Abstract
Down syndrome (DS) is a complex condition associated with multiple medical, developmental, and behavioral concerns. A prospective, longitudinal clinical database was integrated into a specialty Down Syndrome Program, with the goals of better understanding the incidence, course, and impact of co-occurring medical, neurodevelopmental, and mental health conditions in DS. We describe the process of developing the database, including a systematic approach to data collection and database infrastructure, and report on feasibility, challenges, and solutions of initial implementation. Between March 2018 and November 2021, data from 842 patients (ages 4.8 months to 26 years) was collected. Challenges included caregiver form completion as well as time and personnel required for successful implementation. With full integration into clinical visit flow, the database proved to be feasible. The database enables identification of patterns of development and health throughout the lifespan and it facilitates future data sharing and collaborative research to advance care.
Keywords
Introduction
Down syndrome (DS), caused by the presence of all or part of an extra chromosome 21, is a complex condition including multiple co-occurring medical conditions (Bull, 2020; Bull et al., 2011), as well as developmental, behavioral, and mental health needs that can impact functional outcomes (Capone et al., 2006; Dykens, 2007; Palumbo and McDougle, 2018). There is significant heterogeneity in cognitive abilities, language, social, and behavioral difficulties in DS; however, the complexity and interplay of factors that influence functional outcomes is largely unknown. There is a pressing need for longitudinal research to clarify the course and incidence of co-occurring medical, neurodevelopmental and mental health conditions, and to understand the interplay and impact of these conditions in DS. Questions such as the prevalence of sleep apnea in DS, effective treatments for attention-deficit/hyperactivity disorder (ADHD) in this population, early developmental factors predicting autism spectrum disorder in DS, and factors that influence functional outcomes in DS require further investigation (Hendrix et al., 2021).
Previous research has shown the feasibility and value of clinical databases derived from medical records to understand the natural history of relevant conditions to inform guidelines (Barbaresi et al., 2002, 2020; Evenhuis, 1990; Prasher and Gomez, 2007). Such clinical databases have been critical for advancing research in other pediatric diseases, such as inflammatory bowel disease (Atreja et al., 2008; Cashman et al., 1995; McCormick et al., 2002), pediatric cancer (Jeanquartier et al., 2019), and ADHD (Barbaresi et al., 2002).While existing data from specialized DS programs, including collaborative data sharing efforts, have yielded important information (Lavigne et al., 2015), there is still a critical need for systematic collection of comprehensive longitudinal clinical data in DS. The development of research databases, registries, and clinical cohorts has been a high priority for public health research related to DS (Carr and Collins, 2018; Hendrix et al., 2021; Oster-Granite et al., 2011; Ostermaier et al., 2020; Rasmussen et al., 2008) and has been recognized by the National Institute of Health (NIH) through the INCLUDE Project (INvestigation of Co-occurring conditions across the Lifespan to Understand Down syndromE), which outlines areas of needed research and provides NIH funding (National Institutes of Health, 2021).
DS-Connect (Peprah et al., 2015), is a national registry funded by the NIH that contains demographic and health information about individuals with DS provided by these individuals or their families. Participants can periodically update their information and researchers can use the database to identify potential participants for studies. This is an important step, but more precise and clinically-confirmed longitudinal data is needed.
Excellent clinical care involves taking detailed history from patients and considering the complexity and interplay of many factors. Systematic collection and analysis of these clinical data from multiple individuals affords an opportunity to learn more about how complex conditions play out over time. Given the relatively small population of individuals with DS, estimated at 8 per 10,000 live births (Presson et al., 2013), and smaller numbers still for each co-occurring condition, it will be important to have collaborative projects at multiple sites and to collect data throughout the lifespan. Building and sharing data collection systems can facilitate collaboration among clinical DS programs.
This paper describes the creation of a prospective, longitudinal clinical database for children with DS that is integrated into the clinical workflow of an existing specialized pediatric DS clinic. The long term goals are to: 1) improve the quality and consistency of data acquired for clinical visits to advance care; 2) describe the time course and incidence of co-occurring medical, neurodevelopmental, and mental health conditions in children and adolescents with DS, as well as the interplay of these conditions; 3) make discoveries related to patterns of development and the impact of sociodemographic, psychosocial, and medical factors; and 4) understand the impact of interventions on co-occurring conditions. Here, we describe the process of developing the database, including the systematic approach to clinical data collection and database infrastructure. We report on the feasibility, challenges, and solutions of the initial implementation, and describe the sociodemographic distribution of participants enrolled in the database during the first three years after its creation.
Methods
Setting
Founded in 1967, the Boston Children’s Hospital Down Syndrome Program (DSP) provides specialized health care for individuals with DS from birth through young adulthood. Most patients are from Massachusetts and the surrounding New England area; however, since the DSP is situated within a regional referral center for pediatrics, patients also come from across the United States and the world. The DSP is a multidisciplinary clinical and research program with team members consisting of developmental-behavioral pediatricians, a neurodevelopmental disability and child neurology specialist, a psychologist, a pediatric nurse practitioner, a clinic coordinator, a self-advocate patient liaison, two research assistants, a self-advocate research intern, and a research manager. Clinical care for younger patients in the DSP also includes a physical therapist, speech language pathologist and feeding specialist, and an audiologist. Consistent with principles of community engaged research, a parent of a child with DS plays an essential and central role in our clinical and research programs, including participation in the design and implementation of the DSP longitudinal clinical database (“Database”). In addition, parents of individuals with DS were involved in piloting and revising caregiver-completed data collection forms (“Caregiver Forms”) from families. Individuals with DS who are employed in the program also assist with collection of Caregiver Forms from families, as well as data entry.
Routine clinical visits occur approximately every 6 months in the DSP, while children and adolescents with complex medical needs (e.g., multiple co-occurring medical conditions needing follow-up; complex mental health conditions; and/or the need for medication checks), have more frequent clinical visits. Clinicians address patient and caregiver concerns, follow published healthcare supervision guidelines (e.g., annual screening for anemia/iron deficiency, thyroid dysfunction, hearing problems, screening for obstructive sleep apnea), screen for and manage co-occurring medical, neurodevelopmental, and mental health conditions, and monitor developmental and educational progress. Psychological and/or neuropsychological assessments are also offered in the program, particularly when there are behavioral changes, when a child is going through major transition(s), when a child is making limited progress with current supports, or when diagnostic clarity is needed for co-occurring mental health conditions.
Study population
All children and adolescents with a diagnosis of DS as determined by clinical examination and/or karyotyping, who are seen in the DSP and had caregivers who completed any portion of the Caregiver Forms, were eligible to be included in the Database. Out of the 892 patients seen in the DSP from March 1st, 2018 - November 1st, 2021, 761 (85.3%) unique patients, ages four months to 26 years, were included. The remaining 131 patients were not included in the Database because their caregivers did not complete any Caregiver Forms. Information about non-responders was reviewed, and the primary reasons for not completing Caregiver Forms were language barriers and challenges accessing interpreter services in advance of the visit. Sociodemographics in our population were compared to general Massachusetts population sociodemographics from the United States Census (United States Census Bureau, 2021), and also compared to specific sociodemographics of individuals with DS that are available via March of Dimes Peristats (for 2012-2016) (March of Dimes, n.d.) and the Massachusetts Public Health Information Tool Birth Defects dataset (for 2000-2014) (Mass.gov, 2021).
Introduction of the DSP clinical database
The Database launched in March 2018 and was approved by the Boston Children’s Hospital Institutional Review Board (IRB). A waiver of informed consent was approved for this study, as the data were collected as part of the clinical visit and data analyses were conducted as part of an IRB-approved retrospective review of de-identified clinical data. Figure 1 describes the timeline of creation and implementation of the Database. Timeline of Creation and Implementation of the Longitudinal Clinical Database.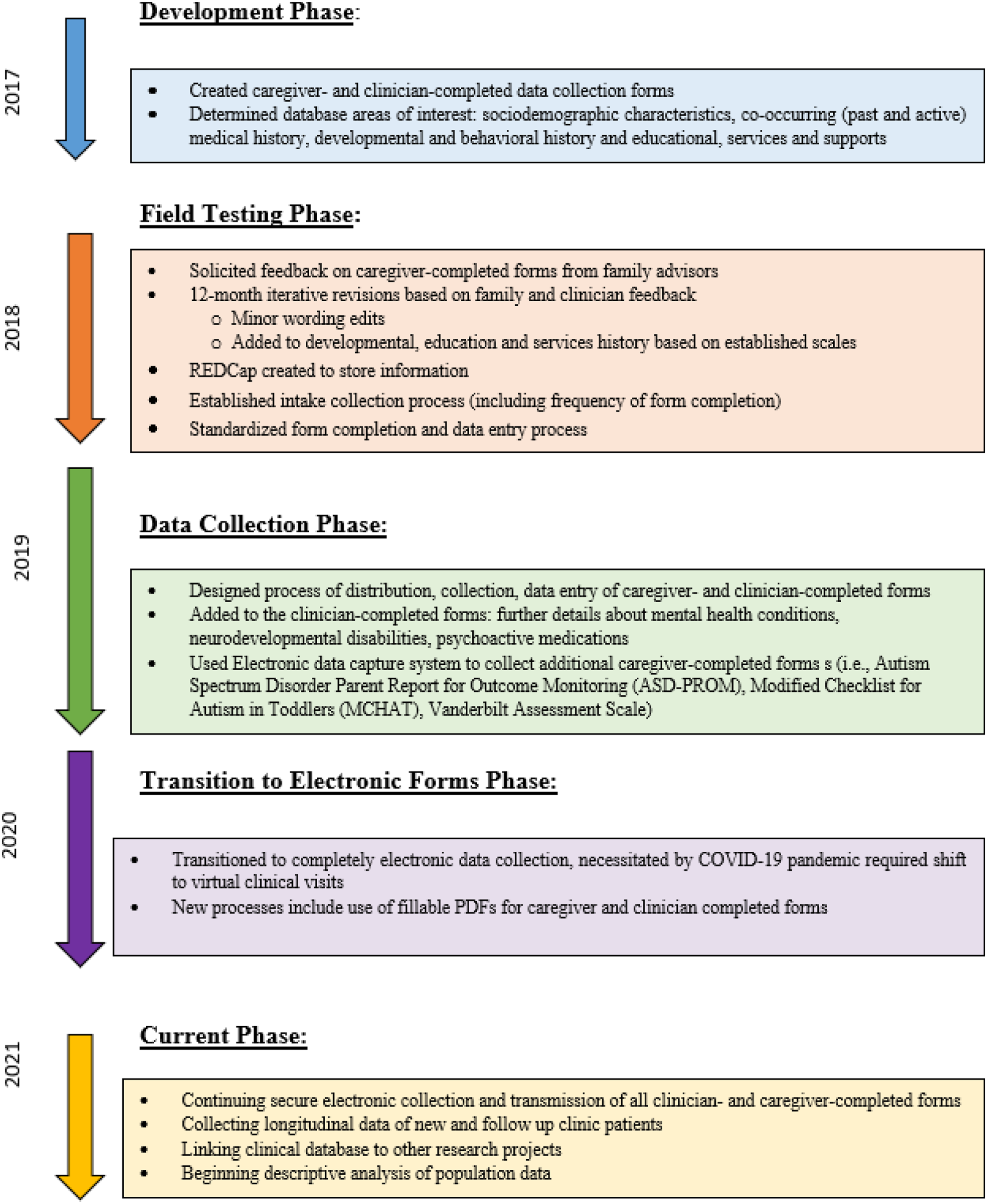
Several data collection forms were created for the Database in order to capture critical information across multiple domains that are relevant to DS. The key areas of information included in the Database are: sociodemographic; medical and family history; psychosocial stressors; developmental and behavioral history; education, services, and supports; co-occurring medical conditions (active and past medical history); and neurodevelopmental disorders and mental health conditions. Standardized data collection forms were created for each domain in order to facilitate efficient, comprehensive, patient-centered care. Following database creation guidelines (Brembilla et al., 2018), forms underwent an iterative six-month testing and editing period that included extensive quality and validity checks. Final versions of the forms were developed within 12 months of the start of this testing period.
To introduce the Caregiver Forms to DSP families and patients, our team developed standardized language that explained the purpose of the Database and accompanying forms, and the process for completing and returning them. This information was shared through a newsletter, via email prior to clinic visits, and discussed during clinic visits. Responses to Caregiver Forms and other structured clinical data (e.g., standardized assessments) were available for review by DSP clinicians and program staff prior to DSP visits so that concerns could be triaged and the visit could be tailored to best meet patient needs.
Form creation
All data collection forms were developed to cover necessary information in as concise, neat, and clear a way as possible. The same formatting style was used across all forms (i.e., age range labels, section headers, grid layouts, text sizes and colors). Section instructions were bolded, italicized, and underlined, and all text remained at a 5th grade reading level. Write-in response boxes were limited to maintain data standardization. Edits from the iterative piloting/feedback process were made in batches when possible, and all versions of the forms were saved so that data changes could be tracked accordingly. The Boston Children’s Hospital medical records/health information team was consulted during the development process to ensure that all published forms met hospital formatting standards.
Database components
A comprehensive outline of the Database domains and their associated data collection forms is shown in Figure 2. Content Areas and Data Sources within the Longitudinal Clinical Database. Note: All caregiver- and clinician-completed data collection forms (Caregiver Forms and Clinician Forms, respectively) are completed on an annual basis, and are for individuals of all ages, unless otherwise noted. White boxes indicate Caregiver Forms; gray boxes indicate Clinician Forms. ASD=Autism Spectrum Disorder; ADHD=Attention-Deficit/Hyperactivity Disorder; OCD=Obsessive Compulsive Disorder. SSRI=Selective Serotonin Reuptake Inhibitor. (a) The Past Medical and Family History form is completed once before the initial clinic visit, or if >3 years have passed since the last visit. (b) The Co-occurring Medical Conditions form is completed by the caregiver when first filled out and then reviewed and verified by the clinician for each subsequent visit. (c) Psychological and Neuropsychological Testing is completed as needed/per clinician referral.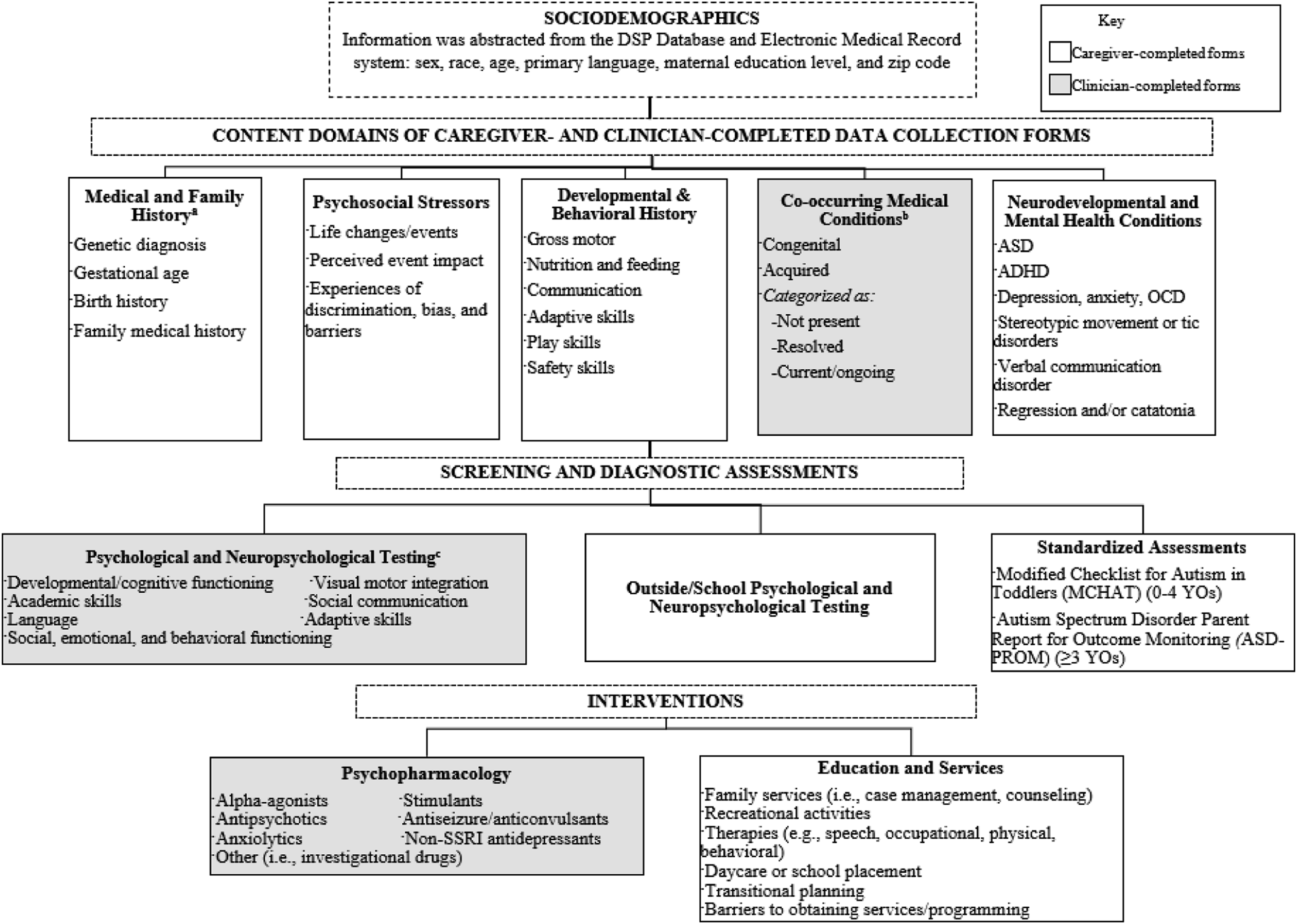
Caregiver-completed data collection forms
Caregiver Forms include information on medical and family history information such as genetic diagnosis, birth history, family history; psychosocial stressors; medical conditions; general development and behavior; and education and services. Caregiver Forms were created by the DSP team based on consensus opinion from experienced clinicians and existing health care guidelines. Caregivers completed the forms independently, except for a limited number with technology and/or literacy barriers who completed the forms with the assistance of our program coordinator. Families requiring an English-language interpreter completed the forms with our program coordinator through the use of a trained medical interpreter.
In addition to the DSP Caregiver Forms, families complete a standardized developmental monitoring tool, the ASD-PROM (Autism Spectrum Disorder Parent Report for Outcome Monitoring). The tool, originally developed for clinical use in autism spectrum disorder (ASD) monitoring and administered to DSP families regardless of co-occurring ASD diagnosis, consists of 127 questions that assess key domains of clinically relevant functioning. Items address communication and social skills, behavior, adaptive skills, and sleep, using Likert scale responses from Never to Always. The ASD-PROM was created by a multidisciplinary group of clinicians and researchers at our institution, including two members of the DSP, for children of varying ages and developmental levels (Levin et al., 2020). The ASD-PROM is hierarchical and directs caregivers to the most developmentally appropriate and relevant questions, which can be more palatable to complete than other available standardized developmental questionnaires.
Caregiver-completed, clinician-confirmed medical conditions form
The Caregiver-Clinician Form is a collaborative form that evolved over the course of the database creation process. Caregivers are asked to provide medical history information for their child, which is then updated and verified with the clinician during their visit. The form covers a comprehensive list of congenital (e.g., congenital heart disease, congenital hypothyroidism, duodenal atresia, Hirschsprung’s disease) and other co-occurring health conditions, organized by body system (i.e., cardiac/heart, respiratory, endocrinology, dental, gastrointestinal, feeding, dermatological, musculoskeletal, hearing/otolaryngology, vision/eyes, blood/hematology, kidney/genitourinary, allergy, neurological, sleep, weight). Once an initial round of medical conditions information has been verified, clinicians re-assess that same information at future visits to identify any changes or new diagnoses. The medical history form used is available in appendix A.
Clinician-completed data collection forms
The neurodevelopmental disorders and mental health Clinician-Completed Data Collection Form (“Clinician Form”) captures whether the child has been diagnosed with any of the following conditions: ASD, ADHD, communication disorder, depression, anxiety, obsessive compulsive disorder, stereotypic movement or tic disorder, regression and/or catatonia, behavior disorder, and psychosis. The Clinician Form also captures treatment with therapies and/or behavioral interventions for each condition, and the classes of psychopharmacological treatments that are prescribed.
For some patients, information is also collected through psychological and/or neuropsychological assessment batteries. Depending on the individual’s age and needs, the battery may include measurement of cognitive, language, motor, academic, behavioral, and adaptive skills, with additional measures added for social communication, mood, anxiety, and executive functioning when needed for diagnostic clarification. Score reports and age equivalents are provided in a standardized form that enables a unified framework for our interdisciplinary team of providers to compare patients’ assessment data to that of other children within the population norms (Klin and Volkmar, 1997; Oakland et al., 2015), in the absence of standardized norms for individuals with DS.
Medical record
Sociodemographic information including sex, race/ethnicity, age, primary language, maternal education level, insurance type, and zip code are obtained from the electronic medical records. A variable was created for annual household income quartiles based on patient zip code: low (< $34,282/year); moderate ($34,282 ≥ x < $54,850); middle ($54,850 ≥ x < $82,276); and upper (≥ $82,276) (Johnson et al., 2013).
Developing and piloting data collection
To ensure smooth integration into the clinic workflow, the complete data collection and entry process was explained to clinicians alongside a written reference guide that outlined the purpose and content of each form; the patient age range for each form; the frequency at which each form should be completed; how each form is disseminated; and how/where forms and data are stored. Study staff observed clinical visits and took notes on clinical flow, and clinicians were able to provide feedback at weekly team meetings.
The forms described above were developed using an iterative feedback process with caregivers and clinicians. A research assistant collected verbal feedback from caregivers following their completion of the Caregiver Forms at home or in the clinic waiting room before a clinic visit. Cognitive interviews were conducted with caregivers who piloted forms in order to assess usability, comprehension, and time to complete the forms. During DSP weekly meetings, clinicians shared feedback regarding ease and length of time needed to review Caregiver Forms, discussed information during clinical visits, and completed clinical and research documentation. Qualitative feedback from caregivers and clinicians was transcribed by the research assistant. Quantitative data such as patient visit time, form completion time, and documentation time were logged. All feedback was reviewed by the team on a weekly basis and used to make adjustments to the intake process to improve clinical flow and efficiency.
During the implementation process, limitations of the caregiver-reported medical conditions process were found (described below). Therefore, a clinician verification process was implemented. The Caregiver-Clinician Form is initially completed by the caregiver, then reviewed by a research assistant, who reviews available clinical documentation and pre-fills information into the Clinician Form prior to the clinic visit. Finally, the clinician reviews and verifies the information before, during, or after the clinic visit, through discussion with the patient and/or caregiver and review of the medical record. Figure 3 describes the timeline and system of data collection and verification associated with each clinical visit. Timeline of Data Collection Steps Associated with Clinical Visits.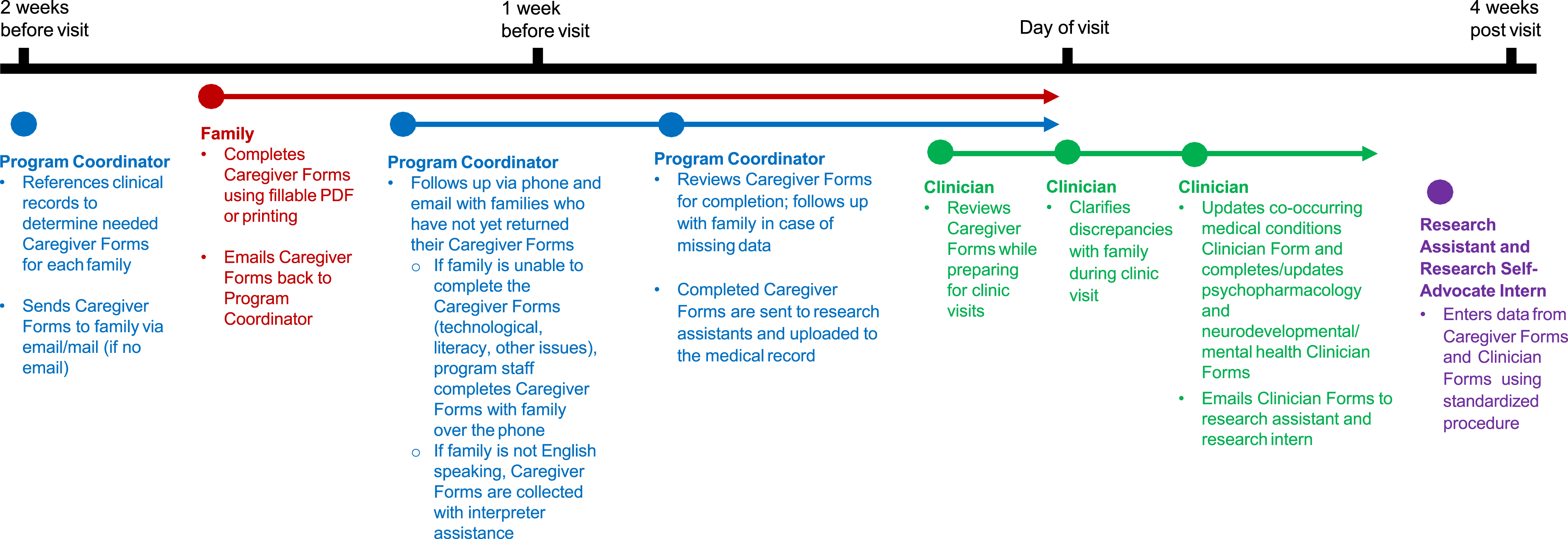
Data collection, entry, and management
Data were stored and managed using REDCap (Research Electronic Data Capture) (Harris et al., 2009, 2019). REDCap is a secure, web-based software platform designed to support data capture for research studies. The REDCap database is protected for study participants’ privacy and is fully compliant with HIPAA regulations. All changes to forms and REDCap instruments after implementation were logged and data were coded accordingly.
Data entry and data quality control were performed by study staff and overseen by the Principal Investigator. All data were entered into REDCap by a research assistant and research intern. A data entry standard operating procedure was created, and data checking and validation procedures (such as branching logic and field-specific validation tools to alert the team if an entry was not within the expected range) were employed to ensure strong data integrity (Harris et al., 2009, 2019). Every twentieth database record was verified by another research team member to ensure data entry reliability. Data reports were shared regularly with the clinical team to discuss progress, discrepancies, challenges/barriers, and feedback for improvement.
Results
Patient sociodemographics
Sociodemographic Characteristics of Participants in the Longitudinal Clinical Database from March 1st 2018 – November 1st 2021.
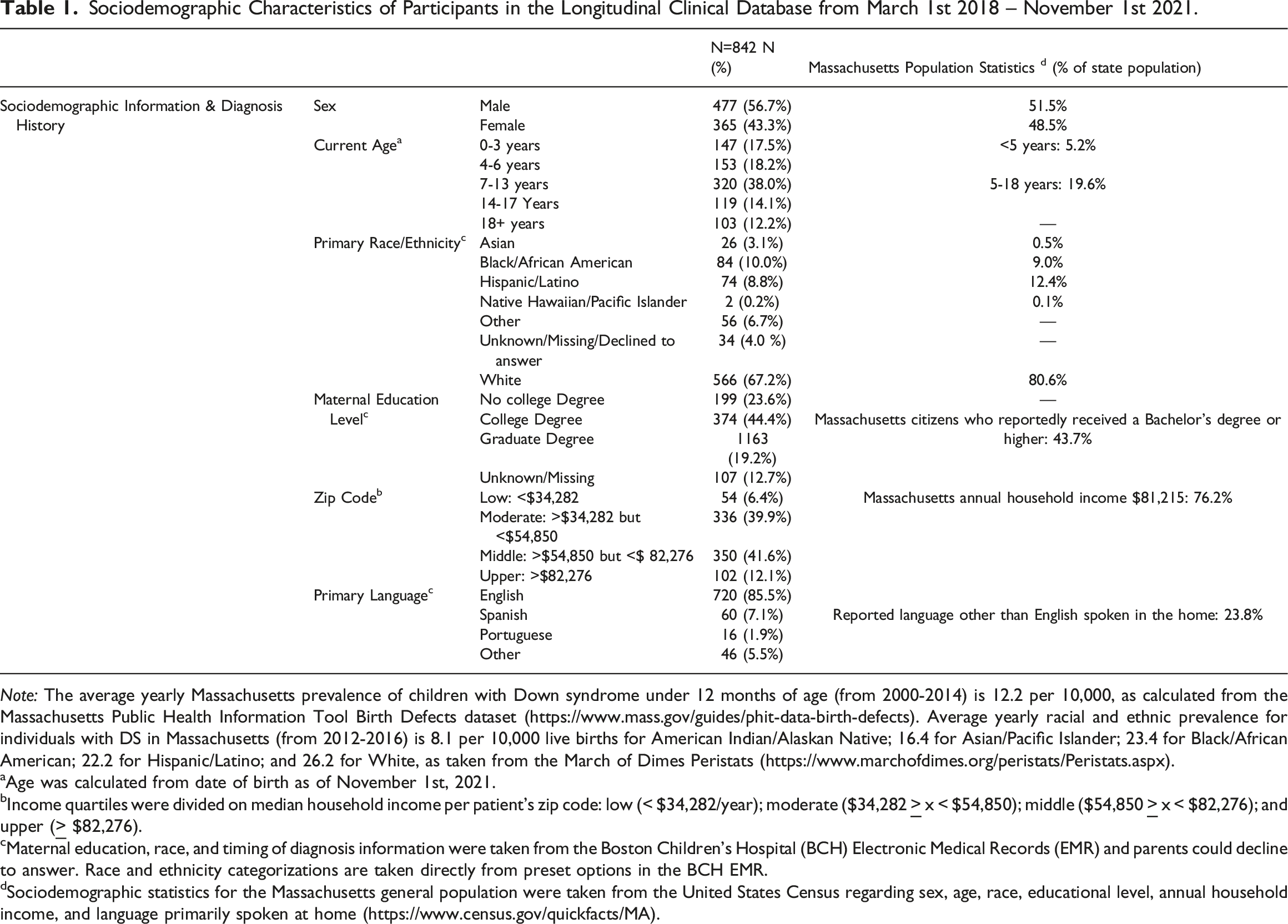
Note: The average yearly Massachusetts prevalence of children with Down syndrome under 12 months of age (from 2000-2014) is 12.2 per 10,000, as calculated from the Massachusetts Public Health Information Tool Birth Defects dataset (https://www.mass.gov/guides/phit-data-birth-defects). Average yearly racial and ethnic prevalence for individuals with DS in Massachusetts (from 2012-2016) is 8.1 per 10,000 live births for American Indian/Alaskan Native; 16.4 for Asian/Pacific Islander; 23.4 for Black/African American; 22.2 for Hispanic/Latino; and 26.2 for White, as taken from the March of Dimes Peristats (https://www.marchofdimes.org/peristats/Peristats.aspx).
aAge was calculated from date of birth as of November 1st, 2021.
bIncome quartiles were divided on median household income per patient’s zip code: low (< $34,282/year); moderate ($34,282
cMaternal education, race, and timing of diagnosis information were taken from the Boston Children’s Hospital (BCH) Electronic Medical Records (EMR) and parents could decline to answer. Race and ethnicity categorizations are taken directly from preset options in the BCH EMR.
dSociodemographic statistics for the Massachusetts general population were taken from the United States Census regarding sex, age, race, educational level, annual household income, and language primarily spoken at home (https://www.census.gov/quickfacts/MA).
Caregiver form completion
Completion Rates of Caregiver-completed Forms in the Longitudinal Clinical Database from March 2018 – January 2022.
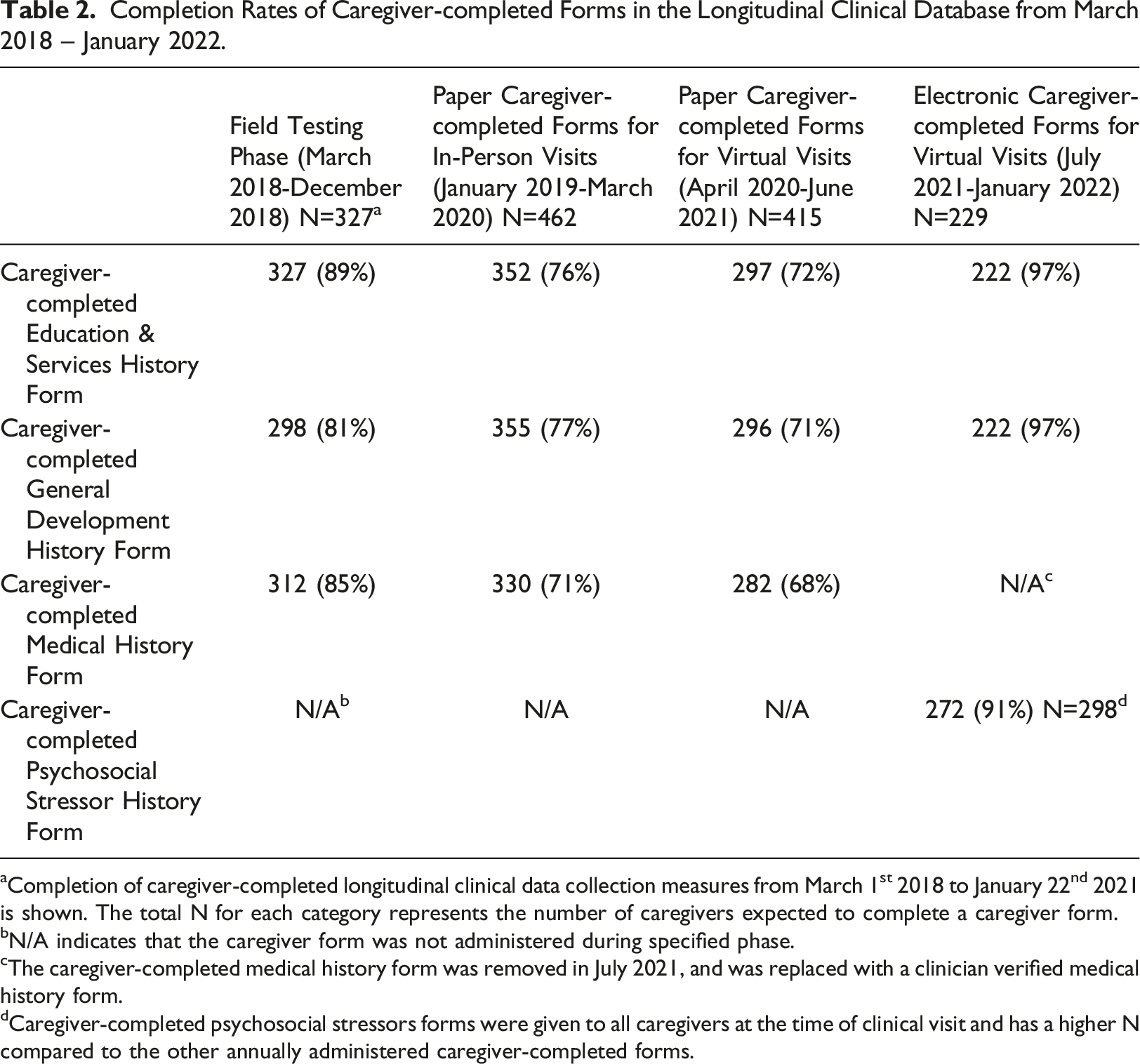
aCompletion of caregiver-completed longitudinal clinical data collection measures from March 1st 2018 to January 22nd 2021 is shown. The total N for each category represents the number of caregivers expected to complete a caregiver form.
bN/A indicates that the caregiver form was not administered during specified phase.
cThe caregiver-completed medical history form was removed in July 2021, and was replaced with a clinician verified medical history form.
dCaregiver-completed psychosocial stressors forms were given to all caregivers at the time of clinical visit and has a higher N compared to the other annually administered caregiver-completed forms.
Caregiver feedback
Caregiver concerns centered around form layout and clarity, completion time, and completion logistics. They emphasized the importance of readability (e.g., that text needed to fit together concisely but not be too crammed), additional section delineation and instructions, use of familiar medical phrases and/or layperson's terms, and that examples should be provided. Caregivers reported that Caregiver Forms took approximately 40 minutes to complete, with this range decreasing over time as caregivers became more familiar with the form layout and content. Logistically, they shared that the process of printing, completing, scanning, and returning forms was time-consuming.
To address Caregiver Form access and timing concerns, section and item instructions were reworded and further visually highlighted. Items were condensed into grids where possible. Items that were found to be out of caregivers’ scope of knowledge (e.g., exact number of hours of each kind of in-school service) were eliminated. Small notes and icons were added to the bottom right corner of intake pages indicating how many pages remained. Additionally, all of the fillable Caregiver Forms were ultimately merged into one single file, rather than multiple separate forms. These changes, in addition to increased familiarity over time, resulted in improved completion rates and efficiency over time.
Clinician feedback
Introduction of the Database system and the Caregiver, Caregiver-Clinician, and Clinician Forms into pre- and post- clinic visit workflow posed initial challenges for clinicians. Clinician concerns included time to review forms as well as impingement on clinic visit time when Caregiver Forms were not completed by the start of the visit. Challenges included caregivers skipping items or incorrectly completing items (e.g., marking more than one answer where only one answer should have been selected), and caregivers not completing sections of the form towards the end. Additionally, as Clinician Forms cannot be automatically uploaded into the medical record, clinicians are responsible for both completing Clinician Forms, and manually documenting and updating the medical history in the clinical chart, which is time consuming. Overall, the amount of time to complete forms decreased over time.
Several benefits were reported by clinicians. They reported that the use of Caregiver and Clinician Forms allowed for standardization of practice among clinicians in the DSP, to ensure that all clinicians, including trainees, cover the full breadth of complex needs for individuals with DS. Reviewing Caregiver Forms completed by families prior to visits improved clinic visit efficiency by decreasing time for medical history taking and information gathering. This allows clinicians to prioritize patient and family concerns and spend more time on recommendations, in addition to reviewing health care guidelines.
Lessons from data collection form and database creation
Several practical tips emerged over the course of development of our Database and data collection forms. Organization based on topic domain (e.g., education and services) made it easier to customize sections of Caregiver Forms for different age groups where appropriate. All data collection form items were evaluated for the clarity of data they would output, as well as the ability of that data to contribute to clinical and research questions.
Our longitudinal Database was created to maximize REDCap features like event arms, branching logic, automatic age and date calculations, and coded data pulls that can be used repeatedly as data are updated. A comprehensive standard operating procedure has been essential for training new team members and maintaining a unified data entry system. A parallel clinic appointment database tracks the Caregiver Form completion process and allows us to track anticipated versus completed statistics.
Discussion
In this paper, we described the process of creating and implementing a comprehensive, longitudinal clinical database for children and adolescents with DS. Observational standardized clinical data from routine visits to the DSP were collected and captured for a large number of patients with DS. Multiple data collection iterations and process phases were required to obtain standardized, comprehensive clinical data while also reducing burden on caregivers, clinicians, and maintaining efficient clinical flow. Ultimately, establishment of a clinical DS database is feasible, and a critical step to address gaps in our knowledge about DS.
Our findings revealed both benefits and challenges to implementation of the Database. While our Database represents the racial and ethnic diversity of the general population in Massachusetts (United States Census Bureau, 2021), when compared to available sociodemographic data of individuals with DS in Massachusetts (Mass.gov, 2021), our population has more White non-Hispanic/Latino participants.
Form completion was most challenging for non-English speaking families, families with low levels of English literacy, and those without an email address and/or access to technology, even with support from our DSP program coordinator. We also found that a primary limitation to our Database was the accuracy and completion of certain medical information provided exclusively by caregivers. For example, when cross-checked against the medical records, inaccuracies were noted for the specific type of congenital heart disease the child had, with caregivers often missing diagnoses or indicating a current (rather than past/resolved) diagnosis. This trend is consistent with previous reports that caregivers may less accurately report health events and diagnoses outside of the previous year, and/or conditions that are considered less severe (Pless and Pless, 1995), or are long-term chronic diagnoses, such as obesity or obstructive sleep apnea (Akinbami and Ogden, 2009; Giménez et al., 2018; Goodman et al., 2000; Maris et al., 2016). This may also explain lower than expected rates of these conditions in our Database. For these reasons, a clinician verification process for medical conditions was implemented, requiring additional time on the part of the treating clinician, particularly for the initial clinician verification of medical history.
Additionally, we encountered challenges with caregivers completing individual items incorrectly or missing/skipping sections. The final sections of Caregiver Forms were not always fully completed, which may be due to respondent fatigue, a common dilemma in questionnaire-based research that is primarily attributed to questionnaire length, delivery, and content clarity and density (Rolstad et al., 2011). Clinic coordinator review of Caregiver Forms was initiated to ensure that necessary information was provided to clinicians; however, reviewing forms and following up on missing information requires substantial staff time and effort. The level of staffing required to ensure completion of the forms could represent a barrier to implementing a similar database in other settings. Future iterations will involve secure, web-based forms that perform automatic completion checks and require completion prior to advancing to subsequent sections.
A central benefit of caregiver-reported data is that caregivers are well positioned to observe their children across a range of settings and situations (Sachse and von Suchodoletz, 2008). This advantage is particularly important for assessing and documenting early developmental milestones and delays that may not be exhibited as clearly (if at all) in clinical settings (Miller et al., 2017). Similarly, caregivers of older children and adolescents with DS are able to provide essential information about development, behavior, and services in school and social settings.
Key elements to successful implementation of the Database included: 1) multidisciplinary collaboration and an iterative creation process among the clinical and research team, as well as caregivers of patients and self-advocates employed in the DSP; 2) collaborative family/clinician approach to completing the Caregiver-Clinician medical conditions form during clinical visits; and 3) use of electronic fillable form technology to capture comprehensive data across multiple clinical domains. Our comprehensive approach to database development allowed us to identify and include high priority areas for clinical research and ensure integration of clinical and research goals with community needs.
In the future, we also hope to have integrated clinical and research systems, such as web-based forms that can be directly entered into the medical record and automatically integrated into the Database. Future iterations will also include Spanish language forms, as Spanish is the second-most commonly spoken language in the DSP, as well as additional staff support for families who have challenges completing the Caregiver Form. Additionally, we will investigate differences between those who complete forms and those who do not. Inclusion will also be enhanced with piloting of patient-report forms for individuals with DS. Additionally, in the future we will link this Database with other research projects within our program that have collected extensive phenotypic data and biomarkers, such as genetic, neurophysiological, and neuroimaging assessments, through assignment of unique study participant identifiers.
Conclusion
Our findings demonstrate the feasibility of a prospective, longitudinal clinical database in a large, pediatric subspecialty Down Syndrome Program that enables close tracking of development, medical conditions, education and services, and functional outcomes over time. This represents an important first step in the use of clinical data to address pertinent questions about health and development in children and adolescents with DS. The clinical database infrastructure can serve as a model for other specialty clinics, and facilitates future data sharing and collaborative research to advance care.
Footnotes
Acknowledgements
The authors would like to acknowledge our colleagues, Sherry Tsai, Jennifer Guan, and Nathan Simon, for their contributions; and to the patients and families in the Boston Children’s Hospital Down Syndrome Program.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was received by NTB from the Eleanor and Miles Shore Faculty Development Awards Program (![]() ). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
XXX Medical Conditions Form
*Clinical Site Header / Copyright Removed*
Congenital Conditions
Congenital Conditions
Check if Present / Positive
CARDIAC / HEART
Complete Antrioventricular Canal Defect (CAVC)
Patent Foramen Ovale (PFO)
Patent Ductus Arteriosus (PDA)
Atrial Septal Defect (ASD)
Ventricular Septal Defect (VSD)
Valve problem(s)
Tetralogy of Fallot
OTHER
Tracheal esophageal fistula
Congenital Hypothyroidism (Diagnosed with hypothyroidism < 2 weeks of age)
Duodenal atresia/stenosis
Esophageal atresia
Hirschsprung’s
Transient myeloproliferative disorder
Acquired Conditions
Acquired Conditions
Check if Never had condition
Check if History of condition, but resolved at current visit date
Check if New / or established ongoing at current visit
CARDIAC / HEART
High blood pressure
Lipid disorder
Cardiac surgery
Other cardiac:
RESPIRATORY
Asthma
Aspiration pneumonia
Pneumonia
Respiratory infections
Laryngeal cleft
Other respiratory:
ENDOCRINOLOGY
Hypothyroidism
Hyperthyroidism
Type 1 Diabetes
Type 2 Diabetes
Early puberty onset (before 8 for female; before 9 for male)
Low vitamin D
Other endo:
DENTAL
Cavities
Periodontal disease
Orthodontic treatment (e.g. braces, palate expander)
Dental surgery
Other dental:
GASTROINTESTINAL
Constipation
Gastroesophageal reflux
Celiac
GI surgery
Other GI:
FEEDING
G-tube
Dysphagia WITHOUT aspiration
Dysphagia WITH aspiration
Special dietary needs
Behavioral feeding problem
Other feeding:
DERMATOLOGICAL
Dry skin
Eczema
Hair loss/Alopecia
Folliculitis/Hidradenitis suppurativa
Acne
Toenail problem
Other skin/derm:
MUSCULOSKELETAL
Diagnosed hip problems
Diagnosed knee problems
Diagnosed foot problems
Cervical spine problems/Atlantoaxial instability
Scoliosis
Musculoskeletal surgery
Other musculo:
HEARING / OTOLARYNGOLOGY
Hearing loss
Hearing aids (wears or prescribed)
Eustachian tube dysfunction
Tympanostomy tubes
Tonsillectomy
Adenoidectomy
Wax build-up
Ear infection
Other hearing/ENT:
Other ENT surgery:
VISION / EYES
Refractive errors/Astigmatism
Esotropia/Eye crossing
Cataracts
Glasses (wears or prescribed)
Eye surgery
Other vision/eyes:
BLOOD / HEMATOLOGY
Anemia
Leukemia
Other blood/hem:
KIDNEY / GENITOURINARY
Kidney/Urinary infections
Hydronephrosis
Vesicoureteral reflux
Undescended testicle(s)
GU surgery (e.g. orchidopexy, hernia)
Other kidney/GU:
ALLERGY
Immunodeficiency
Other allergic reactions
Seasonal allergies
Other allergy:
NEUROLOGICAL
Abnormal weakness
Generalized seizures
Partial seizures
Infantile spasms
Other neuro:
SLEEP
Obstructive sleep apnea
Diagnosed sleep disorder
Other sleep:
WEIGHT
Low weight/Failure to gain weight
Overweight/Obese
Other weight:
