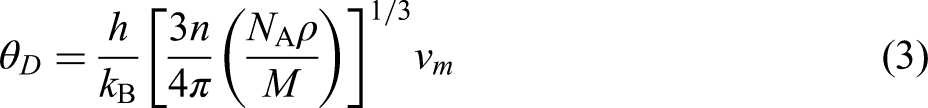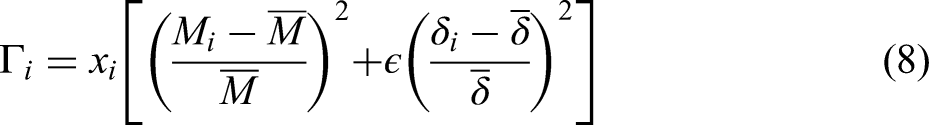Abstract
Advancements in materials discovery tend to rely disproportionately on happenstance and luck rather than employing a systematic approach. Recently, advances in computational power have allowed researchers to build computer models to predict the material properties of any chemical formula. From energy minimization techniques to machine learning-based models, these algorithms have unique strengths and weaknesses. However, a computational model is only as good as its accuracy when compared to real-world measurements. In this work, we take two recommendations from a thermoelectric machine learning model, TaVO5 and GdTaO4, and measure their thermoelectric properties of Seebeck coefficient, thermal conductivity, and electrical conductivity. We see that the predictions are mixed; thermal conductivities are correctly predicted, while electrical conductivities and Seebeck coefficients are not. Furthermore, we explore TaVO5’s unusually low thermal conductivity of 1.2 Wm−1K−1, and we discover a possible new avenue of research of a low thermal conductivity oxide family.
Keywords
Introduction
Accelerating worldwide energy demand and increasing interest in preventative measures regarding global climate change have led to the development of diverse methods of energy production. One such method is the use of thermoelectric generators to capture waste heat. 1 Thermoelectric generators rely on thermoelectric materials, which can generate an electric potential difference (i.e. a voltage) from a temperature gradient with no moving parts. This unique set of properties makes these materials promising for a wide variety of engineering applications. However, current thermoelectric devices suffer from low efficiency and often rely on rare and expensive elements, so they have thus far been relegated to use in mission-critical applications where no other options are available, such as space applications including rovers and probes.2–5 Indeed, some of the highest-performing thermoelectric materials utilize toxic elements, such as PbTe, rendering them unsuitable for many applications. New high-efficiency and low-cost thermoelectric materials will need to be discovered to enable broader device applications.
Unfortunately, discovering new high-efficiency materials has proven challenging. Device efficiency scales with
The large potential compositional and structural space available for exploration makes this challenge perhaps well-suited for data-driven approaches. In recent years, materials informatics, or data science tools directed towards materials research, have proven valuable in the rapid discovery and development of a variety of different materials including photovoltaics, superhard materials, metal alloys, and more.7–9 Pertinent to the field of thermoelectrics is the use of machine learning to predict thermal conductivity, which has become well-established in the last few years. Many have used machine learning to predict the lattice thermal conductivity of crystalline solids,10,11 and a recent report used machine learning models to identify a new aperiodic structure transition metal oxide, Ba10Y6Ti4O27, with thermal conductivity among the lowest ever measured for an oxide. 12 Still, relatively few works have taken their predictions out of the computer and onto the bench to synthesize and characterize new materials with low thermal conductivity. 13
In the field of thermoelectrics, materials informatics is finding increasing application.14–19 A common refrain among articles concerning materials informatics is that these tools can help to explore chemical whitespace in an accelerated and structured manner, allowing for the discovery of remarkable compounds with desirable yet previously unknown properties.11,10,14–19,12,20,21 Many thermoelectric materials informatics articles present impressive performance metrics over existing data and compare their results to calculations, yet few, if any, present the recommendation, synthesis, and characterization of hitherto unexplored thermoelectric materials. 18 We aim to address this gap in the literature by synthesizing and characterizing novel thermoelectric materials recommended by a previously published machine learning model. 16 In this article, we evaluate two new oxides, TaVO5 and GdTaO4, as candidates for low thermal conductivity materials and measure their thermoelectric properties, relating the observed transport to their structures.
Materials and methods
Machine learning algorithm
The machine learning model used to make materials recommendations was described previously.
16
Briefly, a random forest algorithm was trained on a combined dataset created using experimental data taken from literature values, the NIMS database, and the Materials Project. Rather than predicting materials properties via regression, the model instead treated predictions as classifications of whether or not they would have values above or below a given cut-off. For a given chemical formula, the model assigns a confidence value with a score
Synthesis of TaVO5
In accordance with Wang et al., 22 stoichiometric powders of Ta2O5 and V2O5 were mixed with ethanol and ball milled for 10 h with zirconia media. This mixture was then allowed to dry overnight. The dried powder mixture was then ground with an agate mortar and pestle and pressed into 13 mm diameter discs. These were buried in sacrificial powder and underwent reaction heating at 800 ∘C for 24 h. Sacrificial powder was used to reduce vanadium loss during synthesis as vanadium would diffuse into the alumina crucible boats during reaction heating. The discs were then crushed into powder with an agate mortar and pestle, and the powder was spark plasma sintered at California Nanotechnologies Inc. under a load of 5.7 kN for 20 min at 1000 ∘C to achieve maximum density for thermal and electrical measurements.
Synthesis of GdTaO4
In accordance with Yang et al., 23 GdTaO4 was synthesized with starting materials Gd2O3 and Ta2O5, which were calcined at 800 ∘C for 8 h. Stoichiometric powders of these calcined starting materials were mixed with ethanol and ball milled for 10 h with zirconia media. This mixture was allowed to dry overnight. The dried powder mixture was then ground with an agate mortar and pestle and pressed into 13 mm diameter discs. These discs underwent reaction heating at 1400 ∘C for 12 h. The discs were then crushed into powder with an agate mortar and pestle, and the resulting powder was spark plasma sintered at California Nanotechnologies Inc. under a load of 5.7 kN for 29 min at 1500 ∘C to achieve maximum density for thermal and electrical measurements.
Material characterization
Powder X-ray diffraction measurements were taken with a Bruker D2 Phaser diffractometer. Quantitative phase analysis was performed by the Rietveld refinement method using GSAS-II software. 24 Heat capacity measurements were taken in small portions of the synthesized powder using a Netzsch 3500 DSC differential scanning calorimeter.
The sintered discs were mounted in epoxy and cut with a diamond saw to a thickness of one to 2 mm for the measurement of the Seebeck coefficient, electrical conductivity, and thermal diffusivity. Prior to measurement, discs were sanded with a 1200 grit sandpaper on a Struers TegraPol-11. The electrical conductivity and Seebeck coefficient were measured using a Netzsch Nemesis 458 which has a 7% standard error for the Seebeck coefficient and a 5% standard error for electrical conductivity. The thermal diffusivity was measured via the laser flash technique on a Netzsch LFA 457, which has a standard error of 10%. The bulk density was measured with the Archimedes method in an immersion medium of deionized water. Thermal conductivity was calculated using the standard relationship:
Results
X-ray diffraction spectra for the TaVO5 sample along with Rietveld refinement are shown in Figure 1. A good fit,
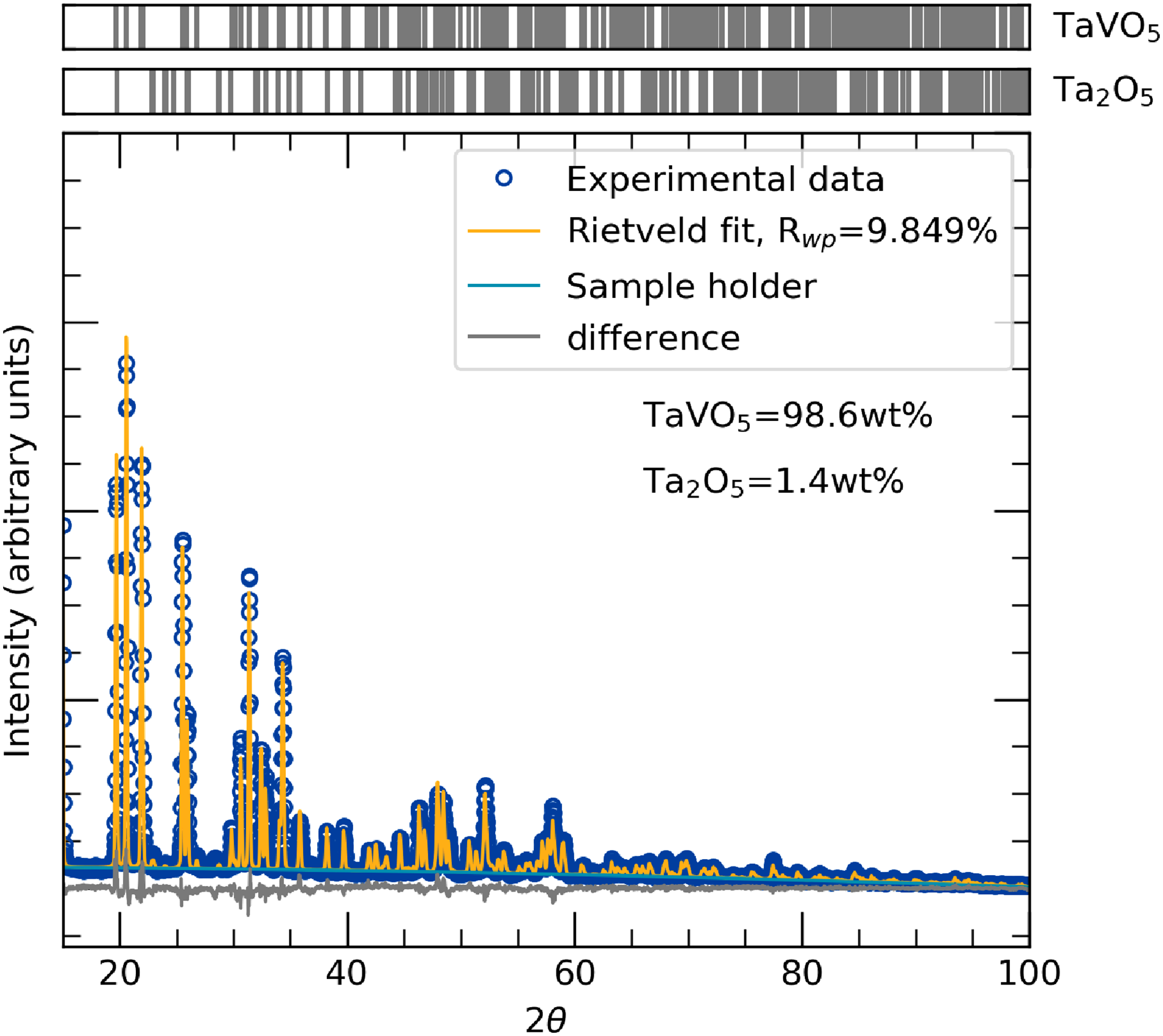
Rietveld refinement of powder X-ray diffraction data from TaVO5 samples showing a high purity sample with <2 wt% Ta2O5 impurity.
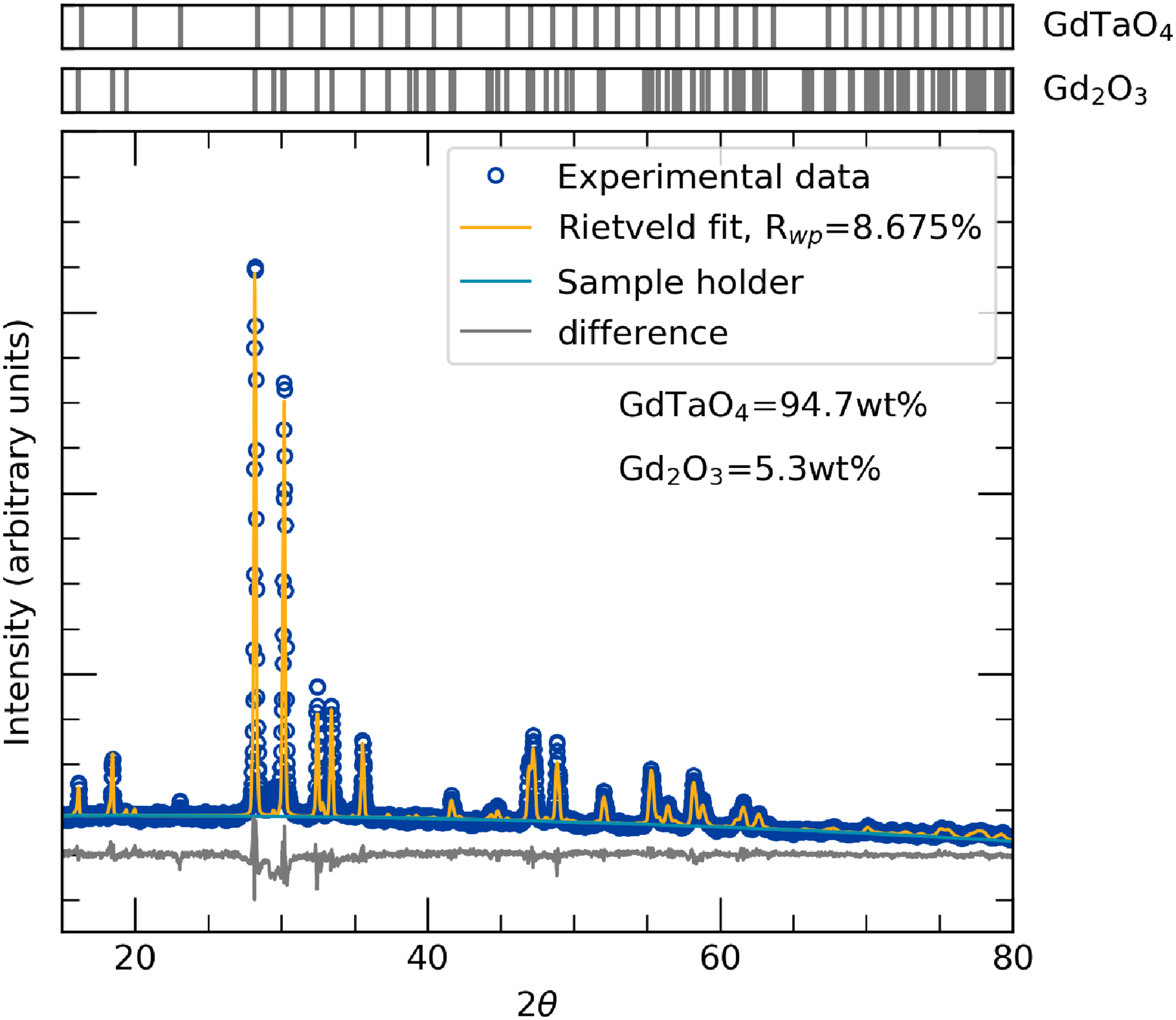
Rietveld refinement of powder X-ray diffraction data from GdTaO4 samples showing a high purity sample with
Since this study was focused specifically on machine learning predictions of thermal conductivity, stoichiometric oxides of both TaVO5 and GdTaO4 were synthesized. By simple electron counting one would rightly predict both Ta[
Thermal conductivity measurements, shown in Figures 3 and 4, confirm the predictions that TaVO5 is a very low thermal conductivity material, particularly among oxides, while GdTaO4 is not. The thermal conductivity of TaVO5 is both low and nearly independent of temperature. TaVO5 reaches a maximum value of 1.34 W
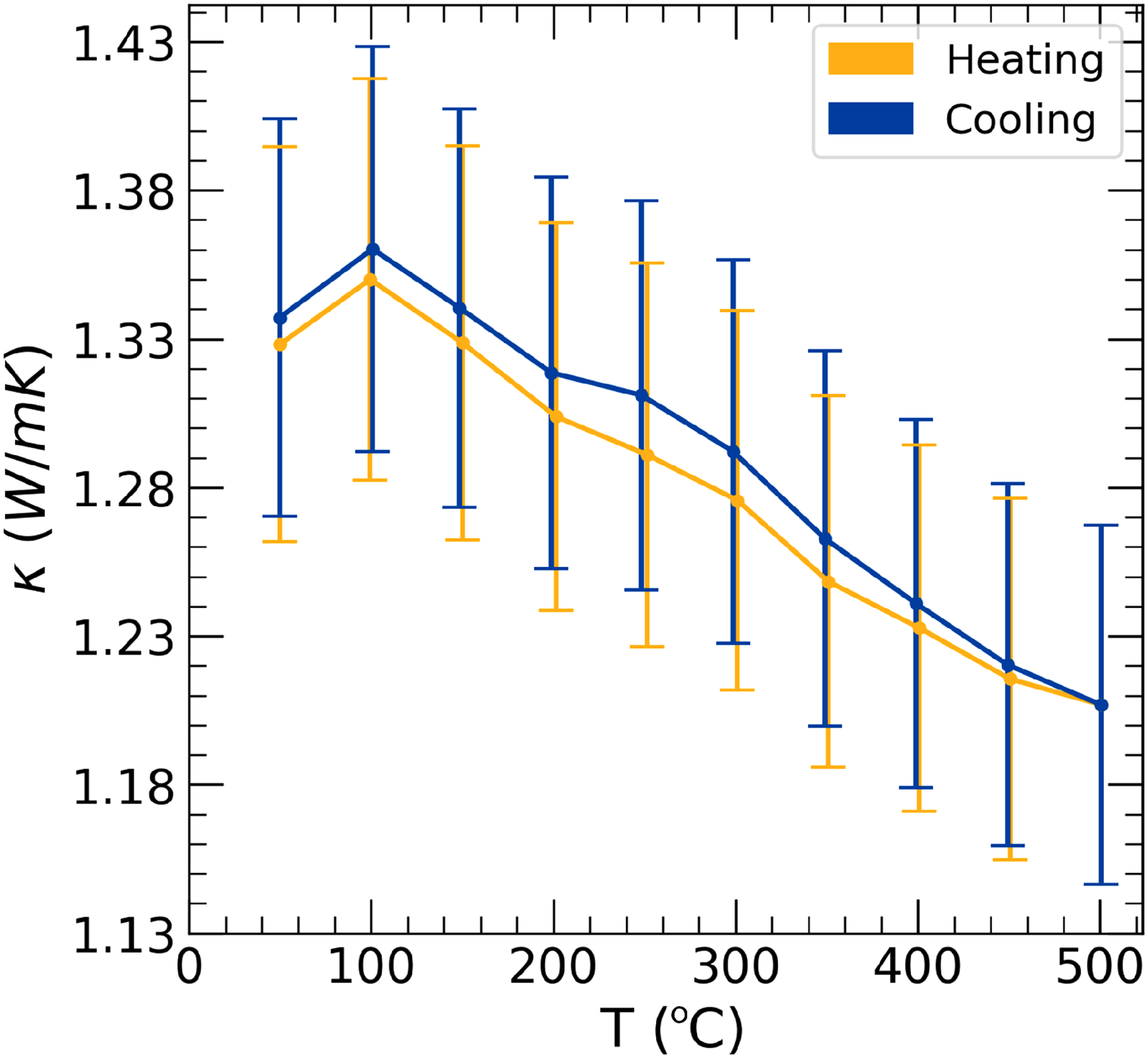
Thermal conductivity of TaVO5. Orange is thermal conductivity during heating while blue is during cooling.
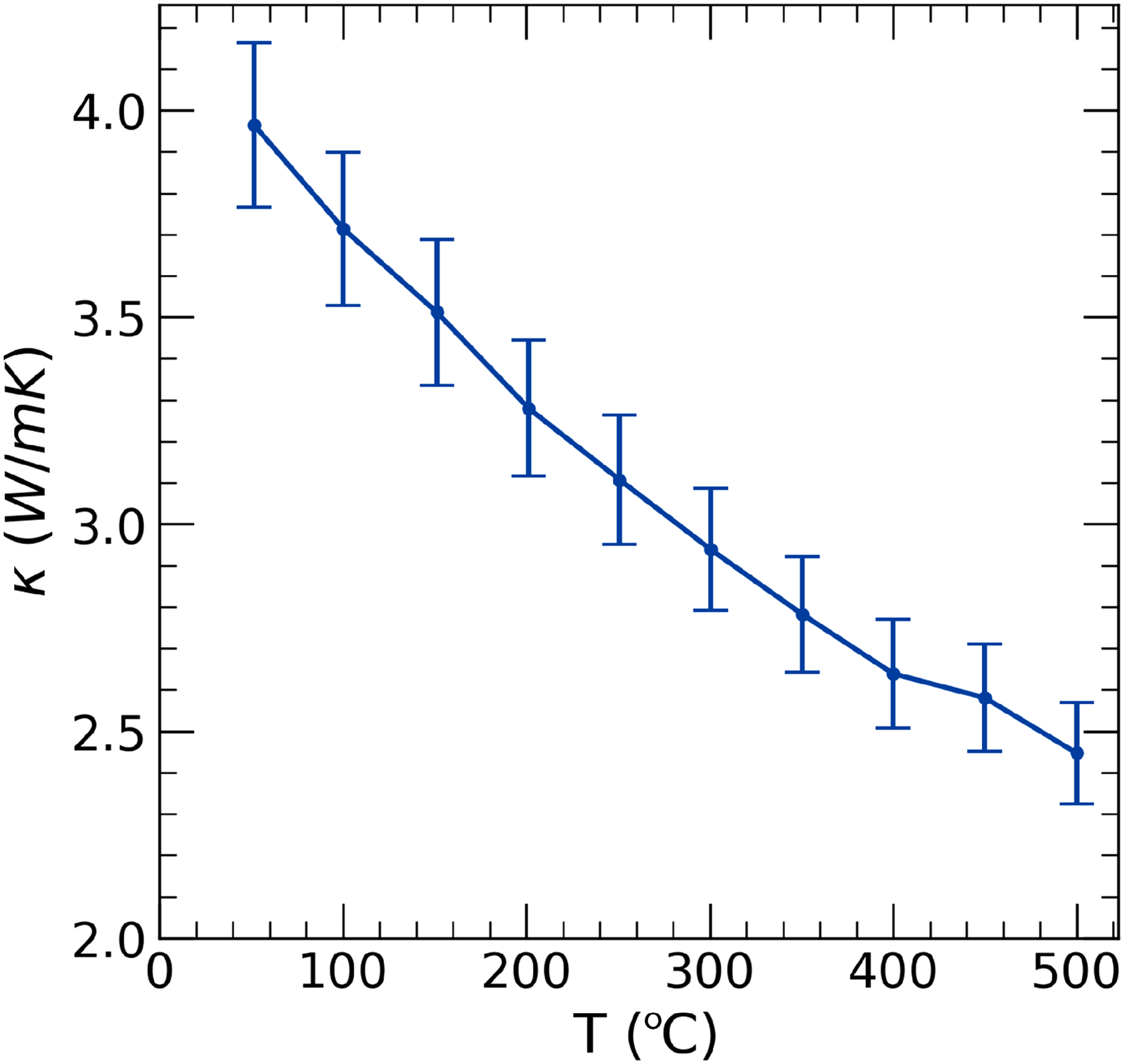
Thermal conductivity of GdTaO4 during heating.
In TaVO5, we observed during initial laser flash measurements that at higher temperatures, vanadium loss became apparent with TaVO5 decomposing into Ta9VO25. Therefore, samples were only measured up to 500 ∘C in order to maintain TaVO5 phase stability. Samples measured at higher temperatures had large hysteresis between heating and cooling which we attribute to vanadium volatilization. New samples were synthesized, and laser flash measurements were taken up to 500 ∘C to avoid volatilization and hysteresis.
TaVO5 was measured to have a bulk density of 4.47 g
Discussion
Given the low electrical conductivity of both TaVO5 and GdTaO4, the electrical contributions to the overall thermal conductivities were calculated to be negligible. TaVO5 ranks lower than many well-known low thermal conductivity oxides as outlined by Winter et al. 27 This is noteworthy considering that most low thermal conductivity oxides achieve low lattice thermal conductivities by relying on phonon scattering due to aliovalent doping producing vacancies on the cation or anion sublattices.28,29
Why is the thermal conductivity of TaVO5 so much lower than that of GdTaO4? There are several possible factors that could be responsible. Let us first consider the Debye temperature,
While the two estimated Debye temperatures are quite close, the Debye temperature of TaVO5 exceeds that of GdTaO5. This is unexpected given that the thermal conductivity of TaVO5 is considerably lower than that of GdTaO4. However, the Debye temperature is not the only material property that influences thermal conductivity. Other metrics, such as mass contrast and polyhedral connectivity of the crystal structure itself, can also dictate thermal conductivity.
Let us first consider atomic mass. It is known that large atomic mass atoms, such as Ta, and compounds with large mass contrast between atoms result in lower thermal conductivity.35,36,28 If we assume a Debye phonon spectrum, the thermal conductivity is dependent on the inverse square root of the defect phonon scattering coefficient at temperatures above the Debye temperature:
The low thermal conductivity is likely intrinsically related to the crystal structure of TaVO5 itself. As shown in Figure 5, the TaO6 octahedra is only corner-shared with the VO4 polyhedra. This flexible linkage implies that the structure is more likely to twist and change volume during acoustic phonon propagation, lowering its thermal conductivity.27,28 Additionally, the large mass contrast between vanadium and tantalum within the lattice is likely to cause phonon dampening, limit the phonon’s degrees of freedom, and interrupt the possible phonon modes. 29 This determination is further supported by previous Raman scattering measurements showing limited vibrational nodes as well as distortion of octahedra and polyhedra at higher temperatures. 6
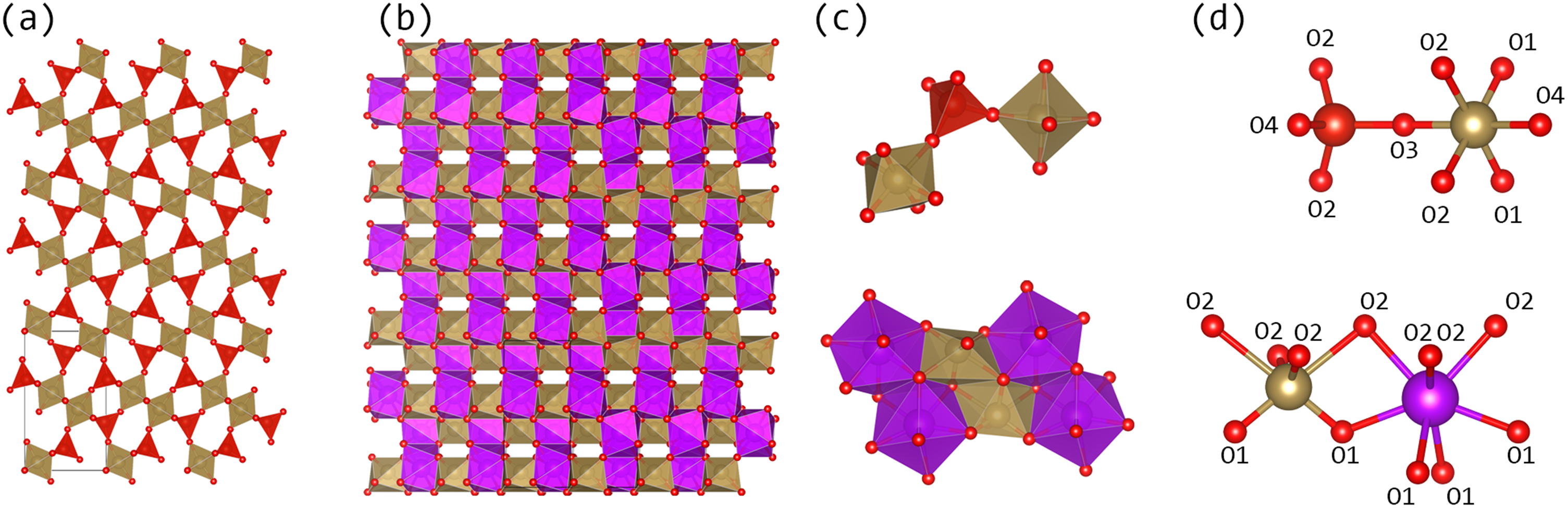
Extended crystal structure views of (a) TaVO5 and (b) GdTaO4 and local polyhedral connectivity diagrams (c) and atomic labels (d).
TaVO5 and other comparable oxides in the same Pnma space group, such as NbVO5 and TaPO5, are negative thermal expansion (NTE) materials.38–40 This is unsurprising as the same anharmonicity of low-frequency phonon modes that gives rise to NTE also results in the coupled phonons and shorter mean free path lengths that are typically associated with low thermal conductivity. 41 The literature review shows a gap in thermal conductivity predictions and measurements for this family of oxides. These Pnma corner-shared oxides, such as NbVO5 and TaPO5, could represent a new avenue of low thermal conductivity materials that have yet to be fully explored.
Seebeck coefficient, as shown in Figure 6, was measured to be around
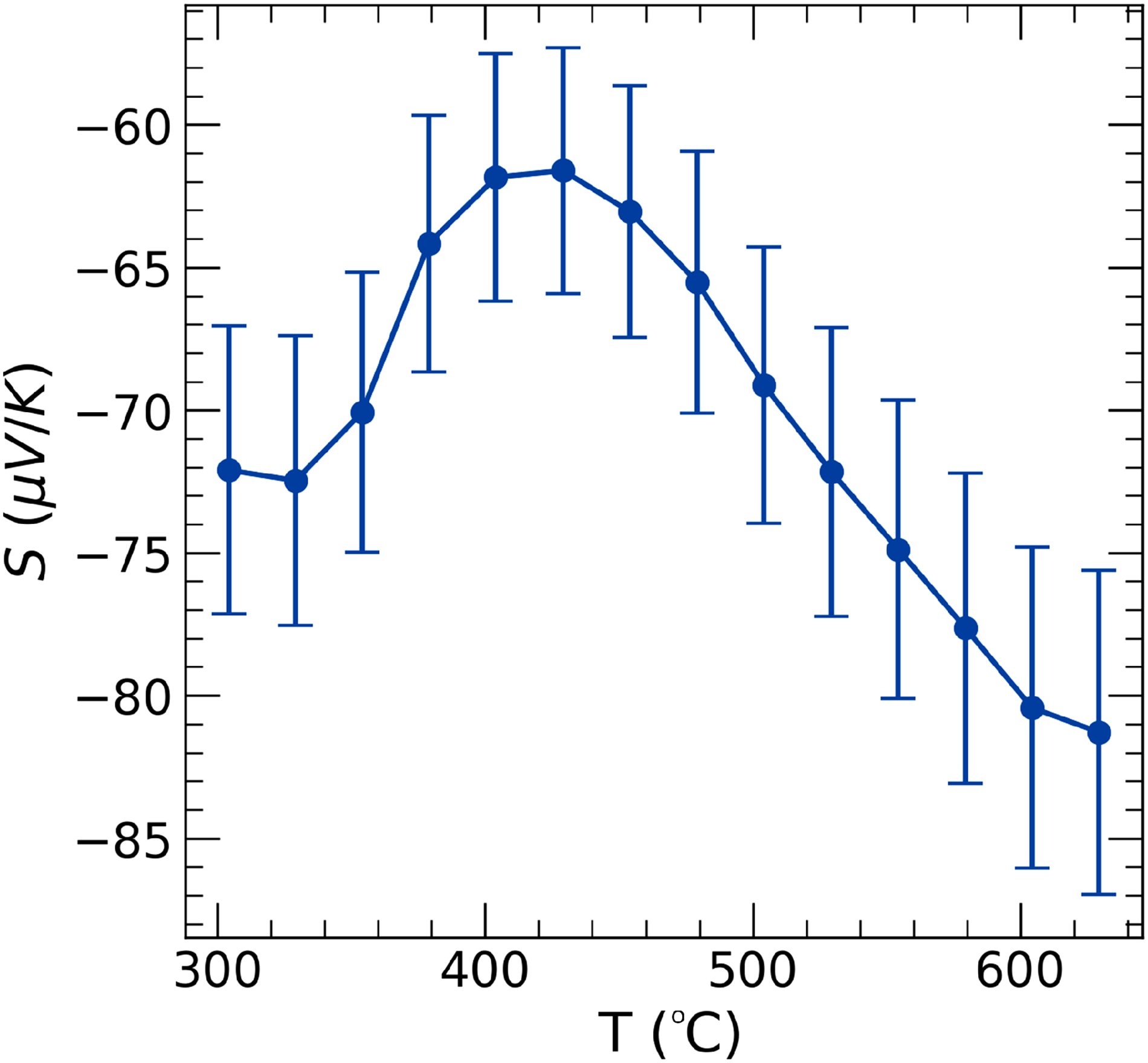
Seebeck coefficient of TaVO5.
The electrical conductivity of GdTaO4 was unmeasurable on the Nemesis 458, suggesting its conductivity to be somewhere below the limitations of 0.05 S
Thermal conductivity, as shown in Figure 4, was shown to start at 4 W
The higher than expected thermal conductivity of GdTaO4 can be explained by having a more interconnected crystal structure, as well as the similar atomic weights of gadolinium and tantalum. This allows a greater number of phonon modes to exist within the structure. Single crystal studies support this theory, showing similar sloping decreases in thermal conductivity as well as low thermal anisotropy.
23
Despite the fact that the GdTaO4 exhibits thermal conductivity well below the cutoff value of 10 W
The Seebeck coefficient measurements of GdTaO4 showed large variation and lack of continuity and are thus not reported. The large amount of noise and lack of linearity in the measurement suggests that the Seebeck coefficient is far below the limitations of the Nemesis 458 of 10
It should be noted that these sources have similar shallow band structures but show contradictions for band gap. The Topological Materials Database43–45 lists GdTaO4 as a semimetal while others such as the Materials Project 46 and Ding et al. 26 suggest a large band gap ranging from 3.26 to 4.86 eV, respectively. Our electrical conductivity measurements support the notion that this material is a wide-gap semiconductor and is not a semimetal. The curvature of the band structure indicates similar effective masses for both carriers which, in conjunction with a large band gap, means the bipolar effect further constrains the magnitude of the Seebeck coefficient. 23
Conclusion
By using predictions to excite and motivate scientists, machine learning gives us a promising direction to explore chemical whitespace. However, these tools are very dependent on training data and the choice of algorithm, and as such require scrutiny and verification. In the case of the Citrination engine model, we chose two of the recommended oxide compounds to test as thermoelectric materials: TaVO5 and GdTaO4. We have shown that for predicting thermal conductivity, the model performed as expected, with both compounds being well below the cutoff of 10 W
Footnotes
Acknowledgements
The authors gratefully acknowledge funding and support from the National Science Foundation through Award NSF DMR 1651668. Special thanks to Clayton Cozzan and Dr Ram Seshadri for preliminary assistance in sintering trial runs prior to final synthesis. We would like to thank Eric Eyerman at California Nanotechnologies Inc. for his assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.