Abstract
Flash sintering (FS) is of great interest today, as it allows the production of high-density ceramics in seconds at lower temperatures than conventional sintering. In this study, the effects of conventional sintering at 1250°C for 4 h and FS at various electric fields (12.5, 25, 50 and 75 V/mm) on copper smelting slag (CSS), which is a multiphase material system, are investigated. The results show that FS occurs at substantially lower temperatures with much shorter times than conventional sintering, in conjunction with improved density and hardness values for CSS. The optimum condition for better densification is observed for the sample applied the 25 V/mm electric field in 554 s, and the maximum power density of about 76.49 mW/mm3. On the other hand, the shortest FS duration is achieved with the sample applied 75 V/mm in 114 s. Flash sintering at lower temperatures also stabilizedthe magnetite-hematite the phase transition, which arouses at higher temperatures. Overall, FS has made a huge contribution to reducing sintering time and temperature while providing better microstructure integrity and mechanical properties in multiphase materials systems.
Introduction
Copper is one of the most common metals that is widely used in a variety of industries today. The demand for the copper industry along with the amount of copper production to meet that demand is increasing by the day, and global annual copper production has reached approximately 21 million tonnes in 2021. 1
Slag is a waste product acquired during a variety of metal production processes. 2 The repurposing of these waste products is critical for reducing pollution and creating a sustainable economy. Copper smelting slag (CSS) is a by-product of copper production by smelting that typically contains oxides such as iron oxide, calcium oxide, aluminum oxide, silicon oxide, and minor amounts of other elements (e.g., zinc, titanium and lead). The chemical composition of copper slag is determined by the ore source and the copper production process. 3 Iron oxide and silicon oxide, as well as fayalite (2FeO.SiO2) formed by the combination of these two oxides, dominate among these components. 4 It is estimated that about 2.2 tonnes of copper slag are produced for just one tonne of copper production.5,6 The reuse of such an over-produced product has become inevitable for several applications. 2 Copper smelting slag can typically be used in products such as abrasive tools, roofing granules, road foundation construction, railway ballasts, and fine aggregate in concrete because it contains oxides such as iron oxide and silicon oxide in high amounts. 7
Materials with high melting temperatures, such as ceramics, can be produced via a technique called sintering, which is carried out at 2/3 or 3/4 of the melting temperature of the sample. Sintering is a high-temperature process that densifies and consolidates the material and enhances mass transport.8,9 There are also different types of sintering besides the traditional ones, such as electric field assisted sintering. 10 Conventional sintering consumes large amounts of thermal energy, requires hours to days of processing, and increases CO2 emissions significantly, increasing its manufacturing cost and making it harmful to the environment. Long-term sintering at very high temperatures also increases the probability of grain coarsening and adversely affects the strength.8–10 To minimise such negative effects, a new sintering method called flash sintering (FS) was first developed by Rishi Raj of the University of Colorado Boulder in 2010. 11 The main feature of the FS method is that it allows high-density ceramics to be densified in just seconds, at temperatures that are considerably lower than those required for conventional sintering. Flash sintering can be applied to many material systems. In this method, the pressed pellet with its nature of semiconductor behaviour is exposed to both thermal and electric field at the same time. Initially, no current flows through the sample are observed at a lower temperature under a DC electric field. However, when a certain temperature is reached in the sample, the current suddenly passes over the sample, and an extremely rapid mass transfer is observed in the sample in a very short time period. As a result, sintering is completed in a shorter duration and at lower temperatures compared to conventional sintering.8,11–13 This study examined the effects of different electric fields of FS on pelleted CSS. The microstructural, mechanical, and chemical properties of the flash-sintered and conventionally sintered pellets are compared in conjunction with the investigation of phase transitions triggered during the high-temperature firing process.
Experimental
Copper smelting slag in powder form was supplied by Eti Copper Inc., the copper smelter plant in Samsun, Turkey. XRF analyses were performed to determine the chemical composition of CSS powder using a Rigaku-NEX-CG, Austin, TX, USA, multi-element analyzer. The chemical composition of CSS powder is shown in Table 1. XRD analysis was performed for the mineral phases’ determination with CuKα radiation over a 2θ range of 20–80° with a scan rate of 2°/min by using a Europe 600 Benchtop XRD Instrument, Novara, Italy.
The chemical composition of the CSS materials system.

The slag powder, which was of 3.6 µm average particle size, was first mixed with an aqueous solution of polyethylene glycol (Merck KGaA, Darmstadt, Germany) as a binder and then pressed with a uniaxial hydraulic press (Hidroliksan, Konya, Turkey) under a pressure of 100 bars (10 MPa) to form cylindrical identical pellets (diameter of 13 mm and height of 2 mm). The binder burnout process is applied to these pellets at 600°C for 1 h to remove the binder. Then identical pellets were sintered with conventional and FS methods.
First, the conventional sintering process is carried out as follows; the pellet was heated to 1250°C at a heating rate of 3°C/min, then sintered for 4 h at this temperature, and then cooled to room temperature in the furnace.
Second, the FS experimental setup is given schematically in Figure 1. The surfaces of the pellets were first contacted with platinum foils, an inert metal, to create ohmic contacts. For better contact, ceramic insulators are covered by platinum foils tight enough to keep them in contact during the whole sintering process. The platinum foils were then linked to the power supply next to the furnace via copper wiring.
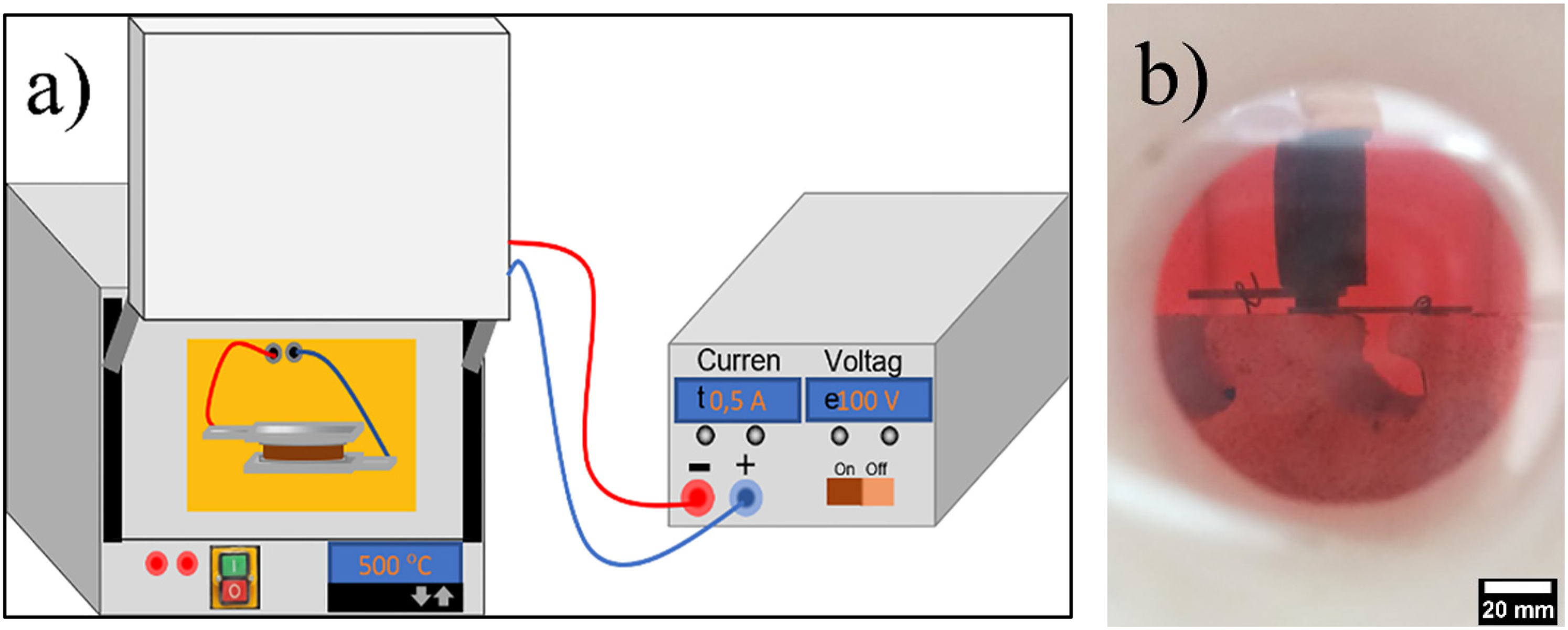
(a) The schematic of the flash sintering setup, (b) a real photograph of the experimental setup.
After the experimental setup was completed, four identical samples were individually exposed to four different electric fields – 12.5, 25, 50, and 75 V/mm – which were generated by a DC power supply (Ametek, XG600–2.6 model 1500 W, 2200 VA, San Diego, CA, USA) while the furnace temperature was gradually increased at a heating rate of 20°C/min. The current cut-off value was set to 3.775 mA/mm2 to prevent overheating when maximum current flowed through the pellet. During the experiment, voltage and current values were recorded by the computer-aided system as a function of time and temperature. In order to enable visual documentation of the sintering procedure, a quartz glass cover was positioned in front of the furnace (Figure 1(b)).
A scanning electron microscope (SEM) was used to observe the microstructure evolution after both the conventional and FS processes. The elemental composition of sintered pellets was determined using energy-dispersive X-ray spectroscopy (SEM-EDS). Zeiss Evo LS 10, Carl Zeiss. NTS, Germany microscope was used for SEM and SEM-EDS analyses. XRD analysis was performed to determine any phase evolution along with any phase transition triggered by the sintering process. Microhardness (HV) test was carried out to measure the hardness of sintered pellets. Hardness measurements were performed at least five times using a load of 100 grams for a dwell time of 10 s at room temperature with the Leeb Hardness Tester, Thl280 Plus, Beijing, China. The Archimedes method was used to calculate the densities of the sintered pellets.
Result and discussion
The result of the XRF analysis of CSS is given in Table 1. As can be seen from the table, the amount of Fe3O4 in this material system is the highest, and the second-largest phase is SiO2. The table demonstrates that CSS also includes a variety of other phases in addition to these main ones.
The results of the XRD analyses of CSS powder and conventionally sintered powder are given in Figure 2. According to XRD analysis results, the existing phases in the CSS powder are magnetite, fayalite and SiO2. The phases with lower amounts were not detected with XRD diffractometry due to the resolution limit. After conventional sintering, it is seen that the magnetite phase partially transforms into the hematite in conjunction with decreasing peak intensity in the magnetite phase. The transformation of magnetite to hematite is an expected situation according to the standard Gibbs free energies of the formation of the oxides of iron in the Ellingham diagram. 14 In addition, an improvement in SiO2 peak reflections was also observed accordingly.
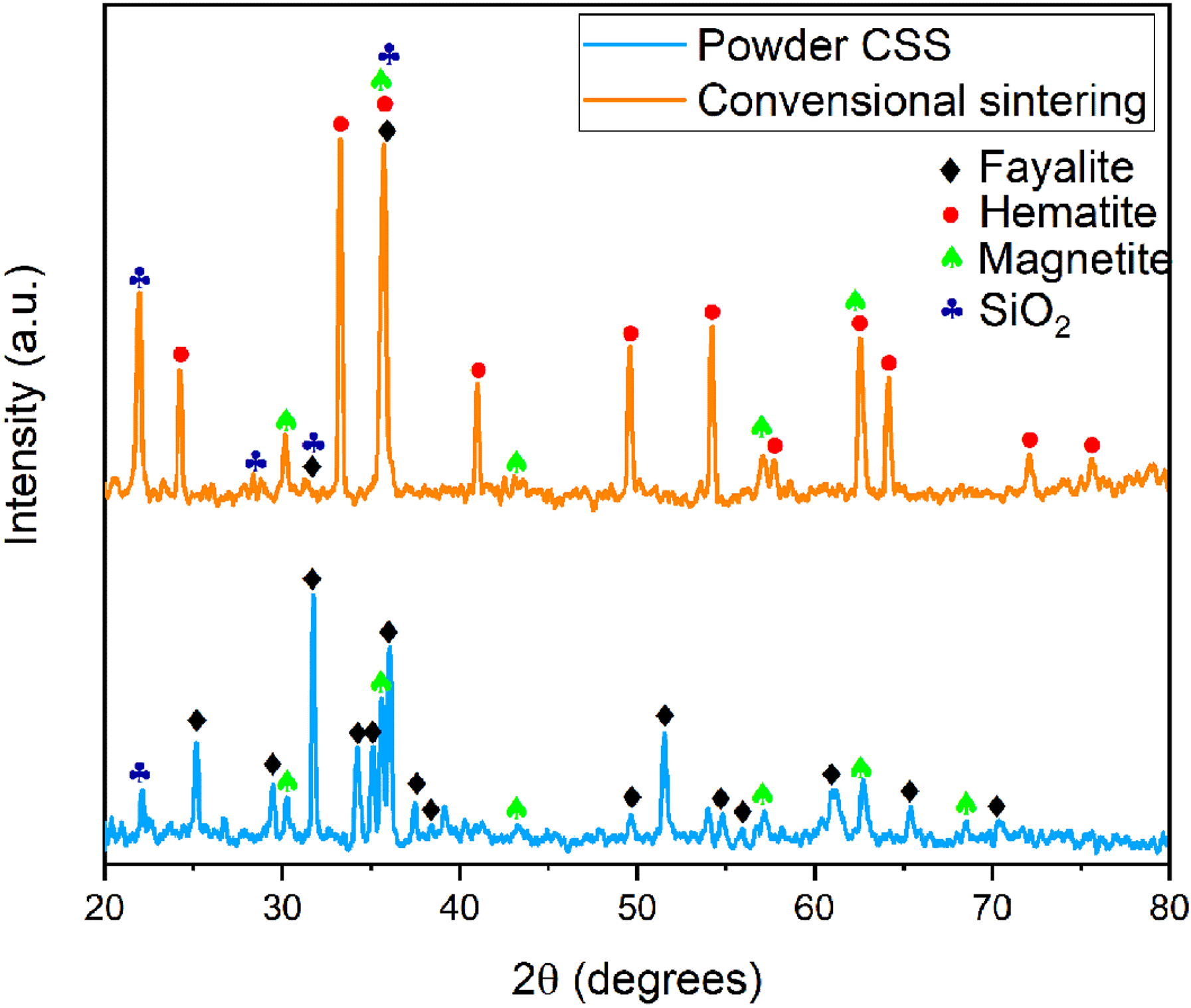
XRD graph of powder and conventionally sintered CSS.
Scanning electron microscope images of powder and conventionally sintered CSS are shown in Figure 3(a) and 3(b). It is seen that CSS powders have angular shapes and bimodal particle size distributions before sintering (Figure 3(a)). After conventional sintering, two different structures were observed due to the different melting temperatures of each phase in the CSS material system. The phases with lower melting points transformed into liquid phases while the phases with higher melting temperatures kept their less dense form after the sintering process at 1250°C; therefore, the grain boundaries have largely disappeared and grains have begun to appear as a whole. 15 Nevertheless, there are still areas where no sintering has occurred and structural voids exist (Figure 3(b)). Even though the sintering was done at 1250°C for 4 h, it is understood from Figure 3(b) that the sintering temperature and/or holding time are not sufficient for some phases in this system. Overall, the conventional sintering method is not an efficient way to acquire uniform and highly dense bulk CSS due to the different melting temperatures of each phase.

SEM images of (a) powder CSS, (b) conventionally sintered CSS.
There are three stages in FS: the incubation stage, the transient state, and the steady state. The incubation stage is stated when current leakage is detected. The current flow is increased in monolithic increments. In the transient state, the material's conductivity suddenly increases, which leads to an abrupt increase in current density and power dissipation throughout the samples. In the steady state, the applied electric field and current cut values are stabilised at certain numbers.15–17
Figure 4 shows how the electric field, current density, and power density varied over time during FS under various electric fields. Figure 4(a) shows the graph of the sample with an applied electric field of 12.5 V/mm. In the experiments with an electric field of 12.5 V/mm, no current flowed through the pellet up to 600°C with a 20°C/min heating rate. In other terms, the sample behaved as an insulator between 25°C and 600°C owing to the absence of particle–particle interactions in the green dense sample and limited electrical conductivity (incubation stage). When the furnace temperature reaches the temperature of 600°C, a current of 0.001 mA/mm2 begins to flow through the sample. As the furnace temperature continues to increase and reaches 818°C, the current value increases slowly up to 0.88 mA/mm2. After this point, the current flow increases rapidly, and when the furnace temperature reaches 850°C, the current passing through the sample reaches the cut-off value of 3.775 mA/mm2. At this point, the sample absorbed a maximum power density of 38.42 mW/mm3. After the first current leakage, reaching the highest current cut-off value took about 25 min which is very high compared to other experiments with a relatively higher electric field. So, 12.5 V/mm electric field is not enough to trigger FS, so this experiment is classified in FAST sintering processes.18–21 After the system experienced the current cut-off value, the power source automatically decreased the electric field down to 7.65 V/mm. The current density is fixed at 3.78 mA/mm2 to avoid excessive joule heating.
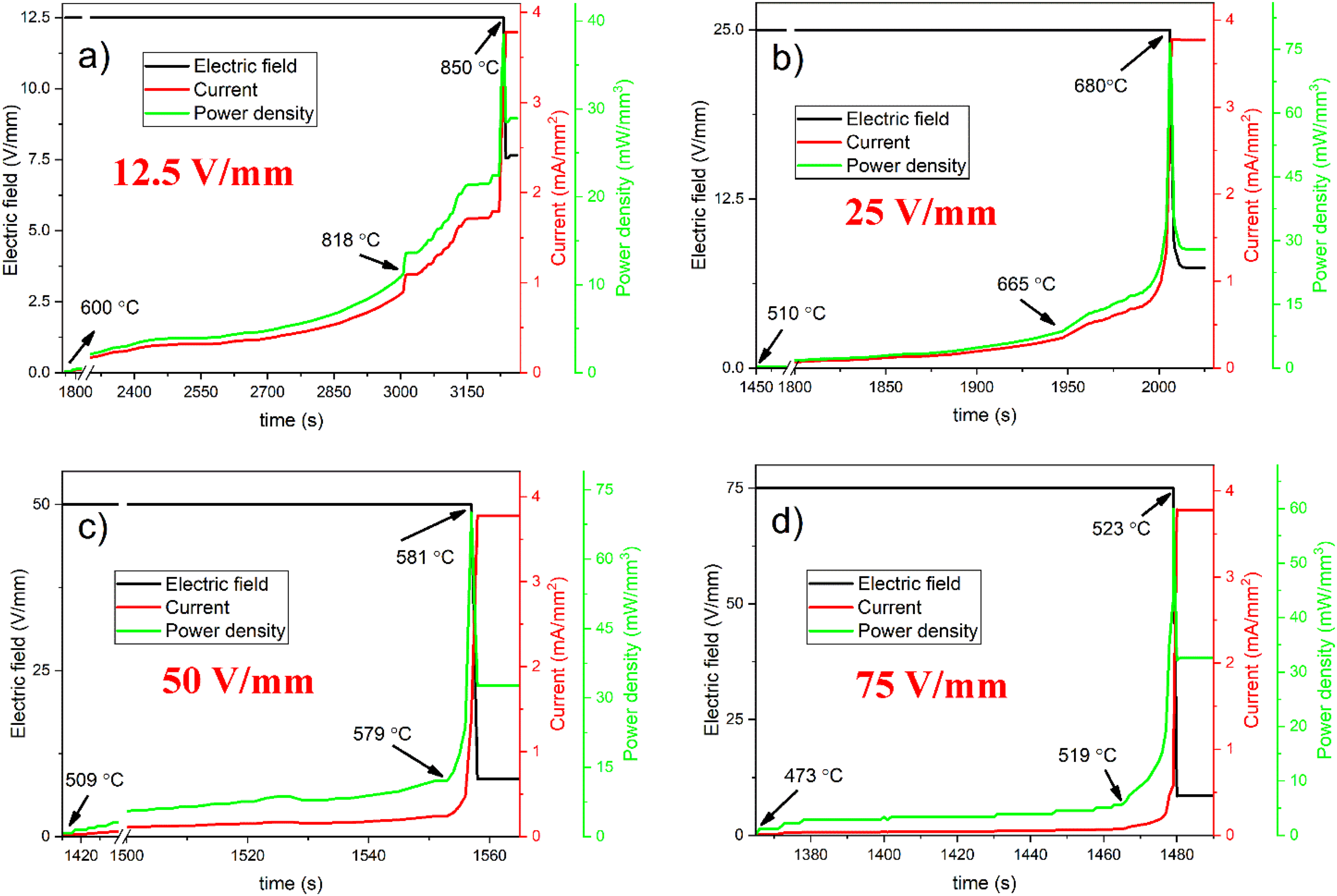
Graphs of current density and power density over time of flash sintered CSS at (a) 12.5 V/mm, (b) 25 V/mm, (c) 50 V/mm, (d) 75 V/mm.
As can be seen in Figure 4(b), 4(c) and 4(d), a similar trend is observed in FS at 25, 50 and 75 V/mm electric field values. In these experiments, the furnace temperatures at which the current starts to pass through the samples are 510°C, 509°C, and 473°C, respectively. In other words, the pellets exhibited insulating properties from 25°C to these temperatures. Increasing the value of the applied electric field leads to a decrease in the current leakage temperature. 20 The reason for this phenomenon is the same as the sample in which the electric field of 25 V/mm is applied. For these experiments, the furnace temperatures at which the transient state begins to noticeably increase are 665°C, 579°C and 519°C, respectively. And the furnace temperatures at which the current values passing through the pellets reach the cut-off value are 680°C, 581°C, and 523°C, respectively. The same trend is also available for the current cut-off temperature which is decreased by applying higher electric fields.20–22
Increasing the electric field value leads to a singularity for the incubation time period. Applying a higher voltage to the sample decreases the incubation time by one order of magnitude. 20 For electric fields of 12.5, 25, 50, and 75 V/mm, the times elapsed between the first current passing through the sample and the time the current reaches the cut-off value are 1500, 554, 150, and 114 s, respectively.
Many researchers state that FS is mainly governed by joule heating, which refers to the instantaneous heating of the sample by creating a thermal effect of the current passing through it, thus accelerating the movement of the ceramic particles and contributing to the sintering.9,13,15 Based on this mechanism, it is possible to establish the following equation (equation (1)) between ceramic sintering rate and temperature (presuming temperature is the only variable in FS).13,23
In FS experiments, there are differences between the furnace temperature and the sample temperature. The sample temperature is computed using the black body radiation model (equation (1)) based on the furnace temperature that was measured during the experiments13,23,24: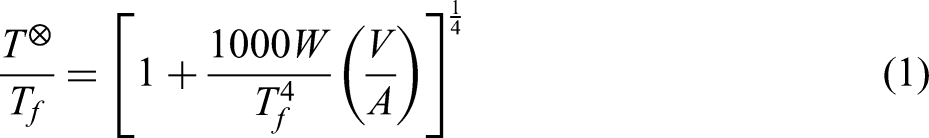
The joule heating on bulk samples is stabilised in the 10% range compared with the furnace temperature.8,10 Rather than heating phenomena on the bulk sample, joule heating on localised particle–particle contact could be much higher than calculated. 25
Figure 5 shows the relationship between the furnace temperature and the power density for the samples with various electric fields observed during the FS experiment. The maximum power density of about 76.49 mW/mm3 is reached under electric fields of 25 V/mm which suggests that there could be an optimum FS temperature and electric field combination to acquire a highly dense sample. If a higher electric field is applied to the sample, the flash point reduces accordingly, but the diffusivity of the sample at a lower temperature is not enough to initiate proper mass transport. If lower electric fields are applied to the sample, the flash point is increased; however, the electron flow and its momentum are insufficient for proper mass transport even though the increased diffusivity coefficient of the sample is at higher temperatures.18,24,25
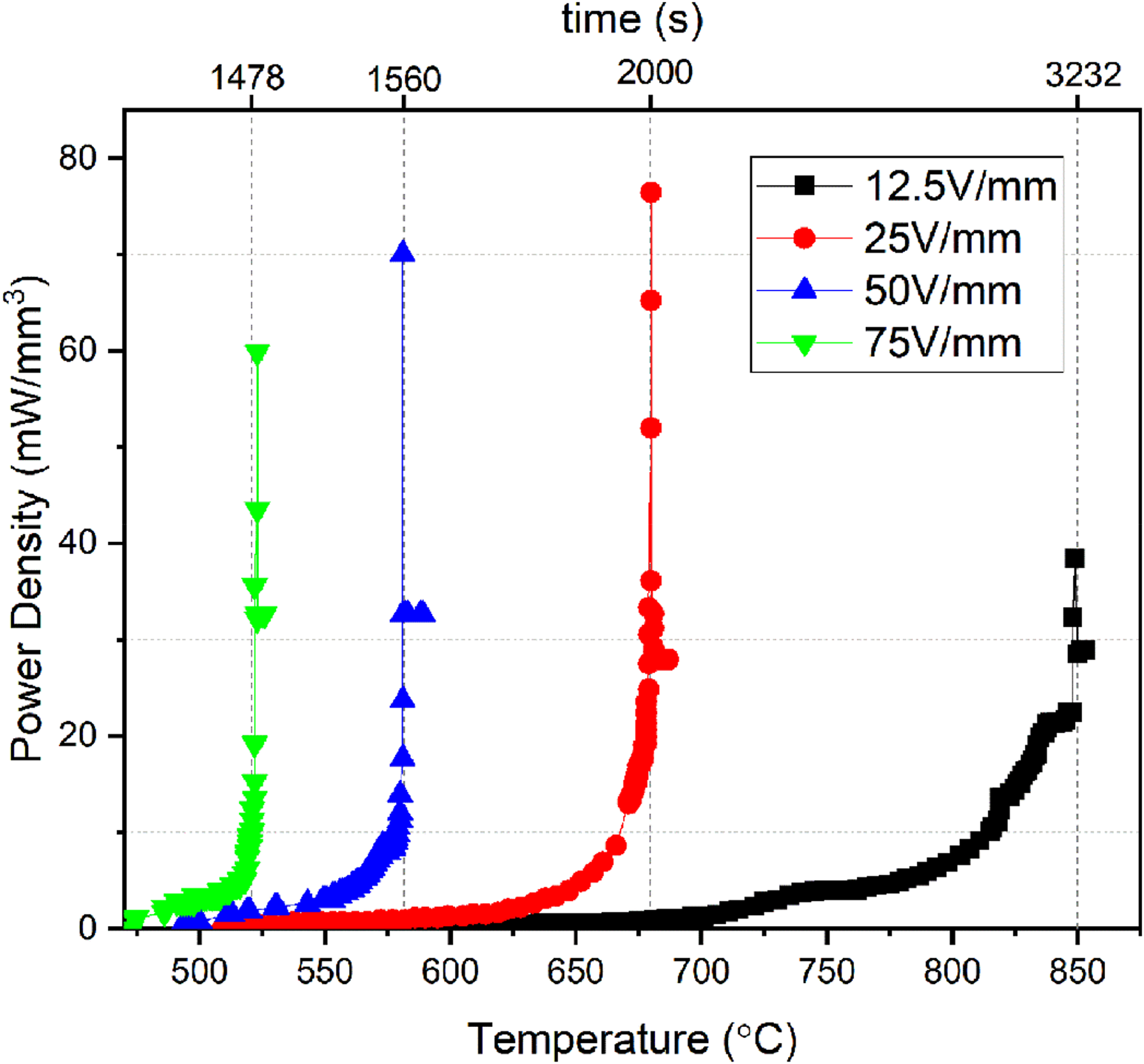
Power density of flash sintered CSS samples as a function of furnace temperature and time.
The results of XRD analyses of flash-sintered pellets are given in Figure 6. It is observed that hematite, magnetite, fayalite, and SiO2 phases are formed after FS by applying different electric fields. Applying 12.5 and 25 V/mm electric fields to samples leads to FS at higher temperatures. Consequently, the transition from magnetite to hematite and/or from fayalite to hematite is observed commonly according to the XRD data. Therefore, higher intensity on hematite reflection is observed for 12.5 and 25 V/mm conditions. However, applying a higher electric field reduces the flash temperature.20,22 Therefore, the phase transition to hematite phases is stabilized for these conditions. It is safe to say that FS at lower temperatures leads to mitigation of phase transition in the CSS materials system.
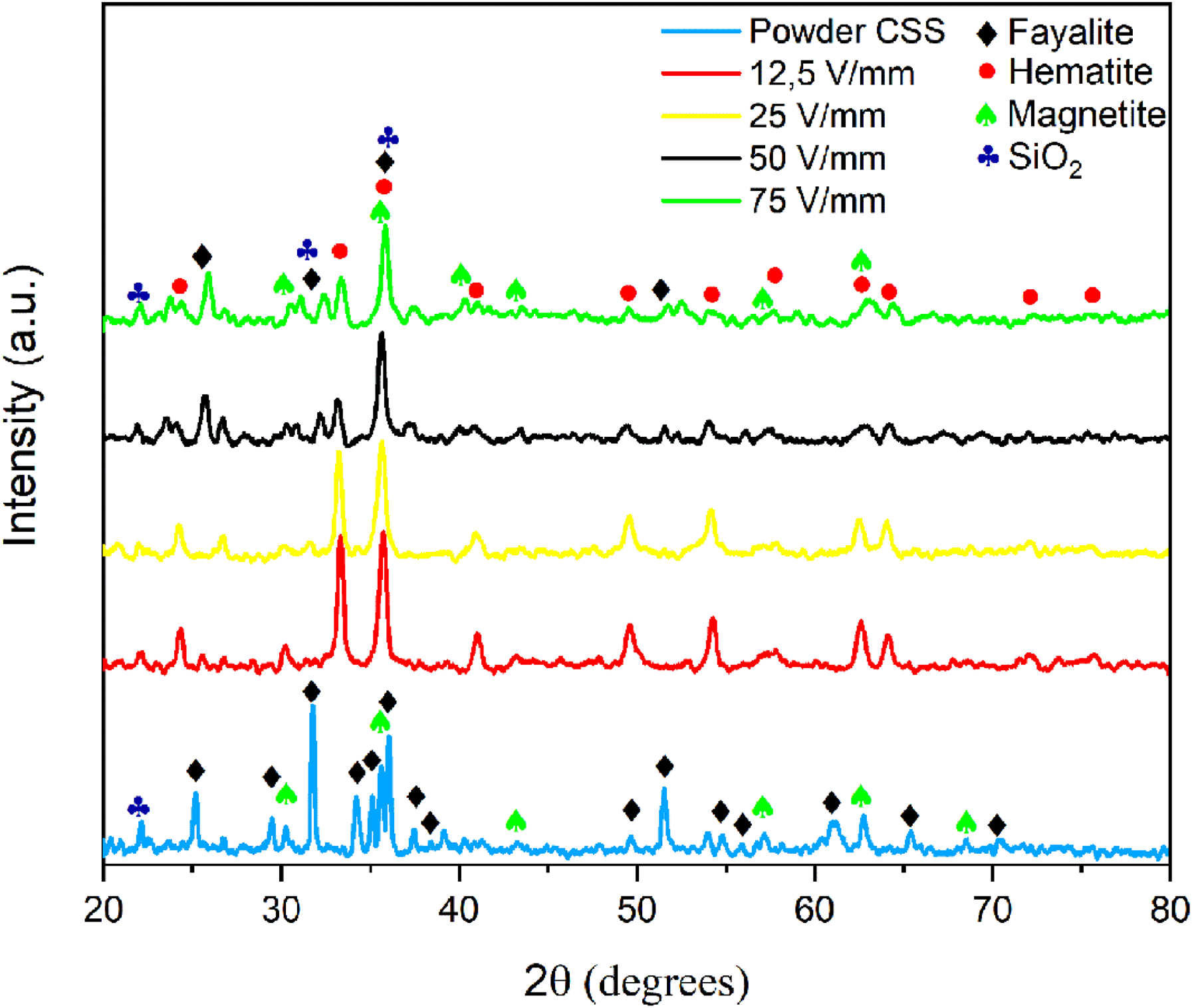
XRD graph of flash-sintered CSS and powder CSS.
Figure 7 shows the SEM images of the flash sintered samples with different applied electric fields. The SEM images reveal that all samples were effectively sintered by FS. However, the sintering of the 25 V/mm applied sample, in which the grains are not fully combined with good integrity and close pore surface structure suggests higher density values. A closer look at the 12.5 V/mm condition shows that the microstructure exhibits inhomogeneous grain boundary formation, with liquid phases covering unsintered or lower-density phases. The same formation on the microstructure is also observed in conventionally sintered samples, too. Overall, FS at 25 V/mm electric field reveals the best microstructure along with good interface integrity for all phases in the CSS sample, which is not achievable with conventional sintering methods. So, FS enables the mass transport mechanism for all phases in the CSS materials system at the same time by injecting massive amounts of electrons into the system in a very short time period. The excessive electron presence around atoms leads to an increment in both interatomic distance and diffusion coefficient concomitantly. So, all phases in this multiphase material system can be forced for mass transport at the same time thanks to the FS method.25,26 For conventional sintering, all phases have different diffusion coefficients with respect to different melting temperatures. So, it is hard to convey a homogenous density distribution throughout the sample.27,28 Overall, FS makes it possible to obtain a homogenous and dense microstructure for a multiphase material system.8–10

SEM images of the flash sintered samples at (a) 12.5 V/mm, (b) 25 V/mm, (c) 50 V/mm and (d) 75 V/mm.
The densities and hardness values of flash sintered samples are consistent with the microstructure examination, as represented in Figure 8. Density values exhibit a tendency that is similar to that of hardness. The lowest hardness and density are seen in the conventional sintered sample. Under the electric field of 25 V/mm, where the hardness and density are highest, sintering is most successful.
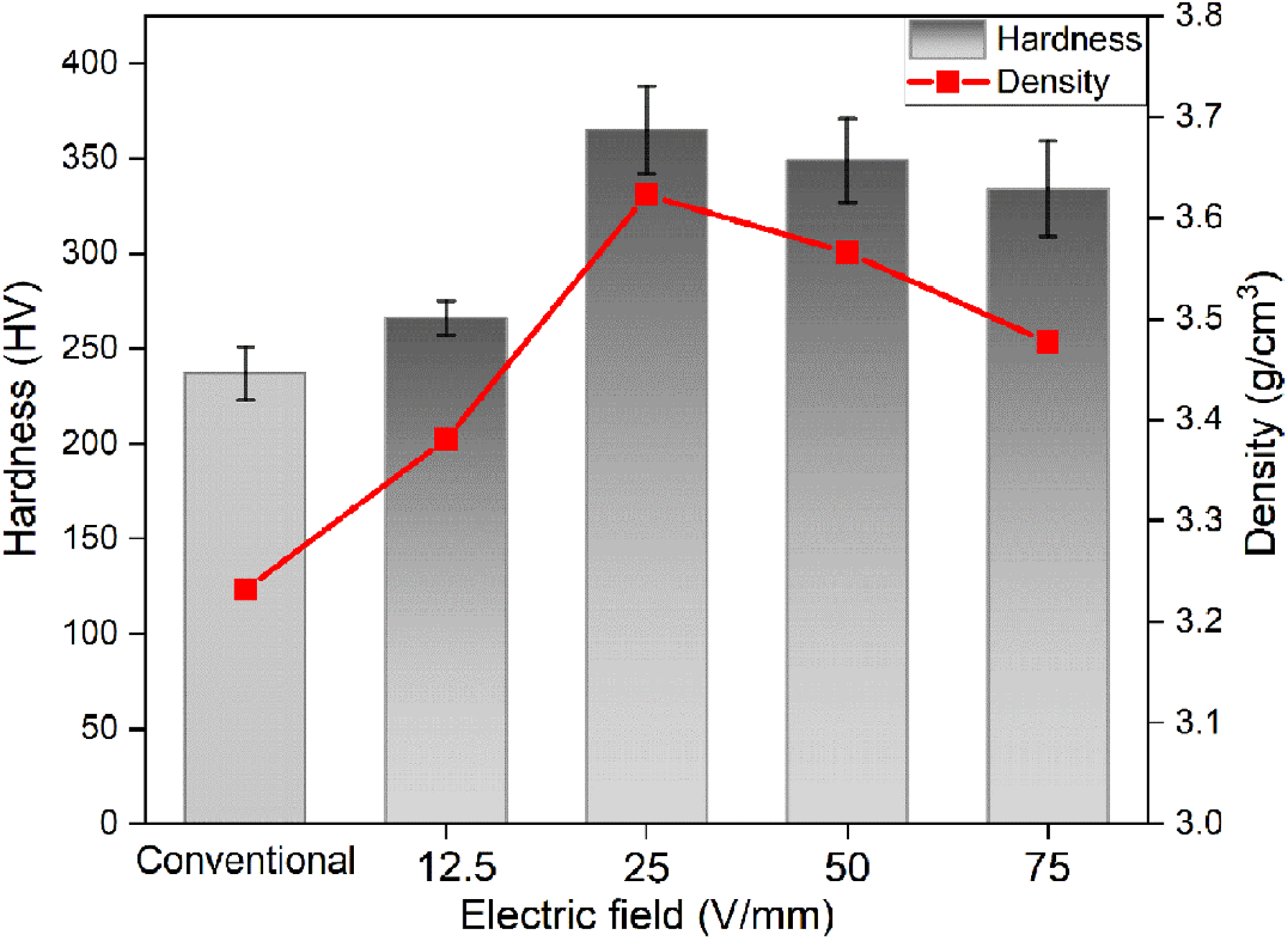
Hardness and density values of sintered samples.
Figure 9 shows the image of the EDS surface analysis. In the analysis, the coloured regions show the existence of the element of that colour. Energy-dispersive X-ray spectroscopy analysis suggests a homogenous distribution of each phase throughout CSS, which is evidence for a good experimental procedure. It is seen that the results of the SEM-EDS analysis are compatible with the XRF results of the analysis. The quantities of the elements determined by EDS analysis are shown in Table 2.

The image of the EDS surface analysis.
The elemental distribution of (in wt. %).

Conclusion
In this study, a comparative study is presented between conventional and FS for CSS multiphase materials systems. The following outcomes are acquired in this work:
Conventional sintering at 1250°C for 4 h reveals lower density values compared to flash sintered samples at various electric fields (12.5, 25, 50 and 75 V/mm) Flash sintering makes it possible to reduce the sintering temperature down to 523°C for 75 V/mm condition and the sintering time to 114 s. It is observed that FS at 25 V/mm electric field was an optimum condition for better densification and microstructure integrity in a multiphase system. In this condition, materials show the highest density and hardness values. Flash sintering also helps to reduce magnetite-hematite phase transition, which happens at higher temperatures. Overall, FS has made a huge contribution to reducing sintering time and temperature which could be considered as a valid green processing method.
Footnotes
Acknowledgements
The author wishes to thank Dr İlyas Şavklıyıldız and Dr Zeynep Çetinkaya for their valuable technical feedback and for providing raw materials and flash sintering setup.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
