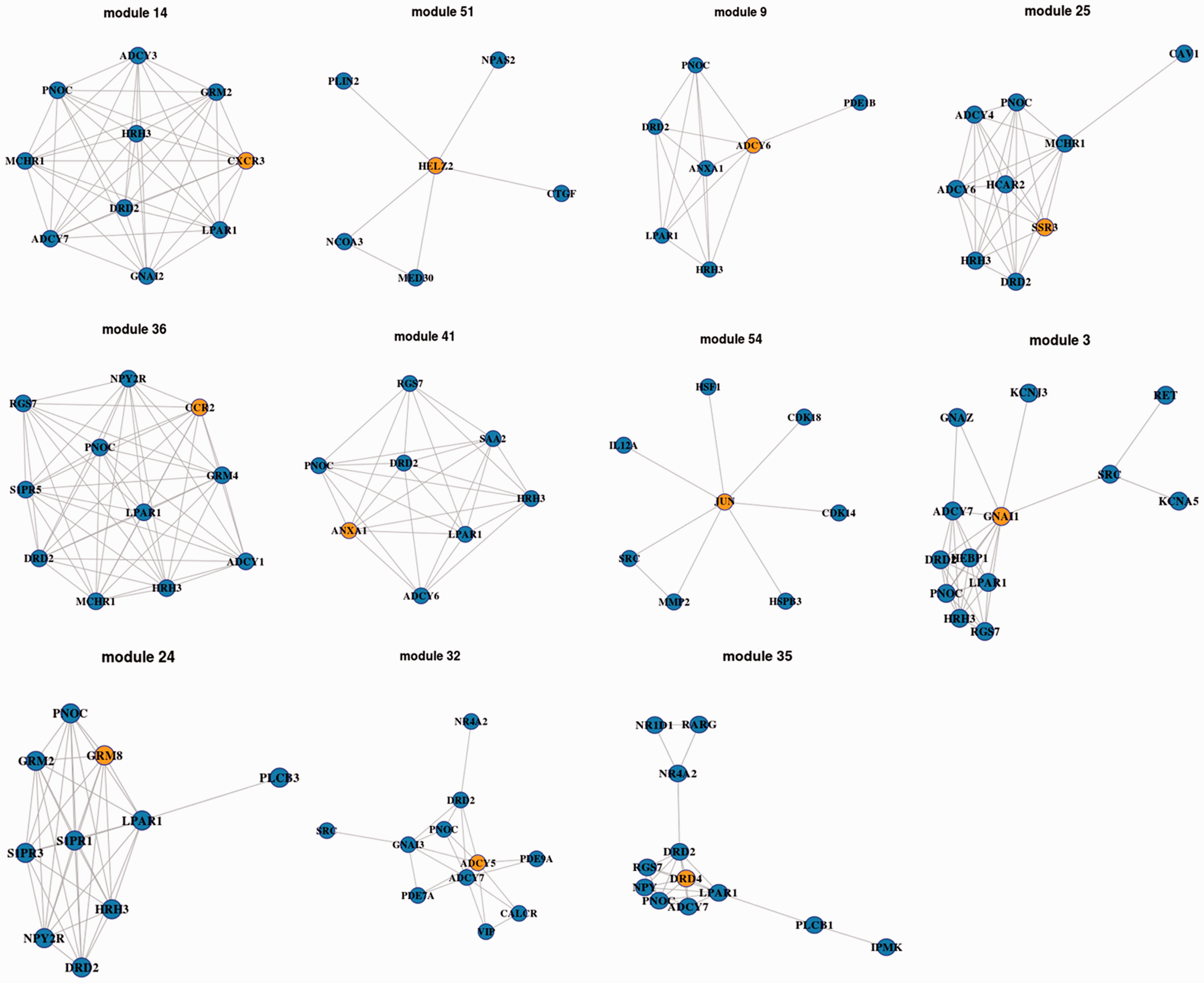
Subnetwork for anxiety and nociceptive sensitivity. Note: The nodes represent important genes for anxiety and nociceptive sensitivity, and edges represent their relationships between these genes. The blue and yellow genes represent normal and ego genes, respectively.
Discussion
In this study, an ego network was given, which selected subnetworks for predicting genes related to anxiety and nociceptive sensitivity. The main advantage of ego network was to screen potential genes that were not differentially expressed but with important functions by interacting with differentially expressed genes. Based on this method, a total of 11 modules including ego genes and potential genes were identified. Thereinto, module 3 was with highest accuracy, and genes in this module were G protein subunit alpha I1 (GNAI1), DRD2, prepronociceptin (PNOC), and adenylate cyclase 7 (ADCY7). GNAI1 was the ego gene in this module. Similarly, module 36 included genes of C-C motif chemokine receptor 2 (CCR2), DRD2, PNOC, and lysophosphatidic acid receptor 1 (LPAR1). CCR2 was the ego gene in this module.
Besides, we identified ADCY2 and ADCY6 to be the top five ego genes that are related to anxiety and nociceptive sensitivity. These genes are members of the family of ADCYs, which are membrane-associated enzymes that catalyze the formation of the secondary messenger cyclic adenosine monophosphate. Chen et al. 10 built a mice model and found that ADCY5 was significantly highly expressed in striatum. In addition, the deletion of ADCY1 in mice clearly reduced nociceptive behaviors and chronic pain-induced anxiety.11,12 Previous studies also confirmed that ADCY1 contributed chronic pain-induced long-term synaptic plasticity in the anterior cingulate cortex of mice.13,14 Moreover, the polymorphism in 3′ untranslated region of ADCY7 was confirmed to be closely related with the risk of anxiety, especially in males. 15 Therefore, ADCYs were closely related with anxiety and nociceptive sensitivity.
GNAI1 was a heterotrimeric signal-transducing molecule consisting of alpha, beta, and gamma subnits. The alpha subunit binds guanine nucleotide, could hydrolyze GTP, and also interacts with other proteins. 16 GNAI1 was commonly enriched in GTP binding, signal transducer activity, and peptide ligand-binding receptors. In rat models, both JTC-801 and Ro64-6198 were confirmed to affect GTPγS binding in brain and to further monitor the anxiety and nociceptive sensitivity.17,18 In addition, mice with deficiency in corticotropin-releasing factor receptor 1 was found to be with lower anxiety sensitivity. 1 Similarly with GNAI1, DRD2 was also mainly enriched in signal transducer activity and peptide ligand-binding receptors. Besides, it was also enriched in G-protein-coupled receptor activity. Fındıklı et al. 19 demonstrated that G-protein-coupled estrogen receptor 1 level in serum could be used as an independent predictor for anxiety. In this study, PNOC and DRD2 were found to be with closely relationship (weight = 0.4838). This gene encoded a protein which could generate various protein products, including nociception, nocistatin, and orphanin FQ2. 20 It is widely known that orphanin FQ2 could bind to the nociception receptor and further increase pain sensitivity. 21 Diseases associated with PNOC included neonatal abstinence syndrome and pain agnosia. Thereby, GNAI1, DRD2, and PNOC in module 3 were closely related with anxiety and nociceptive sensitivity.
Furthermore, CCR2 encoded two isoforms of a receptor for monocyte chemoattractant protein-1, a chemokine which specially mediates monocyte chemotaxis. 22 This protein was associated with monocyte infiltration and inflammatory response. In addition, the receptors of CCR2 mediate agonist-dependent calcium mobilization and inhibition of adenylyl cyclase. 23 As shown in previous studies, diabetes might induce changes of calcium mobilization in nociceptive neurons. 24 In addition, the recovery of Ca2+ and Mg2+ could regulate the endoplasmic reticulum and plasmalemma and further affect spinal dorsal horn and dorsal root ganglia neurons. 25 Gover et al. 26 also confirmed that external and internal Ca2+ could alter the pattern of discharge and affect the excitability of sensory neurons. In this study, DRD2 and CCR2 were found to be with association (weight = 0.2327). This gene encodes the D2 subtype of the dopamine receptor. This G-protein-coupled receptor inhibited adenylyl cyclase activity. Mutation in this gene causes myoclonus dystonia and schizophrenia. In mouse model, deficiency in D3 dopamine receptor was confirmed to reduce anxiety. 27 Moreover, chronic deficiency of dopamine receptor D3 might induce depressed spirit and even anxiety. 28 Interestingly, de la Mora et al. 29 found that D1 and D2 dopamine receptors have a differential effect on anxiety modulation. LPAR1 in this study was also found to be involved in module 36. This gene encoded a protein which regulated various biologic functions, including proliferation, platelet aggregation, inhibition of neuroblastoma cell differentiation, and chemotaxis. 30 Although no evidence confirmed that LPAR1 was directly related with anxiety and nociceptive sensitivity, LPAR1 was found to be closely related with CCR2, DRD2, and CCR2 in this study. Then, LPAR1 might be a potential, but indirect, gene for anxiety and nociceptive sensitivity.
In conclusion, ego network analysis was a useful and comprehensive method for biomarkers screening. Several modules such as module 3 and module 36 were important subnetworks. Potential genes in these modules including ADCYs, GNAI1, DRD2, PNOC, CCR2, DRD2, and LPAR1 might be important genes in the research of anxiety and nociceptive sensitivity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


