Abstract
Three types of alumina powders and a polymer electrolyte Isobam system were used to fabricate translucent alumina ceramics via gelcasting, and each powder had the highest attainable solid loading. The effects of alumina particle size on slurry dispersion, microstructures and properties of green bodies and ceramics were investigated. All of the high-solid loading slurries contained strong aggregates, which could bridge with each other to form weak agglomerates. The smaller the initial particle size was, the more the contained particle number per aggregate. In addition, the smaller the aggregate size was, the lower the sintering activation energy of the ceramic and, finally, the higher the relative density. The initial particle size of the powder may not be the critical factor determining ceramic sintering, but the aggregate size and effective volume fraction of the aggregates may be the critical factors. Finally, the in-line transmittance of the ceramic with a higher density reached 22% at a wavelength of 650 nm and a thickness of 0.8 mm.
Introduction
Translucent alumina has been widely applied as an arc tube for high-pressure sodium lamps and ceramic metal halide lamps, benefiting from its satisfactory optical performance and excellent chemical durability. 1 Many processes have been developed to fabricate translucent alumina. Typically, gelcasting exhibits distinct advantages in the fabrication of green bodies with complicated shapes and composite ceramics.2–4 The Isobam system, as a new additive agent, is often used as both a dispersant and gelling agent in gelcasting, and it is a copolymer of isobutylene and maleic anhydride, has no toxicity and enables performance under aqueous and room-temperature casting conditions.5–10 Due to the low additive amounts of Isobam, slurries can achieve high density after densification for structural ceramics11,12 and transparent ceramics, such as Al2O3, YAG, Y2O3, MgAl2O4 and AlON.13–17
The pursuit of a slurry with high-solid loading and low viscosity is significant for fabricating high-performance ceramics by gelcasting. A gel is usually recognised as a wet cake with a three-dimensional network structure by which the well-dispersed slurry with independent powder particles is solidified in situ via the reaction of the polymer additive. 18 However, the limited volume capacity makes particles inclined to form aggregates containing more particles under high-solid loading conditions. Thus, a high-solid loading slurry frequently consists of dispersed particles, aggregates and agglomerates.19,20 Both aggregates and agglomerates are assembled by powder particles. Strong and dense particle assemblies are referred to as aggregates, which are formed in the primary minimum and are nonfriable, gritty, and not readily disperse, even after considerable shear forces are applied; whereas agglomerates are formed from the loose bridging of aggregates, which are friable and readily dispersed into primary particles or aggregates. 20 Aggregation is the process of formation of clusters of particles via gathering small particles by forming strong chemical bonds between the particles. Agglomeration is the process of formation of clusters of particles via gathering small particles by forming weak physical interactions with each other. It has been reported that an increase in the solid volume fraction in a slurry can cause particle aggregate, and the sizes and shapes of aggregates have a large impact on the packing density of green bodies and further sintering deformation of ceramics.21,22 However, the influence of initial particle sizes on aggregates, particularly in high-solid loading slurries dispersed by the Isobam system for ceramic fabrication, has scarcely been reported.
The formation of strong aggregates is probably due to the insufficient distribution of polymer dispersant molecules due to high-solid loading. The molecules of the dispersant do not completely cover all particles, and aggregate takes place. 23 On the other hand, agglomerate has been attributed to the direct mutual attraction between nanoparticles via van der Waals forces, occurring when van der Waals forces are stronger than electrostatic and steric forces; the above forces dominate the interaction between particles and interparticle distance, which is related to the electric double-layer and adlayer thickness. 24 Yamamoto et al. 25 used colloidal probe atomic force microscopy to directly measure such interactions between alumina particle surfaces in an aqueous solution containing a copolymer of Isobam (Isobam104, MW = 55,000–65,000 g·mol−1). Both repulsive forces and attractive forces existed between dispersed alumina particles at first, and the repulsive force disappeared, with only long-range attractive forces remaining as the aging time increased beyond 3 h. This may be the reason why Isobam104 has both dispersive and gelling abilities, resulting in weak agglomerate. In addition, Di et al. 21 used the dispersant agent PAANH4 (A-30SL, MW = 6000 g·mol−1) to prepare an alumina slurry and found that the particles also aggregate at a high-solid loading of 56–60 vol.-%. Aggregate and agglomerate may be inevitable in slurries with high-solid loading, and thus, their influences on the sintering and densification of ceramics cannot be ignored. However, few studies have focused on the causes of agglomeration and the effect of the microscopic characteristics of ceramics.
In our previous study, aggregates from alumina slurries were diluted and separated, the morphologies of aggregates were shown to be related to the types of dispersants, and aggregate affected the particle packing density and structural uniformity of green bodies and the sintering properties of ceramics. 21 Michálková et al. 23 reported that larger aggregates can decrease the relative density and induce a higher average grain size when increasing the solid loading to a higher magnitude. Although the effect of aggregate in colloid formation on sintering has been investigated, the influences of initial particle sizes on aggregate formation should be considered during slurry fabrication and subsequent ceramic sintering.
The objective of this study is to elucidate the influence of aggregate and agglomerate on the densification and transparency of alumina ceramics obtained by gelcasting. The effects of the initial powder particle sizes on the properties of slurries with high-solid loadings and consolidated compacts are explored. The microstructures of both green bodies and sintered ceramics, as well as densification and grain size, are deeply investigated. Finally, the optical transmittance of the sintered ceramics is examined to study the impacts of the initial powder features.
Experiments
Materials and fabrication
The high-purity alumina powders CR10, CR6 (4 N purity, Baikowski, Annecy, France) and SMA6 (4 N purity, Baikowski, Japan) with Brunauer, Emmett and Teller (BET) surface area of 7, 6 and 5 m2·g−1, and with transmission electron microscopy-derived ultimate particle sizes of approximately 350, 400 and 700 nm, respectively, were employed as initial powder particles. Copolymers of isobutylene and maleic anhydride with different molecular weights (5500–5600 and 55,000–65,000, Isobam600AF and Isobam104, Kuraray, Osaka, Japan) were used as dispersants and gelling agents. Slurries with solid loadings of 44 vol.-%, 45 vol.-% and 55 vol.-% were prepared by adding 0.2 wt-% Isobam 600AF as a dispersant and 0.1 wt-% Isobam 104 as a gelling agent relative to the weight of alumina powder. 11 MgO with a content of 600 ppm (4 N purity, Sinopharm Chemical Reagent Co., Ltd, China) was used as a sintering aid. 26
Deionised water, dispersants, the sintering aid and alumina powder were first mixed by ball milling at a speed of 300 r/min for 4 h to obtain high-solid loading slurries, and then, the slurries were degassed before casting into a plastic mould (Φ29 mm × 8 mm). After gelling for 3 days, they were dried in an environment at 25 °C with 85% relative humidity for 3 days and further heated at 120 °C for 8 h to obtain dried green bodies. Subsequent sintering was carried out in a vacuum furnace at a heating rate of 2 °C·min−1 and held at 1850 °C for 6 h.
Characterisation
Rheological behaviour was characterised using a stress-controlled rheometer (Pear MCR302, Anton, Graz, Australia) with a parallel plate (25 mm in diameter). The viscosities of the slurries were measured with the continuous shear mode increasing from 1 s−1 to 500 s−1 at 25 °C. After ball milling, to explore the aggregate/agglomerate of alumina particles in the as-prepared slurries during gelling, 1 g of slurry was taken and added to 2500 ml of deionised water to form diluted suspensions, and the diluted suspension was ultrasonically vibrated by ultrasound at 40 kHz to separate the loosely assembled particles at different gelling times (0–1440 min). Then, the particle size was characterised using a laser dynamic scatterometer (Zetasizer Nano ZS ZEN 3600, Malvern, UK).
The linear shrinkage after drying/sintering was estimated by the ratio of the size of wet cakes/green bodies to that of green bodies/ceramics, and the final value was averaged from three samples. The pore size distribution of the green bodies was determined by a mercury intrusion porosimeter (AutoPore IV 9500, Micromeritics, USA). The microstructures of the specimens were observed using scanning electron microscopy (SEM, JSM-5600LV, JEOL, Japan). The bulk densities of alumina ceramics were measured in deionised water at room temperature after boiling for 3 h according to Archimedes’ principle.
The sintered ceramics were polished to a final cylindrical thickness of 0.8 mm. An ultraviolet‒visible spectrometer (TU1810, Purkinje General, China) was employed to record the in-line transmittance in the wavelength range from 200 to 1100 nm. The as-prepared ceramics were thermally etched at 1700 °C for 2 h in a vacuum, the grain size was measured using a metallographic microscope (10XB-PC, SHOIF, China), and at least 1500 gains were counted for each sample.
Results and discussion
Formation of aggregates and agglomerates
The key to high-density green bodies and high-performance ceramics by gelcasting is to provide a slurry with as high solids loading as possible. Sun et al. 27 prepared a slurry with 55 vol.-% solids loading using SMA6 powder by the addition of 0.1 wt-% Iosbam 104 and 0.2 wt-% Isobam 600AF, and a relatively low viscosity of the slurry about 1 Pa·s at 100 s−1 shear rate was obtained. Similarly, 45 vol.-% solids loading was obtained by using the CR10 powder. 27 Using the same method, the solid loading of the slurries prepared by CR10, CR6 and SMA6 are the highest with 44 vol.-%, 45 vol.-% and 55 vol.-%, respectively. If the solid loading is higher, the slurries will undergo coagulation, form wet pastes and lose fluidity.
Figure 1(a) displays the rheological characteristics of slurries, each of the slurries exhibits pseudoplastic behaviour at a low shear rate and shear thinning behaviour. Figure 1(b) shows the rheological characteristics of the slurries prepared by CR10, CR6 and SMA6 at a shear rate of 100 s−1, which are 54 Pa·s, 24 Pa·s and 75 Pa·s, respectively. The viscosity is higher than the previous work, 27 which may be relative to the lower humidity of the open environment.
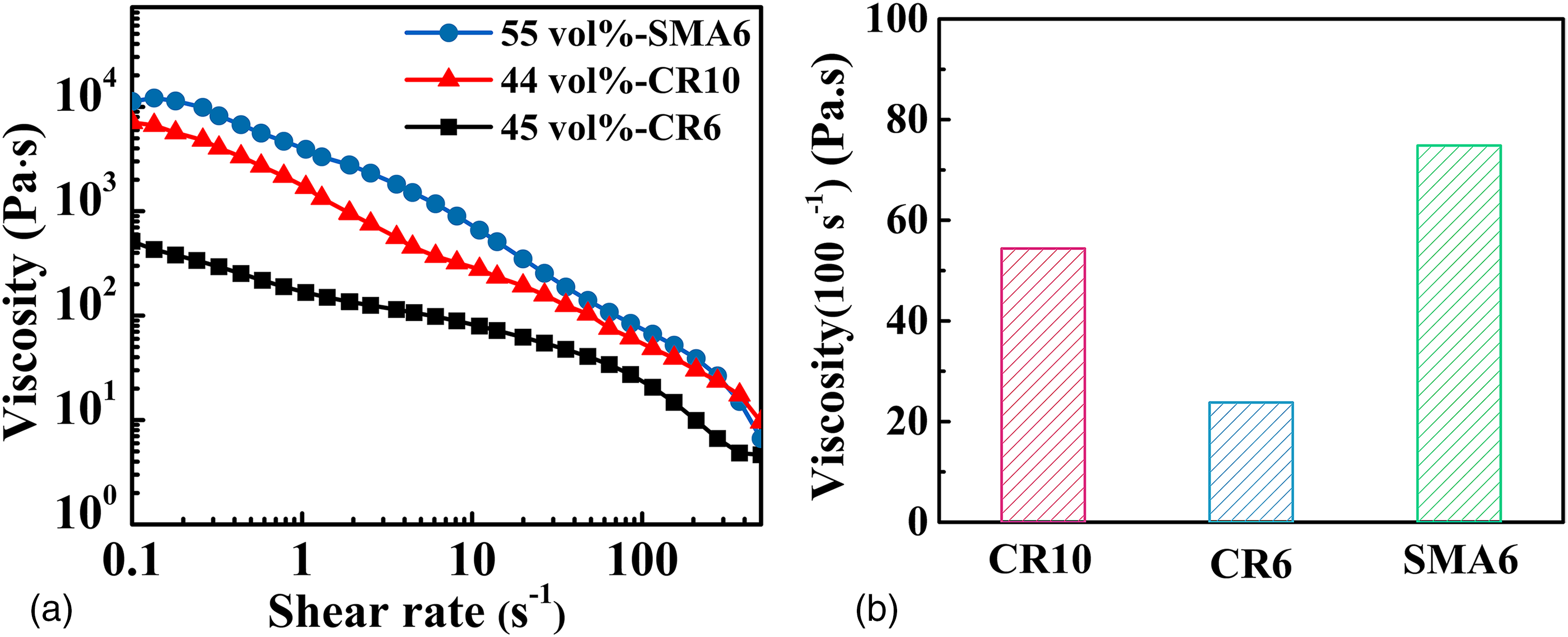
(a) Viscosities of the slurries prepared by CR10, CR6 and SMA6. (b) Viscosities of slurries prepared by CR10, CR6 and SMA6 at a shear rate 100 s−1.
To scrutinise the evolution of the dispersing condition of particles in the slurry, the particle sizes of the diluted suspensions were measured during gelling at different times. Figure 2 shows the size distribution of the particles in slurries. After ball milling (gelling for 0 min), as shown in Figure 2(b), the particle size (d50) achieved with CR6 powder is the same as the initial particle size, 406 nm, indicating that the initial powder particles were adequately dispersed during ball milling. When the gelling time is 30 min, the particle size reaches 722 nm, which is twice the size of the initial particle. This indicates that the particles tend to combine to form strong aggregates during gelling, and the coagulated aggregate cannot be broken by ultrasonication. When gelling for 24 h, the particle size is comparable to that obtained by gelling for 30 min, implying that the aggregate size is stable enough and will not increase with time. Moreover, when the ultrasonic time was <30 min, that is, 20 min, there was a second peak distribution at the size of 5.56 μm for the slurries prepared with CR10 and CR6, as shown in Figure 3. There is a much larger agglomerate in the slurry, which is often formed by aggregate bridging and can be separated by ultrasonic vibration. Obviously, the particles in the slurries contain aggregates and agglomerates, and both play a large part in the solidification of the slurry.
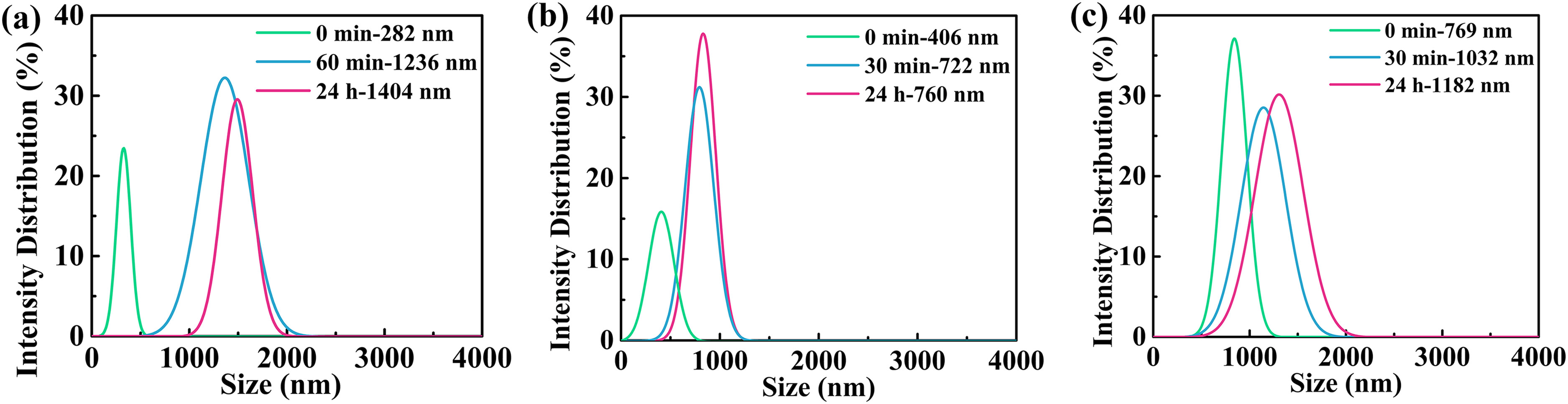
Particle size distribution of aggregates in diluted suspensions formed by CR10 (a), CR6 (b) and SMA6 (c) with ultrasonic vibration for 30 min.
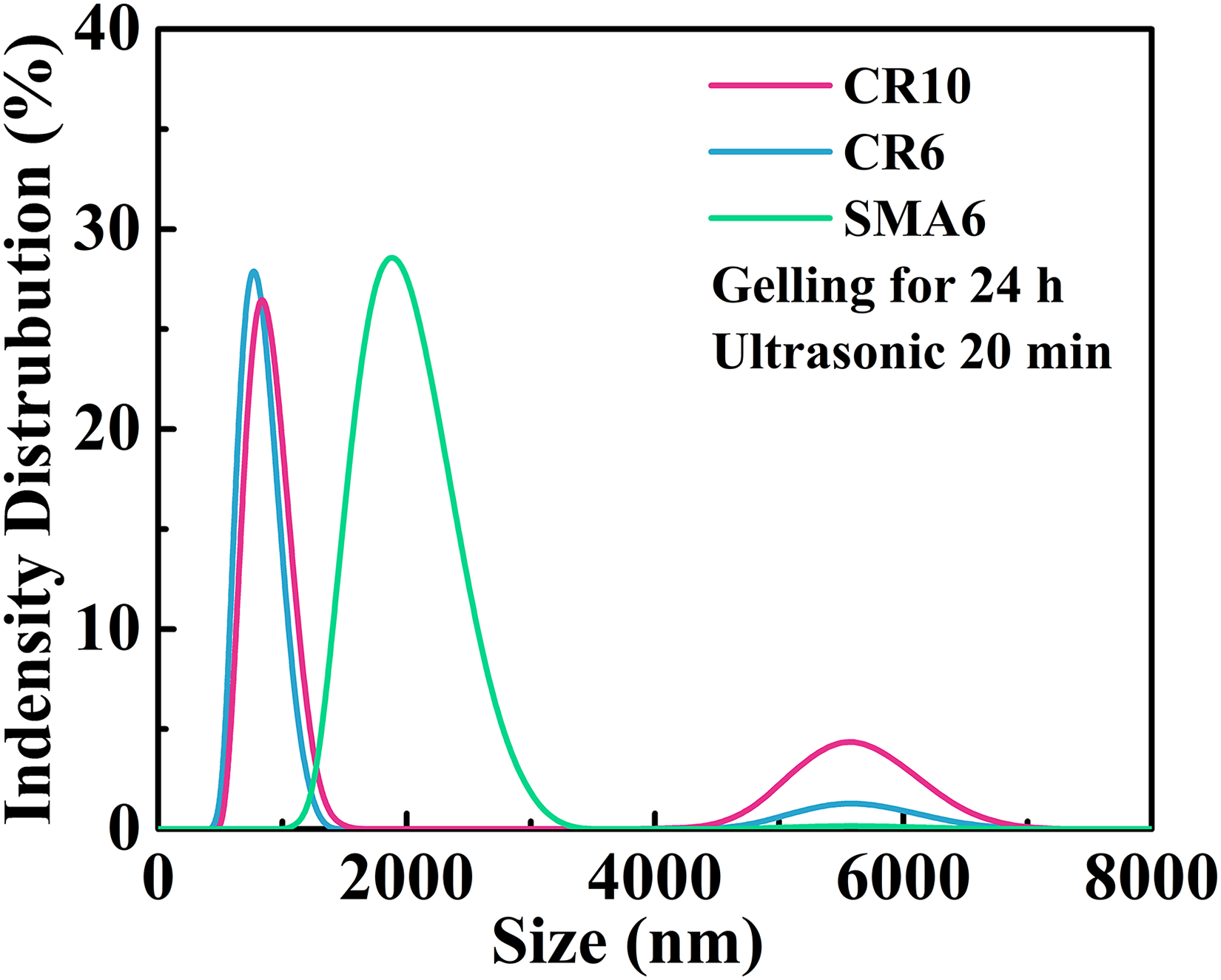
Particle size distribution of aggregates in diluted suspensions prepared with ultrasonic vibration for 20 min.
The particle size achieved with CR10 was 282 nm after ball milling, reached 1.24 μm after gelling for 60 min and reached 1.40 μm after 24 h (Figure 2(a)), being almost five times larger than the initial powder size; the particle size achieved with SMA6 was 769 nm after ball milling, reached 1.03 μm after gelling for 30 min, and reached 1.18 μm after 24 h (Figure 2(c)), being 1.5 times larger than the initial particle size. Similar to the slurry with CR6, both of the particle sizes in the slurries with CR10 and SMA6 increase with increasing gelling time. Notably, the smaller initial particle size of CR10 is involved with a larger aggregate size because of the higher surface energy.
From the above results, aggregates formed during the gelling of slurry, and they bridged each other to form agglomerates and then interconnected into a network, forming a gel. 28 Although the particles can be uniformly dispersed via appropriate dispersant addition and moderate ball milling or more efficient milling methods, that is, wet-jet milling, aggregates can still spontaneously form in the high-solid loading slurry without external mechanical disturbance, resulting from the deficient space and close particle‒particle distance.19,29 Likewise, LaSalle et al. 30 clearly observed agglomerates in submicron alumina slurries using low-temperature SEM (cryo-SEM). In addition, agglomerates were also observed in a 30 vol.-% solid-loading slurry with γ-Al2O3 (43 nm, PAANH4 as the dispersant) after drying at room temperature for 24 h. 31
The particles in the slurries were monodisperse according to Derjaguin–Landau–Verwey–Overbeek (DLVO) theory. To coagulate two approaching particles, they must have enough kinetic energy (due to Brownian motion) to overcome the energy barrier (kB· T > Umax) and fall in the primary minimum. Isobe et al.
29
found that the aggregate of particles depended on the particle surface spacing (SDP); while increasing the solid loading, the SDP gradually decreased and then remained constant at approximately 10 nm. van der Waals interactions prevail at very small interparticle distances (1–10 nm). To accommodate more particles, tightly packed aggregates formed in the slurries.
29
According to the Woodcock expression for the SDP (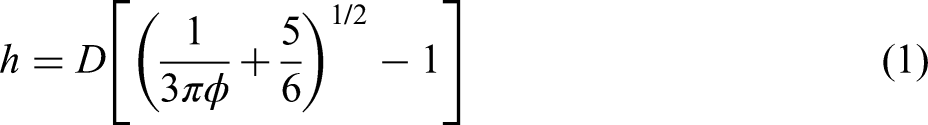
In detail, the adsorbed layer of the particles is dependent on steric hindrance from the adsorbed Isobam dispersant and electrostatic repulsive interaction between particles. The conformation of the Isobam dispersant on the surface of the Al2O3 particles, such as loop and tail segments, 32 plays an important role in the thickness of the adsorbed layer of the particles. Furthermore, charged long-chain polymers favour bridging to stimulate aggregate/agglomerate owing to the influence of charge induction and/or chain entanglement. 33 Weakly attractive interactions occur between aggregates with a shallow secondary minimum to form agglomerates. In fact, the interparticle force determines whether two colliding particles aggregate. Colloidal suspensions at equilibrium can be either dispersed or aggregated, depending on whether the particle interaction energy is repulsive or attractive, respectively. Then, SDPs are crucial in determining particle‒particle interactions, as well as the arrangement order and phase transitions.
The magnitude of aggregate in suspensions has a large impact on the properties of the solidified compacts. The effective volume fraction of aggregated particles in a suspension (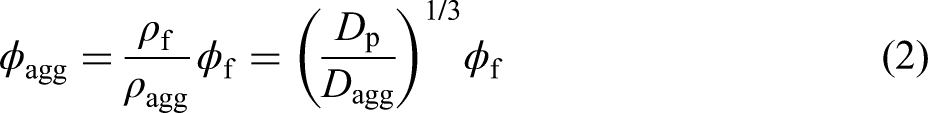
Related parameters of the particles and aggregates.

Moorehead et al. 35 investigated several degassing and deagglomerate approaches and found that adding a degassing dispersant combined with ball milling was effective in mitigating agglomerate. In this work, the polymer Isobam600AF, which has a better dispersion effect than Isobam104, 36 is used as a dispersant, but agglomerate still occurs in the slurries. In short, aggregate and agglomerate are inevitable in slurries with high-solid loadings, and their effects on the consolidation of the resultant compacts should be evaluated.
Pore volume and size in the green bodies
The slurries were cast into a mould and solidified for 3 days with sealing, and the obtained jelly-like wet cake was soft and translucent. Then, the wet cakes were dried in the air, and shrinkage occurred for the cake with water evaporation and particle rearrangement. The ultimate linear shrinkages of the wet cakes after drying in an 85% humid environment at 25 °C were determined. The linear shrinkages of the wet cakes prepared by CR10, CR6 and SMA6 were 4.08%, 3.54% and 3.56%, respectively. The larger shrinkage of the wet cake prepared with CR10 can be attributed to the finer particles having greater capillary force, which causes greater rearrangement in particle packing.
Figure 4 shows the microstructures of the particles in the green bodies. There are both monodispersed particles and aggregates in the green bodies, which corresponds to the particle size distribution in Figure 2. The number of particles per aggregate is 2–5 initial powder particles, and this value may originate from packing, as in a crystal lattice, to form the densest, closest-packed aggregates. The size of the aggregates may follow the fractal law of a certain shape. Thus, the reduction of the specific surface area of the particles is related to the geometric shape of the aggregate, and the size of the aggregates will not be so too large that its stability is lost. Therefore, the morphologies of the aggregates formed by the three powders look very similar.

Microstructure of the particles in green bodies obtained with CR10 (a), CR6 (b) and SMA6 (c).
Figure 5(a) shows the relative density of the obtained green bodies. The green body obtained by CR10 had a lower relative density (50.38%) than the green bodies obtained by CR10 (50.69%) and SMA6 (61.99%). Higher solid loading can achieve a higher relative density of green bodies, and the higher volume fraction of aggregates corresponds to a higher relative density of green bodies, that is, the one obtained with SMA6. Figure 5(b) presents the pore size distribution of the relative green bodies. The green bodies obtained with CR10 and CR6 have the same cumulative pore volume, 0.23 ml·g−1, which is consistent with the similar relative densities of their green bodies. However, the green body obtained with CR10 has a smaller average pore size (100 nm) than that obtained with CR6 (119 nm), which is related to the smaller SDP and lower effective volume fraction of aggregates in the slurry prepared by CR10. The green body obtained with SMA6 has the lowest cumulative pore volume (0.14 ml·g−1) and the smallest pore size (68 nm), which is also consistent with it having the highest relative density and the smallest SDP. This suggests that increasing the initial particle size may decrease the porosity and pore size and, to some extent, increase the relative density of the green body.
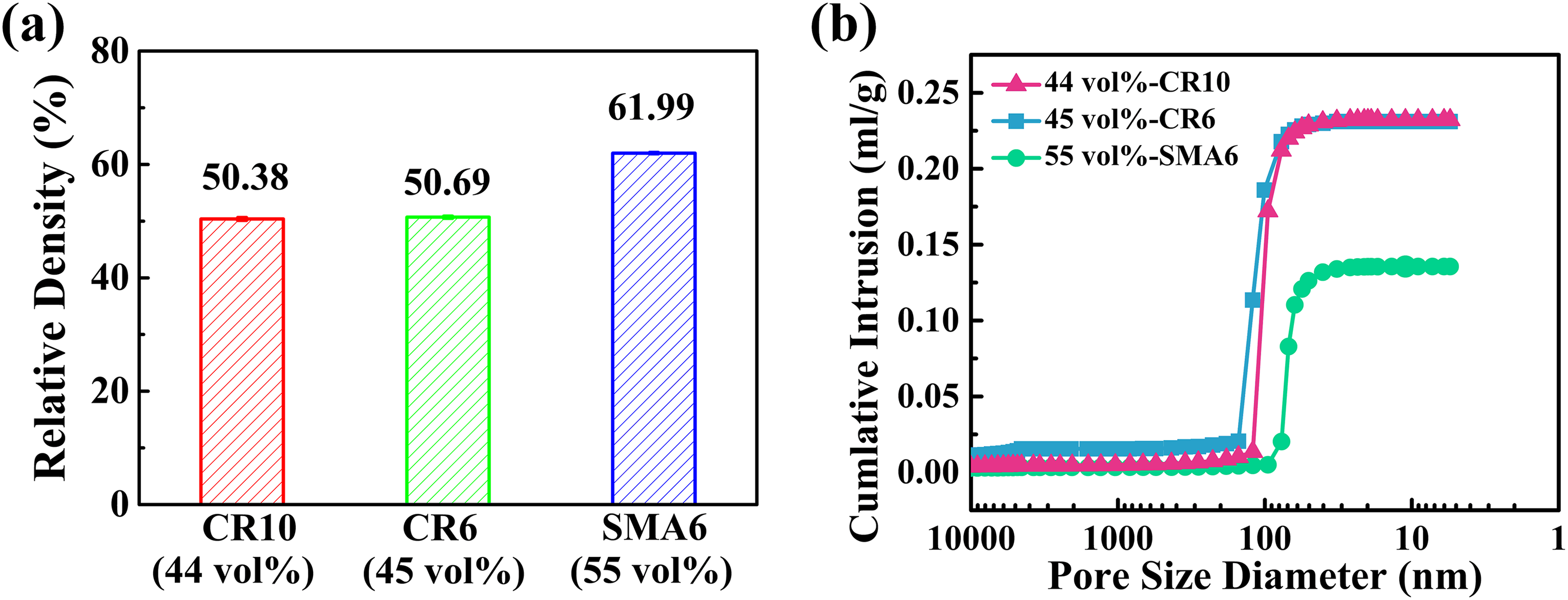
Relative densities (a) and pore size distributions (b) of green bodies obtained by CR10, CR6 and SMA6.
Mechanisms of ceramic sintering
The kinetics of the initial stage of sintering can be determined by the relationship between linear shrinkage and the diffusion mechanism. If the densification of the green bodies is controlled by lattice diffusion or grain boundary diffusion at the initial stage of sintering, the relationship between the linear shrinkage (Δ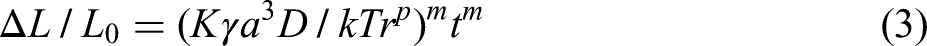
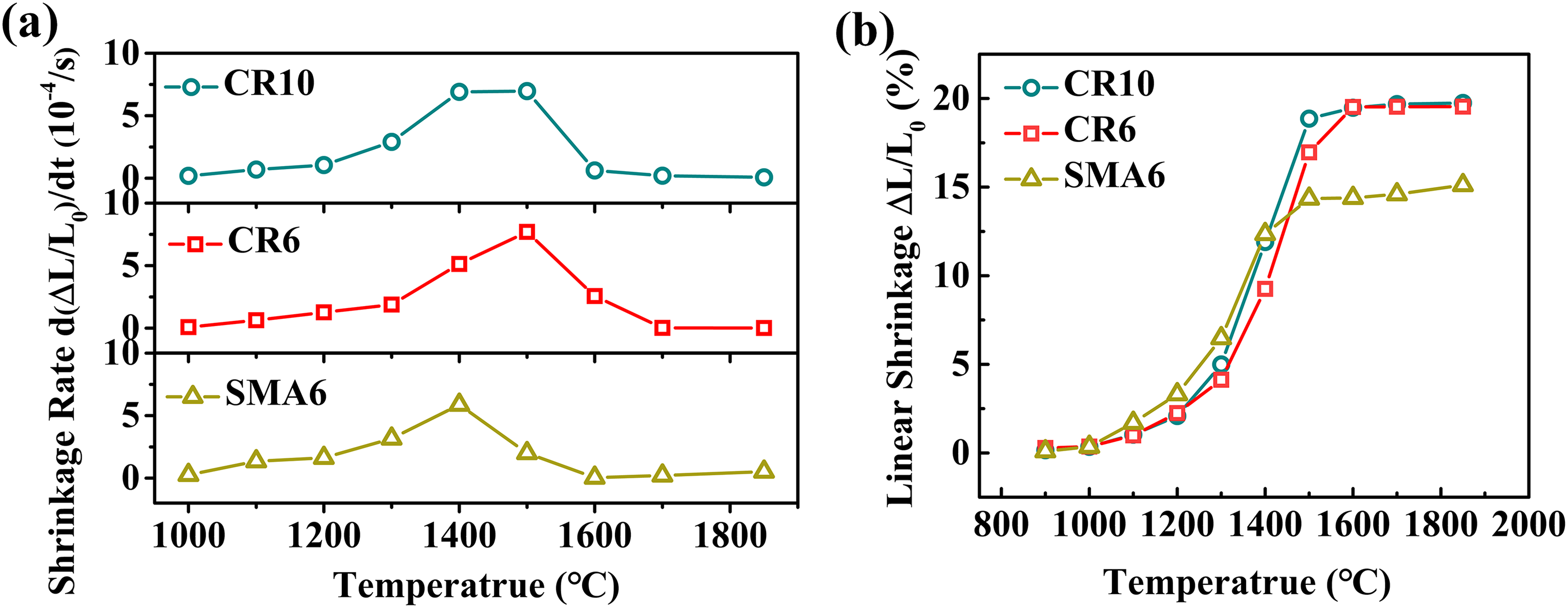
Linear shrinkage rate (a) and linear shrinkage (b) of the specimens as a function of temperature during constant-heating rate sintering.
According to Johson's sintering model,
38
with a constant-heating rate, when densification is controlled by grain boundary or volume diffusion, the relationship between the shrinkage of the green body and temperature can be expressed as
39
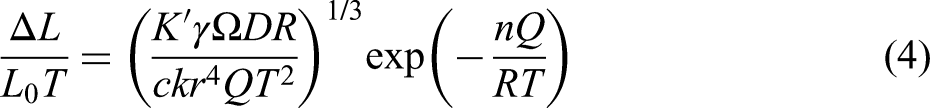
In formula (4), the influence of temperature with exp(−
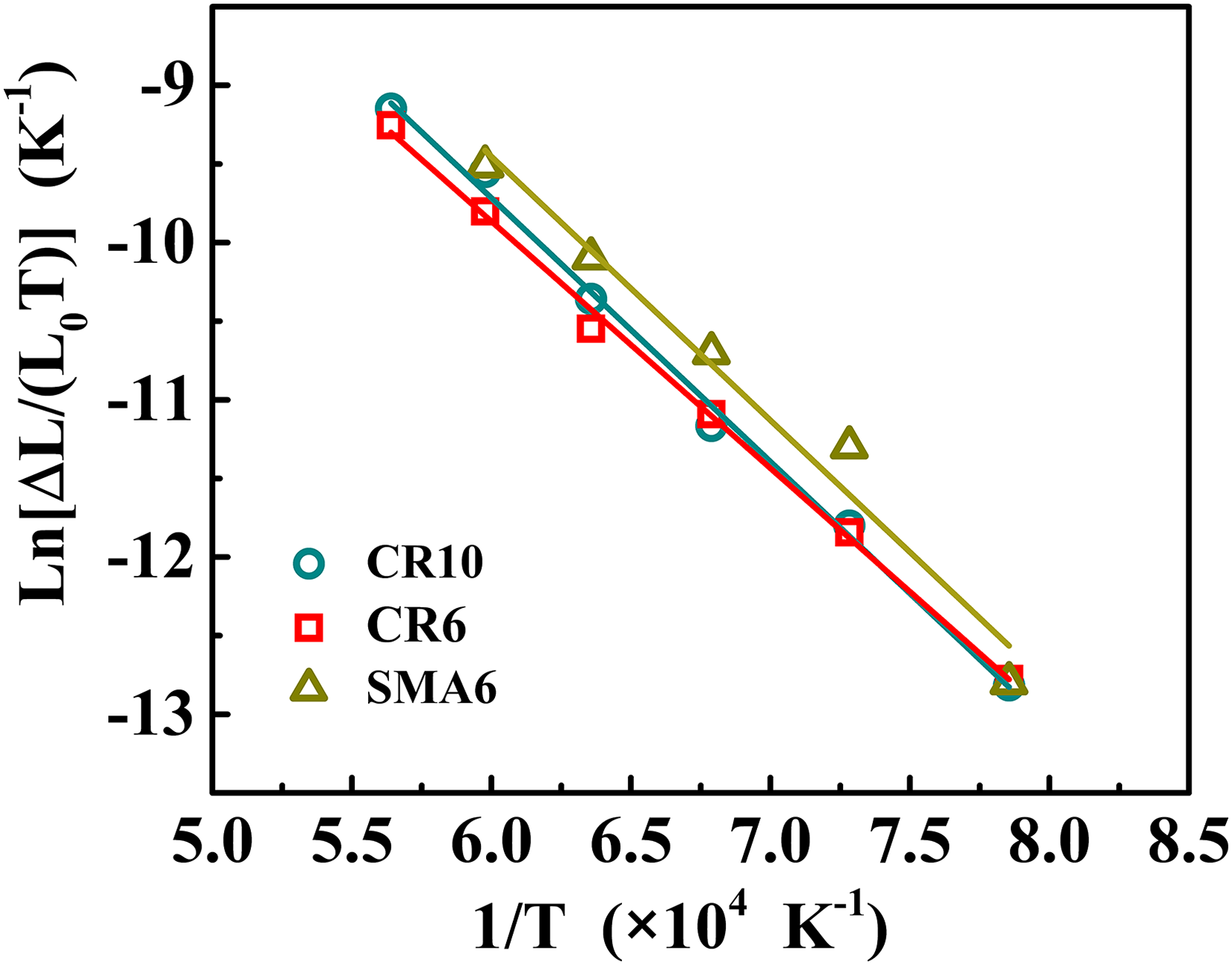
Relationship between Ln[Δ
Related parameters and activation energy in the initial stage of sintering.

The morphologies of green bodies sintered at 1400 °C prepared by CR10, CR6 and SMA6 are shown in Figure 8. During sintering, the particles became spherical due to surface diffusion, and the individual particles or aggregates acted as sintering units when shrinkage occurred. Large aggregates can be seen in the sintered microstructures in Figure 8(a) and (c), while relatively smaller aggregates were contained in the specimen prepared by CR6 in Figure 8(b), which is consistent with the smaller size distribution of the aggregates in Figure 2(b). Typically, the microstructure of the green body obtained by SMA6 exhibited evident densification, as shown in Figure 8(c), which can be attributed to the smaller pore size of its green body (Figure 5(b)) and largest shrinkage rate at 1400 °C (Figure 6(a)).

Fracture morphologies of green bodies obtained with CR10 (a), CR6 (b) and SMA6 (c) and sintering at 1400 °C.
Effects of ceramic microstructures
Finally, the green bodies were sintered in a vacuum furnace at 1850 °C for 6 h. The ceramic prepared by CR6 has a higher relative density (99.60 ± 0.03%) than the other ceramics, whereas the ceramic prepared by SMA6 has the least densification (99.48 ± 0.06%). Figure 9(a) to (c) displays the cross-sectional images of the sintered ceramics prepared by CR10, CR6 and SMA6, respectively. The driving force of sintering is derived from the reduction in particle surface free energy, which also drives the elimination of the interparticle pores 40 ; the smaller the curvature radius of the particle is, the larger the pressure difference caused by surface tension, and then, it is easier for small particles to transfer mass and eliminate interparticle pores.41–43 From this point of view, the initial particle size of CR10 is smaller, yet the relative density of its ceramic is not as high as that of CR6. This result suggests that the initial particle size is not the critical factor determining the ceramic density. Notably, for the compacts obtained with CR6, the aggregates in the slurry are the smallest, and the relative density is the highest for the ceramic. On the other hand, the relative density of the green body obtained by SMA6 is the highest, but the resultant ceramic density is not. This indicates that the smaller aggregate size contributes more to the higher relative density of the ceramic, and the relative density of the green body may play a lesser role in sintering densification. 44
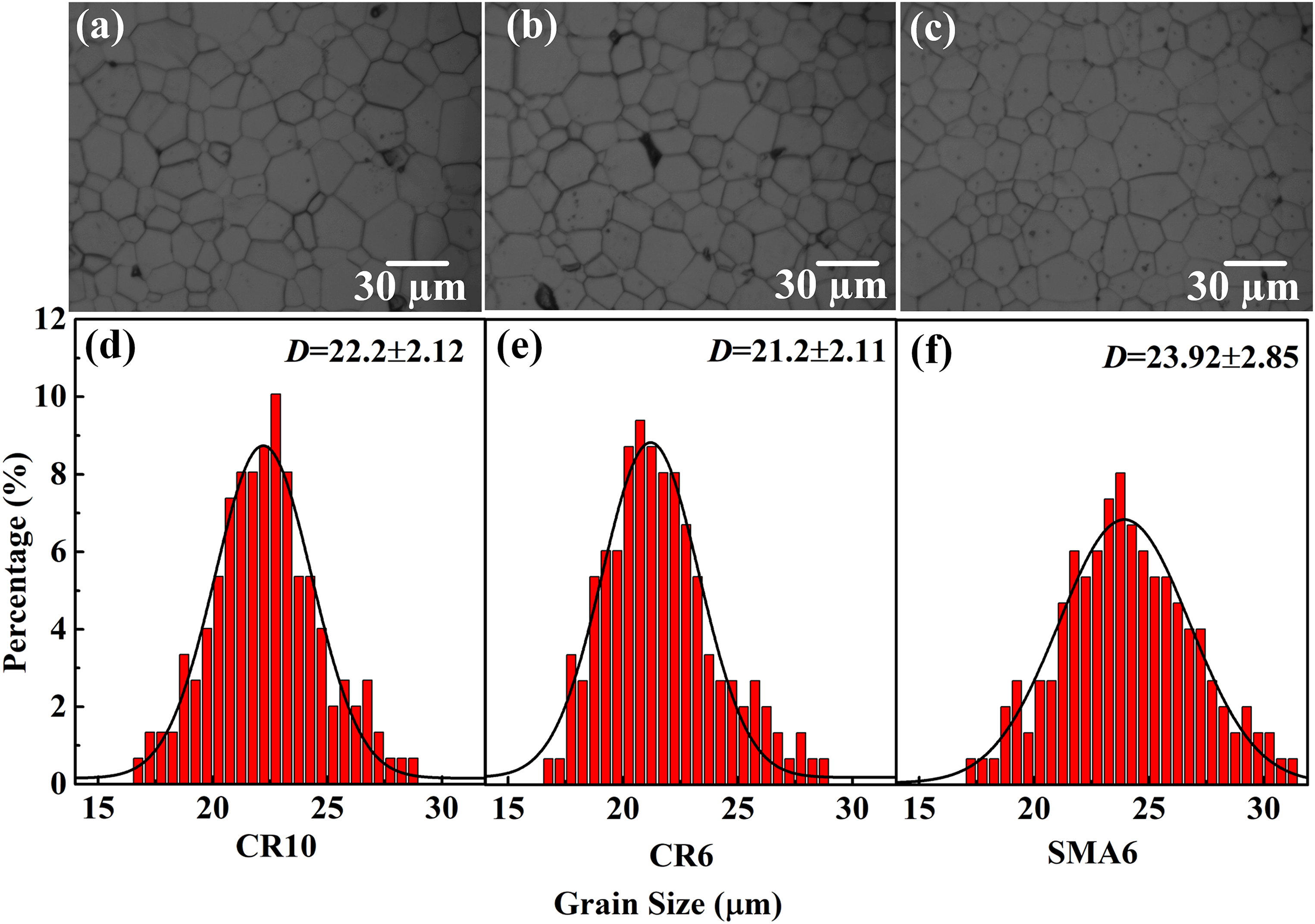
Corrosion morphology of ceramics prepared by CR10 (a), CR6 (b) and SMA6 (c), the panels (d), (e) and (f) show the grain size distribution of (a), (b) and (c), respectively. The grain size distribution was obtained from the corrosion morphology, and the histograms were fitted by Gaussian functions. A total of 1500 grains were counted.
Figure 9(d) to (f) shows the grain size distributions of the ceramics. Corresponding to the relative density, the ceramic prepared by CR6 exhibits the smallest average grain size, and the ceramic prepared by SAM6 has the largest average grain size. Compared to the green bodies of CR10 and CR6, the green body of SMA6 has a lower cumulative pore volume and pore size, which can promote mass transfer between particles to stimulate grain growth. 45 In addition, the specimen prepared by SMA6 shows the highest sintering shrinkage rate at 1400 °C, which indicates that the densification of the specimen occurs earlier and that a relatively longer time is needed for grain growth, resulting in larger grain size.
Figure 10 shows the fracture morphology of the sintered ceramics. Pores with sizes of ∼2.3 μm and ∼2.0 μm can be observed at the grain boundaries of the ceramics prepared by CR6 and CR10, respectively. However, for the ceramic prepared by SMA6 (Figure 10(c)), most of the pores with a size of ∼1.9 μm are trapped in the crystal grains. It is obvious that the pores in the ceramics originate from the green bodies and are controlled by the SDPs in the slurries. As mentioned above, SMA6 has the highest content of aggregates in the slurry. Consequently, the aggregates can trap many pores. Moreover, the largest shrinkage rate started at a lower temperature, the aggregates enabled faster densification, and mass transportation along the grain boundary were orders of magnitude faster than that within the grain. The pores may have insufficient migration times, so they are trapped inside the grain under the effect of pore–boundary separation, 46 leading to internally trapped pores (Figure 10(c)). According to the results, Figure 11 shows the schematics of the compact densification. The lower aggregate magnitude can make the pores migrate to the grain boundaries for densification from CR6 and CR10 (Figure 11(a)), but the higher aggregate magnitude for SMA6 traps pores inside grains during sintering (Figure 11(b)).

Fracture morphologies of ceramics prepared with CR10 (a), CR6 (b) and SMA6 (c).
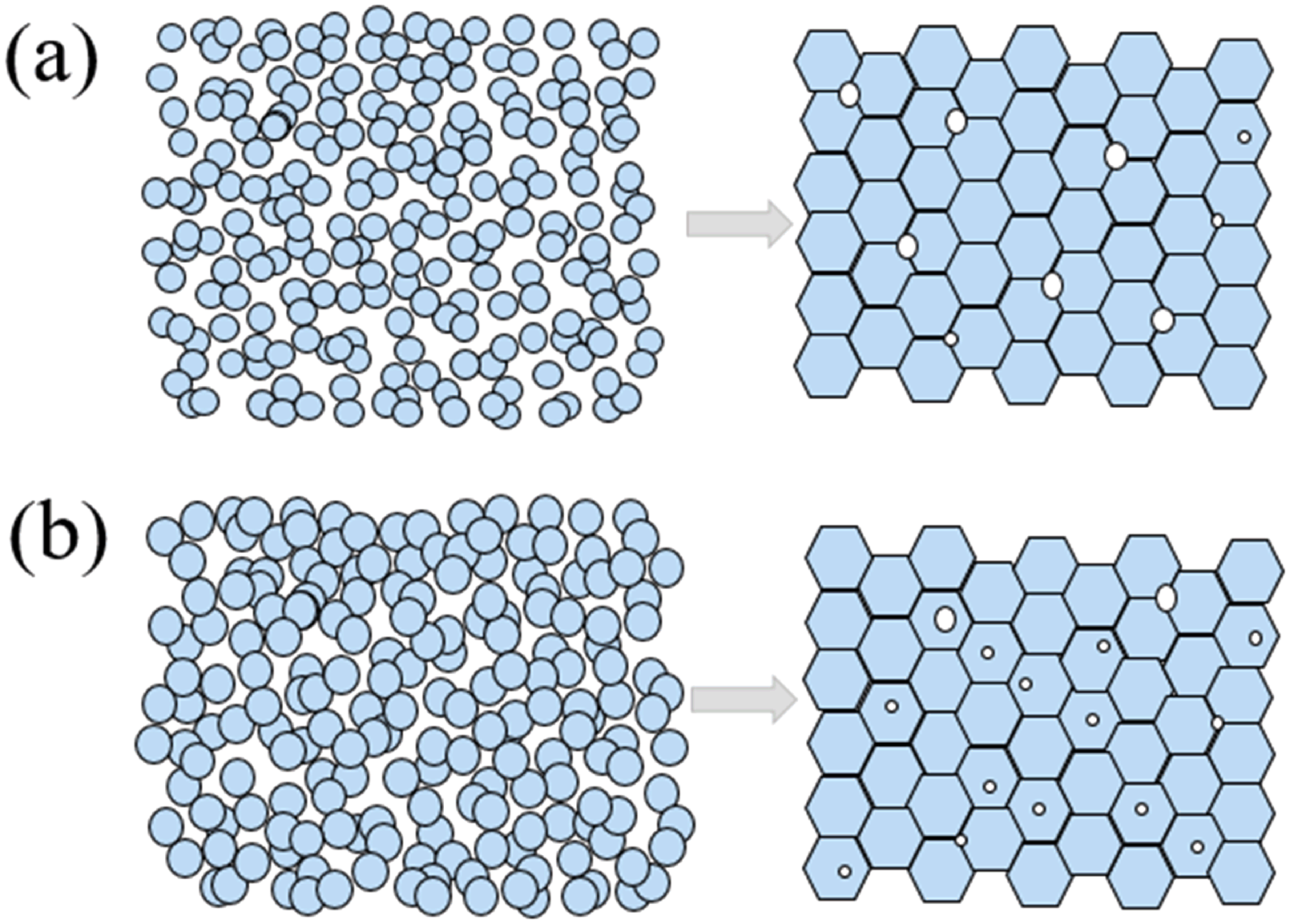
Schematic diagrams of compact densification for CR10/CR6 (a) and SMA6 (b).
The in-line transmittance of polycrystalline Al2O3 is significant for applications in optical components. Figure 12 shows the in-line transmittance curves and optical images of the sintered ceramics with a thickness of 0.8 mm. The in-line transmittance of the ceramic prepared with CR6 is higher (22%) than the in-line transmittance values of the ceramics obtained with CR10 (21%) and SMA6 (18%) at a light wavelength of 650 nm. Light scattering is the main source affecting ceramic optical transmittance. Microstructural features, such as pores, grain boundaries, and impurities, dominate light scattering, where pores contribute more to scattering. 47 The cumulative pore volume of the green body obtained by CR10 is the same as that obtained by CR6 (0.23 ml·g−1), but the green body obtained by CR10 has a smaller average pore size (100 nm), which indicates that the green body obtained by CR10 has a larger number of pores than that obtained by CR6. It is reasonable to conclude that the ceramic produced by CR10 has a higher porosity than that produced by CR6. Therefore, this contributes more to the higher in-line transmittance of the ceramic prepared by CR6.
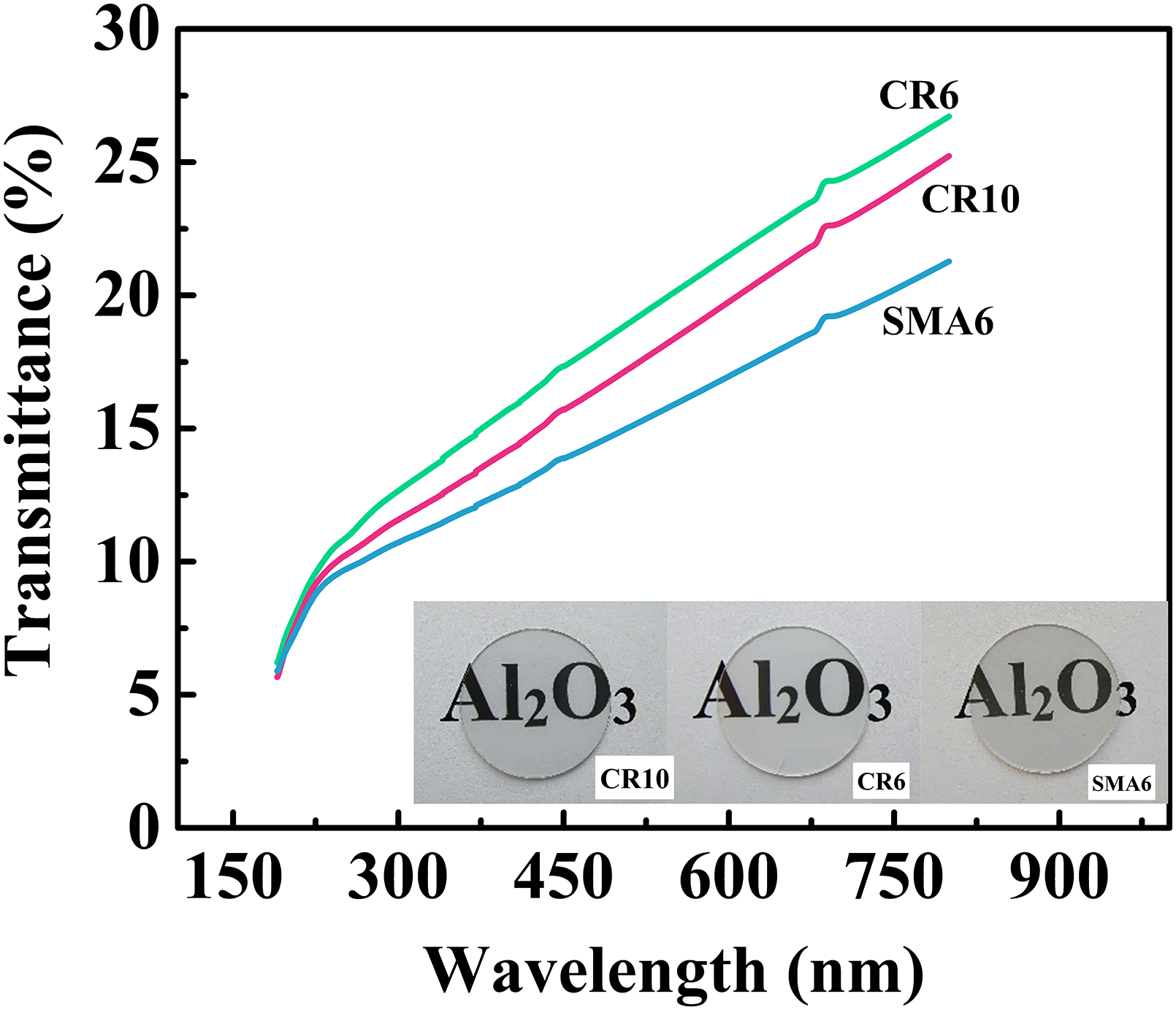
In-line transmittance curve and optical images of alumina ceramics (thickness: 0.8 mm).
It is generally accepted that the properties of final ceramics are related to the features of green bodies that are dominated by slurry qualities. However, the results of this work demonstrate that densification is more influenced by aggregate. A previous study showed that pores are also trapped inside grains in terms of the high-solid loading (50–56 vol.-%) with inadequate degassing. 48 Michálková et al. 23 reported that higher solid loading will decrease densification and increase grain growth. Therefore, as the features of the unavoidable aggregates are appropriately controlled within slurry fabrication, the solidified green body can be sintered to achieve alumina ceramics with higher density and better optical quality.
Conclusion
Three types of powders at their highest attainable slurry solid loadings were used to fabricate alumina translucent ceramics by gelcasting and vacuum sintering. The properties of the slurry, green body and ceramic were investigated. The initial particles were monodispersed in the slurries using an Isobam system as a dispersant after ball milling. As the gelling time increased, aggregate/agglomerate occurred in all of the slurries prepared by CR10, CR6 and SMA6 powder particles. The larger size of the initial particle corresponded to larger aggregates, and the small particle size of CR10 particles increased the particle number per aggregate. Although the higher solid loading of SMA6 achieved a green body with a higher relative density, the product with the smaller aggregate size and low sintering activation energy achieved by CR6 was sintered into products with higher densification that were involved in the growth of smaller grains and lower porosity, thus reaching a higher in-line transmittance of 22% at a light wavelength of 650 nm.
Footnotes
Acknowledgements
We gratefully acknowledge the assistance of the Key Laboratory of Transparent and Opto-functional Inorganic Materials at the Shanghai Institute of Ceramics.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present work was supported by Frontier Exploration Projects of Longmen Laboratory (No. LMQYTSKT017), the State Key Laboratory of New Ceramic and Fine Processing, Tsinghua University (KF201911), and the West Light Foundation of the Chinese Academy of Sciences (2021).
