Abstract
Background
Clinical management of neuropathic pain, which is pain arising as a consequence of a lesion or a disease affecting the somatosensory system, partly relies on the use of anticonvulsant drugs such as gabapentinoids. Therapeutic action of gabapentinoids such as gabapentin and pregabalin, which act by the inhibition of calcium currents through interaction with the α2δ-1 subunit of voltage-dependent calcium channels, is well documented. However, some aspects of the downstream mechanisms are still to be uncovered. Using behavioral, genetic, and pharmacological approaches, we tested whether opioid receptors are necessary for the antiallodynic action of acute and/or long-term pregabalin treatment in the specific context of neuropathic pain.
Results
Using the cuff model of neuropathic pain in mice, we show that acute pregabalin administration at high dose has a transitory antiallodynic action, while prolonged oral pregabalin treatment leads to sustained antiallodynic action, consistent with clinical observations. We show that pregabalin remains fully effective in μ-opioid receptor, in δ-opioid receptor and in κ-opioid receptor deficient mice, either female or male, and its antiallodynic action is not affected by acute naloxone. Our work also shows that long-term pregabalin treatment suppresses tumor necrosis factor-α overproduction induced by sciatic nerve constriction in the lumbar dorsal root ganglia.
Conclusions
We demonstrate that neither acute nor long-term antiallodynic effect of pregabalin in a context of neuropathic pain is mediated by the endogenous opioid system, which differs from opioid treatment of pain and antidepressant treatment of neuropathic pain. Our data are also supportive of an impact of gabapentinoid treatment on the neuroimmune aspect of neuropathic pain.
Keywords
Background
Neuropathic pain is defined as a direct consequence of a lesion or disease affecting the somatosensory system. 1 It can result from a wide range of conditions including diabetes, nerve root compression, herpes zoster infection, cancer, stroke, thus affecting millions of persons worldwide. This complex syndrome involves maladaptive changes in injured sensory neurons and along the entire nociceptive pathway within the central nervous system. 2 The recommended pharmacotherapy for neuropathic pain includes the use of anticonvulsant drugs, such as the gabapentinoids, pregabalin, and gabapentin. 3
Despite their structural similarity to the inhibitory transmitter γ-aminobutyric acid (GABA), neither gabapentin nor pregabalin binds to GABAA or GABAB receptors or interact with GABA uptake transporters.4,5 Their therapeutic effect is mediated through binding to the α2δ-1 subunit of voltage-dependent calcium channels (VDCCs).6,7 The interaction between gabapentinoids and the α2δ-1 subunit inhibits calcium currents, thus decreasing excitatory transmitter release. 5 This subunit also plays a role in trafficking VDCC complexes to cell surface 8 and in synaptogenesis, and these functions are blocked by gabapentin. 9
The opioid system is involved in the action of different pain medications. This implication concerns on one hand the direct analgesic action of opioids targeting the μ-opioid (MOP) receptor 10 and on the other hand the indirect requirement of opioid receptors for the action of antidepressants against neuropathic pain.11–13 During the past decade, it has been preclinically and clinically proposed that gabapentinoids and opioid drugs can have a synergistic action in neuropathic pain.14–17 However, this does not mean that gabapentinoids require the endogenous opioid system. A potential role of the opioid system has been recently suggested in the central, acute, analgesic effect of a high dose of pregabalin in the tail flick test in naive mice, 18 and in the antinociceptive response induced by acute gabapentin in a model of acute inflammatory pain, the orofacial formalin test in mice. 19 On the contrary, previous pharmacological studies reported no effect of opioid antagonists on gabapentinoid action.20–22 For example, naloxone do not block acute pregabalin action on abdominal constrictions in the lipopolysaccharide (LPS)-induced rectal hypersensitivity model of visceral pain; 21 and naloxone do not block acute gabapentin action in the formalin test, a model of inflammatory pain. 22 However, these studies did not really model the specific clinical use of gabapentinoids, i.e. in a neuropathic pain context, and did not either address the consequences of a long-term treatment.
Gabapentinoids have also been proposed to act on inflammatory mechanisms. Gabapentin may, for example, decrease the expression of pro-inflammatory cytokines;16,23,24 this action has been associated with an upregulation of the anti-inflammatory cytokine interleukin (IL)-10. 24 Interestingly, experimental evidence supports a role of glial and/or immune cells in the pathophysiology of neuropathic pain, particularly through the recruitment of cytokines. 25 In sustained neuropathic pain, some pro-inflammatory cytokines such as tumor necrosis factor α (TNF-α) still display enhanced expression,26–29 and blocking TNF-α has been preclinically postulated to relieve neuropathic pain symptoms.26,30 It is, however, not known whether the expression of TNF-α is also targeted by pregabalin in a context of neuropathic pain.
In the present study, we used both genetic and pharmacological approaches to evaluate whether opioid receptors are critical for the antiallodynic action of acute and/or long-term pregabalin treatment. We demonstrate that neither the acute nor the long-term antiallodynic effect of pregabalin requires the endogenous opioid system. We also show that long-term pregabalin treatment inhibits the neuropathy-induced TNF-α overproduction in dorsal root ganglia (DRG).
Methods
Animals
Experiments were performed using male C57BL/6J mice (Charles River, L’Arbresle, France) with ages between 8 and 10 weeks at surgery time, or with mice lacking μ-opioid (MOP), δ-opioid (DOP), or κ-opioid (KOP) receptors and their littermate controls. The generation of mice lacking MOP, DOP, or KOP receptors has been previously described.31–33 All mice were under a C57BL/6J background for over 10 generations. Heterozygote mice were bred in our animal facilities (breeders were kindly provided by Pr Kieffer and Pr Gavériaux-Ruff), genotyping of the litters was done, and the experiments were conducted on adult male and female wild type and knockout littermate mice weighing 20–30 g. We used the same number of males and females in each experimental group. As the wild type animals have the same background and the same behavior, they were pooled to form the control groups. Mice were group housed two to five per cage and kept under a 12 hr light/dark cycle with food and water ad libitum. A total of 104 C57BL/6J mice, 43 MOP-related, 43 DOP-related, and 43 KOP-related transgenic mice were used for the experiments. All animals received proper care in agreement with European guidelines (EU 2010/63). At the end of the experiments, mice were killed by cervical dislocation for immunoblot experiments, or by CO2 inhalation (CO2 Euthanasia programmer 6.5 version, TEMSEGA, Pessac, France) followed by cervical dislocation for other experiments, according to the institutional ethical guidelines. The animal facilities Chronobiotron UMS3415 are registered for animal experimentation under the Animal House Agreement A67-2018-38. All protocols were approved by the “Comité d’Ethique en Matière d’Expérimentation Animale de Strasbourg” (CREMEAS, CEEA35).
Model of neuropathic pain
Neuropathic pain was induced by cuffing the main branch of the right sciatic nerve.34,35 Surgeries were performed under ketamine (68 mg/kg)/xylazine (10 mg/kg) intraperitoneal (i.p.) anesthesia (Centravet, Tadden, France). The common branch of the right sciatic nerve was exposed and a cuff of PE-20 polyethylene tubing (Harvard Apparatus, Les Ulis, France) of standardized length (2 mm) was unilaterally inserted around it (Cuff group). The shaved skin was closed using suture. Sham-operated mice underwent the same surgical procedure without implantation of the cuff (Sham group).
Measure of mechanical allodynia
Mechanical allodynia was tested using von Frey hairs, and results were expressed in grams. Tests were done during the morning, starting at least 2 hr after lights on. Mice were placed in clear Plexiglas boxes (7 cm × 9 cm × 7 cm) on an elevated mesh screen. Calibrated von Frey filaments (Bioseb, Vitrolles, France) were applied to the plantar surface of each hindpaw until they just bent, in a series of ascending forces up to the mechanical threshold. Filaments were tested five times per paw, and the paw withdrawal threshold (PWT) was defined as the lower of two consecutive filaments for which three or more withdrawals out of the five trials were observed.35–37 The person who conducted the tests was blinded to the treatments.
Treatment procedures
The long-term treatment with pregabalin began two weeks after the surgical procedure (cuff implantation or sham surgery). Pregabalin (Lyrica®, Pfizer, Sandwich, UK), 300, 100, 50, or 5 µg/mL, was delivered per os through the drinking water with ad libitum access as sole source of fluid. This anticonvulsant drug was dissolved in water with 0.02% saccharin to increase palatability, and control mice were given a solution of 0.02% saccharin in water (vehicle solution). For acute administration, pregabalin was dissolved in 0.9% NaCl and administered intraperitoneally (30 mg/kg, 5 mL/kg). The injection of naloxone hydrochloride (Sigma–Aldrich, St. Quentin Fallavier, France), a competitive non selective MOP, DOP, and KOP receptors antagonist at high dose, was performed 25 days after surgery, i.e. after 11 days of pregabalin treatment; or 30 min after the acute administration of pregabalin. Naloxone hydrochloride was dissolved in 0.9% NaCl and administered subcutaneously (s.c., 1 mg/kg, 5 mL/kg). Long-term and acute treatment experiments were conducted on independent sets of mice.
Immunoblot analysis
In a separate experiment, DRG were collected from Sham-vehicle, Cuff-vehicle, and Cuff-pregabalin (300 µg/mL) group after two weeks of oral treatment. Mice were killed by cervical dislocation, the back was dissected, and a midline incision was done in the lumbar vertebrae to extract the L4, L5, and L6 DRG ipsilateral to the surgery. The three DRG were pooled per animal, quickly frozen, and stored at −80℃ until protein extraction.
Total proteins were extracted in 150 µL lysis buffer (20 mM Tris pH 7.5; 150 mM NaCl; 10% glycerol; 1% NP-40; Protease Inhibitors Cocktail, Roche), quantitated with Bio-Rad Protein Assay Dye Reagent Concentrate and stored in Laemmli buffer (2% sodium dodecyl sulfate (SDS); 25% glycerol; 0.01% bromophenol blue; 0.125 M Tris pH 6.8); 10 µg of total protein from individual animals was resolved by 12% SDS-polyacrylamide gel electrophoresis under reducing conditions, and then transferred to polyvinylidene fluoride (PVDF) membrane (Immobilon, transfer membranes, Millipore, IPVH00010). The blots were incubated for 1 h in blocking agent (ECL kit, Amersham Biosciences), overnight with the antibodies specific for either TNF-α (1:500, R&D Systems, AF-410-NA) or β-tubulin (1:50,000, Abcam, ab108342), followed by rabbit anti-goat horseradish peroxidase (HRP)-conjugated secondary antibodies (1:12,000, Abcam, ab97100) or goat anti-rabbit HRP-conjugated secondary antibodies (1:10,000, Millipore, AP307P), respectively. Blots were revealed by chemiluminescence (ECL Prime Western Blotting Detection Reagent, Amersham Biosciences, RPN 2232) using Hyperfilm substrates (Amersham Biosciences, RPN 1674K). Relative protein expression was determined using the densitometry tool of Adobe Photoshop CS5 software. The bands were evaluated in grayscale, subtracting the background value, and the TNF-α/β-tubulin ratio was calculated for each sample.
Statistical analysis
Mechanical thresholds measured with the von Frey test provide discrete values corresponding to filaments’ values, thus limiting the relevance of classical parametric multi-factor analysis of variance (ANOVA). An ANOVA-type multiple-factor nonparametric methodology for longitudinal data, which can take into account both within and between factors, has recently been developed 38 as a package (nparLD) for R (version 3.2.1). We used the nparLD function to analyze the effects of time, side (left vs. right paw), sex (male vs. female), and of treatment (e.g. surgery and/or drug dose). The asymptotic ANOVA-type statistic (ATS) is provided as ATS(d.f.), with its adjusted degrees of freedom (d.f.) and p value. Multiple comparisons between groups at a given time point were performed with the two-sample Wilcoxon test, with the corresponding Bonferroni adjustment. The Wilcoxon test was also used for comparison of the mechanical sensitivity thresholds between males and females. Immunoblotting experiments were analyzed with the nonparametric Kruskal–Wallis test, followed by multiple comparisons with the Wilcoxon test. The significance level was set at p < 0.05. Data were represented as mean ± SEM.
Results
Antiallodynic action of chronic oral pregabalin: Dose response
The mechanical sensitivity of the C57BL/6J mice was assessed using von Frey hairs. Although sham surgery did not influence mechanical thresholds (Figure 1(a) and (b)), cuff implantation induced an ipsilateral mechanical allodynia (Figure 1(a); surgery × time interaction, ATS(2.9) = 3.9, p < 0.005 on postsurgery days 1–19). We did not observe any change in the nociceptive threshold of the left paw, contralateral to the cuff implantation; 19 days after surgery, we started treatment with different doses of pregabalin (300, 100, 50, or 5 µg/mL) or with vehicle solution (0.02% saccharin). Pregabalin treatment at doses 100 and 300 µg/mL alleviated the cuff-induced allodynia after about three days of treatment (Figure 1(a); group × time interaction, ATS(13.9) = 2.8, p < 0.001; multiple comparisons: “Cuff Vehicle” < “Cuff Pregabalin 100 µg/mL and Pregabalin 300 µg/mL” at p < 0.05 on postsurgery days 22–40). A partial antiallodynic effect was also present with the 50 µg/mL dose of pregabalin after eight days of treatment (Figure 1(a); multiple comparisons: “Cuff Vehicle” < “Cuff Pregabalin 50 µg/mL” < “Sham Vehicle” at p < 0.05 on postsurgery days 27–40). Treatments at different doses did not affect the contralateral nociceptive thresholds (Figure 1(a)). The 5 µg/mL dose of pregabalin had no significant effect (Figure 1(a)).
Chronic pregabalin treatment. (a) Two weeks after unilateral cuff insertion around the right sciatic nerve, chronic oral treatment with pregabalin started and lasted three weeks. The animals (n = 5 per each group) freely drink pregabalin (5, 50, 100, or 300 µg/mL) with 0.02% saccharin, or vehicle composed of 0.02% saccharin in water, as sole source of fluid. Mechanical PWT were evaluated at indicated time points using von Frey filaments. Vehicle treatment did not affect mechanical sensitivity of either Sham or Cuff mice. Pregabalin treatment was ineffective at dose 5 µg/mL, partially effective at dose 50 µg/mL, and reversed the cuff-induced allodynia at doses 100 and 300 µg/mL. (b) Pregabalin treatment at dose 300 µg/mL had no effect per se on sham-operated mice. (c) Histogram showing the equivalence between µg/mL and mg/kg/day of the different doses. (d) Time course of changes in the body weight of the animals throughout the experiment. Data are expressed as mean ± SEM.
Chronic oral treatment with pregabalin at 300 µg/mL suppressed cuff-induced allodynia (Figure 1(a)), but it did not affect mechanical thresholds of mice of the Sham group (Figure 1(b)).
The drinking bottles were regularly weighed during the experiment. Considering the volume of solution drank by the mice per 24 h, the 5 µg/mL solution was equivalent to 0.78 ± 0.05 mg/kg/day, the 50 µg/mL solution was equivalent to 8.09 ± 0.38 mg/kg/day, the 100 µg/mL solution was equivalent to 15.64 ± 0.65 mg/kg/day, and the 300 µg/mL solution was equivalent to 44.63 ± 1.39 mg/kg/day (Figure 1(c)). These amounts were in fact mostly taken over the 12 h night period, period during which mice usually drink.
Body weights of mice treated chronically with different doses of pregabalin or vehicle were also assessed throughout the experiment. Cuff animals showed a difference in weight gain in the days following the surgery compared to Sham animals. This difference persisted in Cuff mice treated with vehicle or pregabalin at doses of 5 and 50 µg/mL. Pregabalin treatment at doses of 100 and 300 µg/mL, which relieved neuropathic allodynia, reversed this deficit in weight gain (Figure 1(d); group × time interaction, ATS(11.2) = 6.2, p < 0.001; multiple comparisons: “Cuff Vehicle, Pregabalin 5 µg/mL and Pregabalin 50 µg/mL” < “Sham Vehicle” at p < 0.05 on postsurgery days 7–40, “Cuff Pregabalin 100 µg/mL and Pregabalin 300 µg/mL” < “Sham Vehicle” at p < 0.01 on postsurgery days 7–19 and “Cuff Vehicle” < “Cuff Pregabalin 100 µg/mL and Pregabalin 300 µg/mL” at p < 0.01 on postsurgery days 25–40).
Response to pregabalin: Male/female comparison in wild-type mice
Mechanical sensitivity thresholds of female mice were significantly lower than in males (baseline threshold values of paws are equal to 4.67 g ± 0.19 for males and 3.28 g ± 0.13 for females, male vs. female: W = 79.5, p < 0.001). Both male and female mice developed mechanical allodynia after cuff implantation and pregabalin treatment suppressed the cuff-induced allodynia in both sexes (Figure 2(a); Male mice: group × time interaction, ATS(6.1) = 7.5, p < 0.001; multiple comparisons: “Cuff Vehicle” < “Sham Vehicle” at p < 0.05 on treatment days 0–12 and “Cuff Vehicle” < “Cuff Pregabalin 300 µg/mL” at p < 0.05 on treatment days 9–12; Female mice: group × time interaction, ATS(5.9) = 5.1, p < 0.001; multiple comparisons: “Cuff Vehicle” < “Sham Vehicle” at p < 0.05 on treatment days 0–12 and “Cuff Vehicle” < “Cuff Pregabalin 300 µg/mL” at p < 0.05 on treatment days 9–12).
Effect of chronic oral pregabalin in opioid receptor deficient mice. Pregabalin treatment (300 µg/mL i.e 44.63 mg/kg/day in the drinking water, with 0.02% saccharin) or control treatment (0.02% saccharin) started two weeks following surgery and lasted 12 days. Mechanical allodynia was tested using von Frey hairs. (a) The mechanical sensitivity threshold (PWT) of female mice is lower than that of male mice. However, both sexes developed mechanical allodynia similarly and pregabalin was effective in reversing the cuff-induced allodynia in both male and female mice. Males and females were then pooled in each experimental group. (b) Chronic pregabalin treatment abolishes the ipsilateral cuff-induced allodynia in wild type mice, as well as in MOP, DOP, or KOP receptors-deficient mice (c). (Data are pooled from three independents experiments, each final group includes the same number of male and female mice, *p < 0.05 as compared with Sham-operated control group drinking vehicle). Data are expressed as mean ± SEM.
Chronic oral pregabalin treatment in opioid receptor deficient mice
The MOP, DOP, or KOP receptors-deficient mice displayed baselines for mechanical sensitivity that were similar to the wild-type littermates (Figure 2(b)). We controlled in our facilities that morphine has no more action in MOP-deficient mice. 36 Two weeks after surgery, we started the oral treatment with either pregabalin (300 µg/mL) or vehicle (0.02% saccharin) solutions. Pregabalin treatment alleviated cuff-induced allodynia in wild-type mice (Figure 2(b); group × time interaction, ATS(6.9) = 13.1, p < 0.001; multiple comparisons: “Cuff Vehicle” < “Cuff Pregabalin” at p < 0.05 on treatment days 9–12). The same antiallodynic effect was also present in MOP receptors (Figure 2(c); group × time interaction, ATS(5.2) = 10.4, p < 0.001; multiple comparisons: “Cuff Vehicle” < “Cuff Pregabalin” at p < 0.05 on treatment days 9–12), DOP receptors (Figure 2(c); group × time interaction, ATS(7.1) = 8.8, p < 0.001; multiple comparisons: “Cuff Vehicle” < “Cuff Pregabalin” at p < 0.05 on treatment days 9–12), and KOP receptors-deficient mice (Figure 2(c); group × time interaction, ATS(5.5) = 8.4, p < 0.001; multiple comparisons: “Cuff Vehicle” < “Cuff Pregabalin” at p < 0.05 on treatment days 9–12). Thus, pregabalin suppressed cuff-induced allodynia independently of the presence or no of the opioid receptors.
Naloxone effect on long lasting pregabalin treatment
We tested the consequence of an acute injection of the opioid receptor antagonist naloxone (1 mg/kg, s.c.) on the antiallodynic action of pregabalin in C57BL/6 J male mice. After 10 days of oral treatment with pregabalin or vehicle (Figure 3(a); group × time interaction, ATS(11.1) = 9.3, p < 0.001; multiple comparisons: “Cuff Vehicle” < “Cuff Pregabalin” at p < 0.005 on postsurgery days 19 to 24 and “Cuff Pregabalin” = (“Sham Pregabalin” or “Sham Vehicle”) at p = 1.0 on postsurgery days 22 and 24), acute injection of naloxone did not suppress the antiallodynic effect of chronic pregabalin treatment (Figure 3(c)). We also observed that naloxone per se had no effect in mice with Sham surgery or in mice that received vehicle alone (Figure 3(b)).
Acute opioid receptor antagonist in chronic pregabalin treatment. (a) Two weeks after unilateral cuff insertion, the oral treatment with pregabalin, or vehicle control started. Mechanical threshold of hindpaw withdrawal (PWT) was evaluated using von Frey filaments. Pregabalin treatment suppressed the cuff-induced alloynia. (b, c) After at least 10 days of pregabalin (300 µg/mL i.e 44.63 mg/kg/day, 0.02% saccharin) or vehicle treatment, the animals received an injection of the opioid receptor antagonist naloxone (1 mg/kg, s.c.) or the control saline solution. Mechanical threshold for hindpaw withdrawal was measured before 30 and 120 minutes after injection. No effect of naloxone or saline was seen in Sham mice or in pregabalin-treated neuropathic animals (n = 9–10, *p < 0.005 compared to the Sham-operated control group). Data are expressed as mean ± SEM.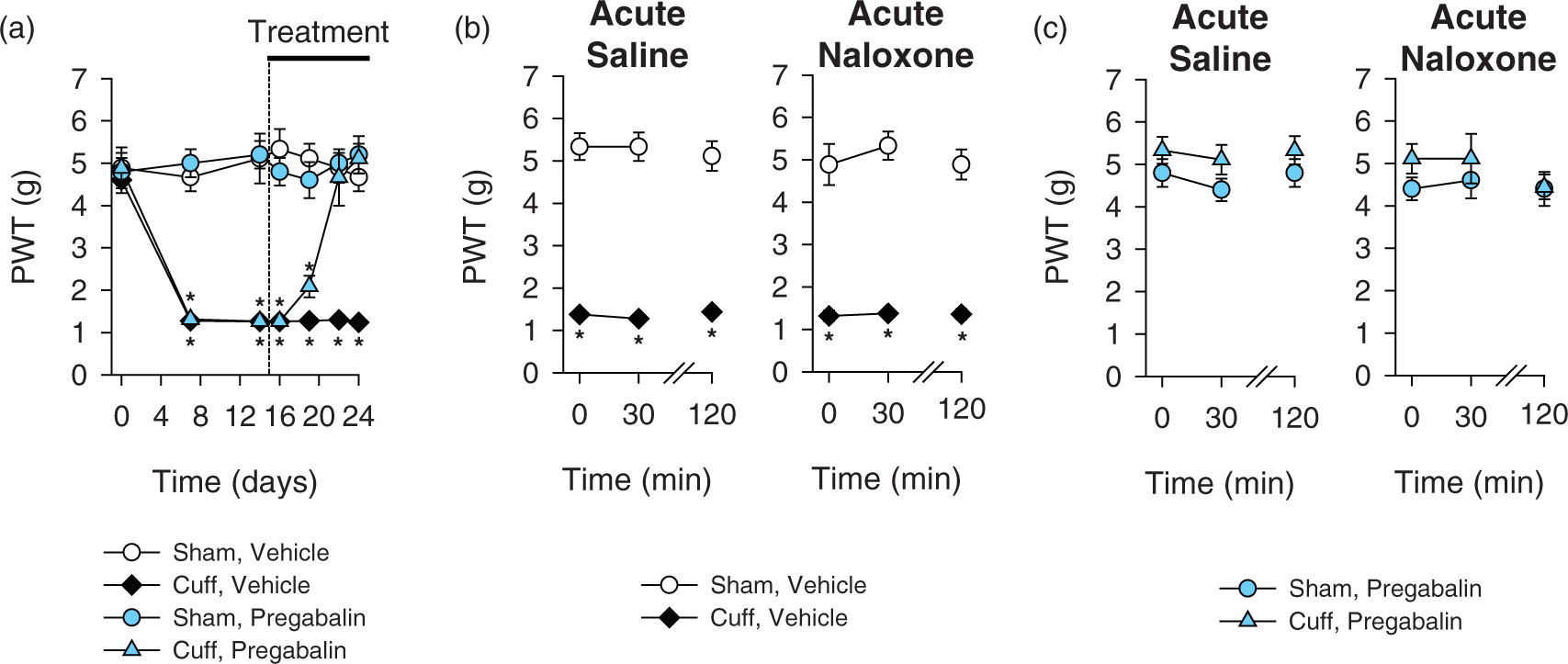
Transitory relief of neuropathic allodynia by acute pregabalin
In wild-type mice, an acute injection of pregabalin at a high dose (30 mg/kg, i.p.) had a transitory antiallodynic effect in Cuff mice, without affecting Sham animals (Figure 4; group × time interaction, ATS(2.7) = 12.3, p < 0.001; multiple comparisons: “Cuff Pregabalin” = “Sham Pregabalin” at p > 0.7 on post-administration time 60 min and “Cuff Pregabalin” < “Sham Pregabalin” at p < 0.001 on post-administration time 0, 30, and 120 min). The same transitory effect was also present in MOP receptors (Figure 4; group × time interaction, ATS(1,.6) = 11.1, p < 0.001; multiple comparisons: “Cuff Pregabalin” = “Sham Pregabalin” at p = 1.0 on post-administration time 60 min and “Cuff Pregabalin” < “Sham Pregabalin” at p < 0.01 on post-administration time 0, 30, and 120 min), DOP receptors (Figure 4; group × time interaction, ATS(2.2) = 12.7, p < 0.001; multiple comparisons: “Cuff Pregabalin” = “Sham Pregabalin” at p > 0.7 on post-administration time 60 min and “Cuff Pregabalin” < “Sham Pregabalin” at p < 0.01 on post-administration time 0, 30, and 120 min), and KOP receptors-deficient mice (Figure 4; group × time interaction, ATS(2.3) = 10.5, p < 0.001; multiple comparisons: “Cuff Pregabalin” = “Sham Pregabalin” at p > 0.6 on post-administration time 60 min and “Cuff Pregabalin” < “Sham Pregabalin” at p < 0.01 on post-administration time 0, 30, and 120 min). These transitory antiallodynic effects disappeared 120 min after injection of pregabalin.
Effect of acute pregabalin in opioid receptor-deficient mice. Two weeks after cuff implantation, the animals received an acute injection of saline (i.p.) or of pregabalin (30 mg/kg, i.p.). Nociceptive mechanical threshold (PWT) was tested before (0 min) 30, 60, and 120 min after these acute injections. Acute pregabalin had a transitory antiallodynic effect in wild type Cuff mice without affecting Sham animals. Similar results were obtained in MOP, DOP, and KOP receptors-deficient mice. (Number of animals are given between brackets, data are pooled from three independent experiments, each final group includes the same number of male and female mice, *p < 0.005 compared to Sham-operated controls receiving pregabalin.) Data are expressed as mean ± SEM.
Naloxone effect on acute pregabalin treatment
Naloxone (1 mg/kg) did not suppress the transitory antiallodynic action of acute pregabalin administration (Figure 5(a); group interaction, ATS(1.0) = 181.7, p < 0.001; multiple comparisons: “Cuff Saline” < “Sham Saline” at p < 0.001 for acute saline administration and at p < 0.005 for acute naloxone administration) (Figure 5(b), acute saline; group × time interaction, ATS(1.0) = 12.7, p < 0.001; multiple comparisons: “Cuff Pregabalin” < “Sham Pregabalin” at p < 0.001 preinjection and “Cuff Pregabalin” = “Sham Pregabalin” at p > 0.5 postinjection; Acute Naloxone; group × time interaction, ATS(1.0) = 13.7, p < 0.001; multiple comparisons: “Cuff Pregabalin” < “Sham Pregabalin” at p < 0.001 preinjection and “Cuff Pregabalin” = “Sham Pregabalin” at p > 0.8 postinjection).
Acute opioid receptor antagonist in acute pregabalin treatment. Two weeks after unilateral cuff surgery, mice received an injection of pregabalin (30 mg/kg, i.p.) or saline control; 30 min later, they received an injection of the opioid receptor antagonist naloxone (1 mg/kg, s.c.) or control saline solution. Mechanical threshold for the right hindpaw (PWT) was measured before the first injection and 30 min after the second injection. (a) Naloxone and saline had no effect in Sham mice and in Cuff mice that received control treatment (n = 7–8, *p < 0.005 compared to the Sham-operated control group). (b) Naloxone and saline had no effect in Sham mice and in Cuff mice that received pregabalin treatment (30 mg/kg, i.p.) (n = 7–8, *p < 0.005 compared to the Sham-operated control group). Data are expressed as mean ± SEM.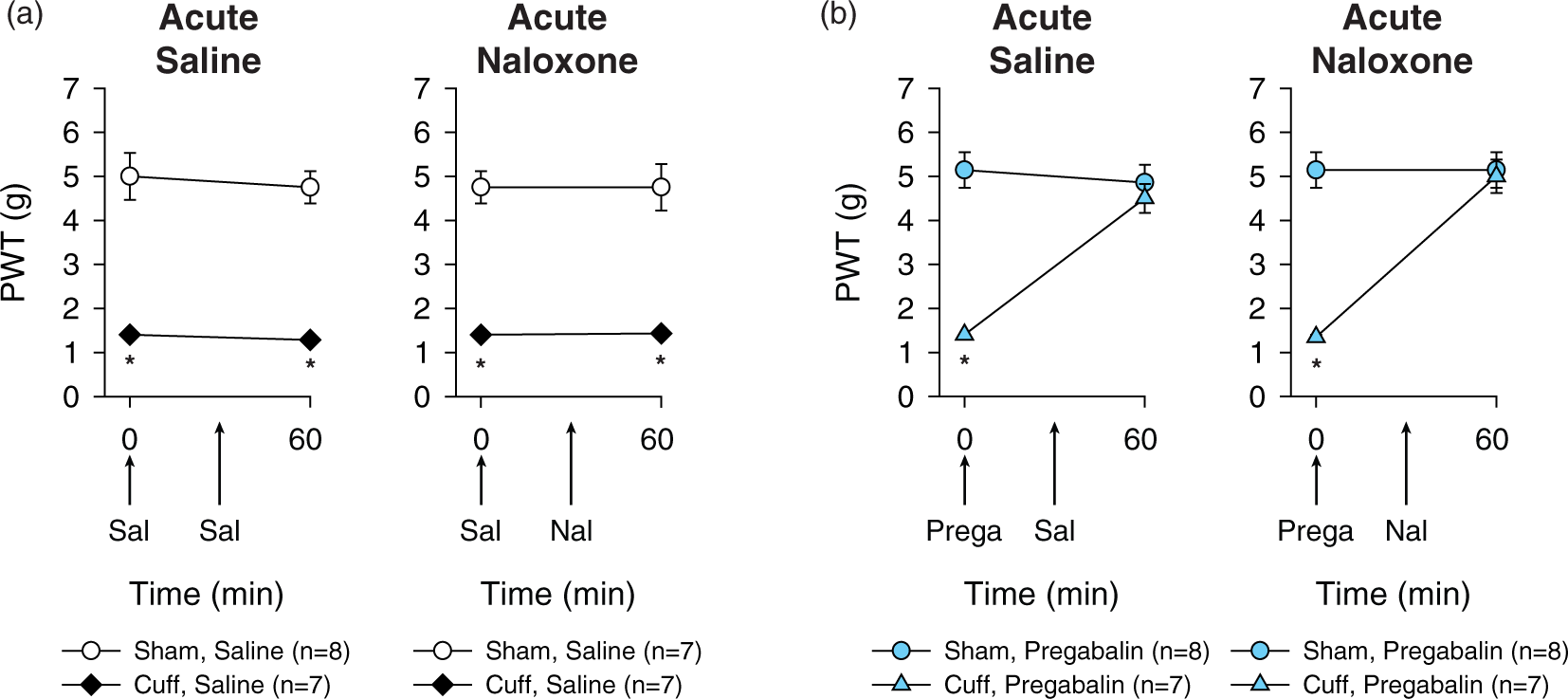
Long-term pregabalin has an anti-TNF-α action
Using Western blot, we observed increased levels of the membrane-bound form of TNF-α (mTNF-α) in the lumbar DRG of C57BL/6J Cuff mice at four weeks postinjury. The long-term treatment with pregabalin reversed this increase in mTNF-α. (Figure 6; H(2.0) = 16.2, p < 0.001; multiple comparisons: “Cuff Vehicle” > (“Cuff Pregabalin” or “Sham Vehicle”) at p < 0.005).
Long-term pregabalin displays an anti-TNF-α action on lumbar dorsal root ganglia of neuropathic mice. (a) Representative picture of Western blot illustrating the increased TNF-α levels in DRG of Cuff mice four to five weeks after induction of the neuropathy, and the anti-TNF-α action of the long-term pregabalin treatment (300 µg/mL i.e. 44.63 mg/kg/day). (b) Histogram presenting the Western blot analysis (n = 8 per group, *p < 0.005 vs. Sham-vehicle). Data are expressed as mean ± SEM.
Discussion
In the present work, we studied the role of opioid receptors in both the long-term and the acute transitory antiallodynic action of systemic pregabalin in a model of neuropathic pain. In both cases, we show that the endogenous opioid system is not necessary for this action. We also show that a long-term pregabalin treatment suppresses the DRG TNF-α overexpression that accompanies neuropathic pain.
Clinically, first line pharmacological treatments to relieve neuropathic pain include anticonvulsants and antidepressants. Gabapentinoid anticonvulsants, which target the VDCCs α2δ-1 subunit, have proved to be effective in a number of neuropathic pain conditions.3,39 Similarly to many reports in various animal models,6,20,40,41 we showed that pregabalin has a short-term transitory antiallodynic action after an acute administration; however, this effect cannot be considered as representative of the main clinical therapeutic effect since the mechanical allodynia reappears within 2 h following the injection. Interestingly, the benefit of pregabalin treatment is sustained after three days of oral administration, which is in agreement with other results obtained with systemic delivery of another gabapentinoid, gabapentin,20,42 or in other neuropathic pain models.24,43 This sustained action may more likely be representative of the clinical use and action of gabapentinoids in neuropathic pain.44,45
Critical aspects of mechanism(s) by which gabapentinoids alleviate neuropathic pain is (are) now well described. Gabapentinoids inhibit calcium currents through direct interaction with the α2δ-1 subunit, thus decreasing excitatory transmitter release and spinal sensitization.8,46 This target subunit is upregulated in the dorsal horn of the spinal cord and in DRG neurons in several models of neuropathic pain and this increase in α2δ-1 correlates with the onset of allodynia. 47 Furthermore, experiments performed in transgenic mice overexpressing the α2δ-1 subunit showed enhanced calcium currents recorded in DRG neurons, as well as nociceptive behavior characterized by hyperalgesia in the absence of nerve damage. 48 In contrast, α2δ-1 deficient mice display reduced DRG calcium currents, have lower baseline mechanical sensitivity, and show delayed mechanical hypersensitivity after partial sciatic nerve ligation. 49 In DRG neurons, α2δ-1 upregulation recruits mitochondrial Ca2+ to prolong intracellular Ca2+ signals evoked by depolarization. 50 This mechanism may contribute to the aberrant neurotransmission observed in neuropathic pain. Pregabalin antiallodynic effect is associated with decreased trafficking of the α2δ-1 subunit to presynaptic terminals of DRG neurons;8,46 and within the dorsal horn, gabapentinoids also decrease the amplitude of excitatory postsynaptic currents. 51
In addition to these actions, two studies suggested that gabapentinoids may also recruit the endogenous opioid system,18,19 which is well known for playing a crucial role in the control of nociception and pain.10,11,52 Indeed, the opioid antagonist naloxone reversed the acute antinociceptive activity of a high dose of pregabalin in naive mice. 18 Another study also showed an effect of naltrexone on the acute action of gabapentin in a model of orofacial inflammatory pain. 19 These recent data differ from previous studies on gabapentinoid drugs, which mostly reported naloxone to be ineffective in blocking gabapentinoid-induced analgesia in different pain models.20–22 However, most of these studies were not done in models of neuropathic pain, which is the clinical pain condition for which gabapentinoids have legal authorization for prescription in various countries. Beside pharmacological approach, the present study used genetic deletion of opioid receptors for the first time, which further clarifies the involvement of the opioid system in both acute and chronic antiallodynic action of pregabalin in neuropathic pain. We demonstrate that neither acute nor long-term antiallodynic effect of pregabalin requires the presence of opioid receptors. Both our results and previous studies20–22 refute the involvement of the opioid system in the antiallodynic action of pregabalin in neuropathic pain, which does not exclude a possible involvement of these receptors in gabapentinoid action on other types of pain.
The opioid system via MOP, DOP, and KOP receptors plays a crucial role in the inhibitory controls of pain10,52,53 and also participates in the therapeutic action of various pain killers. Thus, MOP receptors are the primary molecular target for the analgesic action of opioids such as morphine, codeine, fentanyl, or tramadol.10,54,55 Indirectly, the opioid system is also necessary for the antiallodynic action of tricyclic antidepressant drugs, which requires DOP receptors, but not MOP or KOP receptors.11,36,56 Our results strengthen the idea that antidepressant and anticonvulsant treatments alleviate neuropathic pain through independent mechanisms. These mechanistic differences may be in favor of combination pharmacotherapy for the management of neuropathic pain using both gabapentinoids and antidepressants,57,58 although the benefit of such a combination is still controversial,3,59 or using both gabapentinoids and opioid drugs.14–17
In the last decade, there has been an increasing number of studies which now provide compelling evidence that neuropathic pain pathogenesis is not simply confined to changes in the activity of neuronal systems, but that it also involves interactions between neurons, immune cells, and glial cells, including the involvement of inflammatory cytokines and chemokines.25,60 Indeed, peripheral nerve injury recruits the immune system at various anatomical locations, including the lesion site, DRG, spinal cord, and supraspinal sites associated with pain pathways. 25 Pro-inflammatory cytokines produced after nerve injury could participate to the initiation and maintenance of neuropathic pain. Among these cytokines, TNF-α has the ability to also favor production of other cytokines. 28 The direct anti-TNF-α drugs infliximab and etanercept are clinically used to treat autoimmune diseases, 61 and these drugs have been shown to have some action on neuropathic pain symptoms both in animal models and in humans.26,30,62–64 In particular, infliximab and etanercept can relieve neuropathic allodynia in the model of neuropathic pain used for the present study. 26 Our results show that pregabalin can display an indirect anti-TNF-α action, as seen on DRG from mice with neuropathic pain. This result is in agreement with previous reports on gabapentin suggesting an indirect action of this drug on cytokines.16,24 Thus, it has been proposed that gabapentin could upregulate the expression of the anti-inflammatory cytokine IL-10 in the spinal cord, leading to the inhibition of the expression of pro-inflammatory cytokines, TNF-α, but also IL-1β and IL-6.16,24
Conclusions
This study demonstrates that none of the three opioid receptors is necessary for the antiallodynic action of acute or chronic pregabalin in a neuropathic pain context. Moreover, long-term pregabalin treatment decreases TNF-α in DRG. Further studies will be needed to elucidate the mechanism by which the direct action of pregabalin on the neuronal VDCCs α2δ-1 subunit may downregulate DRG TNF-α expression, which is mostly produced by non-neuronal cells. While the direct action of pregabalin on its target provides an explanation for acute pregabalin action at high dose, the sustained effect of prolonged treatment suggests the involvement of other downstream mechanisms the elucidation of which may provide new candidates for pharmacological targeting.
Footnotes
Acknowledgments
The authors thank Dr Jean-Luc Rodeau for his help concerning statistical tests, and Stéphane Doridot for animal care and genotyping. They are grateful to Pr Brigitte Kieffer (McGill University, Canada) and Pr Claire Gavériaux-Ruff (IGBMC, France) for providing MOP, DOP, and KOP receptors-deficient mouse breeders.
Authors Contributions
ES and MB equally participated to this work. MK and IY did all surgeries. RAH and RAC performed dose responses. MK, LN, and XW performed behavioral tests on chronically treated opioid-receptor deficient mice. MK performed behavioral tests concerning naloxone and acute pregabalin. MK and DD performed the Western blot experiment. MB, MK, IY, and ES codesigned and supervised all experiments. MK collected and analyzed all data. MK and MB drafted the article. All authors revised the article prior to submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that they have no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Centre National de la Recherche Scientifique (CNRS, contract UPR3212) and the Université de Strasbourg (UPR3212), by a CNRS disability PhD fellowship (to MK), by Fondation d’Entreprise Banque Populaire (to MK), and by Fédéeh-Hewlett-Packard (to MK).
