Abstract
Background
Experimental osteoarthritis entails neuropathic-like changes in dorsal root ganglia (DRG) neurons. Since glial activation has emerged as a key player in nociception, being reported in numerous models of neuropathic pain, we aimed at evaluating if glial cell activation may also occur in the DRG and spinal cord of rats with osteoarthritis induced by intra-articular injection of collagenase.
Methods
Osteoarthritis was induced by two injections, separated by three days, of 500 U of type II collagenase into the knee joint of rats. Movement-induced nociception was evaluated by the Knee-Bend and CatWalk tests during the following six weeks. Glial fibrillary acidic protein (GFAP) expression in satellite glial cells of the DRG was assessed by immunofluorescence and Western Blot analysis; the pattern of GFAP and activating transcription factor-3 (ATF-3) expression was also compared through double immunofluorescence analysis. GFAP expression in astrocytes and IBA-1 expression in microglia of the L3–L5 spinal cord segments was assessed by immunohistochemistry and Western Blot analysis. The effect of the intrathecal administration of fluorocitrate, an inhibitor of glial activation, on movement-induced nociception was evaluated six weeks after the first collagenase injection.
Results
GFAP expression in satellite glial cells of collagenase-injected animals was significantly increased six weeks after osteoarthritis induction. Double immunofluorescence showed GFAP upregulation in satellite glial cells surrounding ATF-3-positive neurons. In the spinal cord of collagenase-injected animals, an ipsilateral upregulation of GFAP and IBA-1 was also observed. The inhibition of glial activation with fluorocitrate decreased movement- and loading-induced nociception.
Conclusion
Collagenase-induced knee osteoarthritis leads to the development of nociception associated with movement of the affected joint and to the activation of glial cells in both the DRG and the spinal cord. Inhibition of glial cell activation by fluorocitrate decreases these osteoarthritis-associated nociceptive behaviours. These results suggest that glial cell activation may play a role in the development of chronic pain in this experimental model of osteoarthritis.
Introduction
Pain associated with osteoarthritis (OA) is yet to be fully understood. Recent studies have been pointing towards the possibility of a neuropathic component in OA-associated nociception.1–4 Recently, our group has shown that OA progression leads to the expression of neuronal injury markers in the dorsal root ganglia (DRG), both in collagenase- 1 and monoiodoacetate (MIA)-induced OA. 2 Using the collagenase model, we also demonstrated that, although inflammation may contribute to nociceptive behaviours at the onset of OA, neuropathic-like changes become preponderant as disease progresses. This is highlighted by the inefficacy of non-steroidal anti-inflammatory drugs at late time points, in contrast to the efficacy of gabapentin when experimental OA is fully developed.1,5
The neuropathic-like features in OA pain may arise from injury of nerve endings in the subchondral bone, which becomes exposed as disease progresses.1,2,5 Peripheral nerve injury (PNI) is known to activate both neurons and glia of the peripheral nervous system (PNS) and central nervous system (CNS).6–9 In the PNS, satellite glial cells (SGCs) that tightly surround neurons in the DRG 10 are activated following PNI, contributing to the maintenance of neuropathic pain.11–13 This activation is characterized, among others, by cell proliferation and hypertrophy, increased number of gap junctions and cell coupling, and altered production of various molecules, including an upregulation of glial fibrillary acidic protein (GFAP) expression.11,14,15 GFAP is present at low levels in non-activated SGCs, 12 but it is overexpressed in SGCs in different models of PNI,12,16–20 being a marker of their activation.21,22 In the CNS, glial cells also participate in normal and pathological processes23–26; there is strong evidence that the activation of both microglia and astrocytes is implicated in the induction and maintenance of neuropathic pain by mediating important neuron-glia cross-talk mechanisms.6–9 Several studies have shown that the selective inhibition of these glial cells’ metabolism attenuates the nociceptive behaviour in various pain models.12,27 One of the drugs currently used is fluorocitrate, which selectively inhibits the tricarboxylic acid cycle in SGCs, astrocytes and microglia by blocking aconitase, a glia-specific metabolic enzyme,28,29 and that has been shown to effectively reduce mechanical allodynia in animal models of neuropathic pain. 12
In this study, we hypothesized that, after six weeks of collagenase-induced OA development, when the neuropathic component of nociception is prominent, glia activation may also contribute to the mechanisms of nociception. To test this hypothesis, we assessed the activation of SGCs in the DRG of OA animals through the expression of GFAP, and we evaluated whether there is a link between PNI and SGC activation by performing a double immunofluorescence analysis of the expression of the neuronal injury marker activating transcription factor (ATF)-3 in neurons 30 and of GFAP in their enveloping SGCs. We also assessed the activation of astrocytes and microglia in the spinal cord (SC) through the expression of GFAP 7 and IBA-1, 31 respectively. Finally, we tested the effect of the intrathecal administration of fluorocitrate on movement- and loading-induced nociceptive behaviour in the collagenase-induced OA animals. With these experiments, we aimed at determining if, at six weeks of OA progression, when neuropathy correlates with nociception, 1 there is also glial cell activation in the DRG and SC, as seen in models of neuropathic pain, and if their inactivation can decrease the nociceptive behaviour.
Material and methods
Animals
Adult male Wistar rats (230 ± 30 g, Charles River, France) were housed with water and food ad libitum, at a constant temperature of 22℃ and controlled lighting (12 h light/12 h dark cycle). Experimental procedures were performed in accordance with the ethical guidelines for the study of experimental pain in conscious animals, 32 and the European Council Directive 2010/63/EU, and were approved by the Ethical Committee for Health of Centro Hospitalar de São João, Porto, Portugal.
Osteoarthritis induction
Intra-articular (i.a.) injections were performed using a Hamilton syringe with a 26-G needle inserted through the patellar ligament into the left knee joint cavity of animals briefly anesthetized with isoflurane (5% for induction, 2% for maintenance). Animals received two 25 -µL injections, one on day 0 and another on day 3, of either sterile saline (control group) or 500 U of type II collagenase from
Nociceptive behaviour
A total of 37 rats injected with saline or collagenase were divided in three experimental groups. One group was used for immunohistochemical analysis (11 animals;
Tissue processing
For the immunohistochemistry group, animals were perfused with 4% paraformaldehyde six weeks after the first injection of saline or collagenase. Their ipsilateral L3, L4 and L5 DRG and the L3–L5 SC segments were dissected, post-fixed in the same fixative (4 h) and kept in 30% sucrose with 0.01% sodium azide. DRG were serially sliced in 12 µm sections using a cryostat, with every tenth section collected in the same slide. Each DRG was always cut longitudinally yielding 8–10 sections per slide, on average. SC segments were serially sliced in 40 -µm free-floating transverse sections using a freezing microtome. The contralateral side was identified with a small cut in the ventral horn before sectioning. Sections were then stored at −20℃ in a cryoprotective solution until further processing. 35
Immunohistochemistry
Immunofluorescence reactions in the DRG were performed in slides containing every tenth section of ipsilateral L3, L4 or L5 DRG of rats sacrificed six weeks after collagenase or saline injection, as previously described.5,36 DRG sections were incubated with either rabbit anti-GFAP (1:1000, Abcam) or mouse anti-ATF-3 (1:200, Abcam), overnight at room temperature. Secondary detection was performed with either Alexa Fluor-568 donkey anti-rabbit or Alexa Fluor-488 donkey anti-mouse secondary antibodies (1:1000, 1 h, room temperature, Molecular Probes). Slides were mounted with glycerol. Negative controls were performed without the primary antibody. Images were acquired in a fluorescence microscope (Axio Imager.Z1), through an AxioCam MRm digital camera with AxioVision 4.6 software (Carl Zeiss MicroImaging GmbH). For GFAP labelling, all neurons having at least half of its circumference surrounded by GFAP-labelled SGCs were considered GFAP-encircled neurons,12,36 and were counted. This number was divided by the total number of neurons counted in the analysed sections. Results are presented as the percentage of GFAP-encircled neurons in L3, L4 and L5 DRG. For ATF-3, the total number of neurons and the number of labelled neurons were counted. Data are presented as the percentage of neurons expressing ATF-3 in L3, L4 and L5 DRG. Double labelling for ATF-3 and GFAP was performed by simultaneous reaction for both antibodies and quantified as described.
Immunohistochemistry for GFAP and IBA-1 in the SC was performed as previously described. 37 Briefly, the endogenous peroxidase activity was inhibited with phosphate-buffered saline (PBS) containing 0.3% hydrogen peroxide for 30 min, and sections were then incubated for 1 h in a blocking solution (glycine 0.15 M and 10% normal swine serum). Immunodetection was performed with either rabbit anti-GFAP (1:2500, overnight RT, Abcam) or rabbit anti-IBA-1 (1:2000, overnight RT, Wako). For secondary detection, sections were incubated for 1 h in biotinylated swine anti-rabbit antiserum (1:200, Dako) and for another hour in avidin–biotin complex (ABC) conjugated with horseradish peroxidase (1:200, Vector Labs). Visualization of the immunostaining was achieved using the 3,3’-diaminobenzidine tetrahydrochloride (DAB) reaction (5 min in 0.05 M Tris buffer containing 0.05% DAB and 0.003% hydrogen peroxide). Sections were mounted on gelatine-coated slides, cleared in xylene and coverslipped with Eukitt (Sigma Aldrich).
Labelling was quantified in random sections from the L3, L4 and L5 segments of the SC (6 sections per segment). Images were captured with a light microscope (Axioskop 40) coupled with an AxioCam MRc5 digital camera and Axiovision 4.6 image software (Carl Zeiss MicroImaging GmbH). The acquisition conditions such as amplification of the objective, light intensity, contrast and hue were maintained constant. Immunoreactivity was quantified by densitometry in the ipsilateral and contralateral dorsal horn using Image J. Laminae I–III were delimited taking as reference the Rat Brain Atlas of Paxinos and Watson, 38 and the same area was used in the ipsilateral and contralateral sides. Results are presented as the ipsilateral/contralateral ratio.
Western Blot
WB experiments were performed to evaluate the expression of GFAP in the DRG, and the expression of both GFAP and IBA-1 in the SC of control and collagenase injected rats, six weeks after the first injection. Animals were euthanized by decapitation under pentobarbital anaesthesia and samples were extracted from freshly harvested L3–L5 ipsilateral and contralateral DRGs and SC segments. For each animal, the L3–L5 SC segments or DRG were pooled separately for the ipsilateral and contralateral sides, and were then lysed in radio immunoprecipitation assay (RIPA) buffer (Sigma Aldrich) containing cocktails of protease and phosphatase inhibitors (1:100, Sigma-Aldrich P8340, P5726 and P0044). The samples were centrifuged (20 minutes at 20,000 g), the pellets were discarded and the supernatants were used for analysis. The proteins were quantified by the bicinchoninic acid protein assay. After heating at 94℃, 20 mg of protein were loaded in each lane and separated on 12.5% sodium dodecyl sulphate-polyacrylamide (SDS/PAGE) gels. The proteins were then transferred into nitrocellulose membranes which were blocked with non-fat milk (5% milk powder diluted in Tris buffer saline tween20; TBST buffer) for 1 h, at room temperature, to prevent non-specific bindings. Immunodetection was performed by overnight incubation at 4℃ with mouse anti-GFAP (1:5000, 51 kDa, Millipore) or rabbit anti-IBA-1 (1:500, 17 kDa, Wako). Rabbit anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH, 1:5000, 37 kDa, Abcam) was used as a loading control. Secondary detection was performed by incubation with goat anti-mouse secondary antibody conjugated with horseradish peroxidase (HRP, Santa Cruz Biotechnology, Inc) or donkey anti-rabbit secondary antibody conjugated with HRP (Jackson Laboratories), diluted 1:5000 in TBST with 5% milk powder, for 1 h at room temperature. Antibody binding was visualized with the SuperSignal West Pico Chemiluminescent Substrat kit (Thermo Scientific), and the bands were detected using a ChemiDoc MP System 170-8280 (Bio-Rad). Each blot, containing independent samples, was run in duplicates, and means were used as raw values. Band intensity was determined by densitometric analysis of the signal intensity in the blots using Image J. Both the areas of the lanes and the background signal were used for normalization. The ratio between GFAP or IBA-1 and GAPDH protein levels was calculated for each sample. Additionally, ratios between the ipsilateral and contralateral levels were calculated in control and collagenase-injected animals.
Inhibition of glial activation
The effect of a single administration of the glial activation inhibitor fluorocitrate in the behavioural responses of OA animals was evaluated in a different experimental group. At six weeks after collagenase injection, fluorocitrate (Sigma-Aldrich) was administered intrathecally in order to reach the lumbar spinal segments (
Statistical analysis
Results are presented as mean ± SEM. The normality of all data was assessed by the Kolmogorov–Smirnov test. Behavioural data for movement induced-nociception in control and collagenase-injected groups was analysed by two-way analysis of variance (ANOVA), for factors time and group, followed by Bonferroni post-hoc test for multiple comparisons between groups. The Mann–Whitney test was used for the immunofluorescence and WB data. The pharmacological data were analysed by repeated measures ANOVA followed by Dunnett’s post-hoc test. In all statistical analysis, a level of significance of
Results
Movement-induced nociception
The Knee-Bend (Figure 1(a)) and CatWalk (Figure 1(b)) tests were used to evaluate movement-induced nociception following the i.a. injection of saline or collagenase. In both tests, saline-injected control animals showed, at all time points, behavioural responses similar to those observed prior to the injection. In collagenase-injected animals, the more pronounced changes were observed one week after the first collagenase injection (Figure 1(a) and (b)). Knee-Bend scores (Figure 1(a)) increased from 0.1 ± 0.1 on day 0 to 11.3 ± 0.9 at week 1 ( Nociceptive behaviour. Nociception associated with movement and loading on the joint was evaluated by the Knee-Bend (a) and CatWalk (b) tests in saline-injected control and collagenase-injected OA animals. Knee-Bend score (a) is presented as the difference between ipsilateral and contralateral scores. CatWalk data (b) are expressed as the percentage of total ipsilateral paw print intensity (%TIPPI). Mean ± SEM, two-way ANOVA followed by Bonferroni post-hoc test for comparisons between groups at each time point. ***
Glial activation in the DRG
SGC activation in the DRG was assessed through the expression of GFAP both by immunofluorescence and WB analysis. For GPAP immunolabelling (Figure 2(a) and (b)), the percentage of GFAP-encircled neurons six weeks after OA induction increased from 5.6 ± 1.4% in controls to 21.9 ± 3.2% in collagenase-injected animals ( GFAP expression in the DRG. Representative images of GFAP labelling in SGCs of L4 DRG, six weeks after the first saline (a) or collagenase (b) injection; Scale bar: 50 µm. (c) Immunofluorescence analysis of GFAP expression in L3, L4 and L5 ipsilateral DRG of saline- and collagenase-injected rats, six weeks after injection; GFAP expression significantly increased at six weeks of OA. Data are presented as the percentage of GFAP-encircled neurons in L3, L4 and L5 DRG. (d) Western Blot analysis of GFAP levels in L3–L5 DRG of control and collagenase-injected rats, six weeks after injection. Results are presented as the ipsilateral/contralateral ratio of GFAP levels (GFAP/GAPDH values). Mean ± SEM. Mann–Whitney test, **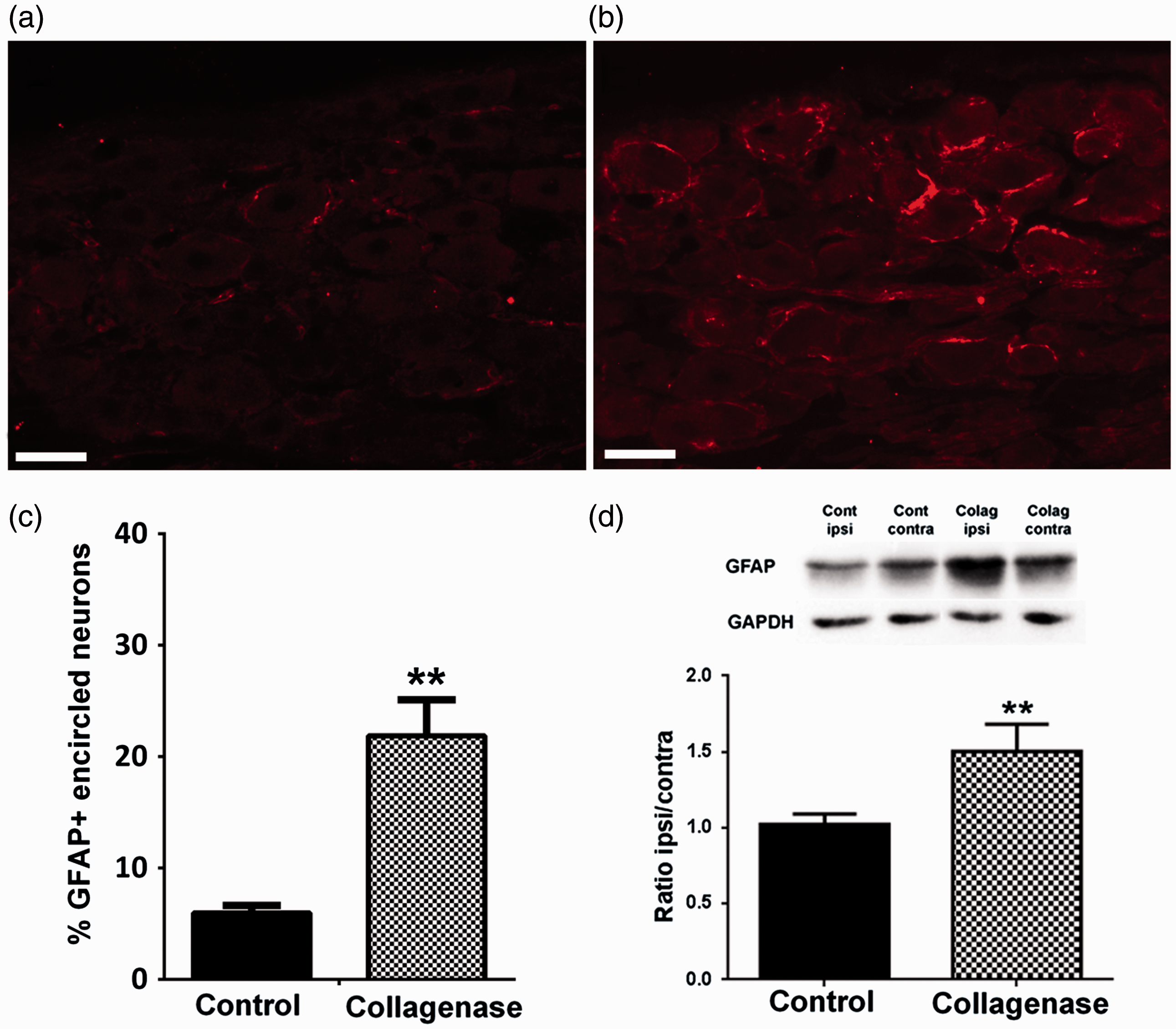
In order to determine if GFAP was preferentially expressed in SGCs surrounding injured neurons, double immunolabelling for GFAP and ATF-3 (Figure 3(a) and (b)) was performed for the six weeks of OA group. While in control animals 7.5 ± 5.0% of ATF-3-positive neurons were encircled by GFAP-labelled SGCs, in collagenase-injected animals this percentage increased to 35.0 ± 4.0% ( ATF-3 and GFAP double labelling. Representative images of double immunofluorescence labelling for ATF-3 and GFAP in L4 ipsilateral DRG of saline-injected (a) and collagenase-injected (b) rats, six weeks after injection. Scale bar: 50 µm. (c) The percentage of GFAP-encircled neurons within the ATF-3 positive population increased in collagenase-injected animals in comparison with control animals. Mean ± SEM. Mann–Whitney test; **
Glial activation in the SC
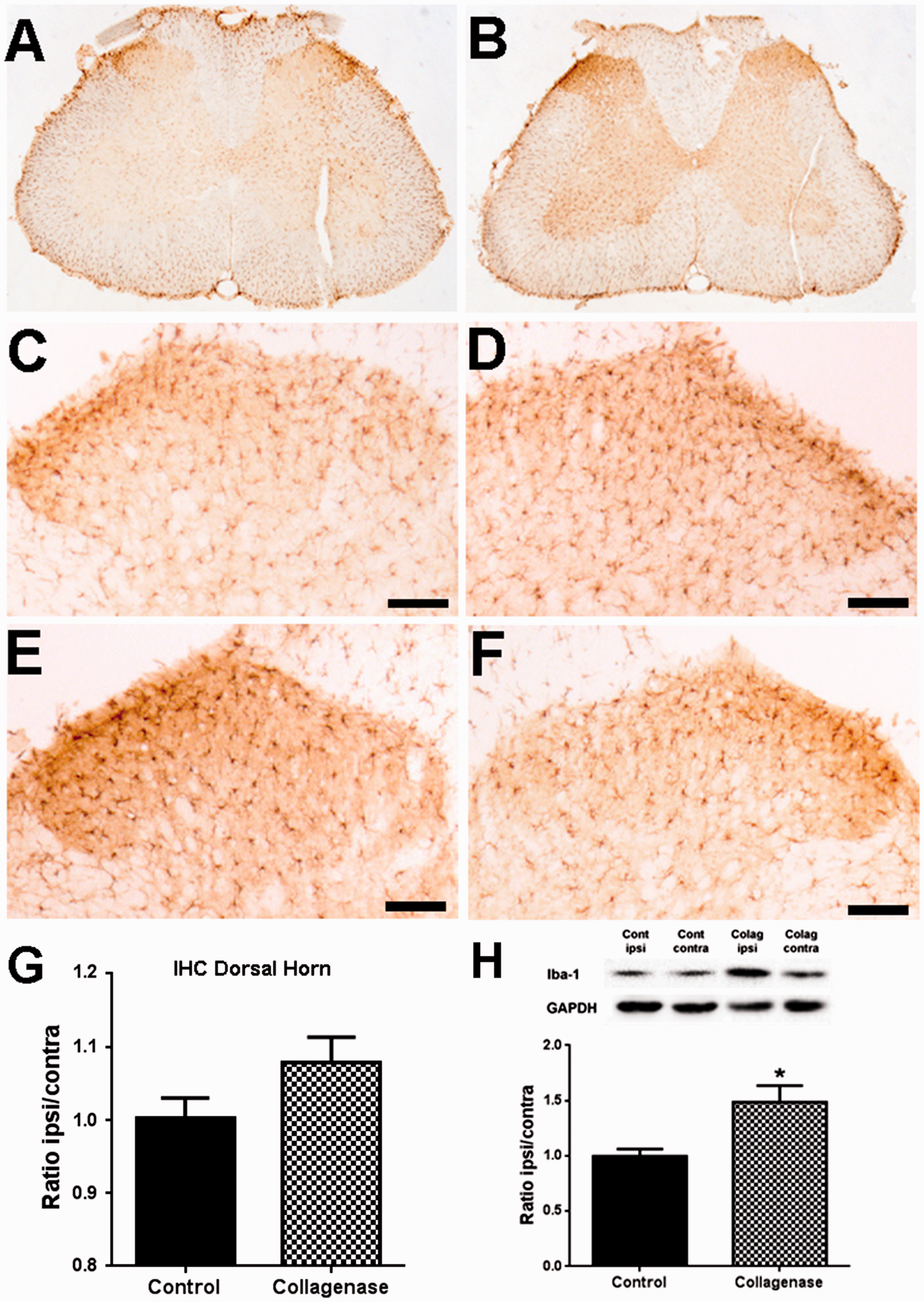
The activation of astrocytes and microglia in the SC was analysed through the expression of GFAP (Figure 4) and IBA-1 (Figure 5), respectively. Immunohistochemistry (Figure 4(a) to (f)) followed by densitometric (Figure 4(g)) analysis was used to evaluate expression changes specifically in laminae I–III of the spinal dorsal horn. There was a small but significant increase in the ipsilateral/contralateral ratio of GFAP expression in the dorsal horn of the L3–L5 segments of the SC of collagenase-injected rats; control animals (Figure 4(c) and (d)) had a ratio of 1.01 ± 0.01, whereas OA rats (Figure 4(e) and (f)) had a ratio of 1.12 ± 0.03 ( GFAP expression in the spinal cord. Representative images of GFAP labelling in the SC, six weeks after the first saline (a) or collagenase (b) injection. Representative images of GFAP labelling in the ipsilateral (c, e) and contralateral (d, f) dorsal horn, six weeks after the first saline (c, d) or collagenase (e, f) injection; Scale bar: 50 µm. (g) Densitometric quantification of GFAP labelling in laminae I-III of the dorsal horn of control and collagenase-injected rats six weeks after injection, presented as the ipsilateral/contralateral ratio. (h) Quantitative WB analysis of GFAP expression in the spinal cord of control and collagenase-injected rats six weeks after injection, presented as the ipsilateral/contralateral ratio of GFAP levels (GFAP/GAPDH). Mean ± SEM. Mann–Whitney test; ** Iba-1 expression in the Spinal Cord. Representative images of Iba-1 labelling in the SC, six weeks after the first saline (a) or collagenase (b) injection. Representative images of Iba-1 labelling in the ipsilateral (c, e) and contralateral (d, f) dorsal horn, six weeks after the first saline (c, d) or collagenase (e, f) injection; Scale bar: 50 µm. (g) Densitometric quantification of Iba-1 labelling in laminae I–III of the dorsal horn of control and collagenase-injected rats six weeks after injection, presented as the ipsilateral/contralateral ratio. (h) Quantitative WB analysis of Iba-1 expression in the spinal cord of control and collagenase-injected rats six weeks after injection, presented as the ipsilateral/contralateral ratio of Iba-1 levels (Iba-1/GAPDH). Mean ± SEM. Mann–Whitney test; **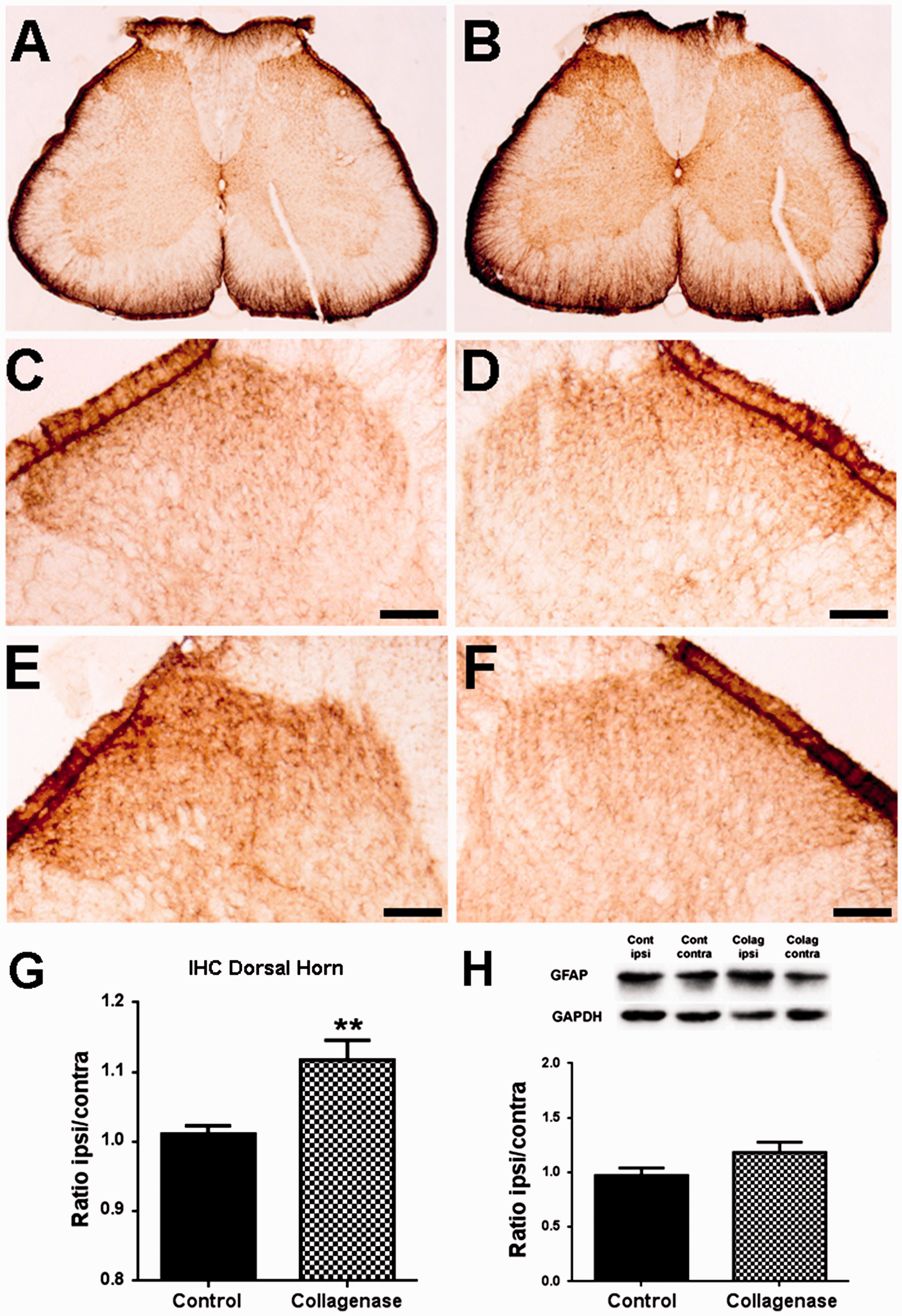
Inhibition of glial activation
The effect of fluorocitrate on movement-induced nociception in collagenase-injected animals with six weeks of OA development was also evaluated (Figure 6). All animals showed significant nociceptive behaviour before drug or vehicle administration ( Fluorocitrate administration. The effect of fluorocitrate administration (30 µL, 0.1 nM, intrathecal) on movement- and loading-induced nociception was assessed by the Knee-Bend (a) and the CatWalk (b) tests six weeks after injection of collagenase. Nociception was assessed on day 0, before the first collagenase injection, and before fluorocitrate administration for determination of baseline values (open circles); the effect of fluorocitrate or vehicle was determined 1, 2, 3, 4 and 6 h after administration (filled symbols). Mean ± SEM. Repeated measures ANOVA followed by Dunnett’s post-hoc test; *
Discussion
Glial activation has been increasingly regarded has a key player in the modulation of nociception. Therefore, this study aimed at determining if glial activation could also contribute to the mechanisms of nociception in knee OA induced by i.a. injection of collagenase in rats. Overall, we observed that there is indeed an increased glial activation both in the DRG and in the SC of OA rats. Furthermore, we observed that the inhibition of glial activation can decrease the nociceptive behaviours associated with movement and loading on OA joints, which mimic patients’ major complaints. 42
In previous studies using the collagenase model of OA, we showed an overexpression of the neuronal injury markers ATF-3 and NPY in the DRG of OA animals. 1 After six weeks of OA progression, there was extensive articular degeneration that correlated with the nociceptive behaviour. 5 The degree of articular degeneration also correlated with the upregulation of neuronal injury markers, 1 as well as with a progressive decrease in fluorogold (FG) backlabelling from collagenase-injected joints, 1 which may be attributed to injury of nerve endings interfering with FG uptake. These findings, along with the observation that gabapentin, effective in neuropathic pain, could reverse the nociceptive behaviour in the collagenase model, 1 strongly pointed to the occurrence of PNI and to a potential neuropathic component in the mechanisms of nociception associated to OA in this model.
Given the known effect of PNI on the activation state of glia in both the PNS and CNS,6–9 we evaluated whether the progression of collagenase-induced OA could have a similar effect on glial activation at six weeks, when all the major changes had been detected and neuropathy correlated with nociception. We observed a significant increase in GFAP expression, a marker of SGC activation, in the DRG of OA animals after six weeks of OA progression, as shown both by WB and immunofluorescence. Aiming at establishing a link between neuronal injury and SGC activation, we evaluated if SGCs overexpressing GFAP were preferentially located around injured neurons identified by ATF-3 expression. 30 A significant increase in the proportion of GFAP-encircled neurons within the ATF-3-expressing population was indeed found in collagenase-injected animals, with around 35% of ATF-3-positive neurons being surrounded by activated SGCs. Nevertheless, increased GFAP expression was also observed around neurons that did not have upregulated ATF-3 expression. Likewise, studies in chronic constriction injury of the infraorbital nerve have shown that, although SGC activation occurs preferentially around trigeminal ganglia neurons expressing ATF-3, GFAP expression associated with SGC activation is not restricted to those neurons. 43 SGC activation leads to an increase in the number of gap junctions, which are known to promote communication between adjacent SGCs enveloping neighbouring neurons and even contribute to nociception.44–46 Furthermore, there are numerous molecules released by neurons whose receptors are found in SGCs, revealing potential pathways of neuroglial crosstalk and of SGC activation.47,48 Indeed, SGCs have been increasingly regarded as more than just supporting cells in the DRG, 15 and evidence for a role of SGCs in direct neurochemical transmission between neighbouring neurons has emerged recently.49,50 Functional changes induced by neuronal injury in collagenase-induced OA may therefore be communicated to neighbouring neurons and SGCs, leading to the activation of SGCs around uninjured neurons.
Glial activation in the CNS also plays an important role in neuropathic pain.6–9 Here, we show that collagenase-injected rats have increased IBA-1 expression at six weeks in the ipsilateral hemisegment of the SC analysed by WB; immunohistochemical analysis of laminae I–III of the spinal dorsal horn showed no significant changes. A different pattern was observed for GFAP expression, which was shown to be increased in laminae I-III of the ipsilateral spinal dorsal horn while WB analysis of the whole ipsilateral hemisegment showed no significant changes. In models of neuropathic pain, microglial activation seems to occur preferentially during the early phases of disease development.9,51 Neuronal injury induces a set of progressive alterations in spinal microglia, with early morphological changes 52 being followed by proliferation in the ipsilateral spinal dorsal horn 53 associated with increased secretion of cytokines, chemokines and neurotrophic factors that alter neuronal excitability. This microglia–neuron communication is increasingly recognized as a key contributor to neuropathic pain. 54 Thakur et al. 3 have previously reported increased spinal microglial activation in both the ipsilateral dorsal and ventral horns in the MIA model of OA. Other studies also showed increased microglial activation specifically in the ipsilateral dorsal horn. The main variable between these studies was the temporal profile of microglial activation, with dorsal horn changes peaking either 7 3 or 14 days 55 after OA induction and decreasing thereafter, or being maintained in later stages of OA, after 28 days 56 or 6 weeks. 57 Although it is possible that dorsal horn specific changes occur at earlier time points of collagenase-induced OA development, at six weeks, significant microglial activation, as evaluated by IBA-1 increased expression, was observed in the whole ipsilateral spinal hemisegment. Microglial cells have a characteristic low threshold for reactivity and are known to respond to a wide range of stimuli. 58 It is possible that initial changes occurring in the spinal dorsal horn may extend to other areas of the SC through intercellular communication, inducing the widespread activation observed in this model. Although it would be interesting to assess if microglial activation occurs earlier in OA progression, this was not our goal in this study since we focused this analysis on a time point when OA-like changes are already fully developed. 5
Astrocyte activation associated with neuropathic pain has been shown to start at later stages after injury and to be long lasting.9,51 Interestingly, a similar pattern has been observed in animal models of OA. Indeed, in the MIA model, no significant changes in GFAP expression in the SC have been observed until 14 days of disease progression, 3 but an increased GFAP expression in the dorsal horn at later time points ranging from three to six weeks has been reported.55,57 Likewise, we observed a significant increase in GFAP expression in the ipsilateral dorsal horn. This preferential increase of GFAP in laminae I–III may be associated with an increased activity of nociceptive neurons leading to an augmented local release of glutamate. Research has shown that an increase in extracellular glutamate causes the activation of astrocytes mediated by the type II metabotropic glutamate receptors mGluR 2/3, which are expressed in astrocytes. 59 The extracellular increase in glutamate also triggers the production of the glial high-affinity glutamate transporter GLAST. Since GFAP acts to anchor GLAST at the plasma membrane of glial cells, 60 an increase in extracellular glutamate can induce an increase in GFAP expression. A similar mechanism may also account for the increased expression of GFAP in SGCs.
Glial and immune cells activation has been gaining ground as a potential pharmacological target for controlling chronic pain. A number of drugs have been shown to suppress the development of neuropathic pain by reducing glial activation, and consequently decreasing the secretion of numerous cytokines known to contribute to the development and maintenance of neuropathic pain. 61 One such drug is fluorocitrate, which has been shown to attenuate nociception in different models of neuropathy. 61 Fluorocitrate acts by selectively inhibiting glial cell metabolism,28,29 with no specific selectivity for different glial cells types, transiently inactivating microglia, astrocytes and SGCs. Therefore, as activation of all those glial cells types had been observed, we evaluated the effect of an intrathecal administration of fluorocitrate in movement- and loading-induced nociception to determine if glial activation could contribute to the nociceptive behaviour in the collagenase model of OA. We observed that fluorocitrate significantly reduced movement- and loading-induced nociception evaluated by the Knee-Bend and CatWalk tests. This is in line with studies in the MIA OA model, in which the glial activation inhibitor minocycline significantly attenuated the nociceptive behaviour. 55 These results suggest a role for glial cells in the development of nociceptive behaviours in the collagenase model of OA, and place these cells as pharmacological targets worth pursuing.
Altogether, the data here presented show that when OA is fully developed, with extensive articular damage associated with movement and loading-induced nociception, there is activation of glial cells both in the DRG and in the SC, and their inactivation can significantly reduce OA-associated nociception. These results therefore suggest that glial activation associated with PNI may play a role in nociception associated with OA.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financed by FEDER funds through COMPETE – Programa Operacional Factores de Competitividade “FCOMP-01-0124-FEDER-021359” and by National Funds through FCT – Fundação para a Ciência e a Tecnologia, within the project “PTDC/SAU-NSC/119986/2010.” This work was also supported by a PhD scholarship attributed to Sara Adães by Fundação Calouste Gulbenkian.
