Abstract
Objectives
Dyadic interventions, involving two persons with a preexisting close relationship, offer the opportunity to activate support persons (SPs) to improve health for adults with chronic conditions. Requiring SP coparticipation can challenge recruitment and bias samples; however, the associations between voluntary SP coparticipation and recruitment outcomes across patient characteristics are unknown.
Methods
The Family/Friend Activation to Motivate Self-care 2.0 randomized controlled trial (RCT) enrolled adults with Type 2 diabetes (T2D) from an academic health system. Participants were asked—but not required—to invite an SP to coenroll. Using data from the electronic health record we sought to describe RCT enrollment in the setting of voluntary SP coparticipation.
Results
In a diverse sample of adults with (T2D) (48% female, 44% minoritized race/ethnicity), most participants (91%) invited SPs and (89%) enrolled with SPs. However, prerandomization withdrawal was significantly higher among participants who did not have consenting SPs than those who did. Females were less likely to invite SPs than males and more Black PWD were prerandomization withdrawals than randomized.
Discussion
Voluntary SP coenrollment may benefit recruitment for dyadic sampling; however, more research is needed to understand if these methods systematically bias sampling and to prevent these unintended biases.
Introduction
Support persons (SP) are friends and family involved in chronic condition management and are critical to the care of the growing population of adults with chronic conditions.1,2 Chronic conditions impact almost half (45%) of Americans3,4 and are the most costly health conditions in the United States.5,6 Chronic conditions can lead to excessive use of the medical system, long-term disability, and reduced quality of life.7–9 Furthermore, the incidence of chronic conditions is expected to continue to rise. 10 Diabetes is one such chronic condition in which dyadic interventions, involving persons with chronic conditions and their SPs, may be beneficial. Diabetes impacts over 34 million Americans.11,12 For persons with diabetes (PWDs), SP involvement is associated with positive outcomes related to diet, exercise, medications, and glucose monitoring.13–15 As a result, there have been efforts to intervene with dyads to improve diabetes outcomes and relationship quality.
While including SPs in chronic condition management has advantages; requiring SP enrollment has setbacks. In dyadic intervention studies for adults with dementia, strict inclusion criteria for the targeted individual and their SP limited enrollment.16,17 Likewise, dyadic interventions for Type 2 diabetes (T2D) often require coenrollment of an SP. Studies requiring coenrollment of an SP, including studies among persons with T2D, resulted in up to one-third of potentially eligible participants being excluded.18,19 Instead of requiring SP enrollment, some studies have made SP enrollment optional. However, studies that made SP coenrollment optional had relatively few PWDs participate with an SP. For instance, in a study with Veterans with T2D and out-of-home SPs, only 39% enrolled with an SP, and attrition was higher among those without SPs. 20 In our own prior work, 48% of PWDs enrolled SPs when given the option, and those who declined cited either concern about adding burden to their SP or not having someone to invite. 21 Limited data on differences between PWDs who do and do not invite SPs are inconsistent; PWDs who invite SPs may have lower health literacy and income 20 or may be younger, more likely to be married and report higher depressive symptoms. 21 The challenges of recruiting dyads are well-documented.16,17,22–24 Therefore, more work describing the recruitment of dyads is needed to optimize the enrollment of SPs without alienating or excluding individuals who do not have an SP readily available.
Family/friend Activation to Motivate Self-care (FAMS) is a mobile phone-delivered intervention designed to help PWDs set and meet health goals and improve family/friend involvement in self-management. 25 FAMS consists of daily automated tailored text messages and monthly coaching calls, plus tailored text messages sent to an SP if enrolled. During recruitment to the FAMS 2.0 randomized controlled trial (RCT), PWDs were asked to invite an SP, although SP enrollment was not a requirement. PWD were then randomized to FAMS 2.0 or control with their SP if enrolled. Within the context of recruitment, we described our recruitment strategies and the associations between SP invitation outcomes (PWDs with a consented SP, PWDs who invited SPs but enrolled alone, and PWDs who did not invite SPs), randomization status (randomized vs. prerandomization withdrawal), and patient characteristics to represent the scope of prerandomization withdrawals in a dyadic sample when SP coenrollment was optional.
Methods
Participants and procedures
All procedures were approved by the Vanderbilt University Institutional Review Board (#190905) and the RCT is registered with ClinicalTrials.gov (NCT04347291).
PWD eligibility criteria
We recruited adults receiving outpatient care for T2D at Vanderbilt University Medical Center (VUMC) primary care clinics in Nashville, TN. Potentially eligible PWDs needed to be between 18 and 75 years old with a T2D diagnosis, had at least one VUMC primary care clinic visit in the last year, and a recent hemoglobin A1c (HbA1c) value ≥ 7.5% within the last 183 days in the electronic medical record (EMR). In addition, PWDs needed to be community-dwelling (e.g., not residing in a nursing facility), be prescribed at least one daily diabetes medication, possess a mobile phone with texting capabilities, and be able to speak and read in English. They were not required to have a coenrolling SP. We excluded PWDs who, per the EMR, appeared to be using hospice or dialysis services, had congestive heart failure, were currently undergoing treatment for cancer, were pregnant, had been diagnosed with dementia or schizophrenia, or disclosed ongoing emotional, physical, or sexual abuse. We also excluded PWDs who were unable to communicate by phone or unable to receive and respond to text messages after training by a research assistant (RA), as they would be unable to participate in the intervention if assigned.
PWD recruitment and informed consent
Recruitment efforts used EMR data to prioritize the identification of PWDs with HbA1c ≥ 8.0%, those of minoritized race or ethnicity, or those with no insurance or public insurance (as a proxy for lower socioeconomic status). Following identification in the EMR, all procedures were identical across potential participants. We mailed potential participants a recruitment letter and study brochure reviewing the study information and eligibility criteria. The materials also informed them that the study involved a family member or friend participating alongside them. PWDs were asked to contact the study team if interested or to opt out. After waiting a week, RAs called PWDs once weekly for four weeks. Once connected, RAs briefly described the study and confirmed eligibility via phone for those interested. RAs then obtained verbal informed consent and administered the two-item Diabetes Distress Scale as a screener to establish high/low distress for randomization purposes. 26 If either item was endorsed as a “moderate problem” or greater, PWDs were classified as distressed.
SP invitation and informed consent
A priori decisions regarding how SPs were recruited were made to maximize SP enrollment. First, consented PWDs were asked to identify and share the contact information of someone to coparticipate with them as their SP, defined as any family member or friend with whom they felt comfortable discussing diabetes management and health goals. Second, we used broad inclusion criteria for SPs. Eligible SPs needed to be at least 18 years of age, able to speak and read in English, and own a mobile phone with text messaging capabilities separate from the PWD's mobile phone. The only exclusion criterion for SPs was the inability to receive and respond to text messages after training by RAs. Cohabitation and relationship type (e.g., spouse, family) were not eligibility criteria.
Third, RAs attempted to reach identified SPs via phone twice a week for up to four weeks until they enrolled or declined, removing the burden from PWDs to describe the study to their SPs. If interested, RAs verified identified SPs’ eligibility and obtained verbal informed consent. Fourth, PWDs were given multiple opportunities to invite an SP during the run-in period allowing them to discuss the opportunity with the SP prior to RAs contacting them. If the first identified SP declined, PWDs were free to invite another SP until one consented or they declined to invite another. PWDs who declined to invite an SP were told they could still participate and given further opportunities to invite an SP. Finally, the intervention placed few demands on SPs (no in-person study components, did not attend intervention with PWD).
Run-in period and randomization
Following consent, all participants (PWDs and SPs) were sent an enrollment (baseline) survey via online REDCap link, US mail, or phone. HbA1c kits were also mailed to PWDs which they were required to return (followed per tracking number) prior to randomization. Baseline data collection was completed prior to randomization and assessed by blinded personnel. Those who did not complete baseline data despite RA weekly contact attempts (calls, emails, and texts) for up to five weeks, were administratively withdrawn. Upon baseline data completion, PWDs and SPs, if enrolled, were randomized to intervention or control with a 1:1 ratio using blocked randomization stratified by key variables for the larger RCT: SP enrollment, recent EMR HbA1c, and high/low diabetes distress. RAs called PWDs via phone to inform participants of what to expect during the study based on the study condition assignment. 27 PWDs were considered randomized once they completed the condition assignment call. RAs attempted condition calls twice a week for three weeks, leaving a voicemail to inform unresponsive PWDs that they must complete this call to continue in the study. PWDs who were unreachable after three weeks were administratively withdrawn and not considered participants in the RCT.
Measures, data handling, and analyses
Demographic and clinical data were extracted from the EMR for all consenting participants including gender, age, race/ethnicity, insurance status, and recent (within 90 days of consent) HbA1c, if available. Therefore, HbA1c data could be missing due to failed HbA1c kits or no recent HbA1c in the EMR. Process data was collected by RAs during the recruitment and consent process, including number of days from initial contact to consent, SP invitation outcome, and randomization status. We used chi-squared tests to describe differences in randomization status by SP invitation outcome. Nonparametric tests were used to compare PWD characteristics across SP invitation outcomes and across randomization status.
Results
Descriptive statistics for the overall sample and across SP invitation outcome and randomization status are in Table 1. HbA1c was missing not at random across SP invitation outcome and randomization status; PWDs without an SP (χ2(2) = 16.1, p < .01) and PWDs who were prerandomization withdrawals (χ2(1) = 101.3, p < .01) were less likely to have HbA1c data. Therefore, results for HbA1c should be interpreted with caution. All other variables had minimal missingness (≤ 0.5%).
Sample characteristics.
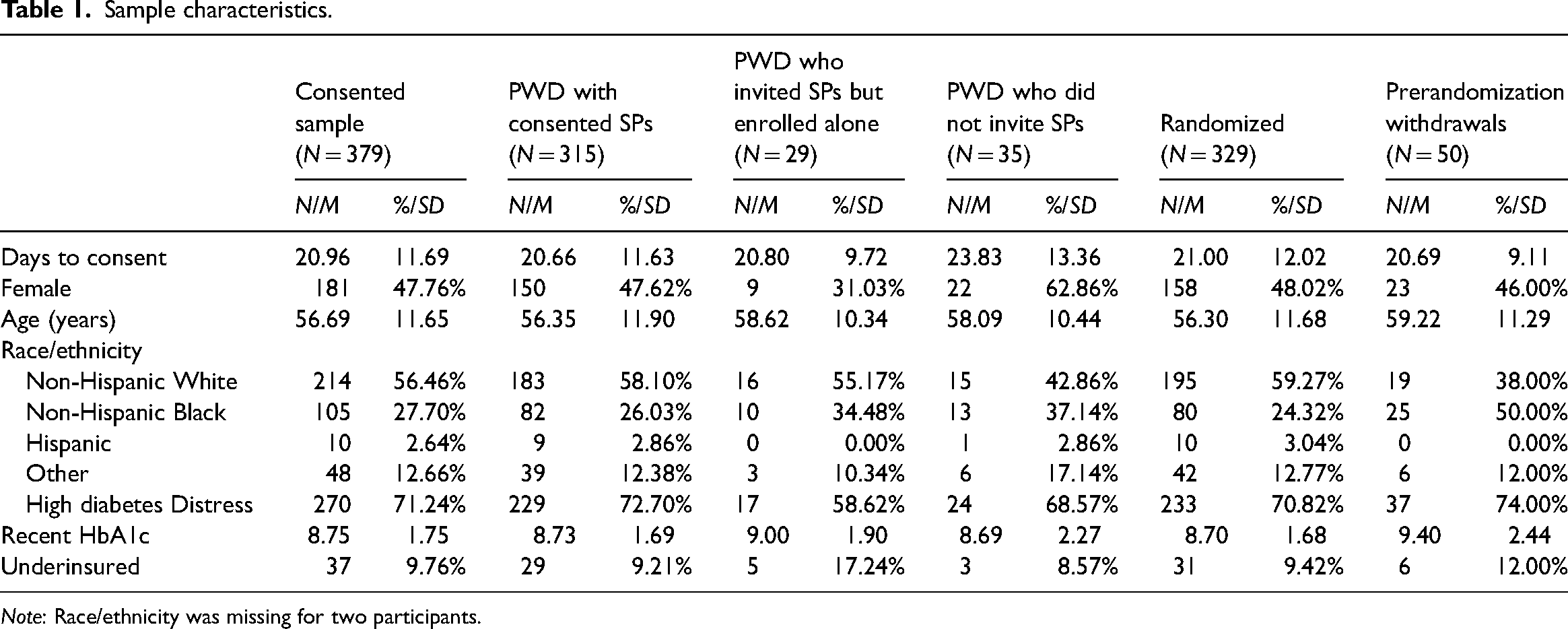
Note: Race/ethnicity was missing for two participants.
Differences in prerandomization withdrawal by SP invitation outcome
Of N = 379 consented PWDs, 344 (90.8%) invited an SP to coenroll. Almost all invited SPs consented to participate (315/344; 91.6%). PWD who did not have a consenting SP were significantly more likely to withdraw prerandomization than PWD with a consenting SP (Χ2(2) = 74.8, p < .01). As shown in Figure 1, prerandomization withdrawals were comparatively unlikely among PWDs whose invited SP consented to participate (21/315; 6.7%). In contrast, around one-third of PWDs whose invited SP did not consent were prerandomization withdrawals (10/29; 34.5%) and over half of PWDs who did not invite an SP were prerandomization withdrawals (19/35; 54.3%).
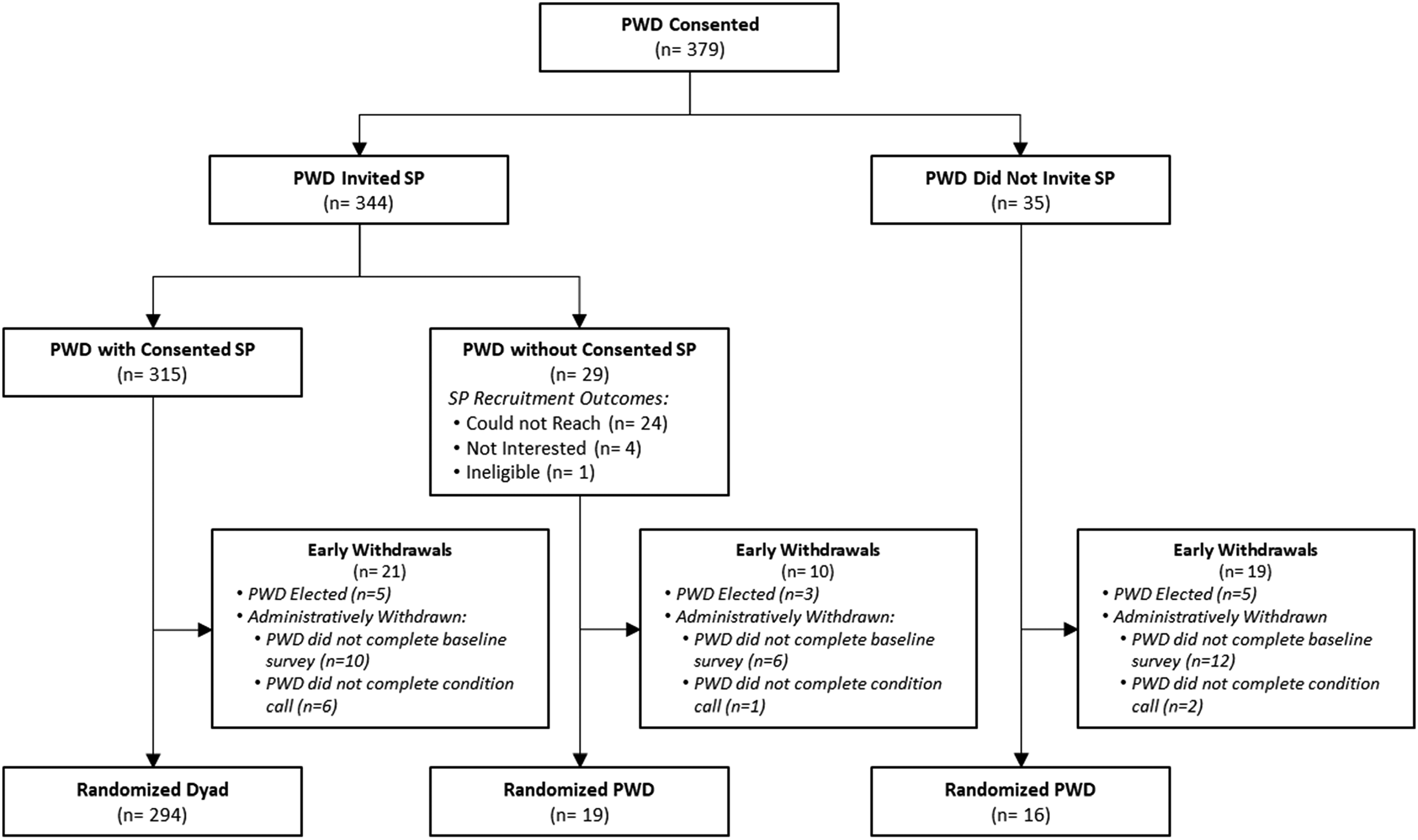
Consort diagram by support person group.
SP invitation outcome differences
Across the three SP invitation outcomes (PWDs with consented SPs, PWDs who invited SPs but enrolled alone, and PWDs who did not invite SPs), there was no evidence of differences in PWD time to consent, age, race/ethnicity, insurance, diabetes distress, or HbA1c value (ps > .24). More females did not invite a SP (22/35; 62.9%) than invited an SP who consented (150/315; 47.6%) or invited a SP but enrolled alone (9/29; 31.0%; Fischer Exact = 6.4, p = .04).
Randomization status differences
There was no evidence of differences in PWD time to consent, gender, insurance, diabetes distress, or HbA1c (ps > .27) among PWDs who were randomized versus prerandomization withdrawals. There were differences by race such that more black PWDs withdrew prior to randomization (25/50; 50.0%) than were randomized (80/329; 24.3%; Fisher's Exact = 13.48, p = .003). Additionally, there was a nonsignificant trend for randomized PWDs to be younger (M = 56.3, SD = 11.7) than those who withdrew prior to randomization (M = 59.2, SD = 11.3, p = .08).
Discussion
Asking PWD to invite an SP, but still allowing PWDs to enroll with or without an SP, during a run-in period for the FAMS 2.0 RCT resulted in 89% of the sample participating as dyads. Indeed, of consented PWDs, more than 90% invited an SP and only 13% of the consented sample withdrew prior to randomization. These observations highlight that our recruitment strategies supported the enrollment of a high number of consented PWDs without substantially diminishing the number of dyads enrolled. In contrast, research studies that require SP coenrollment or place restrictions on SP relationships (e.g., cohabitating, family member) may create biases in or limit the generalizability of samples by excluding individuals who do not have readily available SPs. Prior research efforts that requested adults with cardiovascular disease to explain the study to their caregiver led to higher rates of declining to invite SPs. 28 Notably, about one-third of adults who declined cited not wanting to add a burden to their caregiver. 28 Similar results were found in our prior work with the FAMS intervention when we introduced SP enrollment as a personal preference: around half did not invite a support person and those who did not invite an SP commonly cited their concern about burdening the SP or simply not having someone to invite. 21 Our five strategies (asking PWD to invite an SP rather than asking if they'd like to invite a SP, inclusive SP definition, multiple invitation opportunities, having RAs recruit the SPs, and low SP demands during the intervention) likely decreased the demand which appears to have aided SP enrollment.
There were differences in randomization status (randomized vs. prerandomization withdrawal) by SP invitation outcome. Individuals who declined to invite SPs were more likely to withdraw prior to randomization. They may have had limited social support already. Based on recruitment materials and the request to enroll an SP, they may have felt that the study was not a good fit for them. Further exploration of what drove these prerandomization withdrawals is needed, as is the development of alternative interventions that are potentially individually focused or perhaps aimed at increasing social support. Unfortunately, small sample sizes in several cells prevented us from examining the full factorial of differences by SP group and randomization status; however, collection of participant experiences qualitatively could fill in these gaps.
There were differences between PWDs who invited SPs versus those who did not. Females were less likely to invite an SP. Given females on average have larger social networks than males,29,30 their reticence to invite an SP may be due to fear of burdening their friends/families or to a lack of capable SPs. Prior work enrolling SPs for adults with T2D has mostly been within the Veteran Affairs system with samples that were largely (95+%) male20,31 inviting mostly female supporters. 31 The gender diversity of the current sample highlights important differences when considering SP invitations. Within the National Diabetes Prevention Program, there were benefits for males who participated with another person sharing their address but not for women who participated with another person sharing their address, 32 suggesting there may be gender differences in who benefits from having a coenrolled SP. However, previous analyses of FAMS did not find evidence of gender differences in changes in family/friend involvement or behavior change. 33 Therefore, there may be areas of opportunity in dyadic research to assist women in identifying SPs, or to offer alternatives such as peer support. Unlike prior research on FAMS that found participants who invited SPs were younger than those who did not, 21 we did not find evidence of age differences by SP invitation outcomes. There were nonsignificant patterns suggesting potential variability in SP invitation by racial and ethnic backgrounds, but more work is needed to draw conclusions. We also did not find differences in HbA1c; however, HbA1c data were not missing at random. Therefore, conclusions about differences, or lack thereof, should be considered cautiously.
We also found that prerandomization withdrawals during recruitment for an RCT are an issue worth noting. Prerandomization withdrawals are rarely studied and their impact upon sampling bias is largely unknown. A review of studies for pediatric chronic conditions found the mean prerandomization withdrawal rate was 37% while the mean attrition rate following randomization was 20%. 34 Attrition, defined as failing to complete assessments or study components following enrollment, is a well-documented threat to validity in research studies.35,36 Attrition after randomization undermines the protection that randomization provides against validity threats. However, attrition omits how early (prerandomization) withdrawals during recruitment might ultimately impact the validity and generalizability of results. Similarly, there may be other elements in recruitment materials and processes (e.g., study names, advertising, descriptions by staff) that bias sampling; randomization cannot overcome those threats to generalizability.
Limitations
This work is descriptive and does not have a comparator or control group; future work could systematically vary recruitment methods and features of the run-in period to test the five recruitment strategies discussed here, possibly in a factorial experiment to provide insight into how the strategies function independently and interact with each other. Data was collected as a part of a larger RCT and these secondary analyses were therefore subject to missingness and small sample sizes in some groups which limited our ability to fully describe differences between groups. However, there is an opportunity for other researchers implementing RCTs and other experimental designs to collect data during the recruitment process to replicate and extend the work described here.
Conclusions
Our work highlights the tension between competing goals in dyadic intervention research. On the one hand, we wish to maximize the number of participants who have an SP participate alongside them. On the other hand, finding an SP to participate can feel burdensome to the PWD or simply be impossible. Our study used five strategies to balance this tension that appeared to pay off. First, we encouraged PWDs to identify SPs and provide contact information for them. However, this was not a requirement and we also welcomed PWDs when SPs were not available. Second, rather than asking the PWD to fully inform and enroll the SP, our RAs provided this service. Additionally, our inclusive SP definition, multiple invitation opportunities, and low SP demands likely aided enrollment. The use of a run-in period was another feature of our trial which had several benefits, most notably reducing prerandomization withdrawals, but may have also increased barriers to entry. More barriers to entry may benefit research design but may be associated with more differential fallout, especially for minoritized racial groups. These strategies would benefit from additional research in hopes of offering even further insight into continuous improvement of recruitment and enrollment procedures to maximize both the internal and external validity of findings as well as the health equity of this work.
Footnotes
Acknowledgements
This work was presented to the Association of Behavioral and Cognitive Therapy Annual Conference, November 2022.
Contributorship
MKR contributed to conceptualization, formal analysis, and writing original draft. ME contributed to writing original draft and data curation. LL contributed to data curation. LSM contributed funding acquisition, project administration, and reviewing & editing. JEA, RQW, and RAG contributed to methodology and reviewing & editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Vanderbilt Faculty Research Scholars, U.S. Department of Veterans Affairs, National Institute of Diabetes and Digestive and Kidney Diseases (grant number na, Office of Academic Affiliations, R01DK119282).
Ethical Approval
Ethical approval for this study was obtained from Vanderbilt University Institutional Review Board (#190905).
Guarantor
LSM
