Abstract
Purpose
To explore the association between the degree of Chronic illness management and survival rates at 1-, 3-, 5-years post heart transplantation.
Methods
Exploratory secondary analysis of a cross-sectional, international study (Building Research Initiative Group study). Latent profile analysis was performed to classify 36 heart transplant centers according to the degree of chronic illness management.
Results
The analysis resulted in 2 classes with 29 centers classified as “low-degree chronic illness management” and 7 centers as “high-degree chronic illness management”. After 1-year posttransplantation, the high-degree chronic illness management class had a significantly greater mean survival rate compared to the low-degree chronic illness management class (88.4% vs 84.2%, p = 0.045) and the difference had a medium effect size (η2 = .06). No difference in survival for the other time points was observed. Patients in high-degree chronic illness management centers had 52% lower odds of moderate to severe drinking (95% confidence interval .30–.78, p = 0.003). No significant associations between degree of chronic illness management and the other recommended health behaviors were observed.
Conclusions
The findings from this exploratory study offer preliminary insight into a system-level pathway (chronic illness management) for improving outcomes for heart transplant recipients. The signals observed in our data support further investigation into the effectiveness of chronic illness management-based interventions in heart transplant follow-up care.
Introduction
Despite the significant improvement in physical function and quality of life posttransplantation, 1 heart transplant (HTx) recipients are still considered to have a chronic condition since they have to adhere to a life-long complex therapeutic regimen and regular medical follow-up to monitor graft function and prevent or treat co-morbidities. 2 Given the multifaceted nature of the post-transplant regimen and the impact of behavioral and psychosocial factors on HTx outcomes,1,3 HTx recipients would greatly benefit from a comprehensive follow-up care that not only addresses biomedical parameters but psychosocial and behavioral functioning as well.4,5
Chronic illness management (CIM) is an innovative approach to transplant follow-up care that could improve the health outcomes of HTx recipients. 4 Unlike the prevailing acute care model, wherein the patient initiates the search for care in response to an acute exacerbation of a current condition or the emergence of a new condition, 6 CIM takes a more proactive approach by ensuring continuity of care, preventive measures, and providing the patient with adequate self-management support throughout his/her long-term illness trajectory. 6 The Innovative Care for Chronic Conditions (ICCC) framework provides a roadmap for health system re-engineering to make it more conducive to the needs of patients with chronic conditions. 7 Under the ICCC framework, healthcare systems are encouraged to (1) promote continuity of care and care coordination; (2) improve quality through leadership and incentives; (3) organize and equip healthcare teams; (4) utilize information systems; and (5) support disease self-management and prevention. 6 Changes in this direction would make healthcare systems better equipped to care for individuals with chronic conditions, who require long-term integrated care and support, such as HTx recipients.
Integrated care—defined as health services delivered throughout the health continuum and coordinated across different levels and sites of care 8 —when based on principles of CIM has resulted in improved outcomes in other chronically ill populations. Among people with multiple chronic conditions, comprehensive care based on CIM principles has been shown to improve health behaviors, decrease healthcare utilization and healthcare costs. 9 In HTx, variability in degrees of CIM among centers has been observed, 10 yet the association between CIM and clinical outcomes have not yet been explored.
Therefore, the primary aim is to explore the association between the degree of CIM and survival rates at 1-, 3-, 5-years post HTx at the center-level. Additionally, the associations between the degree of CIM and more proximal outcomes (i.e. patients’ adherence to recommended health behaviors) will be explored.
Methods
This is a secondary analysis of the Building Research Initiative Group: CIM and Adherence in Transplantation (BRIGHT) study, a cross-sectional study that examined the multi-level factors associated with immunosuppressant nonadherence in adult HTx recipients from 36 HTx centers in 11 countries across 4 continents. 11 Information on the methodology of the BRIGHT study has been previously published.11–13 Data were collected between March 2012 and October 2015.
Sampling strategy
The BRIGHT study used a multi-staged sampling approach. A convenience sample of countries and HTx centers were recruited, followed by a random sample of clinicians and patients using a sampling strategy guided by the size of the HTx centers. Centers that performed 50-74 HTx in 5 years were classified as small, centers performing 75–100 HTx as a medium, and those performing >100 HTx as large. 12 The size classification of the HTx centers was determined using the variability in the number of HTx performed by the participating centers and the International Society of Heart and Lung Transplantation (ISHLT) criteria. 14 Twenty-five patients were randomly recruited from small HTx centers, 40 from medium centers, and 60 from large centers. If the center had ≤5 eligible clinicians, all were recruited. In centers with >5 eligible clinicians, a random sample of 5 clinicians was recruited.
Inclusion criteria
Variables and measures
Degree of CIM
The degree of CIM was assessed from two perspectives (patients’ and clinicians’) to obtain a more accurate picture of the CIM practices of each HTx center. The validated short version of the Patient Assessment of Chronic Illness Care (PACIC) was used to measure the extent to which the CIM provided is congruent with the chronic care model from the patient’s perspective (Cronbach’s alpha = 0.955; test-retest reliability (r) = 0.638).15,16 The short self-report version consists of 11 items and uses a 5-point Likert scale from “almost never” to “almost always”. The summary score ranges from 11 to 55 with higher scores indicating a higher degree of CIM. For this study, the patients’ PACIC scores were averaged per center, generating a CIM score per center as perceived by patients being followed up within a given center.
The Chronic Illness Management Implementation-Building Research Initiative Group: CIM and Adherence in Transplantation (CIMI-BRIGHT) instrument was used to assess CIM implemented in the HTx centers from the clinicians’ perspectives. 17 The CIMI-BRIGHT consists of 52 items and uses a 4-point Likert scale from “strongly disagree = 1” to “strongly agree = 4”. 17 The CIMI-BRIGHT allows for a “don’t know” response (coded as “5”); therefore, the “don’t know” responses were treated as missing and the CIMI-BRIGHT scores were averaged per clinician. The clinicians’ CIMI-BRIGHT scores were then averaged per center with higher scores corresponding to a higher degree of CIM implemented.
Behavioral factors
Behavioral factors were analyzed at the patient level. Immunosuppressant adherence was assessed using the Basel Assessment of Adherence to Immunosuppressive Medications Scale (BAASIS).18,19 Any deviation in taking, dosing, or timing was considered nonadherence. 18 Adherence to other medications, including the different implementation dimensions (i.e. dosing, timing),20,21 was assessed using an adapted version of the BAASIS. The level of physical activity was measured using the Brief Physical Activity Assessment Tool. 22 Patients were deemed adherent if they engaged in 20 min of vigorous physical activity ≥3 times/week or 30 min of moderate physical activity ≥5 times/week or 5 or more sessions of any combination of moderate and vigorous-intensity physical activity. 22 Dietary adherence was measured by asking the patients if they were advised to follow a specific diet in the past year and, if so, how strictly they followed the recommended diet (never/rarely/sometimes/ often/ always). The subsample of patients, who were recommended a specific diet, were then classified as adherent if they followed their recommended diets often/always. Patients who were current smokers were classified as non-adherent, and patients who never smoked or quit were considered adherent. Patients who were nondrinkers or mild drinkers (i.e. <1 drink/day for females and <2 drinks/day for males) were classified as adherent and were classified as non-adherent if they were moderate or heavy drinkers. Finally, patients who used sun protection (wear a hat, use sunscreen, stay in the shadows, or limit sun exposure often or all the time) were classified as adherent and those who did not as non-adherent. A detailed description of the measures used is published elsewhere. 11
Survival rates
An investigator-developed, self-report questionnaire completed by the center’s director asked for center-level survival data and mortality risk factors that pertained to all patients at the center and not exclusively for those participating in BRIGHT. Survival rates were requested at 1, 3, and 5 years post-transplantation, congruent with the inclusion criterion (i.e. patient included were 1 till 5 years post-transplant). We assessed specific risk factors of HTx mortality: i.e. the percentage of patients: hospitalized at the time of HTx, with infection requiring IV drug therapy, with defibrillator prior to HTx, on ventilator at time of HTx, on extracorporeal membrane oxygenation (ECMO) at time of HTx, with intra-aortic balloon pump at time of HTx, or on ventricular assist device (VAD) at HTx, etiology of heart failure, and recipient gender/donor gender.
Ethical considerations
Each BRIGHT study HTx center obtained ethical approval from their institutional review board and/or national ethics committee. Written informed consent was obtained from the participating HTx recipients. Finally, completed questionnaires were coded by the HTx center and returned to the investigators in sealed envelopes to maintain confidentiality.
Statistical analyses
For this secondary analysis, data were analyzed at the center-level (i.e. PACIC and CIMI-BRIGHT scores were averaged by HTx center), except in the analysis of the associations between degree of CIM and recommended health behaviors that used patient-level data (n = 1397 patients). Analysis was performed using Stata/SE 15.
Based on the current state of science, we used the most appropriate and innovative method to classify the HTx centers. Latent profile analysis (LPA) is a measurement model in which individuals, or in this case centers, are classified into mutually exclusive and exhaustive groups based on how they perform on a set of continuous indicator variables. The 2-step LPA procedure suggested by K.E. Masyn23,24 was performed to classify the HTx centers into groups based on their degree of CIM (mean PACIC and CIMI-BRIGHT scores). The 2-step approach comprised of (1) identifying latent groups using observed indicator variables (PACIC and CIMI-BRIGHT scores); and (2) assigning centers to groups based on predicted posterior class probabilities. 23 Models with two to four groups were estimated and compared based on goodness-of-fit indices and interpretability. The goodness-of-fit indices evaluated comprised of the Bayesian information criterion (BIC) and Akaike information criterion (AIC), wherein lower values correspond to better model fit. 23
Sensitivity analysis was performed by using the median PACIC and CIMI-BRIGHT scores as the cutoff to classify the centers into two groups (high-/low-degree of CIM), which resulted in similar findings.
Fisher’s exact test was used to compare the center characteristics and HTx mortality risk factors between the two groups identified by the LPA. Meanwhile, logistic regression was used to compare the patients’ adherence to recommended health behaviors between the two CIM groups. Lastly, the mean survival rates at 1-, 3-, and 5 years between the two groups were compared using a random-intercepts regression analysis and partial eta-squared (η2) were calculated to determine effect sizes.
Results
Characteristics of the 36 HTx centers and their respective mean degrees of CIM are presented in Table 1. Over half (n = 19, 52.8%) of the centers were located in Europe, 12 (33.3%) were located in North America, 3 (8.3%) in South America, and 2 (5.6%) in Australia. Twenty-three (63.9%) were large centers (i.e. >100 HTx performed/5 years). Thirty-two (88.9%) of the HTx centers were located in urban areas and the majority (83.3%) were university teaching hospitals. The degree of CIM varied across countries. The overall mean PACIC score was 37.9 ± 4.7 (range: 30.9–42.0) and the overall mean CIMI-BRIGHT score was 2.9 ± .3 (range: 2.7–3.1).
Center characteristics and mean level of chronic illness management (PACIC and CIMI-BRIGHT).
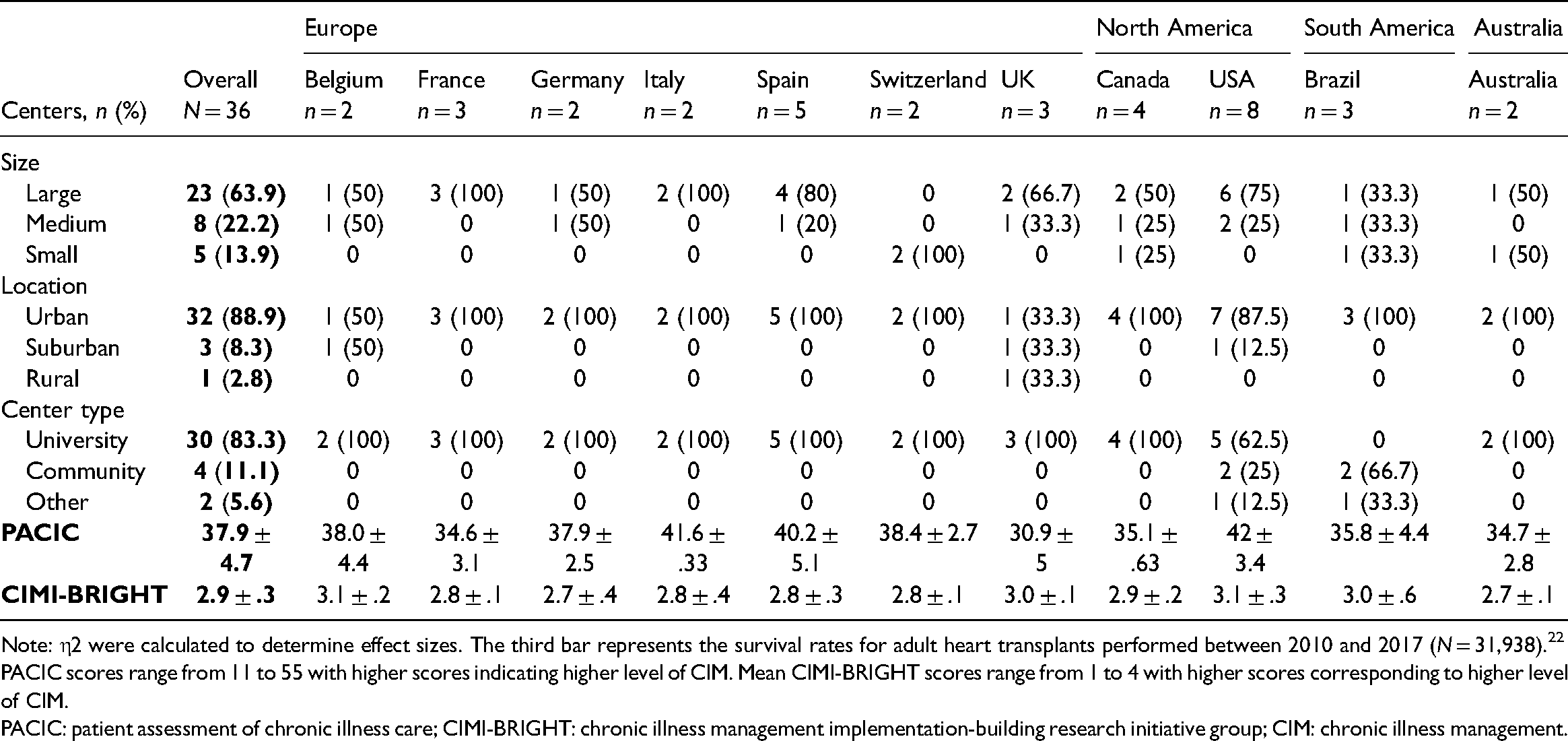
Note: η2 were calculated to determine effect sizes. The third bar represents the survival rates for adult heart transplants performed between 2010 and 2017 (N = 31,938). 22
PACIC scores range from 11 to 55 with higher scores indicating higher level of CIM. Mean CIMI-BRIGHT scores range from 1 to 4 with higher scores corresponding to higher level of CIM.
PACIC: patient assessment of chronic illness care; CIMI-BRIGHT: chronic illness management implementation-building research initiative group; CIM: chronic illness management.
Based on the 2-step LPA, the 2-group model had the best fit (lowest BIC and AIC values). Using the 2-group model, 29 HTx centers were classified under groups 1 and 7 in group 2. After checking the mean degree of CIM for each group, group 1 was labeled as “low-degree CIM” and group 2 as “high-degree CIM”. The mean PACIC score for group 1 was 36.9 ± 4.1 and 42.0 ± 4.8 for group 2. The mean CIMI-BRIGHT score for group 1 was 2.8 ± .2 and 3.3 ± .2 for group 2. There were no significant differences between the groups in terms of center characteristics (i.e. size, location, type), center-level mortality risk factors, etiology of heart failure, and recipient gender/donor gender, except for the percentage of patients with idiopathic (p = 0.034), congenital (p = 0.013), and other etiology of heart failure (p = 0.026). A comparison of the center-level characteristics between the two groups is presented in Table 2.
Characteristic differences between the CIM classes.
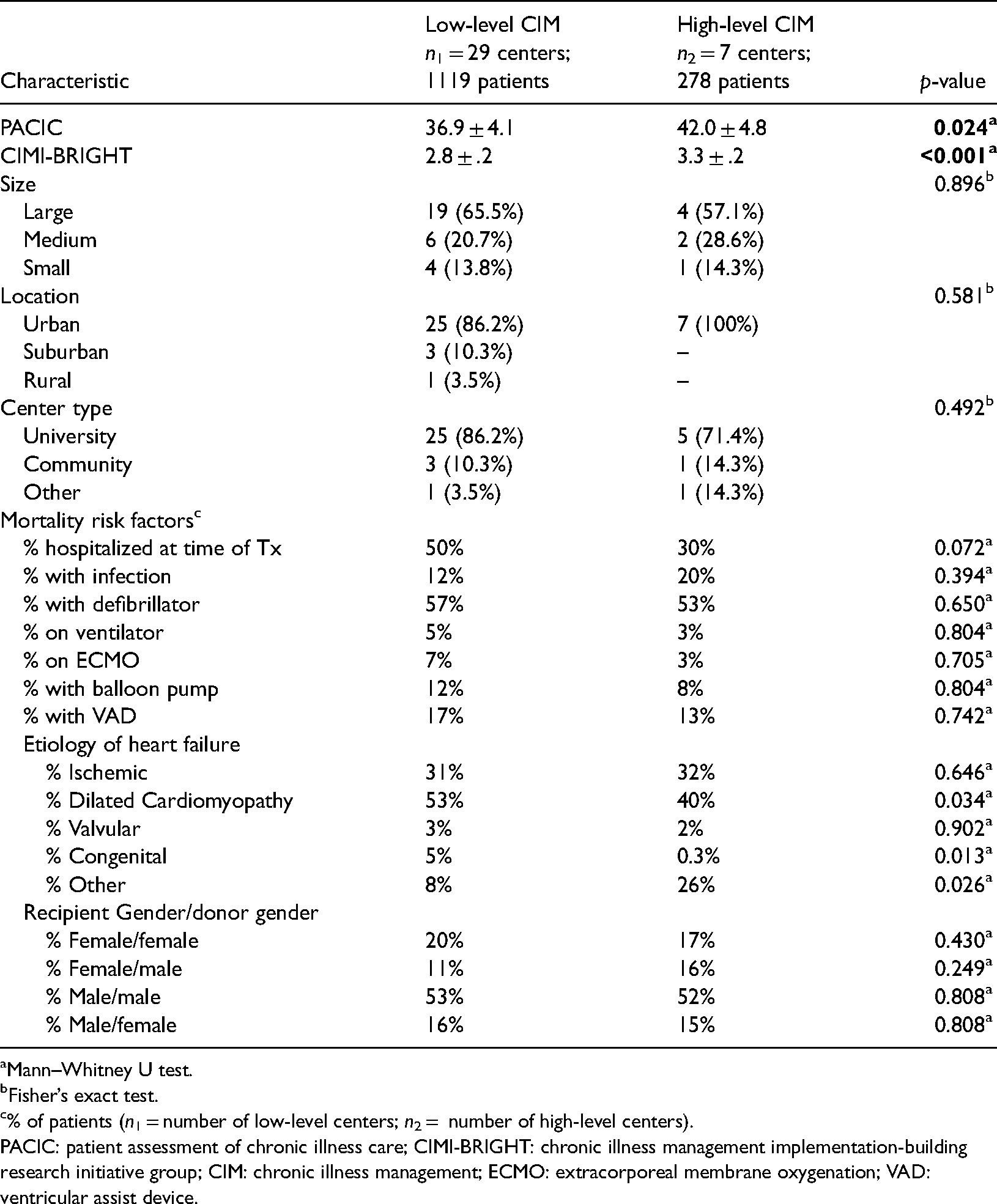
Mann–Whitney U test.
Fisher’s exact test.
% of patients (n1 = number of low-level centers; n2 = number of high-level centers).
PACIC: patient assessment of chronic illness care; CIMI-BRIGHT: chronic illness management implementation-building research initiative group; CIM: chronic illness management; ECMO: extracorporeal membrane oxygenation; VAD: ventricular assist device.
Figure 1 shows the comparison of the survival rates between the low-degree CIM group and the high-degree CIM group. Information on survival rates for adult HTx performed between 2010 and 2017 from the ISHLT registry is also included for reference. 25 Not every HTx center provided information on their survival rate. At year 1, only 31 centers had available information on their survival rates. At years 3 and 5, only 29 and 30 HTx centers, respectively, had available survival rates. It should be noted that all 7 high-degree CIM centers provided information on their survival rates for years 1, 3, and 5; hence, the missing survival rates were all from low-degree CIM centers. At year 1, being in a high-degree CIM center was associated with a 4.8% higher survival rate (95% confidence interval (CI) .11–9.4, p = 0.045) and the difference had a medium effect size (η2 = .06). There were no significant differences in survival rates at years 3 (η2 = .01) and 5 (η2 < .0001).
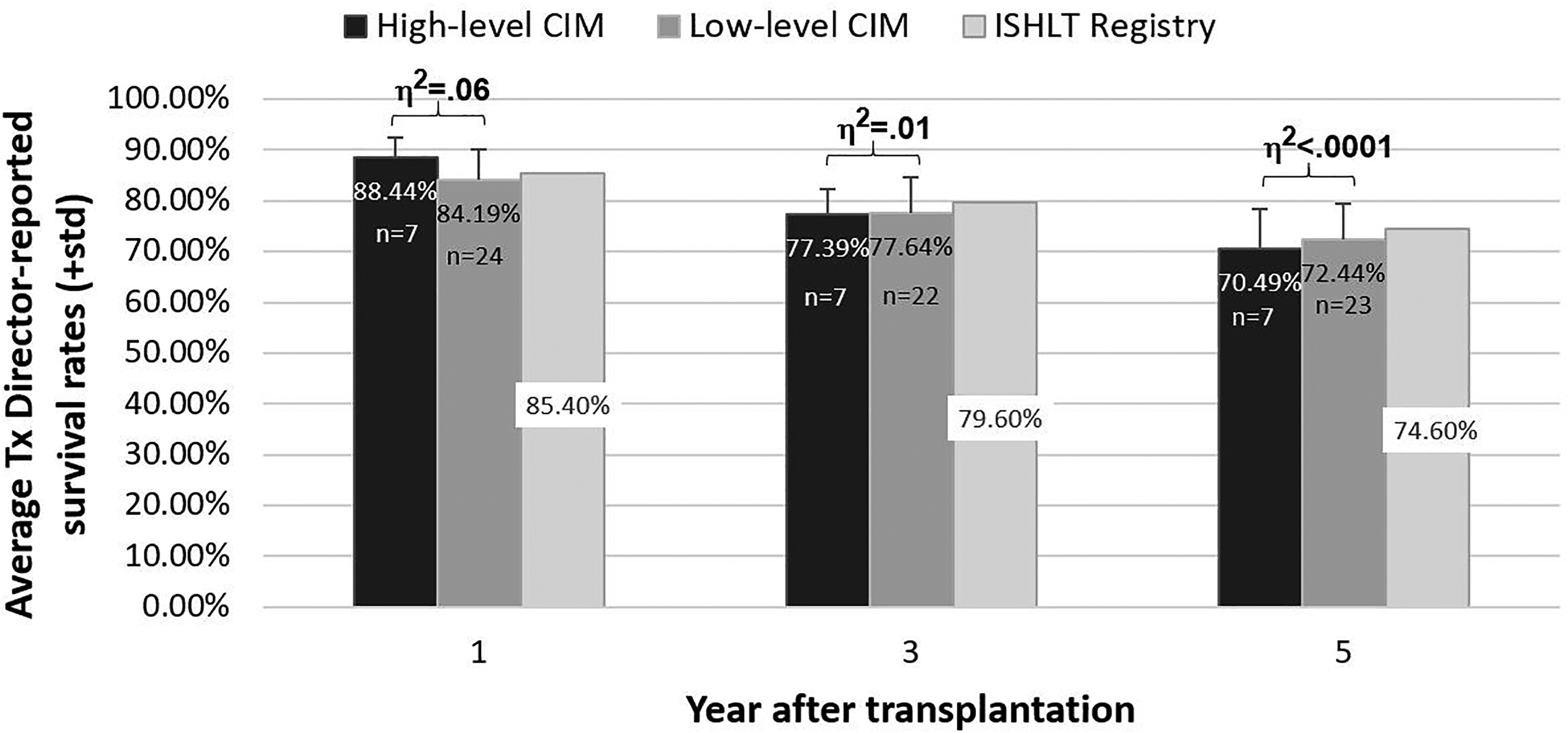
Mean HTx patient survival by level of chronic illness management.
Patients in high-degree CIM centers had 9% higher odds of adhering to their immunosuppressants (95% CI .83 -1.43, p = 0.54) and 8% higher odds of adhering to their other medications (95% CI .76–1.55, p = 0.66) compared to patients in low-degree CIM centers. Similarly, patients in high-degree CIM centers had 33% higher odds of engaging in sufficient physical activity (95% CI .94–1.90, p = 0.11), 34% lower odds of smoking (95%CI .29–1.49, p = 0.32), and 52% significantly lower odds of moderate to severe drinking (95%CI .30–.78, p = 0.003). Conversely, patients in high-degree CIM centers had 21% lower odds of using sun protection (95%CI .54–1.13, p = 0.20). Lastly, patients in high-degree CIM centers had 49% significantly higher odds of being recommended a specific diet (95%CI 1.13–1.96, p = 0.004). However, in the subsample of 450 patients who received diet recommendations, patients from high-degree CIM centers had 21% lower odds of adhering to diet recommendations (95%CI .51–1.22, p = 0.29).
Discussion
The findings from this exploratory study offer preliminary insight into the potential of CIM, as a system-level pathway, in improving short-term survival for HTx recipients. HTx centers with higher degrees of CIM had a better survival rate in year 1 compared to centers with lower degrees of CIM. However, our findings do not support an association between degree of CIM and longer-term survival at 3- and 5-years post transplant. The cross-sectional design of the BRIGHT study meant that the degree of CIM was measured at only one time-point, making it difficult to ascertain the HTx centers’ degrees of CIM at the different time points. It is possible that the centers with high-degree CIM had only recently implemented CIM practices, which could help explain the lack of associations in the later timepoints.
Similarly, when looking into more proximal outcomes—specifically adherence to recommended health behaviors—despite the trend toward better adherence there was a lack of statistically significant associations. Given that CIM is a system-level factor, its association with health behaviors could be indirect in nature through more person-level factors, such as self-efficacy and/or patient activation, which are widely known to impact behavior change.26,27 At an intuitive level, it is logical to expect that the proactive, holistic support provided by a multidisciplinary team of healthcare professionals—the crux of CIM—would increase the patient’s confidence, knowledge, and skills in performing recommended health behaviors. Unfortunately, given the secondary nature of this analysis, the associations between CIM and person-level behavioral factors could not be explored.
Previous studies on renal transplant recipients provide proof of concept that re-engineering transplant follow-up care using CIM principles could be a promising solution to improving long-term transplant outcomes. Bissonnette and colleagues showed that transplant follow-up care based on CIM principles was effective in helping transplant recipients achieve targeted clinical outcomes. 28 Meanwhile, in a randomized-controlled trial, Schmid et al. showed that a telemedically supported CIM intervention resulted in better adherence to immunosuppressants, fewer hospitalizations, and shorter lengths of hospital stay. 29
Admittedly, designing a CIM intervention will require adapted methods beyond the traditional clinical research methods to achieve successful implementation, adoption, and sustainability of an innovative model of care for follow-up of transplant patients. 30 Implementation science could offer a way forward to achieve success in this regard. 31 We are currently investing in developing, implementing, and testing this CIM-based eHealth-powered integrated care model—consisting of a human-component (i.e. care coordinator) as well as technology factors (i.e. SMILE app)—using the principles of user-centered design, agile software development, as well as implementation science to improve outcomes in stem cell transplant patients. 32 This model, should it prove to be successfully implemented in variable contexts and should it prove to be effective in improving clinical outcomes as well, could provide a blue print for solid organ transplantation to reengineer follow-up care based on principles of CIM.
Findings from the present exploratory study should be interpreted within the context of its limitations. First, the cross-sectional design precludes causal inferences. Second, this secondary analysis had a small sample size (center-level), which means it could have been underpowered to detect differences in survival rates. Similarly, we could not rule out the potential impact of survival bias. Moreover, the small sample size (center-level) prevented us from controlling for potential confounders in our analyses as mentioned before. Nonetheless, potential confounders were analyzed descriptively to provide context to our findings. Lastly, the primary outcome (survival rates) was measured using self-report which could limit the validity of our findings. On the other hand, a strength of this study is the assessment of CIM from two independent perspectives (patients and clinicians), and the use of these two measures in the analysis.
Conclusion
Significant association between degree of CIM and short-term survival hints at the potential impact of CIM. The signals observed in our data—combined with the emerging evidence from intervention studies supporting the effectiveness of the CIM approach in renal transplant follow-up care—compels further investigation. Our findings help justify the development and testing of CIM-based interventions for HTx recipients. In addition, future research should explore what degree of CIM and which aspects of CIM are most relevant in view of achieving better outcomes in transplantation. Degree of CIM needs to be regarded as a valuable system-level factor that expresses the extent to which Tx centers have adopted principles of chronic illness care.
Footnotes
Acknowledgements
We would like to thank all the patients and clinicians who shared their time and effort in participating in our study.
Contributorship
SDG, FD, CR, and LB researched literature and conceived the primary study. MIC and KD were involved in data analysis. MIC wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Each BRIGHT study HTx center obtained ethical approval from their institutional review board and/or national ethics committee.
Funding
This study is funded by research grants of the International Transplant Nurses Society in 2008, the International Society for Heart and Lung Transplantation (ISHLT) in 2012, the Swiss Academy of Medical Sciences (SAMW) in 2013 as well as by an unrestricted research grant from Astellas Pharma. None of the organizations that provided funding has access to the data nor were involved in the preparation of the manuscript.
Guarantor
SDG
Informed consent
Written informed consent were obtained from the participating HTx recipients for their anonymized and aggregated information to be published in this article.
