Abstract
The upregulation of various channels and receptors classically linked to sensory transduction from the periphery tightly correspond with changes in the responsiveness of specific subpopulations of primary afferents to mechanical and heat stimulation of the skin at different ages. Previous reports in adults have suggested that the purinergic adenosine diphosphate receptor, P2Y1 can specifically regulate sensory neuron responsiveness to heat stimuli in addition to neurochemical alterations in primary afferents during cutaneous inflammation. To determine if the upregulation of P2Y1 found in the dorsal root ganglia of neonatal mice with cutaneous inflammation initiated at postnatal day 7 (P7) was responsible for the specific alteration in heat sensitivity found in faster conducting (“A”-fiber) nociceptors, we assessed the response properties of cutaneous afferents using an ex vivo hairy hindpaw skin-saphenous nerve-dorsal root ganglion-spinal cord preparation in conjunction with nerve-targeted knockdown of P2Y1. We found that P2Y1 knockdown during neonatal cutaneous inflammation was sufficient to reduce the sensitization of “A”-fiber nociceptors to heat stimuli. Surprisingly, we also found that nerve-specific downregulation of P2Y1 could reduce the observed sensitization of these afferent subtypes to mechanical deformation of the skin. Immunocytochemical analysis of dorsal root ganglia showed that P2Y1 may mediate its effects through modulation of the injury-induced increase of transient receptor potential vanilloid type 1 receptor. This suggests that the upregulation of P2Y1 in cutaneous nociceptors during early life peripheral inflammation can regulate the sensitization of myelinated nociceptors to both mechanical and heat stimuli possibly through modulation of transient receptor potential vanilloid type 1 expression.
Introduction
P2Y1 is a member of the G-protein coupled nucleotide receptor family and is highly expressed in sensory neurons.1–3 During postnatal development (P0–P21), this receptor does not change its expression levels in dorsal root ganglia (DRGs); however, cutaneous inflammation of the skin at P7, but not P14, has been found to significantly increase its expression in sensory ganglia. 4 This specific change is of interest as P2Y1 has been linked to normal thermal sensitivity in sensory neurons. 5 Moreover, inflammation of the skin in adult animals induces the upregulation of P2Y1, which has been shown to regulate afferent sensitization to heat stimuli and modulate behavioral heat hypersensitivity,5–7 possibly through increasing expression of the heat transducing channel transient receptor potential vanilloid type 1 (TRPV1). 8 Together, this suggests that P2Y1 may also play an important role in heat sensitivity in developing animals.
It is known that neonates can experience pain as a result of peripheral injuries; however, insults sustained early in development specifically have the potential to cause long-term effects on nociceptive processing and cognitive function in adulthood.9,10–13 Evidence suggests that the peripheral mechanisms of pain may be unique in neonates compared to later stages of postnatal development or adults.4,5,14,15 One possible reason is that during the early developmental period, the peripheral sensory system undergoes substantial neurochemical reorganization wherein expression of various molecules and receptors that regulate sensory function in the DRG are altered over time.4,16,17,35,36 For example, we have previously found that changes in neurotrophic factor responsiveness of sensory neurons during early postnatal development18,19,35,36,49,65 occur around the same time as age-related expression of distinct receptors/channels involved in sensory transduction in the DRGs. 4 We also determined that the slowly conducting primary afferents (“C”-fibers) that were mechanically sensitive but thermally insensitive (CM) may gain sensitivity to heat and become polymodal (CPM) at this same time period. 4 This suggested that distinct populations of sensory neurons may be sensitized to peripheral stimuli after injury at different postnatal ages.
Studies have suggested that A-fiber sensitization may be crucial for the development of chronic pain during early life.15,20 We have shown that faster conducting “A”-fiber nociceptors are exclusively sensitized to mechanical and heat stimuli after inflammation of the hairy hindpaw skin at postnatal day 7 (P7). The slower “C”-fiber afferents were sensitized to these same stimuli during inflammation initiated at P14. 4 The mechanisms of how these changes may occur in sensory neurons after peripheral inflammation may be due to the distinct age-related changes in DRG gene expression overall leading to the development of pain-like behaviors.21,35,36,65 Thus it is important to determine which receptors are responsible for distinct aspects of afferent sensitization across various ages of development. Here we tested the hypothesis that upregulation of P2Y1 in sensory neurons drives changes in “A”-fiber heat responsiveness during neonatal (P7) inflammation of the hairy hindpaw skin.
Materials and methods
Animals
All experiments were conducted using Swiss Webster mice from postnatal day 6 through postnatal day 10. Specific ages used were approximately ± 0.5 days around the specified time point. All animals were maintained on a 12-h light/dark cycle with a temperature-controlled environment and were housed with the mother, which was provided food and water ad libitum. All procedures were approved by the Institutional Animal Care and Use Committee at Cincinnati Children’s Hospital Medical Center, under Association for Assessment and Accreditation of Laboratory Animal Care approved practices.
Carrageenan injection
For P7 inflammation, mice were first anesthetized by inhalation of isoflurane. Then 1 µL per gram body weight of 3% carrageenan (in 0.9% NaCl) was injected into the right hairy hind paw skin using a 30 gauge needle. As neonatal mice weigh approximately 3 to 4 g at this age, a maximum of 4 µL was injected into the hairy skin of our pups. Carrageenan was introduced under the skin beginning at the ankle and expelled distally as the needle was slowly retracted. All efforts were made to specifically target the medial side of the hairy skin which is the innervation field of the saphenous nerve. A cotton swab was applied at the injection site following needle removal to prevent leakage of carrageenan. Paw edema was approximated by using calipers to measure the thickness of the ipsilateral hindpaw relative to the contralateral hindpaw at one and three days after injection to ensure that all conditions experienced a relatively similar degree of inflammation. All electrophysiological, neurochemical, or molecular analyses were performed one day or three days after injection of carrageenan and compared to P7 or P10 naïve mice as indicated.
siRNA injections
Procedures for saphenous nerve injection of siRNAs into neonatal mice were described previously in the literature. 17 Briefly, mice were anesthetized as described using 2% to 3% isofluorane inhalation. The saphenous nerve was exposed through a small incision made in the right medial mid-thigh. Then approximately 0.05 to 0.1 µL of 90 µM penetratin-1 (MP Biomedicals) linked, non-targeting control (PenCON) or P2Y1 targeting (PenY1) siRNAs were pressure injected into the saphenous nerve using a quartz microelectrode connected to a picospritzer one day prior to carrageenan injection (P6). Specific siRNAs used in the current report were designed and conjugated to penetratin-1 as described previously in literature.6,22–28 Sequences and other information for the selection and specificity of individual siRNAs used in this study are detailed in Queme et al. 27 Penetratin-conjugated siRNAs were first heated to 65℃ for 5 min prior to injection. This strategy has been used extensively by our laboratory in mice and has not been found to injure the axons or cause anti-viral-related off-target effects.6,24,25 This strategy however does provide a decidedly effective means to block injury-induced upregulation of genes in specific nerves at high efficiency that will eventually be studied electrophysiologically and/or immunocytochemically.17,22–28
Ex vivo preparation and recording
The ex vivo preparation has been described in detail previously (e.g., 17,24–26). Briefly, mice were anesthetized with intramuscular injection of ketamine and xylazine (90 and 10 mg/kg, respectively) and perfused transcardially with oxygenated (95% O2/5% CO2) artificial cerebrospinal fluid (aCSF; in mM: 1.9 KCl, 1.2 KH2PO4, 1.3 MgSO4, 2.4 CaCl2, 26.0 NaHCO3, and 10.0 D-glucose) containing 253.9 mM sucrose at a temperature of approximately 12℃. The spinal cord and the right hind limb were then excised from the animal and placed in a bath of the oxygenated sucrose-aCSF. Next, the hairy skin of the right hindpaw, saphenous nerve, L1 to L5 DRGs, and corresponding spinal cord segments were isolated. Then the preparation was transferred to a separate recording chamber containing chilled, oxygenated aCSF in which the sucrose was replaced with 127.0 mM NaCl. The skin was pinned out on a stainless steel grid located at the bath/air border allowing the dermal surface to be perfused with the aCSF while the epidermis stayed dry. The platform served to provide stability during applied thermal and mechanical stimuli. The bath was then slowly warmed to 32℃ before recording.
Sensory neuron somata in L2 and/or L3 DRGs were impaled with quartz microelectrodes (impedance >150 MΩ) containing 1 M potassium acetate. Orthograde electrical search stimuli were delivered through a suction electrode on the side of the nerve to identify sensory neuron somata with axons contained in the saphenous nerve. When the cell was found to be driven by the electrical stimulus, peripheral receptive fields (RF) were then localized by probing the skin with a paint brush, blunt glass stylus, and/or von Frey (VF) hairs which also served to determine mechanical sensitivity. When cells were driven by the nerve but had no mechanical RF, a thermal search was conducted by applying hot (∼52℃) and/or cold saline (∼0℃) to the skin.
Mechanical response characteristics were assessed by applying an increasing series of VF filaments ranging from 0.07 to 10 g to the RF for 1 to 2 s. A mechanical stimulator that delivered digitally controlled mechanical stimuli to the cell’s RF was also employed when possible. This consisted of a tension/length controller (Aurora Scientific, Aurora, ON, Canada) with a 1-mm diameter aluminum tip that applied computer-controlled 5-s square waves of 1, 5, 10, 25, 50, and 100 mN to the RF. In order to compare these results to those of the VF stimulation, VF units in grams were converted to a mN force based on the VF filament diameter. After mechanical responsiveness was determined, a controlled thermal stimulus was applied using a 3 × 5 mm contact area Peltier element (Yale University Machine Shop, New Haven, CT, USA). Cold stimuli consisted of a variable rate cold ramp beginning at 31℃, dropping to approximately 2℃ to 4℃, holding for 4 to 5 s and slowly returning to 31℃. After bath temperature was maintained for approximately 4 to 5 s, a heat ramp was applied which went from 31℃ to 52℃ in 12 s. This heat stimulus was then held at 52℃ for 5 s. The stimulus then ramped back down to 31℃ in 12 s. Adequate recovery times (approximately 20–30 s) were employed between stimulations. In other cases in which thermal data were unable to be obtained using the contact peltier device, after mechanical stimulation, cold (∼0℃) followed by hot (∼52℃) saline was applied directly to the RF on the skin to determine thermal responsiveness. Application duration of these saline stimuli lasted approximately 1 to 2 s.
When fibers were unable to be characterized by computer-controlled mechanical or thermal stimulation but could be characterized by VF and/or saline stimuli, these cells were included to assess the subtype of the afferents. All elicited responses were recorded digitally for offline analysis of thresholds, firing rates, and mean peak instantaneous frequencies (IFs) to the various stimuli using Spike2 software (Cambridge Electronic Design, Cambridge, UK). Firing rates were determined by binning the entire response into 200 ms bins and then determining the peak bin firing for that stimulus. Peripheral conduction velocity (CV) was calculated from the latency from the onset of the stimulus and the conduction distance between stimulating site and recording electrodes (measured directly along the nerve). Recording sessions lasted approximately 4 h. No differences were observed between cells obtained at the beginning of an experiment to those obtained at the end of the recording session, similar to previous reports (e.g., 4,6,17).
A total of 307 primary cutaneous neurons were intracellularly recorded and physiologically characterized at one day or three days postinflammation from 25 Swiss-Webster mice which were inflamed at P7. Of these, 56 cells came from five mice at the one day time point and 43 cells came from four mice at the three day time point in animals that only received hairy skin carrageenan injection; 53 cells were obtained from three mice at the one day time point and 52 cells came from three mice at the three day time point in animals that received carrageenan injections into the hairy skin plus control, non-targeting (PenCON) siRNA injection into the saphenous nerve. The final 103 cells came from five mice at the one day time point (n = 50 cells) and five mice at the three day time point (n = 53 cells) in animals that received carrageenan plus P2Y1 targeting (PenY1) siRNA injections. The results from these three experimental groups at the two time points were compared to previously obtained results from naïve Swiss Webster mice (n = 49 cells from four animals at P7 and n = 53 cells from five mice at P10). 4
Western blotting
For each sample used in western blotting (n = 3–6 per condition), the right L2 and L3 DRGs were excised from two mice of the same condition (naïve, carrageenan only, or inflamed mice with siRNA injections) and homogenized in protein lysis buffer containing 1% sodium dodecyl sulfate, 10 mM Tris-HCl (pH 7.4), and protease inhibitors (1 µg/mL pepstatin, 1 µg/mL leupeptin, 1 µg/mL aprotinin, 1 mM sodium orthovanadate, and 100 µg/mL phenylmethylsulfonyl fluoride; Sigma-Aldrich Biochemicals). Samples (10 µg) were centrifuged, boiled 10 min in a denaturing buffer containing β-mercaptoethanol and sodium dodecyl sulfate, separated on a 12.5% precast polyacrylamide gel (Bio-Rad Laboratories) and transferred to a polyvinylidene difluoride membrane (Millipore) that was blocked in LiCor Blocking Buffer diluted 1:1 in 0.1 M phosphate buffer (PB). Then, membranes were incubated with primary antibodies overnight at 4℃ (rabbit anti-P2Y1, 1:300; Alomone, Cat# APR-009; chicken anti-GAPDH, 1:2000; Pro Sci, Cat# XW-7214). After incubation in primary antibodies, polyvinylidene difluoride membranes were washed and then incubated with secondary antibodies (donkey anti-chicken Dy680RD, 1:20,000, Cat# 926-68075; donkey anti-rabbit Dy800CW, 1:20,000, Cat# 925-32211; LiCor Biosciences). Membranes were scanned using a LiCor Odyssey Imaging System with consistent settings for detection between runs. Immunoreactive bands were then analyzed offline and quantified using NIH Image J software. Band intensity was normalized to GAPDH and reported as a value in arbitrary units (mean ± SEM).
Immunohistochemistry and DRG cell quantifications
L2/L3 DRGs from naïve and carrageenan injected mice with or without siRNA injections were fixed in 3% paraformaldehyde in 0.1 M PB for 30 min at room temperature and then rinsed in 0.1 M PB. After being embedded in 10% gelatin, DRGs were postfixed in 3% paraformaldehyde in 0.1 M PB and then cryoprotected in 20% sucrose; 45 µm frozen DRG sections obtained from a sliding microtome were collected in PB and then processed overnight for TRPV1 immunoreactivity (rabbit anti-TRPV1, 1:2000; Alomone, Cat# ACC-030) and isolectin B4 (IB4) binding (1:200; 647 AlexaFluor-conjugated IB4; Invitrogen, Cat# I21412). After incubation in primary antiserum, tissue was washed and labeled with appropriate secondary antibody (1:200; 594 AlexaFluor-conjugated Donkey anti-rabbit; Jackson ImmunoResearch Laboratories, Cat# 711-585-152). Sections were mounted with Fluorogel (Electron Microscopy Sciences) on gelatin-coated slides and stored in the dark at room temperature.
The expression of TRPV1 and IB4 binding was analyzed here using a previously described methodology for total cell counts in the DRGs.27–29 In brief, three nonconsecutive DRG sections from a single animal were randomly chosen, and Z-stacks were obtained by scanning at 3 µm intervals using a Leica confocal microscope to create 15 -µm thick optical sections. Consistent exposure times were utilized across all sections/conditions during imaging. The number of TRPV1 positive and/or IB4 binding cells were then counted using Neurolucida software and averaged in the top, middle, and bottom optical section of each stack in order to ensure that the same cell was not counted twice. Only cells with nuclei were considered in the analysis. Positive staining was determined to occur when florescence attained was above background intensity detected in negative control sections (no antibody or no lectin controls). Cell counts were averaged across the three optical sections in each mouse and performed in three replicate animals per condition. The number of positive cells per field of view was reported as mean ± SEM. Experimenters performed cell counts blind to the experimental conditions.
Data analysis
All data are presented as mean ± SEM. Mean peak IFs, thresholds to different peripheral stimuli and firing rates were compared via Kruskal Wallis analyses followed by Dunn’s post hoc tests or one-way analyses of variance (ANOVAs) with Tukey’s post hocs when appropriate. Differences in afferent subtype prevalence were determined using a Chi Square test. Percent change in protein expression was analyzed via one-way ANOVA with Tukey’s post hoc test. Cell quantifications were assessed using one-way ANOVA with Holm Sidak post hocs. Critical significance level was set at p < 0.05.
Results
Knockdown of P2Y1 expression in DRGs during carrageenan-induced inflammation
In order to confirm our previous report showing an upregulation of P2Y1 mRNA in the DRGs after cutaneous inflammation
4
at the protein level, we performed western blot analysis on DRGs from mice with carrageenan-induced inflammation with or without saphenous nerve injection of non-targeting control (PenCON) or P2Y1 targeting (PenY1) siRNAs and compared them to naïve controls. We found that P2Y1 protein was enhanced in the L2/L3 DRGs after inflammation of the hairy hind paw skin one day after carrageenan injection (p < 0.05 vs. naive). This upregulation was unaffected in mice that were injected with PenCON siRNAs into the saphenous nerve in addition to cutaneous inflammation (p < 0.05 vs. naive). However, injection of PenY1 siRNAs into the saphenous nerve of mice with hairy hindpaw skin inflammation knocked down the injury-induced upregulation of P2Y1 protein expression in the DRGs (p > 0.05 vs. naïve; Figure 1). This suggests that our siRNA injection strategy is sufficient to block the peripheral inflammation-induced upregulation of P2Y1 in the DRGs of neonatal mice with cutaneous inflammation induced at P7.
Western blot analysis of dorsal root ganglia (DRG) one day following carrageenan-induced inflammation of the hairy hindpaw skin with or without saphenous nerve injection of non-targeting control (PenCON) or P2Y1 targeting (PenY1) siRNAs. (a) Examples of DRG western blots for P2Y1 (arrow) and GAPDH protein in naïve mice, one day following inflammation with carrageenan (Carr. Only), or inflamed mice with injection of control (PenCON+Carr.) or P2Y1-targeting (PenY1+Carr.) siRNAs. (b) The protein isolated from DRGs of mice in the carrageenan only group and PenCON+Carr. groups showed significantly increased expression of P2Y1 versus naive mice; however, injection of PenY1 into the saphenous nerve blocked this inflammation-induced increase. n = 3–6 each; *p < 0.05 versus naïve; One-way ANOVA with Tukey’s post hoc test; mean ± SEM.
Saphenous nerve-specific P2Y1 knockdown reduces carrageenan-induced sensitization of “A”-fiber nociceptors in neonates
Our previous data suggested that cutaneous inflammation sustained at P7 sensitized “A”-fiber but not “C”-fiber neurons to mechanical and heat stimuli one day after injury. We therefore wanted to determine if the upregulation of P2Y1 was responsible for the specific alteration in heat sensitivity observed in the afferents. Similar to our previously published reports and consistent with other classifications, we categorized afferent subtypes based on CV and spike shapes (broad or narrow spike widths). Two categories of fibers can be detected in neonates based on CV since the nerves are still undergoing myelination.4,30,31 “A”-fibers were defined as those conducting at least twice as fast as the other cell types (“C”-fibers) within a given experiment. The average CVs for “A”-fibers were 2.84 ± 0.23 m/s, and average CVs for “C”-fibers were 0.51 ± 0.01 m/s in our groups. These classifications were not altered among the various groups of mice analyzed in this study (not shown).
Consistent with our recent report,4,32 “A”-fiber high threshold mechanoreceptors (HTMRs; faster conducting with broad spikes) that were also responsive to heat stimuli (and sometimes cold) were found to have significantly higher mean peak IFs to heat stimulation of the skin in mice that received carrageenan alone or PenCON siRNA injection with inflammation at one day (p < 0.03 vs. naïve; n = 5). As we were only able to fully characterize one polymodal “A”-fiber from mice with carrageenan injection alone, this cell was added to the data obtained from PenCON injected mice with inflammation (n = 4) for purposes of comparison. PenY1 siRNA injection reduced this inflammation induced change in heat firing when compared to naïves (Figure 2(a); p > 0.05; n = 4). No changes were detected in heat thresholds among the various groups (not shown). Insufficient numbers of A-fiber HTMRs with heat responses were found three days after carrageenan injection in all groups, thus we were unable to assess heat responses in these neurons at this time point.
P2Y1 knockdown reduces hypersensitivity of A-fiber high threshold mechanoreceptor (A-HTMR) sensory neurons to heat and mechanical stimuli during cutaneous inflammation initiated at P7 as assessed with ex vivo recording. (a) At P7, carrageenan injection into the hairy hindpaw skin was found to increase the mean peak instantaneous frequencies of A-HTMR neurons to heat one day after inflammation (Control Carr: combined carrageenan alone and PenCON+Carr; *p < 0.03 vs. naive); however, this was reduced by P2Y1 knockdown (PenY1; p < 0.5 vs. naïve). (b) Carrageenan injection into the hairy hindpaw skin increased the firing rate of A-HTMRs to mechanical stimuli one day after inflammation (Carr only and PenCON+Carr; *p < 0.02 vs. naive). This was also blocked by nerve-specific knockdown of P2Y1 (p < 0.86 vs. naïve, p < 0.05 vs. PenCON). (c) One day after P7 inflammation, mechanical thresholds of “A”-fiber HTMRs were significantly decreased (Carr Only and PenCON+Carr; p < 0.01 vs. naive). PenY1 injected mice with inflammation however were not different than naïve (p < 0.11 vs. naïve) or inflamed mice (p < 0.99 vs. PenCON) at 1 day. #indicates no statistical differences between naïve or control inflammation groups at 1 day. (d) and (e) Examples of responses in A-HTMRs to heat and mechanical stimulation of the skin from the various conditions. Dashed arrow indicates onset of heat stimulus in (d). Horizontal bar over responses indicate duration of mechanical stimulus in (e). Statistics: One-way ANOVA with Tukey’s post hoc; mean ± SEM.
A-fiber HTMRs also had significantly higher firing rates to mechanical stimuli (p < 0.03 vs. naïve; n = 11) at one day in mice that received carrageenan injection alone (n = 9) or in mice with PenCON siRNA injection plus carrageenan (n = 11) similar to that seen in our recent report.4,32 Unexpectedly, downregulation of P2Y1 with saphenous nerve injection of PenY1 siRNAs blocked the inflammation-induced increase in A-fiber firing to mechanical stimuli at the one day time point (Figure 2(b); p > 0.05 vs. naïve; n = 9). No differences were detected in A-HTMR firing to mechanical stimuli at the three day time point in either group relative to naïve mice (Carr Only: n = 10; PenCON: n = 9; PenY1: n = 8). Decreased mechanical thresholds in A-HTMRs were also detected at the one day time point in both carrageenan injected mice and those with PenCON siRNA injection plus inflammation (Figure 2(c); p < 0.01 vs. naïve). A-fiber nociceptors in PenY1 siRNA injected mice with inflammation however were not found to be different than naïve or inflamed controls at one day (Figure 2(c); p > 0.05 vs. naive). No differences were found in any group at the three day time point.
We then wanted to confirm a lack of an effect of inflammation and P2Y1 knockdown on the “C”-fiber nociceptors in mice with hairy hindpaw skin injection of carrageenan at P7. Similar to earlier studies,
4
no changes in mean peak IFs to heat stimulation of the skin were found in the “C”-fiber HTMRs (slower conducting with broad spikes) either one or three days following P7 inflammation in either group (Figure 3(a); p < 0.09; Naïve: n = 6; Control Carr (1 day): n = 10; PenY1 (1 day): n = 6; Control Carr (3 days): n = 8; PenY1 (3 days): n = 10). Cells from carrageenan only animals (n = 1 from 1 day and n = 1 from 3 days) were added to results obtained from PenCON+Carr (n = 9 from 1 day and n = 7 from 3 days) for purposes of comparison among our groups.
Response properties of C-fiber HTMR neurons after cutaneous inflammation initiated at P7 compared to naïve controls as assessed with ex vivo recording. (a) At P7, carrageenan (Carr.) injection into the hairy hindpaw skin was found to slightly increase the mean peak instantaneous frequencies (IFs) of C-HTMRs to heat one day after inflammation; however, this was not found to be statistically significant (p < 0.33). No alterations were found in this afferent subtype in mice with P2Y1 knockdown in addition to cutaneous inflammation. No changes in mechanical firing rates (b) or thresholds (c) were found in the C-HTMRs at any time point after inflammation of the hairy skin initiated at P7 in mice that received carrageenan alone or in mice with PenCON or PenY1 injections plus carrageenan. (d) and (e) Examples of responses of C-HTMRs to heat and mechanical stimulation of the skin from the various conditions. Dashed arrow indicates onset of heat stimulus in (d). Horizontal bar over responses indicate duration of mechanical stimulus in (e). Statistics: Kruskal-Wallis with Dunn’s post hoc test; mean ± SEM.
Similar results were also found in these afferent subtypes in regard to firing rates to mechanical stimuli (Figure 3(b); p < 0.1; Naïve: n = 23; Carrageenan Only (1 day): n = 9; PenCON (1 day): n = 21; PenY1 (1 day): n = 7; Carr Only (3 days): n = 11; PenCON (3 days): n = 16; PenY1 (3 days): n = 28) and thresholds to mechanical deformation of the skin (Figure 3(c); p < 0.33) in mice with carrageenan injection only, or in mice with PenCON or PenY1 injections in addition to cutaneous inflammation at both time points tested. Similar results were obtained if data were separated into mechanically sensitive but heat insensitive (CM) and polymodal C-fiber (CPM) HTMR subpopulations (not shown).
We also detected no differences in response properties of the rapidly or slowly adapting A-fiber low threshold mechanoreceptors (faster conducting cells with narrow spikes) after carrageenan injection at P7. There were also no differences detected in the low threshold mechanically sensitive and cold sensitive C-fibers (slower conducting cells with narrow spikes). Mechanically insensitive but cold or heat sensitive C-fiber neurons (slower conducting, but broad spiking) also displayed no differences among any of our experimental groups (not shown).
Numbers of “A”- and “C”-fiber sensory neurons in mice with cutaneous inflammation, with or without siRNA injections compared to naïve.
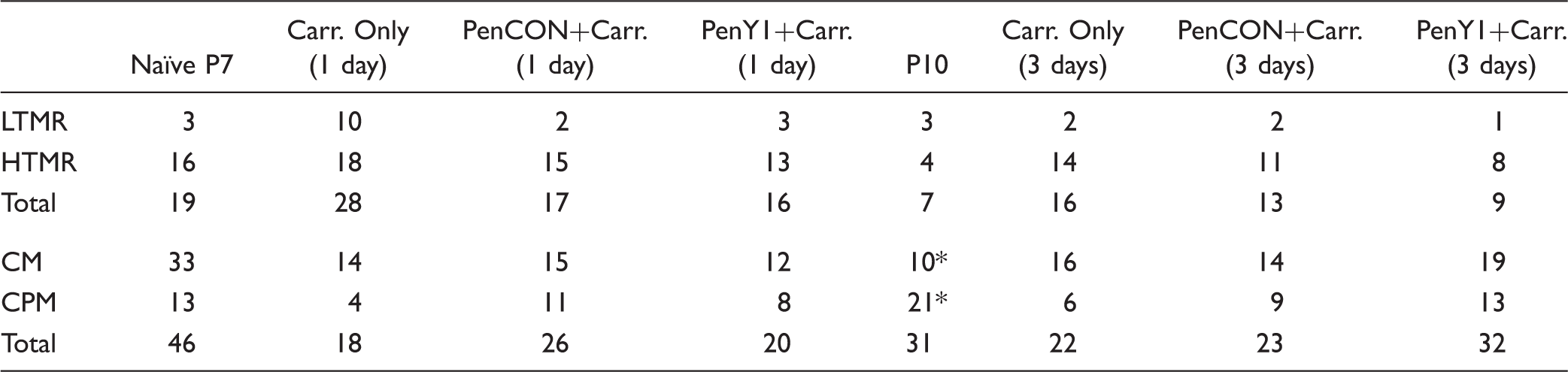
LTMR: low threshold mechanoreceptor; HTMR: high threshold mechanoreceptor; CM: C-mechanoreceptor; CPM: C-polymodal.
p < 0.02, Chi square.
P2Y1 knockdown in vivo blocks carrageenan-induced TRPV1 expression in the DRGs
We then assessed the labeling patterns of TRPV1 and IB4 in the DRGs of mice from our groups (n = 3 each) to assess the role of P2Y1 in neurochemical alterations after inflammation.
4
Immunocytochemical analysis indicated that there was a significant increase in TRPV1-positive cells in the DRGs of mice that received cutaneous inflammation alone or in conjunction with PenCON siRNA injections at one day compared to naïve DRGs (p < 0.045 vs. naïve). P2Y1 knockdown was found to block this de novo expression of TRPV1 (Figure 4). Analysis also revealed differences in IB4 binding after inflammation. Carrageenan-induced inflammation, with or without PenCON injection, increased the number of IB4 positive cells in the DRGs at 1 day compared to naive, but this increase was also detected in mice with knockdown of P2Y1 (Figure 4; p < 0.021). A limited number of cells containing both markers were found in the DRGs of mice from each condition (n < 2), and this was not found to be different between groups (Figure 4; p < 0.6).
Selective knockdown of P2Y1 alters the de novo expression of TRPV1 in the DRGs one day after carrageenan-induced inflammation at P7. (a) Examples of immunostaining in DRGs for TRPV1 and IB4 binding from naïve, PenCON+Carr. and PenY1+Carr. mice. Yellow arrows indicate positive TRPV1 staining, while white arrows designate IB4 binding in sensory neurons of the DRGs. (b) The average number of cells per section that were immunopositive for TRPV1 was significantly increased one day after P7 inflammation in mice with or without PenCON siRNA injections. However, this was completely blocked by P2Y1 knockdown. Significantly increased IB4 binding in the DRGs was found in all groups with inflammation at one day compared to naive. *p < 0.05 versus naïve. One-way ANOVA with Holm Sidak post hoc test; mean ± SEM.
Discussion
Increased P2Y1 expression broadly regulates “A”-fiber HTMR sensitization during neonatal inflammation
We initially hypothesized that the observed upregulation of the purinergic receptor P2Y1 (Figure 1) may play a critical role in primary afferent heat sensitization during neonatal cutaneous inflammation. In adult mice, this receptor has previously been linked to alterations in heat sensitivity in CPMs in addition to phenotypic changes in these afferents during inflammation of the periphery.5,6,33 We found a surprising result here in that specific knockdown of P2Y1 upregulation in the DRGs using our nerve-specific siRNA-mediated knockdown strategy (Figure 1) could blunt carrageenan-induced changes in neonatal “A”-fiber HTMR heat and mechanical responses (Figure 2). This suggests that the upregulation of P2Y1 in sensory neurons may play a broader role in the development of peripheral sensitization during early postnatal inflammation of the periphery.
“A”-fiber primary sensory neurons that are classified as HTMRs, undergo a normal loss of heat sensitivity during the first week of life, around the same time that other sensory neurons are undergoing a switch in their responsiveness to various neurotrophic factors.31,34–36 As a result, these A-fiber nociceptors are almost fully developed at early stages (P7), while the C-fibers are continuing to undergo normal development. It is therefore not surprising that the A-fibers can sensitize in these very young neonates, while A- and C-fibers are often sensitized in older animals.4,5,31,37–39 In essence, different classifications of developed sensory neurons are available to respond to peripheral insults at distinct neonatal ages.
Recent studies have proposed that alterations in neonatal spinal neuron responses to A-fiber input could have a strong influence on dorsal horn circuit development. This would subsequently affect how the nervous system responds to peripheral stimuli at older ages.9,20,40–43 Our previous and current data support this notion in that we specifically found sensitization of “A”-fiber neurons to heat and mechanical stimuli during cutaneous inflammation induced at P7 (Figure 2), but no alterations in the responsiveness of “C”-fiber neurons (Figure 3). Our new results suggest that the upregulation of P2Y1 in cutaneous nociceptors after inflammation during postnatal sensory development may have a profound effect on spinal cord circuit organization due to the observed effects on “A”-fiber sensitization. The hastened time course of central nervous system development in neonates observed under pathological conditions20,44 may be strongly influenced by P2Y1-mediated “A”-fiber responsiveness. This highlights the importance of future studies designed to better understand early life responses in peripheral sensory neurons, and how they influence spinal cord development to subsequently modify the transition from acute to chronic neonatal pain.
Possible role for P2Y1 in sensory neuron phenotypic identity during neonatal inflammation
Our immunohistochemical analysis indicated that there was a significant increase in TRPV1-immunoreactive cells in DRGs within one day of inflammation relative to naïve mice, while knockdown of P2Y1 blocked this Carrageenan-induced increase in TRPV1-positive neurons (Figure 4). The current report however suggests that the effects of P2Y1 knockdown on TRPV1 expression may be in “A”-fiber neurons (Figure 2). One possible explanation for this is that during development TRPV1 may be expressed in a different number of DRG neurons compared to that seen in adult DRGs.45–48 Thus the normal age-related pattern of TRPV1 expression in the DRGs combined with differences in age of inflammation across studies may explain the discrepancy in how P2Y1 may affect TRPV1 expression and possibly peripheral responsiveness postinflammation.
It is well-known that specific populations of sensory neurons switch their neurotrophic factor responsiveness from nerve growth factor to glial-derived neurotrophic factor during the first two weeks of life.49,50 These phenotypic switches accompany alterations in various receptors and channels linked to transduction of peripheral stimuli including TRPV1.35,51 Although our results do not show an effect of P2Y1 on the overall distribution of various fibers types (Table 1), the P2Y1-dependent alterations in TRPV1 expression (Figure 4) may suggest that P2Y1 plays a role in phenotypic identity of neonatal sensory neurons in addition to responsiveness of nociceptors to peripheral stimuli.
TRPV1 expression during inflammation is known to be crucial for the development of thermal hyperalgesia 52 as well as the responsiveness of afferents to heat stimuli after peripheral injuries.26,52,53 Recent studies have assessed how P2Y1 specifically can regulate the expression of TRPV1 in the DRGs; however, the results are somewhat contradictory.6,7,54 Kwon et al. 55 found that the inhibition of P2Y1 receptors can downregulate the expression of TRPV1 during cutaneous inflammation, while other recent reports have suggested that downregulation of P2Y1 during adult cutaneous inflammation can increase the expression of TRPV1 in specific subpopulations of cutaneous nociceptors. 6 In adult mice, P2Y1 receptors can directly regulate TRPV1 expression by modulating p38 MAPK in the DRGs 8 or indirectly by releasing protaglandin E (PGE) and calcitonin gene-related peptide (CGRP).56,57
It is also possible that P2Y1 effects on TRPV1 expression could regulate mechanical response properties in addition to heat in neonates. A few reports have linked the expression of TRPV1 to mechanical responsiveness during injury.58,59 Since neonates appear to have distinct expression patterns of TRPV1 in the DRGs as well as P2Y1-mediated responses compared to adults,45–48 the effects of P2Y1 on TRPV1 expression may also serve to regulate mechanical responsiveness in neonates during cutaneous inflammation. In support of this, MAPK signaling has been suggested to have unique functions in adult DRGs that are not observed in neonates. 60 It is therefore possible that this developmental discrepancy accounts for the differences we observe for P2Y1 function in modulating both mechanical and heat sensitivity in neonatal DRG neurons. P2Y1-mediated regulation of heat sensitivity may be maintained into adulthood,5,6 while its role in mechanical responsiveness may be eliminated. Other possibilities for the observed differences include species (rat vs. mouse) and inflammatory pain model (carrageenan vs. complete Freund’s adjuvant) between the current and previous reports.5,6,8 It is also possible that other targets of P2Y1 signaling regulate mechanical responsiveness that were not tested here. However, this would need to be confirmed in the future in order to fully appreciate the novel role for P2Y1 in developmental sensitization after injury.
Conclusions
Early life injuries in the periphery are known to have long-term effects on nociceptive processing later in life.9,42,43,61,62 As the peripheral and central nervous systems undergo a variety of neurochemical and functional changes during the initial stages of development,4,15,31,35,50,63–66 it is not surprising that injuries sustained in neonates can produce alterations in sensory function that differ from adults.5,6,38,67 Here, we have found a novel role for the upregulation of the adenosine diphosphate responsive G-protein coupled receptor, P2Y1 in the development of both heat and mechanical hypersensitivity in primary “A”-fiber nociceptors during early postnatal cutaneous inflammation. This appears to potentially take effect in part by its regulation of TRPV1 expression in the DRGs during neonatal inflammation of the periphery. Although little is known about the role of P2Y receptors in the development of pain in humans, studies have shown that P2Y1 can play a role in human nociceptor sensitization. 68 Taken together with our current results, this may support a potential role for P2Y1 in the development of neonatal inflammatory pain and could provide a unique direction for the development of specific neonatal pain therapeutics.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NIH (NICHD: R03HD077483-01 and NIAMS: R01AR064551-01A1; MPJ), the Rita Allen Foundation/American Pain Society (MPJ), the Board of Trustees at Cincinnati Children’s Hospital Medical Center (CCHMC), and the Department of Anesthesia at CCHMC.
