Abstract
Magnesium oxide (MgO) porous ceramics with high porosity, compressive strength and low thermal conductivity were prepared by Organic Foam Template Method. The effects of the sintering temperature, polycarboxylic acid (PCE) dispersant and pore size of organic foam template on the properties of MgO porous ceramics were investigated. The experiment results showed that with the increase of sintering temperature, the MgO porous ceramic shrinkage, skeleton density and compressive strength increased. PCE could increase the fluidity of slurry and make the framework clearer, as well as reduce the cracks formed in the process of drying effectively. When the content of PCE was 0.5 wt-%, the porosity, compressive strength and thermal conductivity of MgO porous ceramics were 88.5%, 1.6 MPa and 0.045 W/(m·K), respectively. In addition, as the pore size of the organic foam template decreased, the porosity decreased, and the resistance increased and the thermal conductivity increased.
Introduction
MgO porous ceramic is a kind of ceramic material with three-dimensional mesh structure.1,2 Benefitting from its low thermal conductivity, stable chemical properties, excellent thermal stability, high strength and other excellent performance, porous ceramic is widely used in gas filters, biological materials, thermal insulators, catalyst carriers and other fields.3–6 Hernández-Reséndiz et al. obtained MgO–TiO2 composites by means of two novel different routes. The results showed that the densification and mechanical properties of MgO–TiO2 composites starting from brucite (Mg(OH)2) were better than caustic magnesia (MgO). 7 Gómez-Rodríguez et al. reported that the presence of α-Al2O3 nanoparticles in the magnesia matrix induced the magnesium–aluminate spinel formation (MgAl2O4) and promoted the densification of the magnesia matrix, which improved the mechanical resistance most significantly at 1500 °C. 8 In addition, Gómez-Rodríguez et al. found that the addition of Fe2O3 can improve the corrosion resistance of magnesia refractory matrix more than that of Al2O3. Besides, they found that ZrO2 nanoparticles could enhance the samples’ densification. Compared with those doped with nano-ZrO2, the magnesia bricks were severely corroded.9,10
In the past few years, a number of methods have been adapted to fabricate MgO porous ceramics, including but not limited to Organic Foam Template Method,11,12 Direct Foaming,13–15 Pore-Forming Agent,16,17 Sol–Gel Method,18,19 Freeze–Dry Processing20,21 and Hydrothermal Method. 22 Hao et al. obtained the porous MgO material which was synthesised via an integration of the evaporation-induced surfactant assembly and magnesium nitrate pyrolysis. 22 The composite of MgO porous ceramics and polyethylene glycol (PEG) had excellent heat storage performance, and the apparent heat storage efficiency was 64.6%. Wang et al. prepared MgO by in situ carbonised with magnesium nitrate hexahydrate, ethanol and other raw materials. 23 The porous materials showed excellent enrichment properties for U (VI) by electrostatic interaction between surface hydroxyl groups and uranyl and its hydrolyzate. Liu and Zhu prepared MgO porous materials by Direct Foaming Method. The saponin in single foaming agent had the best foaming effect with the porosity about 74.85%. 24 However, with the increase of compound foaming agent, the pore size, thermal conductivity and compressive strength of the material decreased, and the compressive strength was concerning 0.6 MPa. In addition, Xie et al. prepared MgO foam ceramics with porosity of 70.62–79.47% and compressive strength of 3.27–13.75 MPa by two-step foaming method. 25 The MgO foam ceramics samples prepared with this foaming method had higher compressive strength but lower porosity.
Organic Foam Template Method is to cover the prepared slurry onto the pre-treated polyurethane foam and then squeeze to remove the excess slurry. The operation above is repeated several times to ensure that the slurry can be onto the polyurethane evenly. By drying and sintering the green body, MgO porous ceramics can be obtained.26,27 Due to its controllable process, simple operation and low production cost, organic foam impregnation has become one of the most popular traditional methods. 28 In addition, because of high porosity, large specific surface area, low thermal expansion coefficient, stable size, high temperature resistance, excellent mechanical properties 29 and a wide range of applications of the porous structure ceramics prepared by organic foam impregnation,30–32 this method is widely used in practice. The MgO porous ceramics prepared by this method are widely used in building insulation, magnesium alloy melt filter and other fields. Polycarboxylic acid (PCE) dispersants can be dissolved in water and there are electrostatic repulsion effect and steric hindrance effect between particles and PCE. It has become a research hotspot due to its remarkable performance, such as low dosage, excellent dispersibility, positive stability, and environmental friendliness. Wu prepared a series of porous ceramics by using steel slag kaolin and PCE with Organic Foam Template Method. 33 The porosity of the porous ceramics was high. When the content of water reducing agent increases from 0.2% to 0.6%, the compressive strength increases from 0.2 to 1.5 MPa. Because the addition of PCE can increase the mechanical strength of the materials, we use it as a dispersant of MgO porous ceramics.
In this article, a series of MgO porous ceramics with high porosity, strength and low thermal conductivity were prepared by Organic Foam Template Method. The effects of the sintering temperature, interaction between PCE and MgO, PCE on the properties of MgO porous ceramics were investigated. Furthermore, the effects of different pore sizes on the properties of MgO porous ceramics were also researched.
Experimental
Raw materials
MgO powder (Table 1 shows the elemental analysis results of MgO raw materials), polyurethane foam template (PPI = 15, 20, 25, 30, 40), PCE (purity > 98 wt-%), polyvinyl alcohol solution (PVA, ω = 5 wt-%), Ludox.
XRF analysis of the composition of MgO powder.

Preparation of MgO porous ceramics
The preparation process of MgO porous ceramics is shown in Figure 1 In the experiment, MgO was used as the base raw material, adding distilled water, different contents of PCE, 5 wt-% of polyvinyl alcohol solution and 2 wt-% Ludox to get an evenly mixed MgO slurry by ball milling for 2 hours. The polyurethane foam was corroded in 15 wt-% NaOH solution at 50 °C for 4–6 hours to obtain the foam with rough surface. The prepared MgO slurry was uniformly coated onto the polyurethane foam skeleton and then the excess slurry was extruded. The blank was obtained until the mass is no longer increased. After drying 24 hours in a constant temperature and sintering in muffle furnace, the MgO porous ceramics could be obtained.
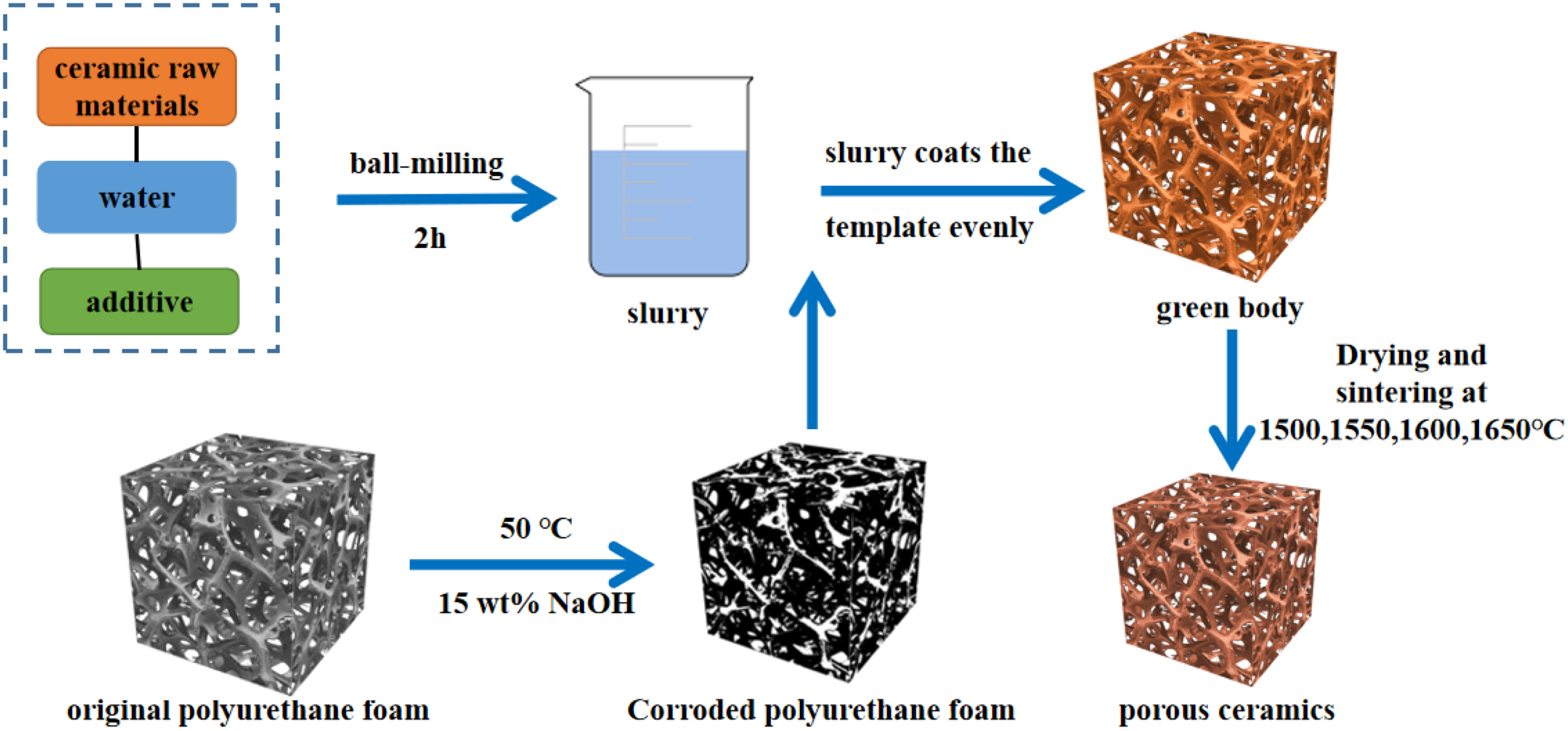
The preparation process of MgO porous ceramics.
Due to the decomposition of polyurethane at high temperature, the porous ceramic skeleton could be destroyed by the gas rushing out. Therefore, in order to ensure the integrity of the porous ceramic structure, it is necessary to formulate a suitable heating rate. Figure 2(a) shows the thermogravimetric (TG) curve of polyurethane foam. It can be seen from the figure that when the temperature reaches 700 °C, the sample weight loss ratio reaches almost 100%. When the temperature continues to rise, the slope of the TG curve is minimal. This phenomenon indicates that the polyurethane is completely decomposed. Therefore, it can be determined that the MgO porous ceramic is slowly heated at 1 °C/min before 700 °C and held for 2 hours to completely remove the polyurethane residue. The foam removal system is shown in Figure 2(b).
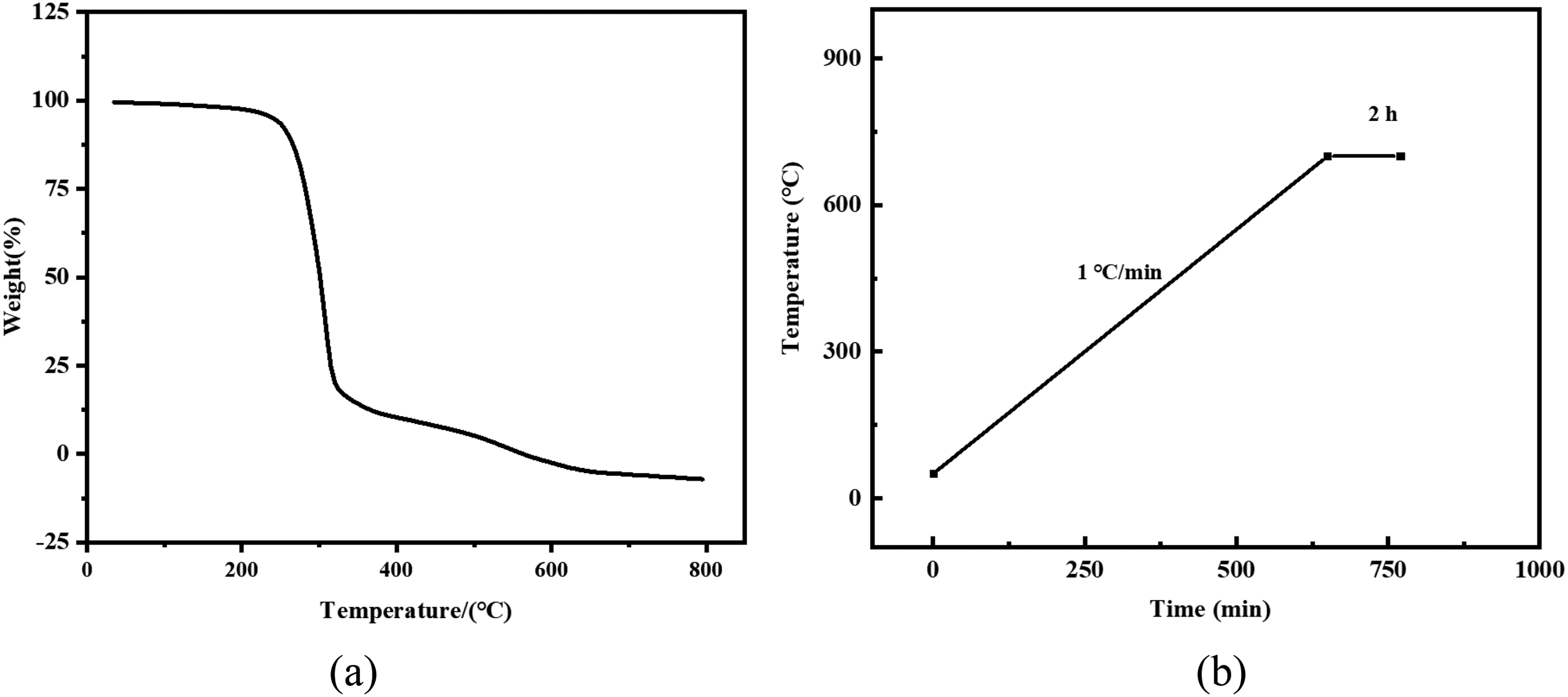
(a) Thermal weight loss curve of polyurethane foam and (b) the burning out system of polyurethane.
Characterisation
The element and oxide composition of MgO were characterised by X-ray fluorescence spectrometer of Shimadzu Limited (XRF-1800). Zeta potential of MgO particles in deionised water was measured by Zeta potential analyser (Zetasizer 300HSA) in the pH values range of 3–11. The crystalline phases of ceramics were determined by X-ray diffractometry (3KW D/MAX-2200 V PC) with Cu Kα radiation. After the gold spray treatment, the samples were placed in the scanning electron microscopy (SEM, JSM-6700F) to observe the microscopic morphology. The compressive strength of porous ceramics was measured by using a universal material testing machine (EM-30A+), and there are three specimens per group were tested for compressive strength. The thermal conductivity was obtained by thermal conductivity tester (TC3000E). The pore size and gain size distribution were calculated by the analysis software Nano Measurer.
The bulk density could be calculated by the mass and volume of porous ceramics according to Equation (1).
The ratio of the bulk density to the theoretical density of MgO was the porosity of the sample, as shown in Equation (2).
The shrinkage of MgO porous ceramics can be determined by Equation (3).
The relationship between slurry solid phase content and particle spacing could be calculated by Woodcock Equation (4).
Results and discussion
Effect of sintering temperature on MgO porous ceramics
Figure 3 is the XRD patterns of MgO porous ceramics at different sintering temperature. It could be seen that the sharpness of the peak of the sample varies from 1500 to 1650 °C, which reflected the growth of periclase during the sintering temperature. As we can see from Figure 3, the strength of crystal plane (200) and (220) increased significantly with the increase of sintering temperature, and reaches the maximum value at 1600 °C. When the temperature rise continuously, the strength of the crystal plane (220) increase, while (200) decreases. This phenomenon indicates that samples present obvious optimal orientation at different temperatures. Schell et al. identified a recrystallisation mechanism occurring during growth as responsible for the preferred orientation. 34 It is attributed to the competing grain growth of adjacent crystals with different orientations which result from anisotropy of surface diffusion. And the preferential orientation is corresponding to the fastest rate of crystal growth. 35
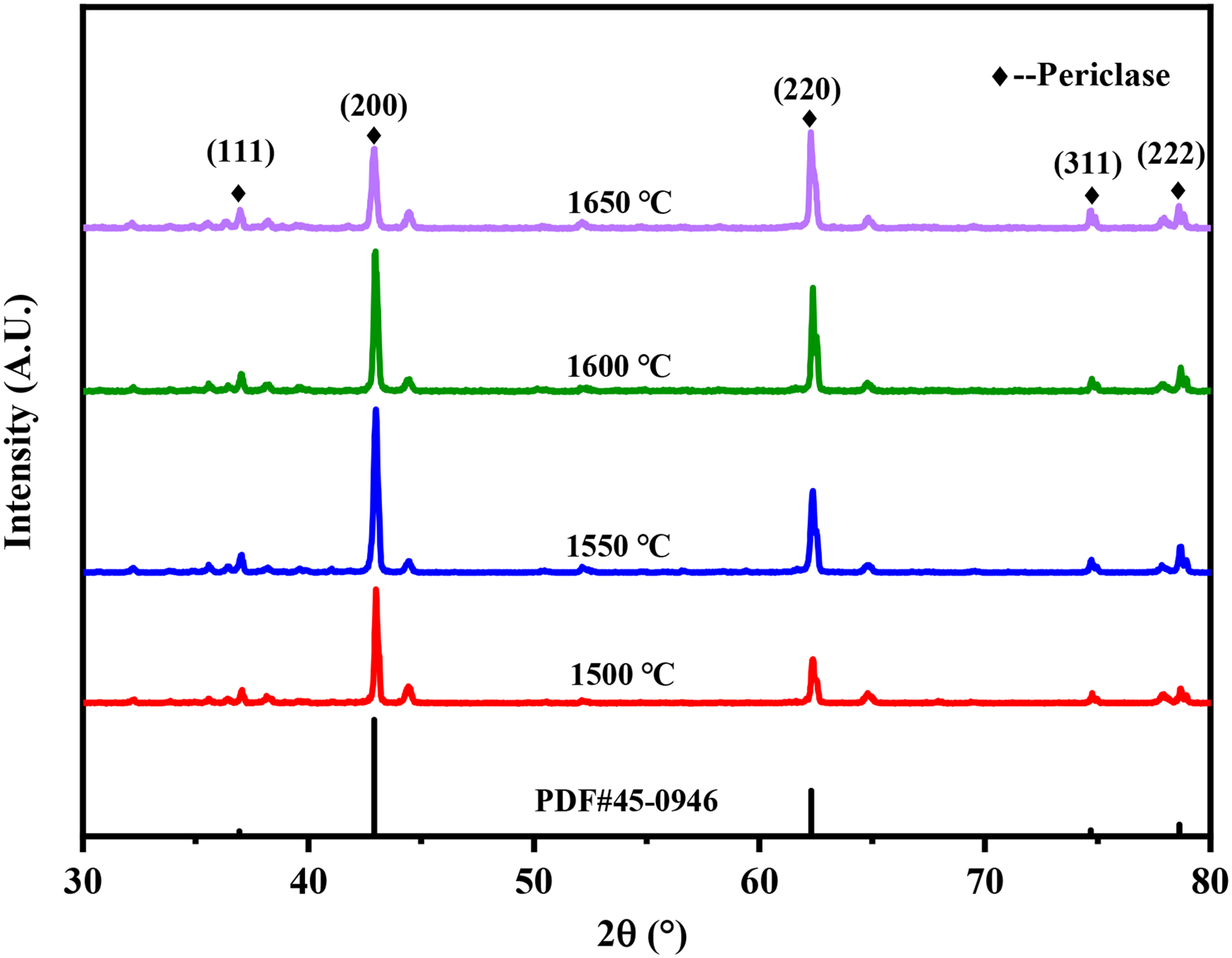
The XRD patterns of MgO porous ceramics with different sintering temperature.
Figure 4 shows the model of sintering process of MgO material. The microstructures and grain distribution of obtained MgO porous ceramics are shown in Figure 5. It can be found that within the range of 1500 to 1650 °C, the MgO porous ceramics exhibited different grain morphologies. As the sintering temperature became higher, the grain size of MgO was more uniform. When the temperature was 1600 °C, the crystalline grains were packed tightly and the intercrystalline pores were less than others. Because as the temperature increases, the provided Joule thermal energy increases. Therefore, in the middle stage of sintering, the rate and degree of densification increased. Since the activation energy of lattice diffusion is faster than that of grain boundary diffusion, the internal sintering mechanism of the ceramic sample changes the grain boundary diffusion to lattice diffusion, so the grain size increases significantly. 36 With the increase of temperature, the grain boundary migration rate increases and the atoms at the grain boundary rearrange faster, which contribute to the elimination of structural defects and energy differences on the grain boundary. And the grains are more uniform at this time.
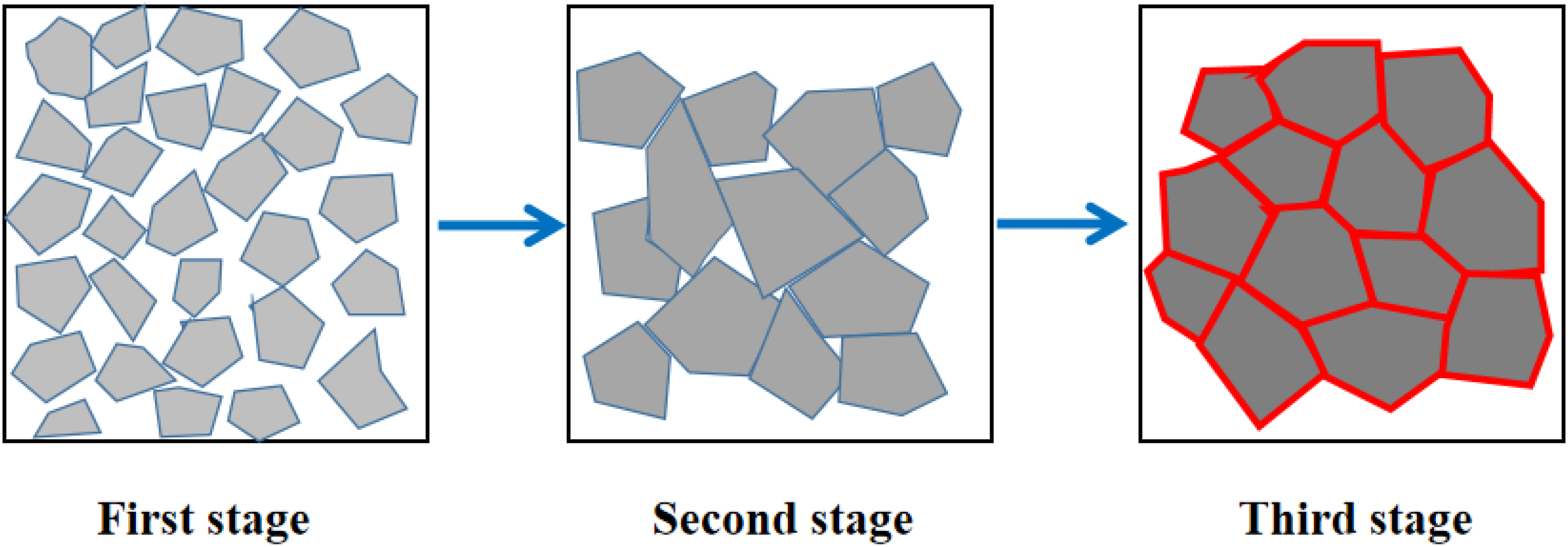
Model of sintering process of MgO material.
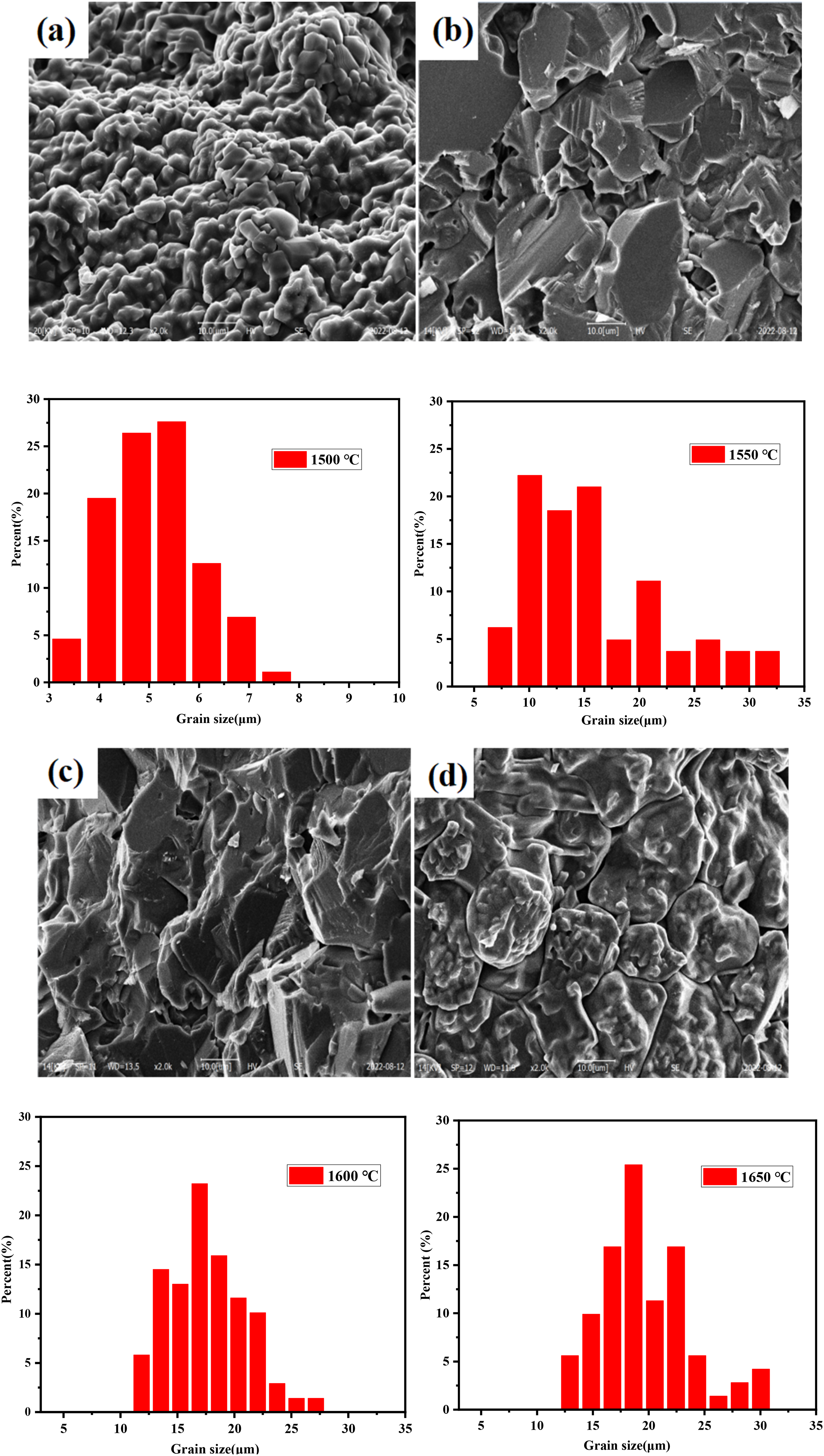
The SEM images of MgO porous ceramics with different sintering temperature: (a) 1500 °C, (b) 1550 °C, (c) 1600 °C and (d) 1650 °C.
Figure 6 shows the shrinkage, skeletal density and compressive strength with different sintering temperatures. According to Figure 4, the compressive strength of MgO porous ceramics increased from 0.63 to 0.96 MPa as the sintering temperature increased from 1500 to 1650 °C. At the same time, the shrinkage rate, skeleton density increase as the sintering temperature increased, and the rising trend slows down after 1600 °C. It could be inferred the optimum sintering temperature is 1600 °C. The result indicated that the sintering temperature have significant effect on the property of MgO porous ceramics.
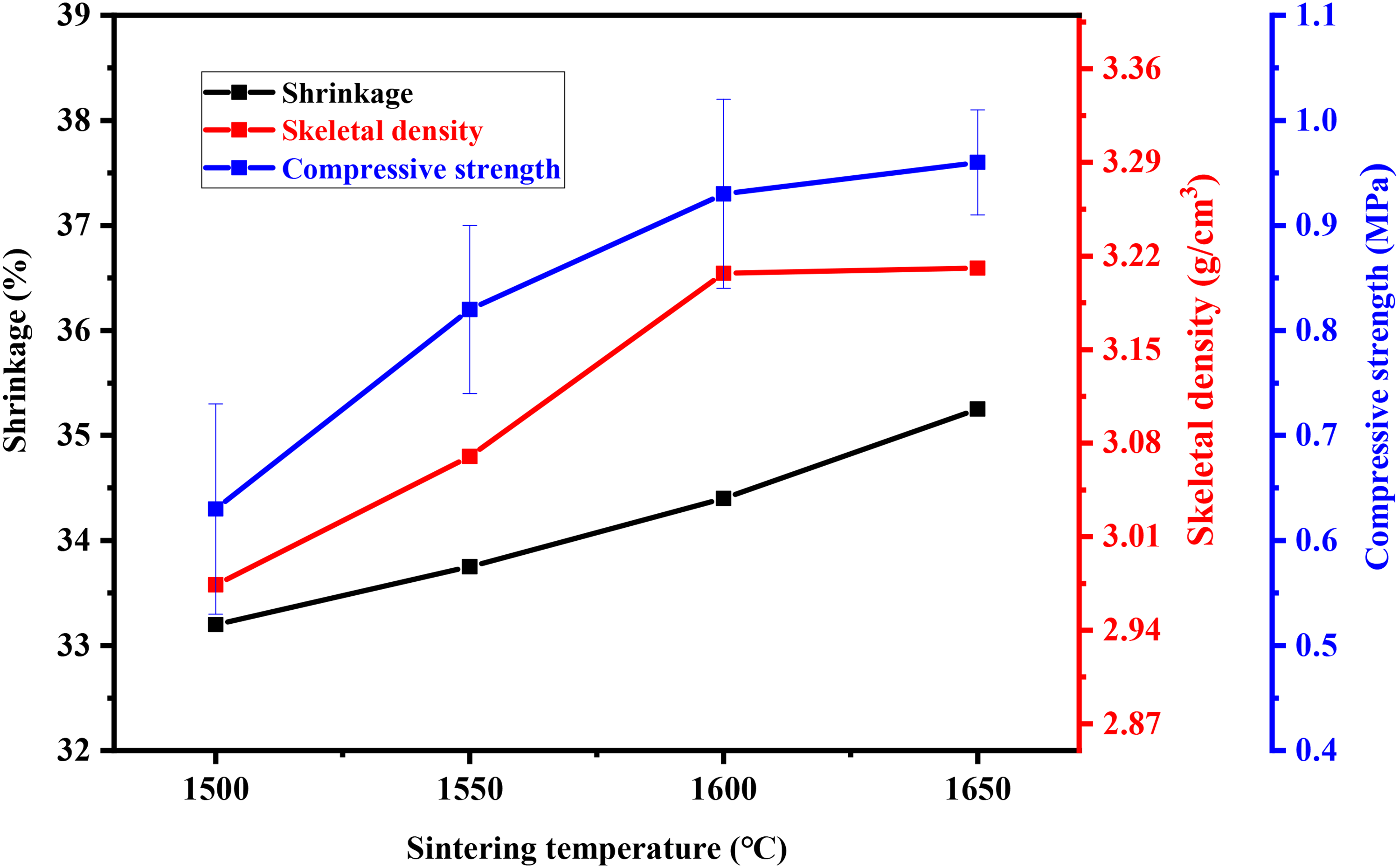
The shrinkage, skeleton density and compressive strength of MgO porous ceramics at different sintering temperatures.
Effect of PCE on MgO porous ceramics
It can be seen that the particle spacing in the slurry is inversely proportional to the solid phase content from Woodcock equation 37 (Equation (4)). As the distance between the particles decreases, the interaction intensifies. Therefore, the viscosity increases as the increase of solid content in the slurry, which is verified in Figure 7. When the solid content of the slurry increased from 55 to 62 wt-%, the viscosity kept increasing without the addition of dispersant. And the fluidity was poor when the solid content of slurry reached 62 wt-%. As is seen in Figure 8, in the range of solid content involved, the slurry distribution on the foam template is not uniform. At the same time, many cracks (circled in red) caused by drying exist and most of the holes are filled with slurry, which resulted in unclear structure.
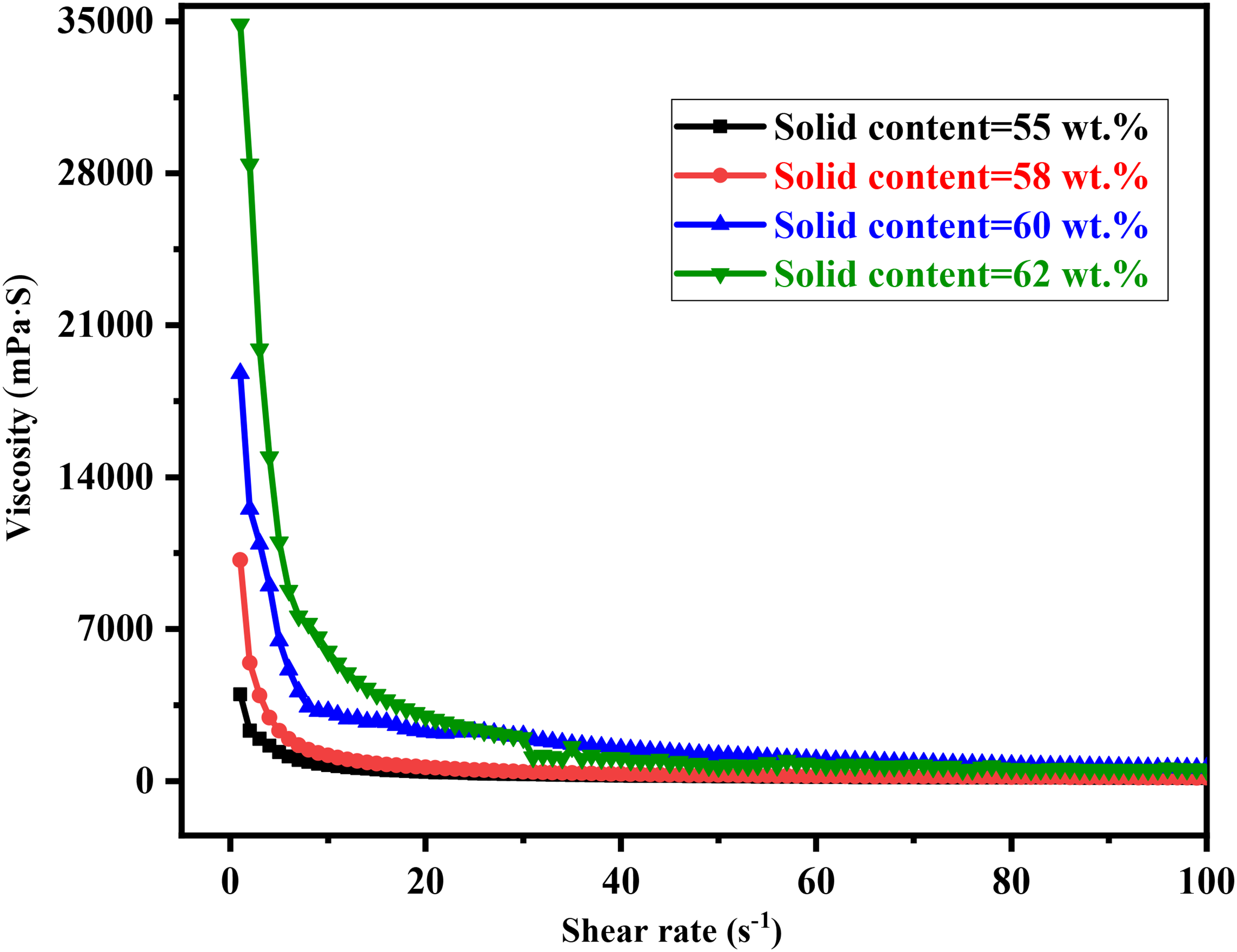
The viscosity of MgO slurries with different solid content.
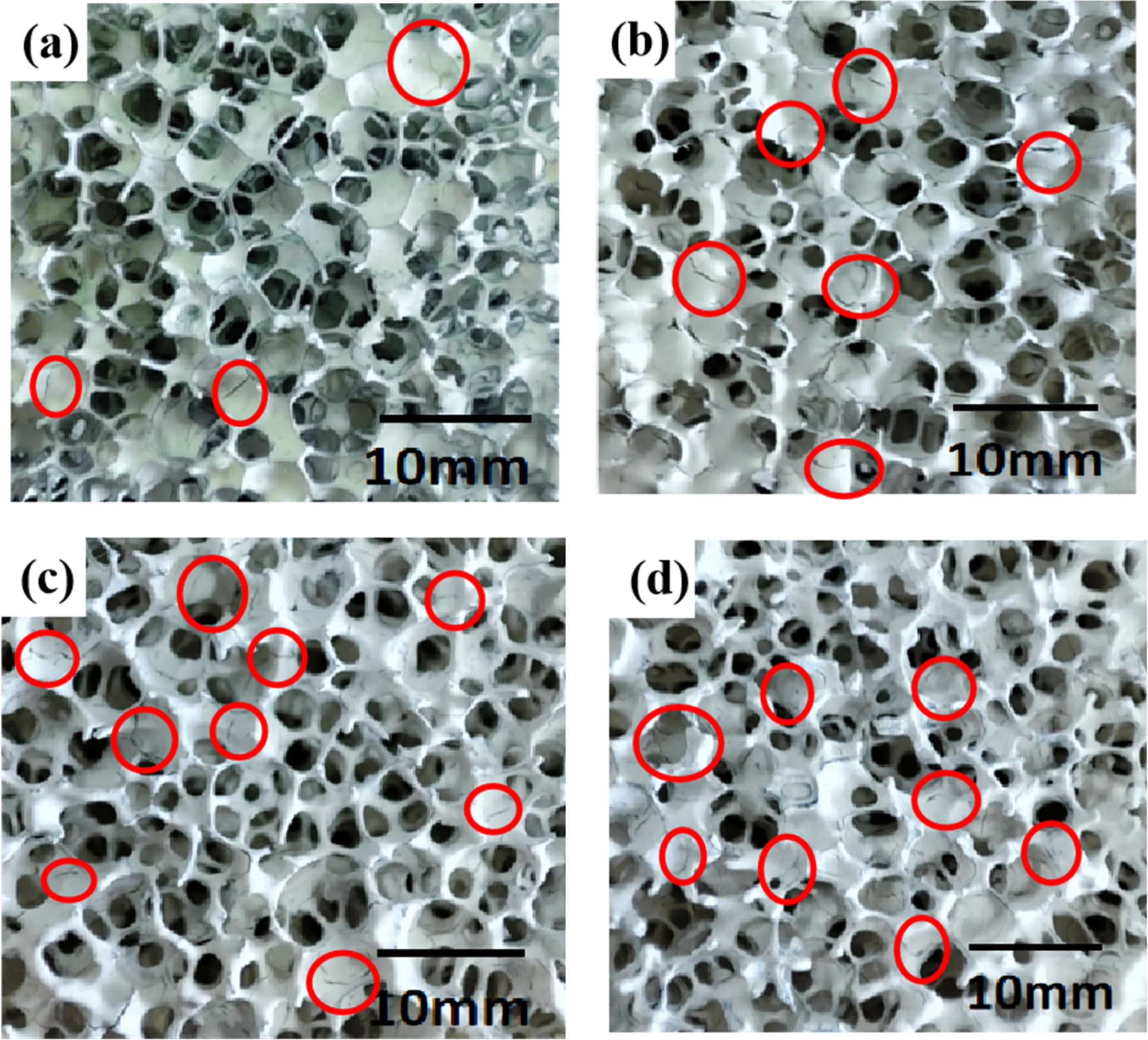
Optical photograph of MgO porous ceramic blank with different solid content: (a) 55 wt-%, (b) 58 wt-%, (c) 60 wt-% and (d) 62 wt-%.
In order to overcome the shortcomings above, anionic polyacrylamide (APAM), sodium hexametaphosphate (SHMP) and PCE were selected in this article. From the results shown in Figure 9(b), it could be seen that the potential of MgO particles was positive within the range of pH measured. 38 The addition of three dispersants led to the anionic group adsorb on the surface of the particle, which made its potential decrease (electrostatic repulsion effect, shown in Figure 9(a)). When the pH is about 10 (pH of saturated MgO slurry), the absolute value of the potential was at a high level relatively. We can conclude that the slurries with these dispersants are more stable than original slurry.
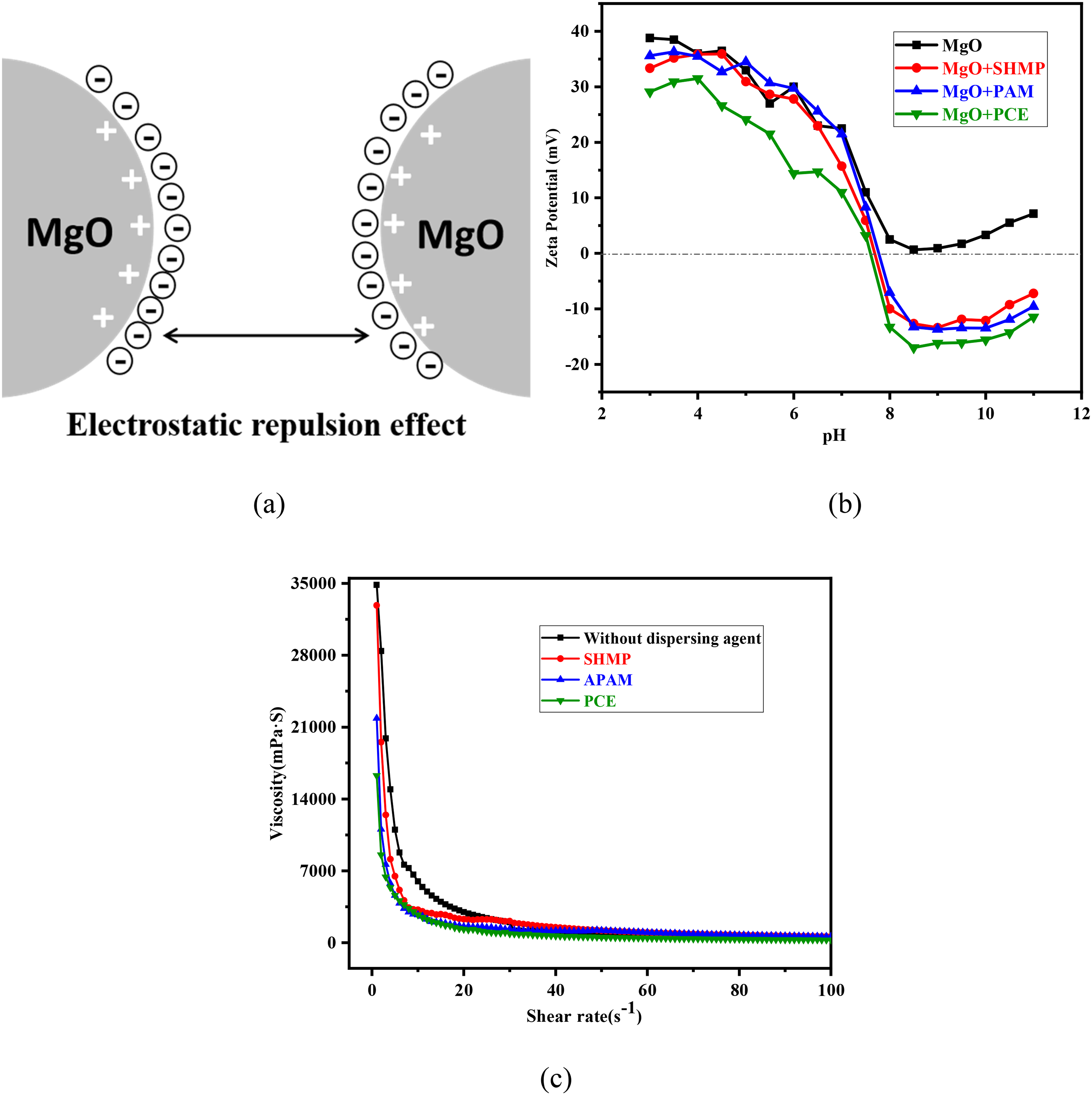
(a) Mechanism of electrostatic repulsion, (b) Zeta potential of MgO particles and (c) the viscosity of MgO slurries with different dispersants.
Then, the viscosity of different slurry was analysed. Due to the effect of electrostatic repulsion, the viscosity of slurries with three dispersions was reduced. Compared with APMA and SHMP, PCE dispersant with both electrostatic repulsion effect and steric hindrance effect increase liquidity effectively, as shown in Figure 9(c).
Figure 10(a) shows the action mechanism between MgO particles and PCE dispersant. In the absence of PCE, the powder will form a suspension system after mixing with water. And the MgO particles attracted each other and form a flocculation structure. The flocculating constituent could wrap a large amount of mixing water, which reduced the fluidity of the slurry.
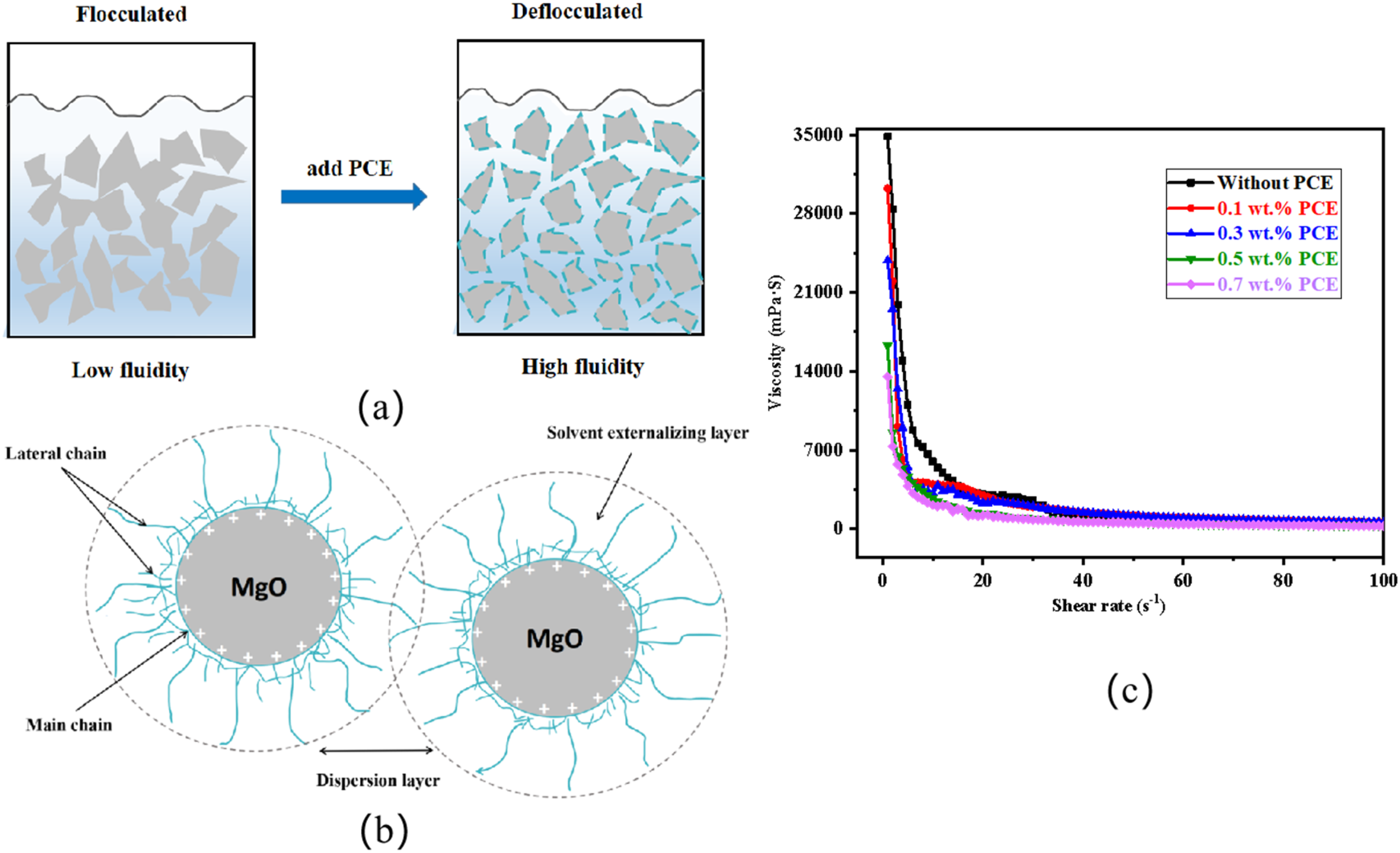
(a) The depolymerisation process after adding PCE dispersant, (b) mechanism of steric hindrance effect and (c) the viscosity of slurry with different content of PCE.
The main chain of the added PCE is adsorbed on the surface of MgO, and the side chain are extended into the liquid phase, which form a polymer molecular adsorption layer on the surface of particles. When the particles’ adsorption layer overlaps each other, the steric repulsion is generated. This is the steric effect, which is shown in Figure 10(b).
Figure 10(c) showed the viscosity of the slurries with different PCE contents. The viscosity of slurry obviously decreased with the increase of PCE content, and the amplitude decreased when the content was added to 0.5 wt-%. It could be inferred that the PCE content has a certain effect on the preparation of slurry. Figure 11 shows the optical photograph of porous ceramic blank with different PCE addition levels. When the content of PCE is 0.1–0.3 wt-%, the cracks on the skeleton were significantly reduced, but the slurry was still blocked in most of holes. As the addition of PCE was increased to 0.5–0.7 wt-%, the foam template could hang the slurry uniformly with few cracks. It is because the addition of PCE can be adsorbed on the surface of MgO particles. These particles have similarly electrical properties to repel each other. This interaction allows the excess water to be released between the particles to disperse the ceramic slurry better. Moreover, PCE could form water films on the surface of the ceramic particles, which effectively reduce hydration rate of the wet green body. Thus, the cracks, resulting from the evaporation of water, were reduced.

Optical photograph of porous ceramic blank with different content of PCE: (a) 0.1 wt-%, (b) 0.3 wt-%, (c) 0.5 wt-% and (d) 0.7 wt-%.
Figure 12 illustrates the effect of PCE content on the properties of MgO porous ceramics. As the content of PCE increased, the porosity increased and the thermal conductivity decreased gradually. Besides, due to the reduction of cracks in porous ceramics, the compressive strength of porous ceramics increased slightly. Because the addition of PCE reduced the cracks of ceramics and improved the compressive strength of MgO porous ceramics When the content of PCE is 0.5 wt-%, the bulk density of the sample is 0.412 g/cm3, the porosity is 88.5%, the compressive strength is about 1.6 MPa, and the thermal conductivity is 0.045 W/(m·K), respectively.
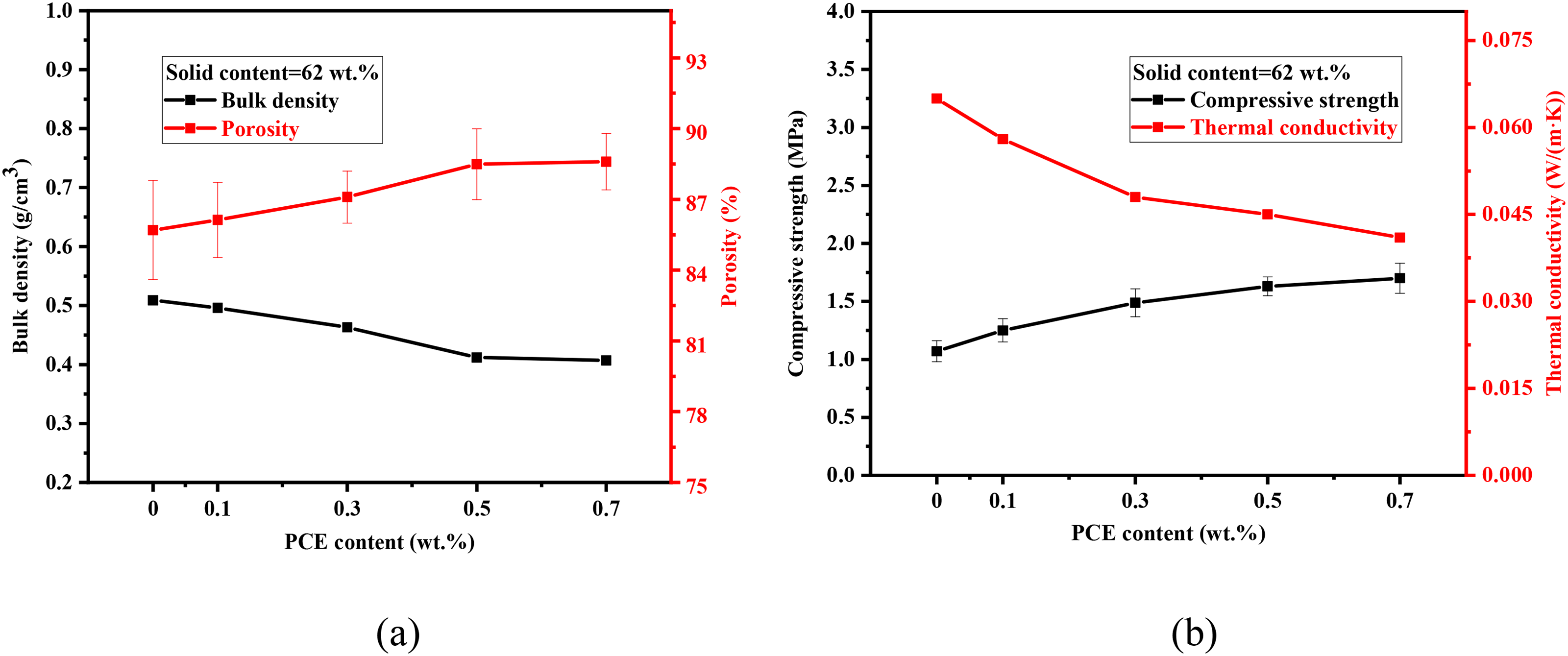
Properties of porous ceramics with different PCE content: (a) bulk density and porosity; (b) compressive strength and thermal conductivity.
Effect of organic foam template pore size on MgO porous ceramics
Figure 13 shows the optical photos and pore size distribution of MgO porous ceramics sintered with different organic foam template pore sizes. It could be seen that the structure of the MgO porous ceramics prepared by larger pore sizes (15PPI, 20PPI) was clear. Apart from this, the aperture distribution is concentrated when the organic foam template pore sizes were 25PPI, 30PPI and 40PPI. However, with the pore size decreased, the holes were partly filled with slurry due to the influence of surface tension. Thus, with the reduction of template pore size, the porosity of the MgO porous ceramics decreased as shown in Figure 14. In general, porous ceramic compressive strength and thermal conductivity were sensitive to the porosity. The initial effective area became smaller with the pore size and porosity larger. Under a certain load, the compressive stress increased, and the load borne by each porous body decreased which led to the decreasing strength. The heat conduction capacity of the solid substance in the material was much larger than that of the air. Accordingly, as the porous material bulk density was large due to the low porosity, the thermal conductivity increased. When the template was 25 PPI, the comprehensive performance is better. The sample bulk density is 0.459 g/cm3, the porosity is 87.5%, the compressive strength is about 1.5 MPa, and the thermal conductivity is 0.047 W/(m·K). As the organic foam template is 40PPI, the porosity of obtained MgO porous ceramics was low, and the heat-insulating property was poor than others. The sample porosity is about 83.0%, the compressive strength is about 2.3 MPa, and the thermal conductivity is about 0.077 W/(m·K).
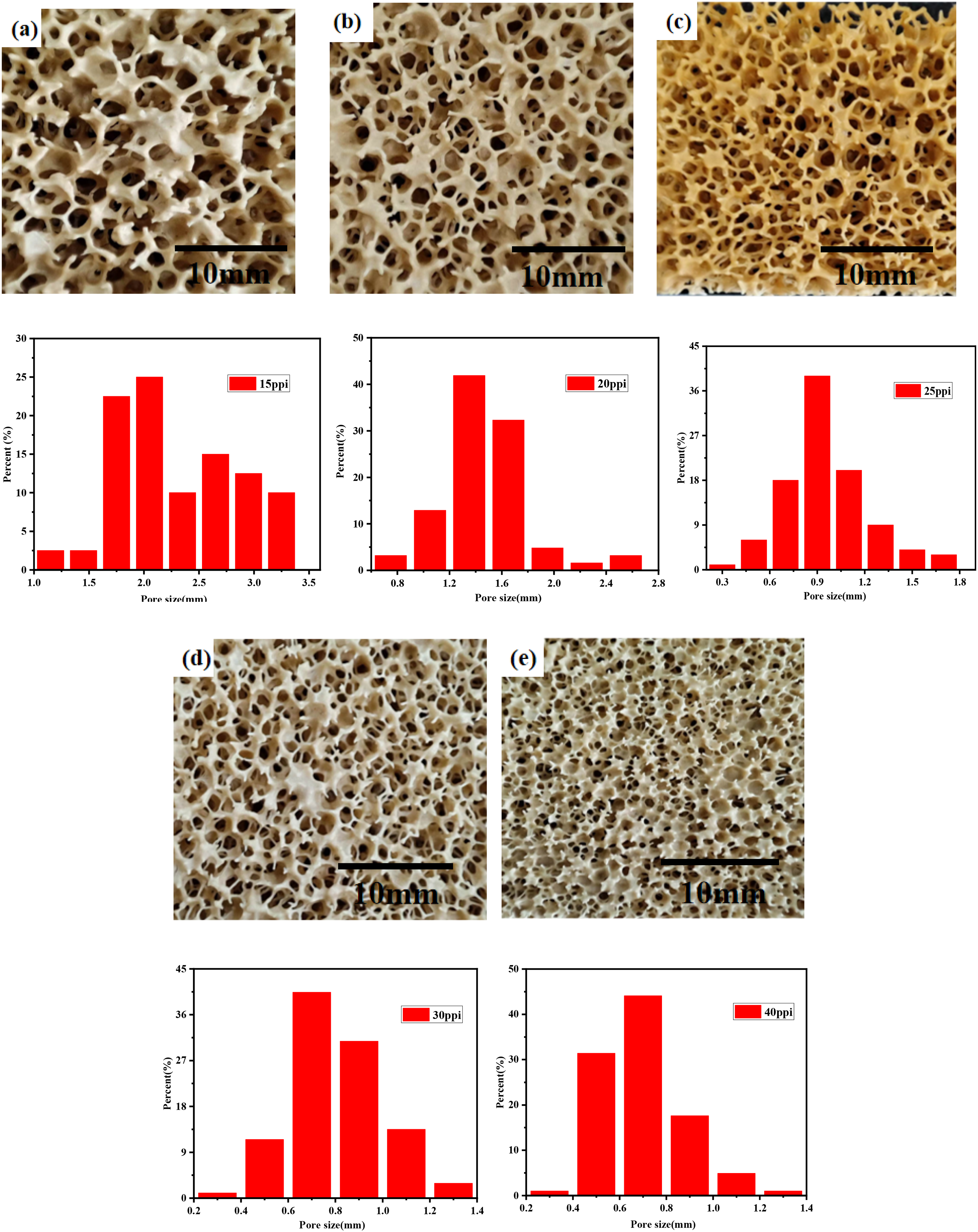
Optical photographs of MgO porous ceramics prepared by different pore sizes of organic foam template: (a) 15PPI, (b) 20PPI, (c) 25PPI, (d) 30PPI and (e) 40PPI.
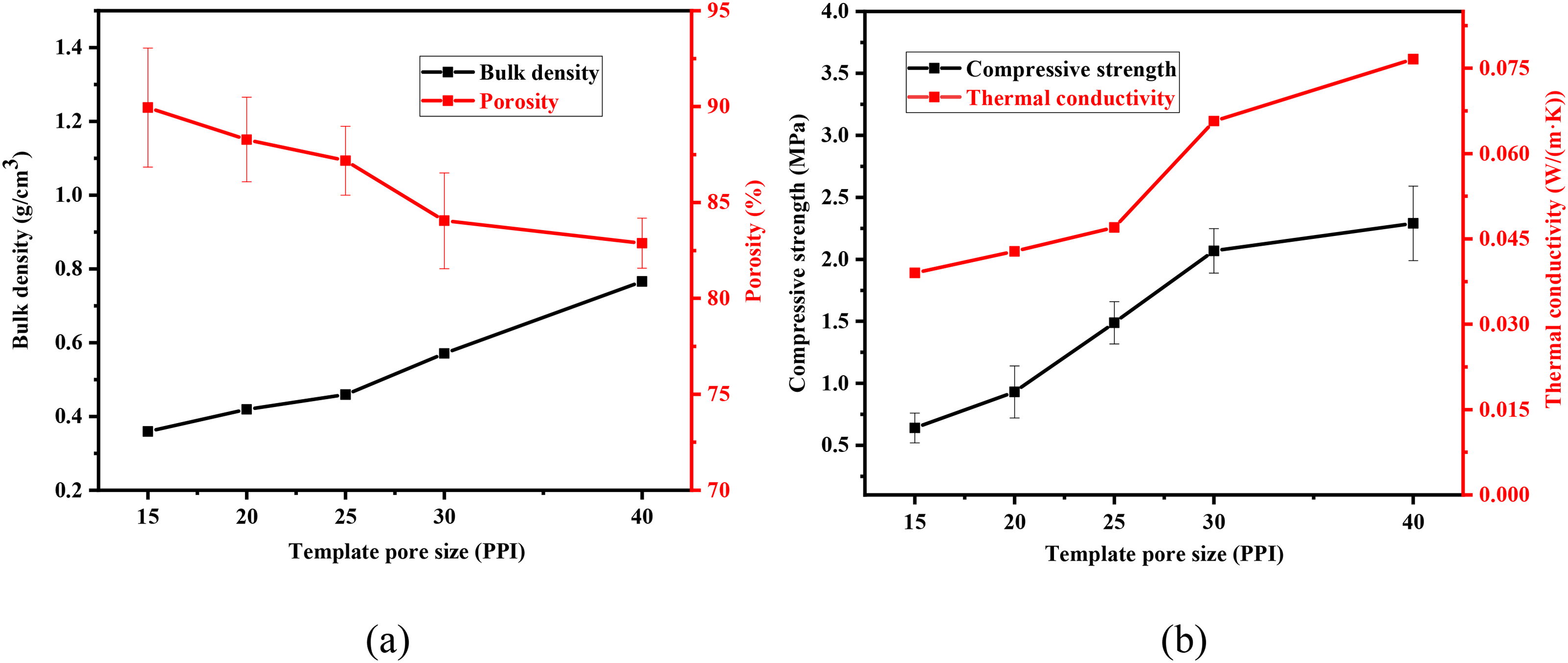
Properties of porous ceramics with different pore sizes of organic foam template: (a) bulk density and porosity and (b) compressive strength and thermal conductivity.
The above results show that the pore size of the template could greatly affect the porosity, compressive strength and heat-insulating property of MgO porous ceramics prepared by organic foam impregnation.
Conclusion
As mentioned above, a series of MgO porous ceramics were prepared by foam impregnation method. In a certain range, the shrinkage rate, skeleton density and compressive strength of porous ceramics increased with the increase of sintering temperature. Compared APAM and SHMP, PCE dispersant could change the surface state of MgO particles in slurry, which not only made the porous ceramic skeleton clear, but also effectively reduced the surface cracks of porous ceramic blank. For MgO porous ceramics samples added with 0.5 wt-% PCE, the bulk density is 0.412 g/cm3, the porosity is 88.5%, the compressive strength is about 1.6 MPa, the thermal conductivity is 0.045 W/(m·K). Moreover, this article points out the influence of templates with different pore sizes on MgO porous ceramics. As the single pore size of template increased, the prepared ceramics porosity increased, the compressive strength and the thermal conductivity decreased. When the template reached 25 PPI, the comprehensive performance was better. The sample bulk density is 0.459 g/cm3, the porosity is 87.5%, the compressive strength is about 1.5 MPa, and the thermal conductivity is 0.047 W/(m·K). The porous ceramics with corresponding pore size could be prepared according to the different requirements in the actual application.
Footnotes
Author contributions
All the authors have equally contributed to the work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the Natural Science Foundation of Shanghai (Grant no. 19ZR1418500).
