Abstract
Background
Fentanyl is often used instead of morphine for the treatment of pain because it has fewer side effects. The metabolism of morphine by glucuronidation is known to be influenced by polymorphisms of the UGT2B7 gene. Some metabolic products of fentanyl are reportedly metabolized by glucuronate conjugation. The genes that are involved in the metabolic pathway of fentanyl may also influence fentanyl sensitivity. We analyzed associations between fentanyl sensitivity and polymorphisms of the UGT2B7 gene to clarify the hereditary determinants of individual differences in fentanyl sensitivity.
Results
This study examined whether single-nucleotide polymorphisms (SNPs) of the UGT2B7 gene affect cold pain sensitivity and the analgesic effects of fentanyl, evaluated by a standardized pain test and fentanyl requirements in healthy Japanese subjects who underwent uniform surgical procedures. The rs7439366 SNP of UGT2B7 is reportedly associated with the metabolism and analgesic effects of morphine. We found that this SNP is also associated with the analgesic effects of fentanyl in the cold pressor-induced pain test. It suggested that the C allele of the rs7439366 SNP may enhance analgesic efficacy. Two SNPs of UGT2B7, rs4587017 and rs1002849, were also found to be novel SNPs that may influence the analgesic effects of fentanyl in the cold pressor-induced pain test.
Conclusions
Fentanyl sensitivity for cold pressor-induced pain was associated with the rs7439366, rs4587017, and rs1002849 SNPs of the UGT2B7 gene. Our findings may provide valuable information for achieving satisfactory pain control and open to new avenues for personalized pain treatment.
Keywords
Background
Opioid analgesics, such as morphine and fentanyl, are widely used for the treatment of moderate to severe pain. However, the analgesic effects of opioids have broad individual differences.
1
This poses a problem for effective pain management. Individual differences may be related to various genetic and nongenetic factors, including gender, age, ethnic origin, hepatic and renal function, and mental status.
2
Several gene-association studies have reported that the analgesic effects of opioids could be influenced by polymorphisms in the genes that encode cyclic adenosine monophosphate response element binding protein 1 (CREB1), calcium voltage-gated channel subunit α1 E (CACNA1E), dopamine D4 receptors, β1 adrenergic receptors (ADRB1), µ1 opioid receptors (OPRM1), and adenosine triphosphate binding cassette subfamily B member 1 (ABCB1).3–7 Fentanyl is widely used instead of morphine because of its route of administration and fewer side effects. Fentanyl is metabolized by cytochrome P450 CYP3A4 in the liver through piperidine-induced oxidative dealkylation and hydroxylation to norfentanyl, which has almost no pharmacological activity. Fentanyl has various metabolic products, including M1 (N-[4-hydroxyphenethyl]propanamide), M2 (N-phenylpropanamide), M3 (N-[1-(p-hydroxyphenethyl)piperidin-4-yl]-N-phenylpropanamide), M4 (N-phenyl-N-4-piperidinylpropanamide; norfentanyl), M5 (4-anilinopiperidine), and M7 (4-anilino-1-phenethylpiperidine). A portion of norfentanyl, which is the main metabolic product of fentanyl, has been shown to be excreted by glucuronidation through urine and bile in dogs
8
(Figure 1). Portions of M2, M3, M4 (norfentanyl), and M5 are metabolized by glucuronate conjugation.
8
Glucuronate conjugation is reportedly influenced by UGT2B7, a uridine diphosphate glucuronic acid transfer enzyme in humans.
9
If the metabolism of various metabolic products like norfentanyl is delayed, then this may affect the metabolism of fentanyl itself. The metabolism of morphine by glucuronidation is well known to be influenced by a gene polymorphism of UGT2B7.
9
The rs7439366 single-nucleotide polymorphism (SNP) is located in the exon 2 domain of the UGT2B7 gene and has been associated with morphine sensitivity. The rs7439366 SNP is a functional SNP that produces an amino acid substitution in UGT2B7.
10
The C allele of rs7439366 is associated with high generative levels of morphine-3-glucuronide and morphine-6-glucuronide, which selectively act on μ opioid receptors to enhance analgesic effects compared with the T allele.
11
Proposed metabolic pathways of fentanyl in dogs. Portions of M2, M3, M4, and M5 are metabolized by glucuronate conjugation. M1: N-(4-hydroxyphenethyl) propanamide; M2: N-phenylpropanamide; M3: N-[1-(p-hydroxyphenethyl)piperidin-4-yl]-N-phenylpropanamide; M4: N-phenyl-N-4-piperidinylpropanamide (norfentanyl); M5: 4-anilinopiperidine; M7: 4-anilino-1-phenethylpiperidine.
Considering the possibility that the genes that are involved in the metabolic pathway of fentanyl may also influence fentanyl sensitivity, we analyzed associations between fentanyl sensitivity and gene polymorphisms of UGT2B7 to clarify the hereditary determinants of individual differences in fentanyl sensitivity. Patients with acute postoperative uniform pain following standardized surgical procedures may be optimal subjects for investigating genetic factors that are associated with individual differences in the analgesic effects of opioids.1,12 To examine whether SNPs of the UGT2B7 gene affect pain sensitivity and the analgesic effects of fentanyl, the present study conducted a standardized pain test and evaluated fentanyl requirements after surgery in healthy Japanese subjects who underwent a uniform cosmetic surgical procedure. Three SNPs of the UGT2B7 gene influenced the analgesic effects of fentanyl in the cold pressor-induced pain test. This suggests the possibility that three SNPs of the UGT2B7 gene, which were significantly associated with analgesic effects, may influence glucuronate conjugation and thus fentanyl sensitivity. The present results suggest that fentanyl sensitivity may be predicted by examining UGT2B7 gene polymorphisms, which may open to new avenues for personalized pain treatment in the future.
Methods
Ethics statement
The study was conducted according to the principles of the Declaration of Helsinki and approved by the Institutional Review Boards of Tokyo Dental College (Chiba, Japan) and Tokyo Institute of Psychiatry (currently Tokyo Metropolitan Institute of Medical Science, Tokyo, Japan). Written informed consent was obtained from all of the patients or their parents if required.
Patients
Enrolled in the study were 353 healthy patients (American Society of Anesthesiologists Physical Status I, age 15–52 years, 125 males and 228 females) who were scheduled to undergo cosmetic orthognathic surgery (mandibular sagittal split ramus osteotomy) for mandibular prognathism at Tokyo Dental College Suidoubashi Hospital. All of the subjects were Japanese. Patients with chronic pain, who were taking pain medication, and who had experienced Raynaud’s phenomenon were excluded.
Preoperative cold pressor-induced pain test
The patients were premedicated with oral diazepam, 5 mg, and oral famotidine, 150 mg, 90 min before the induction of anesthesia. The patients had an intravenous (i.v.) line inserted in the forearm on their nondominant side. The temperature in the operating room was maintained at 26℃. The cold pressor-induced pain test was then performed before and 3 min after an i.v. bolus injection of fentanyl, 2 µg/kg, as previously described.13,14 Crushed ice cubes and cold water were blended 15 min before the test in a 5-L isolated tank, and the mixture was stirred immediately before each test to ensure uniform temperature distribution (0℃) within the tank. The dominant hand was immersed up to the wrist. The patients were instructed to keep their hand calm in the ice-cold water and withdraw it as soon as they perceived any pain. All of the patients were tested by the same investigator. The baseline latency to pain perception, defined as the time of immersion of the hand in the ice water before the i.v. injection of fentanyl (PPLpre), was recorded. A cut-off point of 150 s was set to avoid tissue damage. The hand was warmed with a hair dryer as soon as it was withdrawn from the ice water until the sensation of cold was completely abolished. The patients then received i.v. fentanyl, 2 µg/kg. Three minutes after the injection, the latency of pain perception of the dominant hand (PPLpost) was measured again. The difference between PPLpre and PPLpost (PPLpost–PPLpre) was defined as the preoperative analgesic effect.
Anesthesia and surgery
After the cold pressor-induced pain test, general anesthesia was induced with a target-controlled infusion of propofol using a target-controlled infusion pump (TE-371, Terumo, Tokyo, Japan). Vecuronium, 0.1 mg/kg, was administered to facilitate nasotracheal intubation. After the induction of anesthesia, 10 ml of venous blood was sampled for the preparation of DNA specimens. General anesthesia was maintained with propofol at a target blood concentration of 4–6 µg/ml. Vecuronium was administered at a rate of 0.08 mg/kg/h. The lungs were ventilated with oxygen-enriched air. Local anesthetic block was performed on the right side of the surgical field with 8 ml of 2% lidocaine that contained epinephrine, 12.5 µg/ml, and right mandibular ramus osteotomy was performed. Local anesthetic block was then performed on the left side, and left mandibular ramus osteotomy was performed. The bilateral mandibular bone segments were fixed in appropriate positions. Whenever systolic blood pressure or heart rate exceeded +20% of the preinduction value during surgery, i.v. fentanyl, 1 µg/kg, was administered.
Postoperative pain management
At the end of surgery, rectal diclofenac sodium, 50 mg, and i.v. dexamethasone, 8 mg, were administered at the request of surgeons to prevent postoperative orofacial edema/swelling. After emergence from anesthesia and tracheal extubation, droperidol, 1.25 mg, was administered i.v. to prevent nausea/vomiting, and i.v. patient-controlled analgesia (PCA) with a fentanyl-droperidol combination (2 mg fentanyl and 5 mg droperidol diluted in normal saline in a total volume of 50 ml) commenced using a CADD-Legacy PCA pump (Smiths Medical Japan, Tokyo, Japan). A bolus dose of fentanyl on demand and lockout time were set at 20 µg and 10 min, respectively. Continuous background infusion was not employed. Droperidol was coadministered with fentanyl to prevent nausea/vomiting because our preliminary study showed a high incidence (up to 30%) of nausea/vomiting with PCA fentanyl in young females. PCA was continued for 24 h postoperatively. In the case of treatment-refractory adverse effects or inadequate analgesia, PCA was discontinued, and rectal diclofenac sodium, 50 mg, was prescribed as a rescue analgesic as required. The intensity of spontaneous pain was assessed at 3 h and 24 h postoperatively using a 100-mm visual analog scale (VAS), with 0 mm indicating no pain and 100 mm indicating the worst pain imaginable. Intraoperative fentanyl use, postoperative PCA fentanyl use during the first 24-h postoperative period, and perioperative (i.e., intraoperative + postoperative) fentanyl use were recorded. The doses of fentanyl that were administered intraoperatively and postoperatively were normalized to body weight.
Genotyping
Primers used for genotyping the UGT2B7 SNPs.

SNP: single-nucleotide polymorphism
The sequence of the primer for rs4296738 is not disclosed. The assay ID of rs4296738 is “C—405762-20” (Applied Biosystems Japan, Ltd., Tokyo, Japan).
For further analyses of polymorphisms other than rs7439366 within and around the UGT2B7 gene region, genotype data from whole-genome genotyping were used. 6 Whole-genome genotyping was performed using Infinium assay II and the iScan system (Illumina, San Diego, CA) according to the manufacturer’s instructions. Five kinds of BeadChips were used to genotype 40, 67, 6, 119, and 2 samples, respectively: HumanHap300 (total markers: 317,503), HumanHap300-Duo (total markers: 318,237), Human610-Quad v1 (total markers: 620,901), Human1M v1.0 (total markers: 1,072,820), and Human 1M-Duo v3 (total markers: 1,199,187). Some BeadChips included a number of probes that were specific to copy number variation markers, but most were for SNP markers on the human autosome or sex chromosome. Approximately 300,000 SNP markers were commonly included in all of the BeadChips. After whole-genome genotyping, the data for the genotyped samples were analyzed using BeadStudio Genotyping module v3.3.7 (Illumina) to evaluate the quality of the results, and genotype data for all of the SNPs with the UGT2B7 gene annotation were extracted. In the data-cleaning process, markers that had “Cluster sep” values (i.e., an index of genotype cluster separation) less than 0.4 and in which the three genotype clusters did not sufficiently separate one another were excluded from the subsequent association study.
Of the SNPs within the exon, intron, and approximately 6.8 kbp 5'-flanking region and 1.0 kbp 3'-flanking region of the UGT2B7 gene, SNPs for the association studies were selected based on previously reported tagging strategies.15–17 To identify relationships between the SNPs that were used in the study, linkage disequilibrium (LD) analysis was performed using Haploview v. 4.1. 18 To estimate the LD strength between the SNPs, the commonly used D' and r2 values were pairwise calculated using the genotype dataset of each SNP. LD blocks were defined among the SNPs that showed “strong LD,” based on the default algorithm of Gabriel et al., 19 in which the upper and lower 95% confidence limits on D' for strong LD were set at 0.98 and 0.7, respectively. TagSNPs in the LD block were then determined by the software package Tagger, which is incorporated in Haploview and was detailed in a previous report. 17
Seven of the eight TagSNPs in the UGT2B7 gene that were selected by the LD analysis were additionally genotyped by the TaqMan allelic discrimination assay (rs4296738, rs4235108, rs11931604, rs10028494, and rs12645107) and direct sequencing (rs7668282 and rs6851533) to complement the missing data that was caused by disparities among five kinds of BeadChips, with the exception of rs4587017, for which genotype data of five kinds of BeadChips were available.
To perform the TaqMan assay with a LightCycler® 480 (Roche Diagnostics K.K., Tokyo, Japan), TaqMan® SNP Genotyping Assays (Life Technologies Japan Ltd.) were used that included sequence-specific forward and reverse primers to amplify the polymorphic sequence and two probes that were labeled with VIC® and FAM™ dye to detect both alleles of the UGT2B7 SNPs. The sequences of the primers for rs4296738 were not disclosed. The Assay ID for rs4296738 is C—405762-20. The primers that were used for genotyping are denoted as P3 and P4 in Table 1. The primers that were used for rs4235108, rs11931604, rs10028494, and rs12645107 are presented as P5-P12 in Table 1. Real-time PCR was performed in a final volume of 10 μl that contained 2 × LightCycler® 480 Probes Master (Roche Diagnostics K.K.), 40× TaqMan® SNP Genotyping Assays, 5–50 ng genomic DNA as the template, and up to 10 μl H2O equipped with 2× LightCycler® 480 Probes Master. The thermal conditions were the following: 95℃ for 10 min, followed by 45 cycles of 95℃ for 10 s and 60℃ for 60 s, with final cooling at 50℃ for 30 s. Afterward, endpoint fluorescence was measured for each sample well, and the A/A, A/G, and G/G genotypes were determined based on the presence or absence of each type of fluorescence.
Direct sequencing was adopted to genotype the rs7668282 and rs6851533 SNPs similarly to genotyping the rs7439366 SNP because the TaqMan assay could not be performed. The primers that were used for genotyping are shown in Table 1. PCR was performed with forward and reverse primers for the regions shown in Table 1 (primers P13, P14, P15, and P16). Following the cycle sequencing reaction with a BigDye® Terminator v.3.1 Cycle Sequencing Kit (Applied Biosystems Japan, Ltd., Tokyo, Japan) according to the manufacturer’s instructions and purification of the PCR products, the DNA sequences of the fragments were determined using forward and reverse primers (P13, P14, P17, and P18) and the automated sequencer ABI PRISM® 3100 Genetic Analyzer.
Statistical analysis
The patients’ demographic and clinical data are expressed as mean ± SD or median (interquartile ranges). The statistical analysis was performed using SPSS 19 software. The χ2 test or Fisher’s exact test was performed for all of the genotype frequency data to investigate deviations of the distributions from those in the theoretical Hardy–Weinberg equilibrium. The Kruskal–Wallis test and Mann–Whitney U test were performed to detect possible associations between any of the genomic parameters and clinical endpoints related to pain sensitivity (i.e., PPLpre and PPLpost–PPLpre) or the analgesic effects of fentanyl (i.e., postoperative fentanyl use during 24 h (µg/kg) and VAS pain score at 24 h (mm)). Bonferroni multiple-comparison post hoc tests were used. gPLINK v. 2.049, PLINK v. 1.01 (http://pngu.mgh.harvard.edu/purcell/plink/), 20 and Haploview v. 4.0 18 were used for haplotype-specific tests. In all of the statistical tests, the criterion for significance was set at p < 0.05.
Results
Patients’ demographic and clinical data (n = 353).
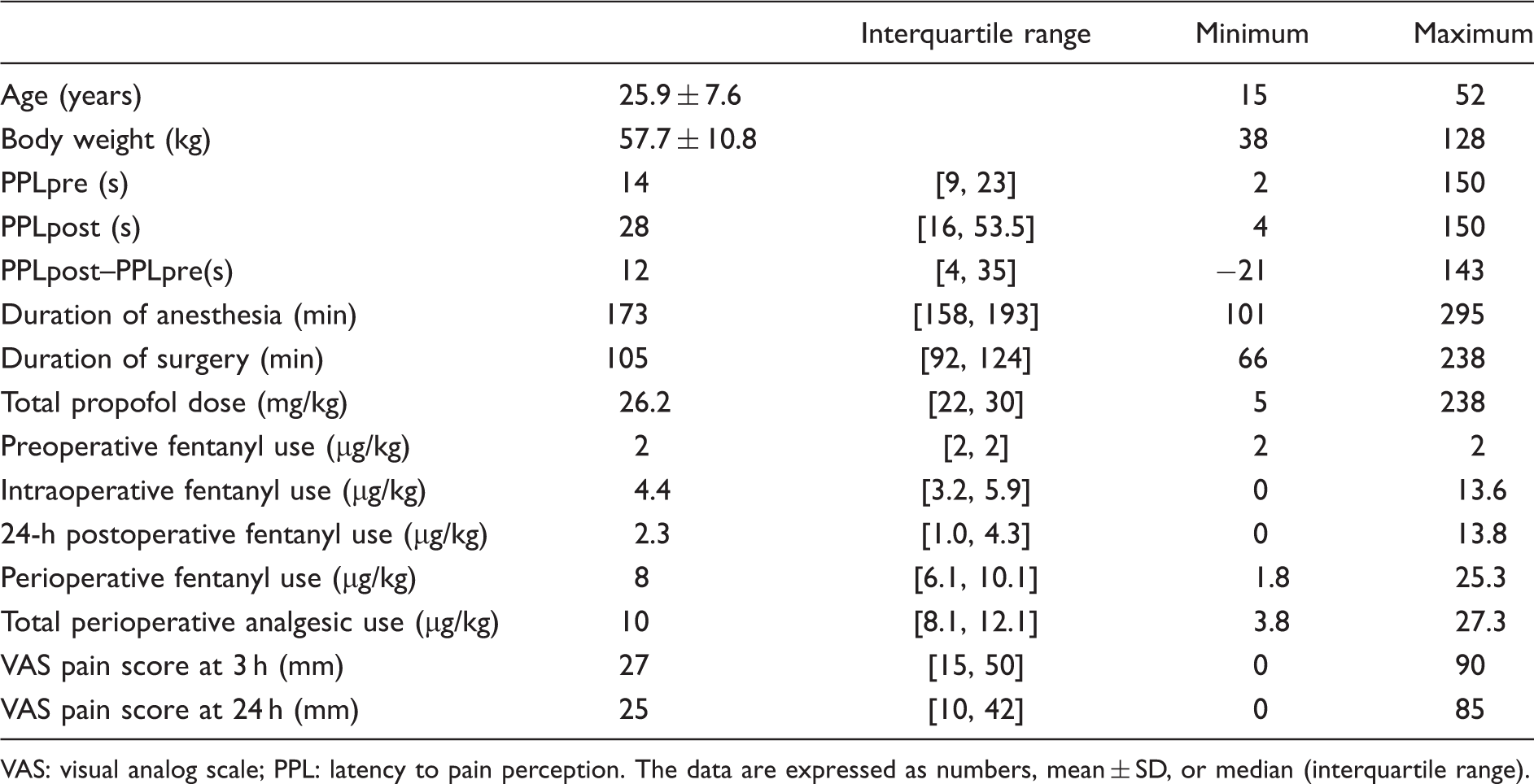
VAS: visual analog scale; PPL: latency to pain perception. The data are expressed as numbers, mean ± SD, or median (interquartile range).
Genotype distribution of the investigated SNPs.
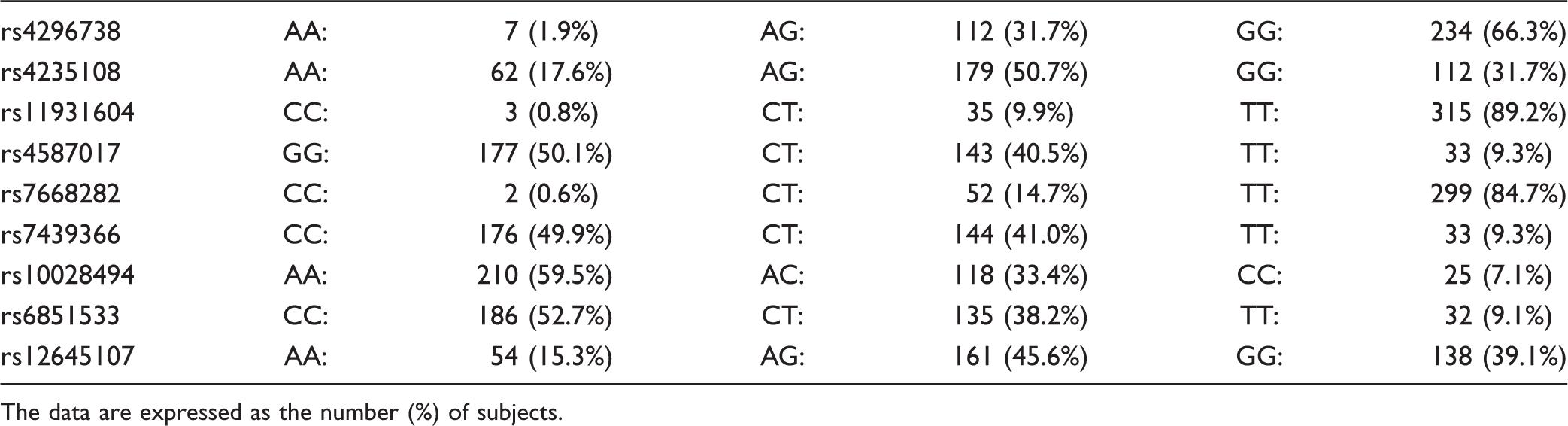
The data are expressed as the number (%) of subjects.
Association analysis between clinical data and one SNP (rs7439366; n = 353).

VAS: visual analog scale; PPL: latency to pain perception.
p < 0.05.

Associations between the analgesic effects of fentanyl in the cold pressor-induced pain test (PPLpost–PPLpre) and genotypes of the rs7439366 SNP. (a) Comparison between three genotype groups (CC: n = 176; CT: n = 144; TT: n = 33). (b) Comparison between two genotype groups (CC + CT: n = 320; TT: n = 33). (c) Comparison between two genotype groups (CC: n = 176; CT + TT: n = 177). The data are expressed by box and whisker plots. The upper and lower ends of the boxes represent the 75th and 25th percentiles. Whiskers represent the 90th and 10th percentiles. The median is depicted by a solid line in the box.
A schematic diagram of the UGT2B7 gene is shown in Figure 3(a). After whole-genome genotyping and the LD analysis of the extracted SNPs within and around the UGT2B7 gene, eight TagSNPs were selected (Figure 3(b)). Exons 2, 3, 4, and 5 contained only coding regions, whereas exons 1 and 6 also contained non-coding regions. Although the rs7439366 SNP was located in the exon 2 region, no TagSNPs were included in the exon region. One TagSNP (rs1002849) was located in the intron 3 region, whereas five TagSNPs (rs4296738, rs4235108, rs11931604, rs4587017, and rs7668282) and two TagSNPs (rs6851533 and rs12645107) were located in the 5'- and 3'-flanking regions, respectively. Therefore, the eight TagSNPs were analyzed for associations between the genotype of the SNP and the clinical data. The genotypes of the rs4296738, rs4235108, rs11931604, rs7668282, rs6851533, and rs12645107 SNPs had no significant associations with any of the clinical data.
The position of the rs7639366 SNP within the UGT2B7 gene region and relative position of the eight TagSNPs (a) selected by linkage disequilibrium (LD) analysis in the present study (b). The blue box and solid line represent exons and introns in the UGT2B7 gene structure, respectively. The SNPs that showed absolute linkage disequilibrium between one another (D' = 1, r2 = 1) are displayed as red squares with no characters.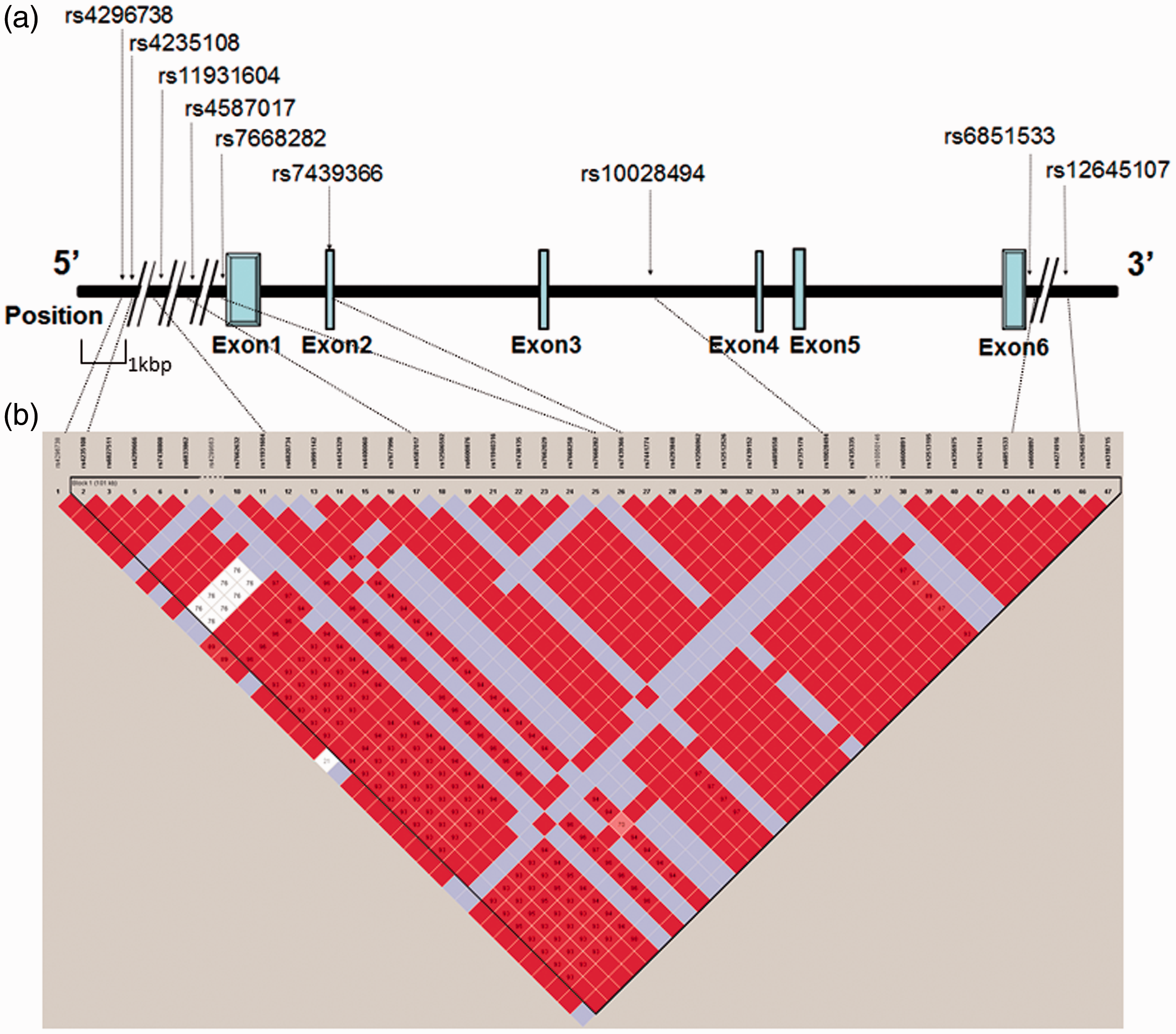
Association analysis between clinical data and one TagSNP (rs4587017; n = 353).

VAS: visual analog scale; PPL: latency to pain perception.
p < 0.00625 (p value after Bonferroni correction for multiple comparisons).

Associations between genotypes of the rs4587017 SNP and the analgesic effects of fentanyl in the cold pressor-induced pain test (PPLpost–PPLpre). (a) Comparison between three genotype groups (GG: n = 177; GT: n = 143; TT: n = 33). (b) Comparison between two genotype groups (GG + GT: n = 210; TT: n = 33). (c) Comparison between two genotype groups (GG: n = 177; GT + TT: n = 176). The data are expressed by box and whisker plots. The upper and lower ends of the boxes represent the 75th and 25th percentiles. Whiskers represent the 90th and 10th percentiles. The median is depicted by a solid line in the box.

Associations between the analgesic effects of fentanyl in the cold pressor-induced pain test (PPLpost -PPLpre) and genotypes of the rs10028494 SNP. (a) Comparison between three genotype groups (AA: n = 210; AC: n = 118; CC: n = 25). (b) Comparison between two genotype groups (AA+AC: n = 328; CC: n = 25). (c) Comparison between two genotype groups (AA: n = 210; AC+CC: n = 143). The data are expressed by box and whisker plots. The upper and lower ends of the boxes represent the 75th and 25th percentiles. Whiskers represent the 90th and 10th percentiles. The median is depicted by a solid line in the box.
For a more detailed analysis, haplotype-based analysis was conducted for the SNPs that were significantly associated with PPLpost–PPLpre. The associations between haplotypes (consisting of the rs4587017, rs7439366, and rs10028494 SNPs of the UGT2B7 gene) and PPLpost–PPLpre were not significant (Table S2).
Discussion
The present study examined whether SNPs of the UGT2B7 gene affect cold pain sensitivity and the analgesic effects of fentanyl, one of the most commonly used opioid analgesics, evaluated by a standardized pain test and fentanyl requirements in healthy Japanese subjects who underwent uniform surgical procedures. 21 The results showed that three SNPs influenced the analgesic effects of fentanyl in the cold pressor-induced pain test (which induces slight pain) but did not influence fentanyl sensitivity for severe postoperative pain. The rs7439366 SNP of the UGT2B7 gene, which is reportedly associated with the metabolism and analgesic effects of morphine through glucuronate conjugation, 9 was associated with the analgesic effects of fentanyl. Furthermore, the rs4587017 and rs1002849 SNPs of the UGT2B7 gene were identified as novel SNPs that can influence the effects of fentanyl. The optimal doses of fentanyl for individuals may be predicted by gene polymorphism analyses, including the UGT2B7 gene. Personalized medicine may be able to manage pain with fentanyl at a minimal dosage, which would thus minimize side effects.
Portions of the M2, M3, M4 (norfentanyl), and M5 metabolites of fentanyl are reportedly metabolized by glucuronate conjugation. 8 The glucuronate conjugation of norfentanyl is influenced by UGT2B7, which is a UDP glucuronic acid transfer enzyme. 9 The gene polymorphism of UGT2B7 may influence the metabolism of norfentanyl. Therefore, the gene polymorphism of UGT2B7 may have influenced the sensitivity to fentanyl. Thus, these three SNPs may also influence the glucuronate conjugation of norfentanyl similarly to morphine in the metabolism of fentanyl.
Several studies have recently shown that UGT2B7 functionally interacts with CYP3A4, an enzyme that N-dealkylates fentanyl and converts it into the inactive metabolite norfentanyl. Miyauchi et al. 22 showed that the activity of CYP3A4 was significantly suppressed by the coexpression of UGT2B7, and this suppressive effect was lost when UGT2B7 was replaced with calnexin, suggesting that UGT2B7 negatively regulates CYP3A4 activity through protein–protein interactions. Gene polymorphisms of UGT2B7 may indirectly influence the metabolism of fentanyl by oxidative dealkylation and hydroxylation through CYP3A4.
The rs7439366 SNP is a functional SNP that exists on the exon 2 domain and produces an amino acid substitution. The rs7439366 SNP was significantly associated with PPLpost–PPLpre. In the cold pressor-induced pain test, subjects with the C-allele of this SNP presented a longer latency of pain perception. Subjects with the C-allele may be more sensitive to the analgesic effects of fentanyl. The metabolism of morphine through glucuronidation has already been reported to be influenced by gene polymorphisms of UGT2B7. Sawyer et al. showed that rs7439366 is a functional polymorphism with regard to morphine metabolism. The C-allele of this SNP resulted in a high generative capacity of morphine-3-glucuronide and morphine-6-glucuronide, which selectively act on μ-opioid receptors to enhance analgesic effects. 11 The present results suggest that the C-allele may also influence the sensitivity to fentanyl, thus corroborating the importance of the C-allele that has been reported in previous studies. According to Innocenti et al., 23 the TagSNP rs7438284 is associated with expression of the mRNA and activation of the enzyme, and rs7439366 is included in the same LD block. This suggests the possibility that rs7439366 is also associated with the enzymatic activity of UGT2B7.
Based on two studies that performed an LD block analysis of UGT2B7 in the Japanese population,24,25 −125T > C (rs7668282) in the promoter region was one of eight TagSNPs that were selected in our study. In this LD analysis, one LD block was formed in a wide domain that spanned the entire gene region. This is consistent with two previous Japanese studies that found that strong LD was formed among many SNPs in the comparatively extensive domain in the gene region of UGT2B7.24,25 To our knowledge, no previous study has analyzed both LD relationships and associations between clinical phenotypic data and UGT2B7 gene polymorphisms. The present study investigated both LD relationships and associations between fentanyl sensitivity and UGT2B7 gene polymorphisms. With regard to TagSNPs that had significant associations with PPLpost–PPLpre, rs4587017 was located in the promoter region and rs10028494 was located in the intron 3 region. These two SNPs (rs4587017 and rs10028494) were associated with fentanyl sensitivity. Carriers of the G-allele of rs4587017 presented higher sensitivity to fentanyl (see Table S1 for the A allele of rs10028494). We selected novel TagSNPs in the LD block that were identified in the UGT2B7 gene region.
Further research on the functional influence of these SNPs is needed. UGT2B7 gene polymorphisms in the promoter domain and a coding region form haplotypes (−45597G, −6682_−6683A, 372A, IVS1 + 9_IVS1 + 10A, IVS1 + 829T, IVS1 + 985G, IVS1 + 999C, IVS1 + 1250G, 801T, and IVS4 + 185C) that may functionally impact the metabolism of morphine. 23 The rs4587017 SNP is also in the promoter region and in strong LD with rs7439366. Therefore, although these SNPs may influence the analgesic effects of fentanyl, the haplotypes were not associated with the analgesic effects of fentanyl in the present study (Table S2). To our knowledge, no previous study has directly shown that the rs10028494 SNP in the intron region is a functional polymorphism with regard to fentanyl metabolism. However, the rs10028494 SNP in the intron region is in strong LD with rs7439366, which may functionally impact the metabolism of fentanyl. Furthermore, introns supposedly affect the transcription level of genes or control the expression of genes by alternative splicing. 26
In the present study, although the polymorphisms of the UGT2B7 gene were not associated with fentanyl sensitivity for severe postoperative pain, the SNPs of UGT2B7 were associated with fentanyl sensitivity in the cold pressor-induced pain test. Genotyping these SNPs may predict the sensitivity of fentanyl for slight pain (e.g., cold pain). Although many studies have reported associations between the analgesic effects of morphine and gene polymorphisms, further research on associations between the analgesic effects of fentanyl and other opioid analgesics (e.g., oxycontin) and gene polymorphisms is still needed in the future.
Conclusion
The present study suggests that three SNPs of the UGT2B7 gene can affect fentanyl sensitivity for cold pressor-induced pain, likely through alterations in glucuronate conjugation. The results indicate that fentanyl sensitivity for slight pain can be predicted by examining UGT2B7 gene polymorphisms, which may open to new avenues for personalized pain treatment in the future.
Footnotes
Authors’ contributions
DN and KI conceived the study and designed the experiments. WM and DN performed the statistical analyses and wrote the manuscript. KF and MH performed the surgeries and experiments. SK and JH performed the genotyping procedures. TN, KW, and KI supervised the experiments and finalized the manuscript. All of the authors contributed to writing the manuscript, and all of the authors read and approved the final manuscript.
Acknowledgments
We acknowledge Mr. Michael Arends for his assistance with editing the manuscript. We are grateful to the volunteers for their participation in the study and anesthesiologists and surgeons at Tokyo Dental College Suidoubashi Hospital for collecting the clinical data.
Declaration of Conflicting Interests
The author(s) declare that they have no competing interests with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan (no. 22790518, 23390377, 24659549, 24790544, 25116532, 26293347, and 26860360), Ministry of Health, Labour and Welfare (MHLW) of Japan (no. H21-3jigan-ippan-011, H22-Iyaku-015, and H25-Iyaku-020), Smoking Research Foundation (Tokyo, Japan), and Astellas Foundation for Research on Metabolic Disorders (Tokyo, Japan).
