Abstract
Background:
Multiple factors impact kidney perfusion peri-operatively. The aim of this study was to evaluate the effect of patient position and size on renal transplant perfusion.
Methods:
Consecutive adult recipients of 123 single renal grafts were studied. Renal artery velocity, renal vein velocity and cortical resistive indices were measured in supine, oblique and decubitus positions on post-operative days 1, 3, 7 and 30, and standing on days 7 and 30.
Results:
Positional resistive indices were significantly lower than the supine resistive indices except in the day 1 oblique scan. Greater reductions in resistive indices occurred in grafts with higher supine cortical resistive indices, higher renal vein velocities and greater change in renal vein velocities. Renal artery velocities, renal vein velocities and resistive indices progressively decreased with greater positional change. Although renal vein velocities correlated poorly with resistive indices in individual patients, mean resistive indices correlated well (r2 = 0.73) with mean renal vein velocities for scans in different positions and on different days, and less so with mean renal artery velocities (r2 = 0.37). Supine abdominal girth and change in girth with position were more strongly associated with larger changes in supine resistive indices than recipient weight, body mass index or peri-operative weight gain.
Conclusions:
Peri-operative renal transplant resistive indices, renal artery velocities and renal vein velocities improve with patient positional change due to reduced compression of the graft and renal vein, with implications for post-operative ultrasound scanning protocols, documentation and reporting. Peri-operative patient position, especially for at-risk grafts, is a modifiable risk factor for poorer graft outcomes. Patients should be nursed in the decubitus position rather than supine. Abdominal girth is more relevant to pre-operative patient assessment than weight or body mass index.
Introduction
Ultrasound is frequently used in the early post-operative period in the assessment of renal transplants. The investigation is non-invasive, can be performed at the bedside, and avoids radiation and nephrotoxic contrast. Ultrasound also provides dynamic results in real time, is easily repeated and can monitor clinical progress with serial scans. While ultrasound does not accurately distinguish between acute rejection and acute tubular necrosis (ATN), the cortical resistive indices (RIs) are increased in both conditions. 1 There are higher RIs in grafts with delayed graft function (DGF) than in those with immediate graft function (IGF), and higher rates of late graft loss 2 in grafts with DGF. There are a small number of case reports of changes in perfusion in renal transplants3,4 and native kidneys 5 when the patient is in different positions.
The aim of this study was to evaluate the association between peri-operative patient position and renal graft perfusion, and the effect of patient factors including anthropometrics 6 on graft perfusion.
Methods
This study was approved by the Northern Sydney Local Health District (2020-ETH00443) HREC. One hundred and twenty-five consecutive adult patients receiving single renal transplants at Royal North Shore Hospital between September 2020 and August 2023 were approached for recruitment with two patients choosing not to participate. Although these two patients underwent routine post-operative supine scans, their results were not included in this research.
All patients had undergone intra-operative duplex scanning with correction of identified major vessel flow abnormalities 7 as required. The intra-operative scan also provided a baseline RI for comparison with the patient’s post-operative scans. Patients underwent scans on days 1, 3, 7 and 30 after transplantation according to a formal protocol. 8 In addition to the standard scan in the supine position, patients were scanned in the 45° oblique and lateral decubitus positions with the side of the kidney upwards, and on post-operative days 3, 7 and 30, patients were also scanned standing. Some positional scans were not performed due to the patient’s condition, including management in intensive care and general patient discomfort. In the early study period, most patients were unable or unwilling to stand during the scan on day 3, and subsequently positional standing scans on day 3 were not routinely performed. No other data were excluded. The nominal day of scanning was that relative to the original day of implantation and was not reset if there was a return to the operating theatre. Scanning was performed using a Canon Aplio I series 800 ultrasound machine (Canon Medical Systems, Tokyo, Japan) with I8CX1 convex matrix and I11LX3 linear matrix transducers.
The cortical RI, ((systolic velocity – end-diastolic velocity)/systolic velocity), was determined by measuring peak systolic and end-diastolic velocities in arcuate arteries in the middle to outer third of the renal cortex. The cortical RI for a scan was defined as the median of the cortical RIs measured in the upper, middle and lower poles, regardless of the number and distribution of renal arteries including accessory renal arteries. No correction to the RI was made for patient heart rate 9 A separate reproducibility study was not performed, noting there is only a small variation in RI with different machines and operators. 10 Multiple sonographers, all experienced in in-patient vascular and transplant ultrasound, performed the intra-operative and post-operative scans. The final report for all scans was formulated by two readers. Complex and unexpected scan findings were also reviewed and discussed contemporaneously with the operating surgeon and renal transplant physician.
Renal artery velocities (RAVs) were measured along the length of the vessel from the anastomosis to the hilum and a representative velocity used. Renal vein velocities (RVVs) were sampled along the length of the renal vein, with the RVV at the point of highest observed velocity used for analysis regardless of site. Mean RAVs, RVVs and RIs were calculated for scans in different positions on different days. RAVs, RVVs and RIs in the same position on different days of scanning were pooled. RAVs and RVVs were not recorded in less than 5% of scans performed.
DGF was defined as the requirement for the patient to undergo haemodialysis within 7 days of implantation regardless of indication. Weight was the ideal dry weight or minimum weight in the preceding 12 months documented in the medical record. The recorded maximum weight in the first week after the transplant was used to calculate the maximum weight gain in the first week post-operatively. The patient’s abdominal circumference was measured at the waist and anterior superior iliac spine (ASIS), in both supine and standing positions post-operatively, with calculation of the change in girth from supine to standing. Data for girth in both positions were not recorded in 23 (19%) patients.
The association of the supine RI and positional change in RI, with patient factors including weight and girth, and graft function, was analysed with unpaired, paired t-tests or analysis of variance (ANOVA) tests according to the data. No adjustments were made for missing data and no outlying data were excluded.
Results
Patient details are shown in Table 1. The numbers and distribution of scans performed are shown in Table 2. All 123 patients underwent supine scans on days 1, 3 and 7. Nine patients did not undergo positional scans on day 1, nor did five patients on days 3 and 7 due to management in intensive care or patient discomfort, with no significant difference in supine RIs between those not undergoing a supine scan and those that did (see Table 3) except at day 30. Twelve patients discharged to satellite units before the day 30 scan. All grafts remained in situ with no grafts removed. All grafts, except one with primary non-function, were functioning at day 30. There was DGF in 36 (55%) of the 65 cadaver donor grafts and 3 (5%) of the 58 live donor grafts. There was a small but statistically significant reduction in girth measurement at the waist level, and a greater and statistically significant increase in girth at the level of the ASIS, when the patient moved from supine to standing.
Patient details (n = 123; n (%); mean (SD)), and for differences in girth (mean (95% CI)).
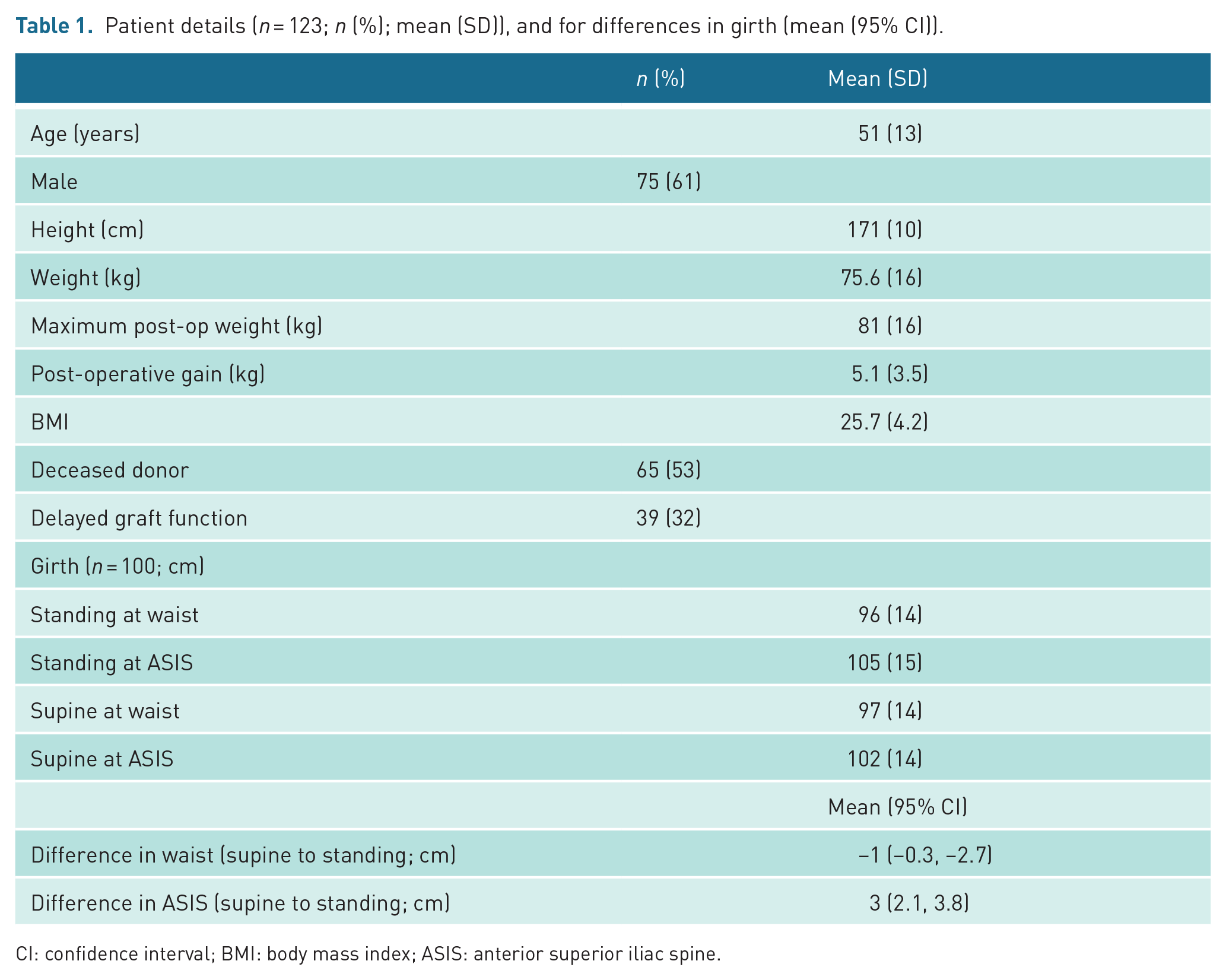
CI: confidence interval; BMI: body mass index; ASIS: anterior superior iliac spine.
Numbers of scans performed according to post-operative day of scan and patient position.

Patient details (mean (SD)) or number (n (%)), grouped according to supine RIs on day 3.
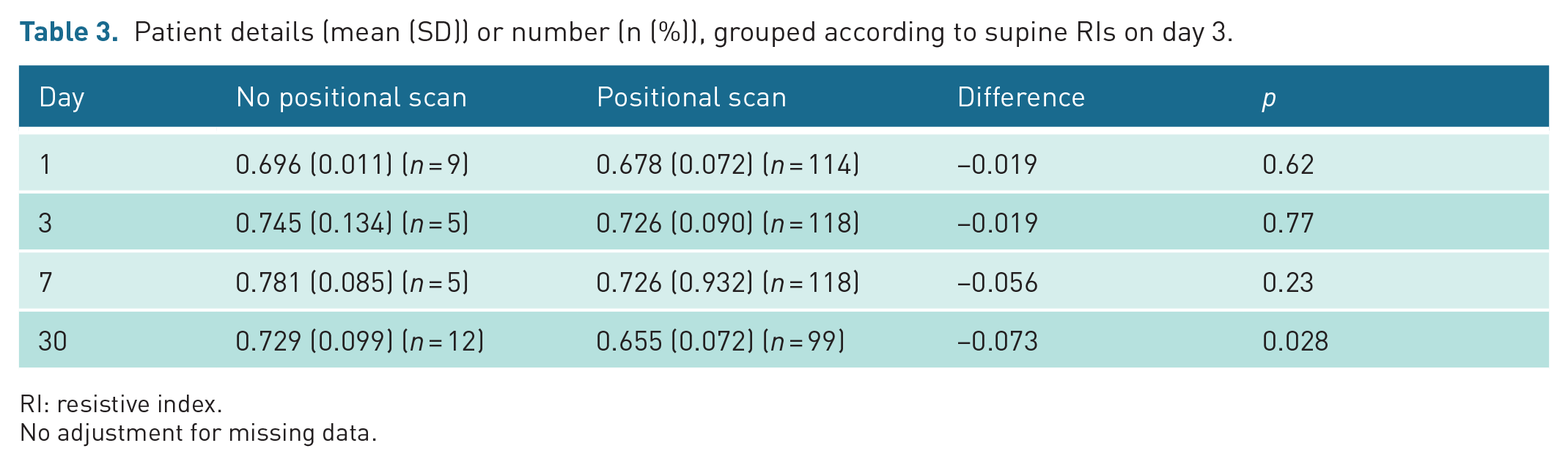
RI: resistive index.
No adjustment for missing data.
There is an uneven distribution of patient factors when categorised according to the day 3 supine RI (see Table 4). Patients with higher RIs were taller, older, heavier, had greater girth, were more likely to have DGF and to have received a cadaver graft, than those with lower RIs.
Patient details (n (%) or mean (SD)) for supine scans classified according to the supine RI on day 3.
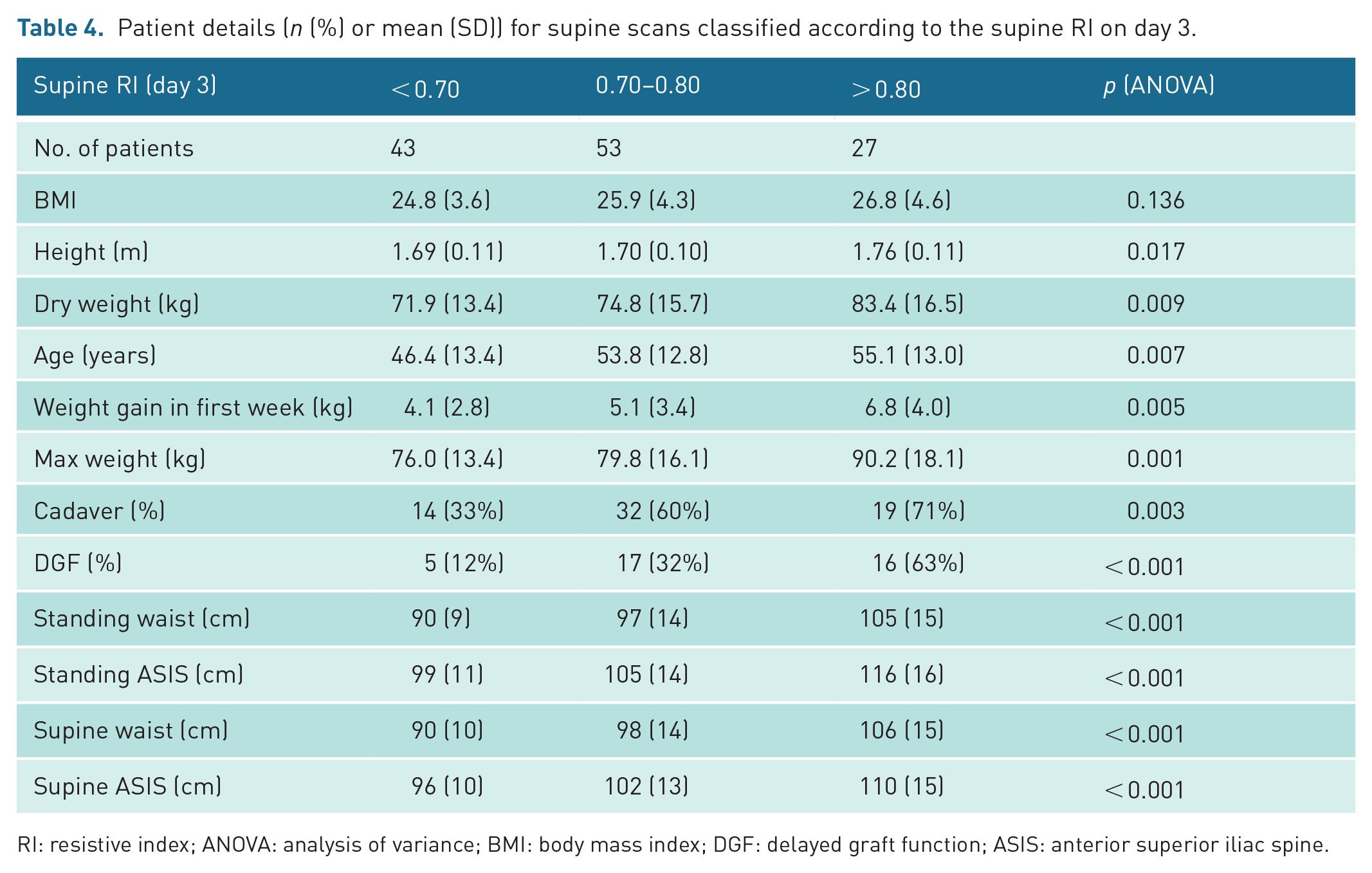
RI: resistive index; ANOVA: analysis of variance; BMI: body mass index; DGF: delayed graft function; ASIS: anterior superior iliac spine.
The mean supine RIs in the small number of patients not undergoing positional scans were slightly higher than those undergoing positional scans (see Table 4) with this difference only statistically significant on day 30. One hundred and one (83%) of the 123 patients underwent at least one standing scan. Exclusion of data from patients who did not undergo the relevant positional scan did not significantly alter comparisons of RIs of positional scans.
Mean cortical RIs reduced significantly with positional change in all positions on different days (see Table 5, Figure 1), except 45° oblique on day 1. There was a greater positional fall in RI with greater positional change in both individual scans and for pooled data. The mean differences (95% confidence interval (CI)) between the pooled supine RI and the oblique, decubitus and standing RIs were –0.016 (–0.027, 0.004), –0.027 (–0.039, –0.015) and –0.075 (–0.091, –0.059), respectively.
Differences of mean cortical RIs between supine and other positions (unpaired t-tests).
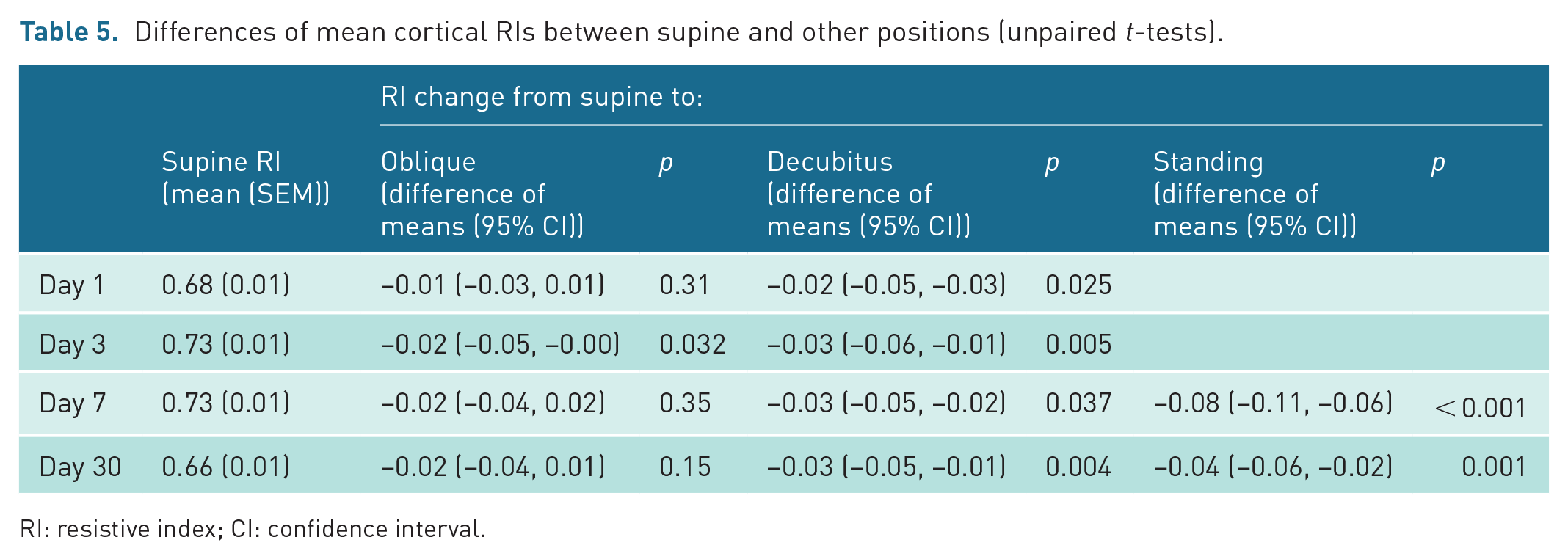
RI: resistive index; CI: confidence interval.
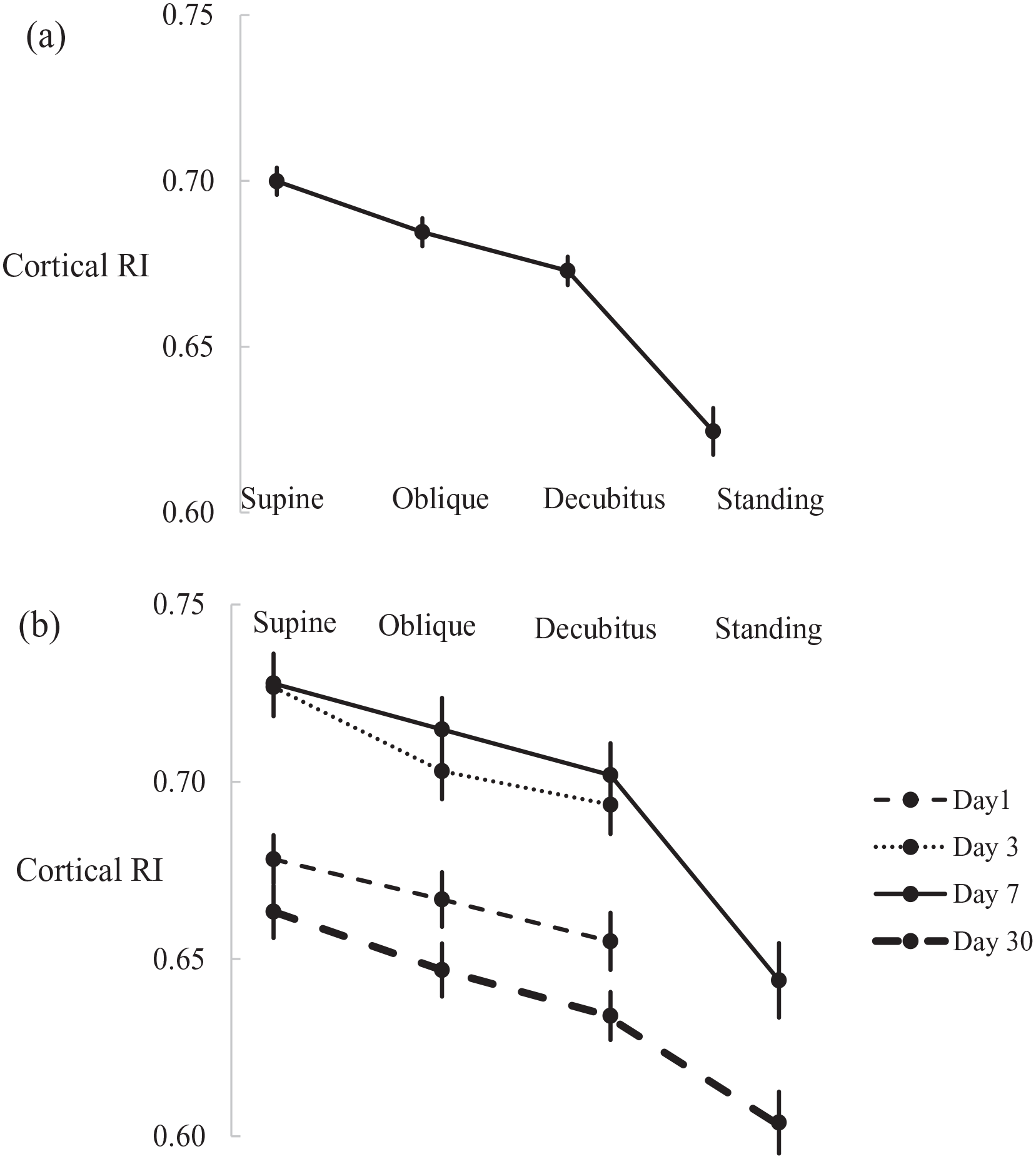
(a) Mean (±SEM) pooled and (b) by post-operative day cortical RIs in different positions.
Post-operatively, the mean supine RI was highest on days 3 and 7. The difference between the supine and decubitus RI was greatest on day 3, when 40 of 118 (34%) of grafts had a decrease in RI from the supine to decubitus of > 0.05. Higher supine RIs, supine RVVs and greater reductions in RVVs were significantly associated with this greater positional change (see Table 6). Representative images of positional changes in cortical perfusion and RIs are shown in Figure 2. Abdominal girth and shift in girth was greater in the grafts with greater reduction in RI from supine to decubitus and were more strongly associated with positional RI change than patient weight or BMI.
Patient factors on day 3 categorised according to change in RI from supine to decubitus of < 0.05 or ⩾0.05 (unpaired t-test).
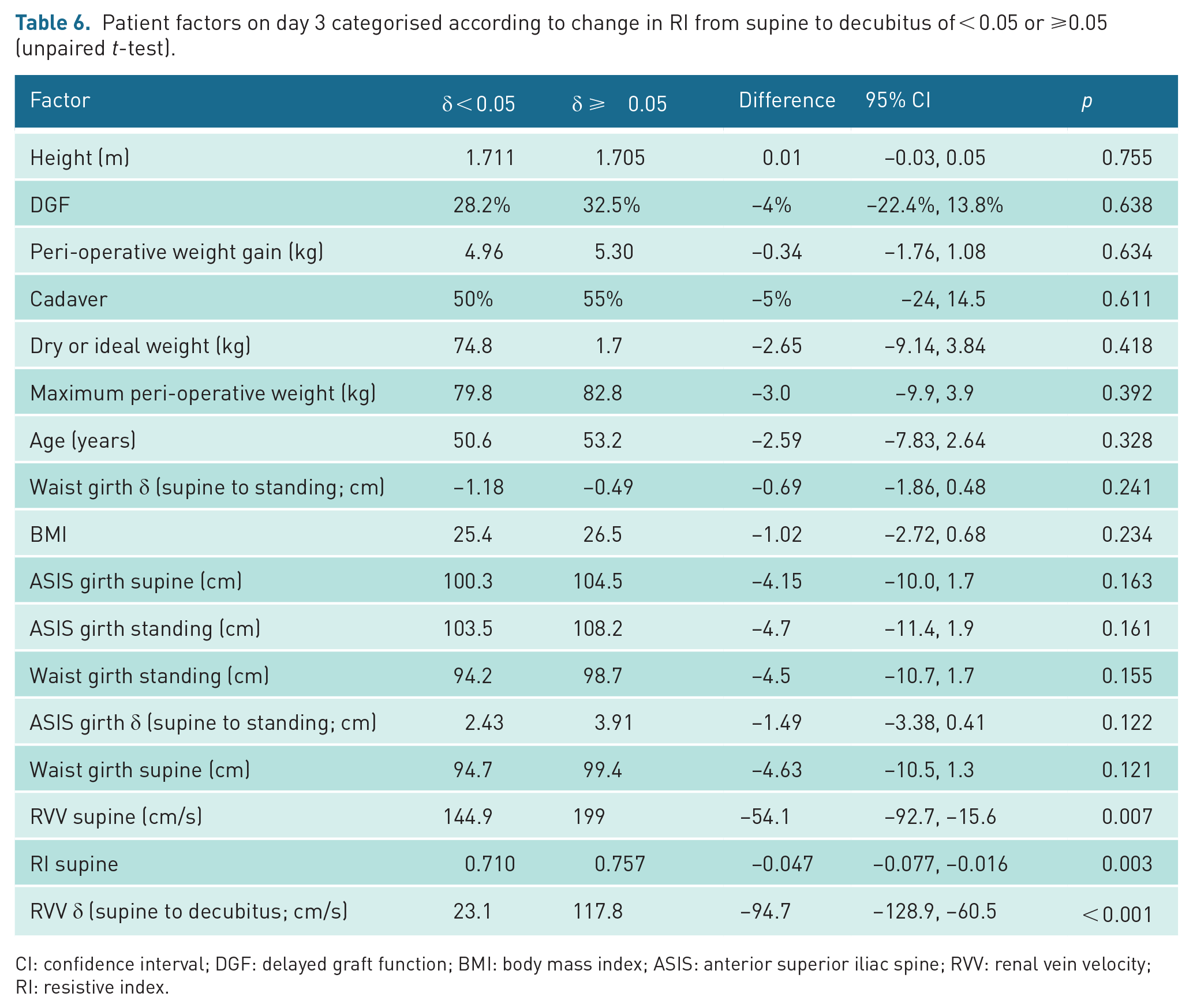
CI: confidence interval; DGF: delayed graft function; BMI: body mass index; ASIS: anterior superior iliac spine; RVV: renal vein velocity; RI: resistive index.
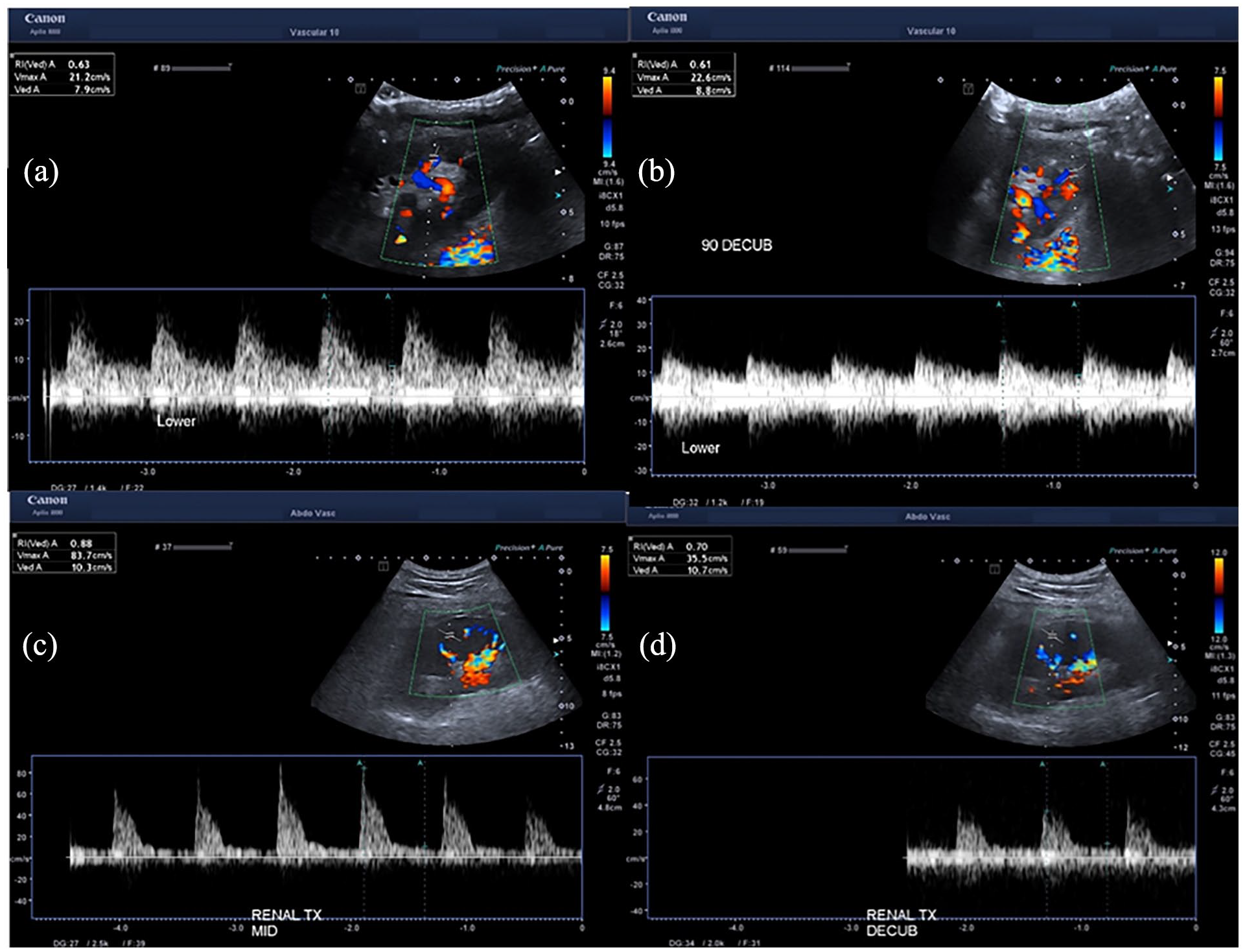
Representative ultrasound images of positional changes in RI. Little or no change in cortical RIs from (a) supine to (b) decubitus in the lower pole day 1 post-implantation, in a live related graft with immediate function in a 45 kg recipient with 75 cm girth at the ASIS; and cortical RIs improving from (c) 0.88 supine to (d) 0.70 decubitus in the mid-pole of a graft with delayed function day 3 post-transplantation, in a 117 kg patient with 139 cm girth at the ASIS.
While RVVs varied considerably for any scan, the mean RVVs for each scan showed a strong association with positional change (see Figure 3). Depending on the day of the scan, the mean RVV in the oblique position was from 35 to 50 cm/s lower than when supine, 35 to 70 cm/s lower when decubitus, with the greatest differences of 50 and 80 cm/s between supine and standing scans on day 7 and day 30. The mean RI and RVV for each supine and positional scan correlated well (r2 = 0.73) with higher mean RIs having higher mean RVVs (see Figure 4). RAVs also correlated with RIs, but less well (r2 = 0.37).
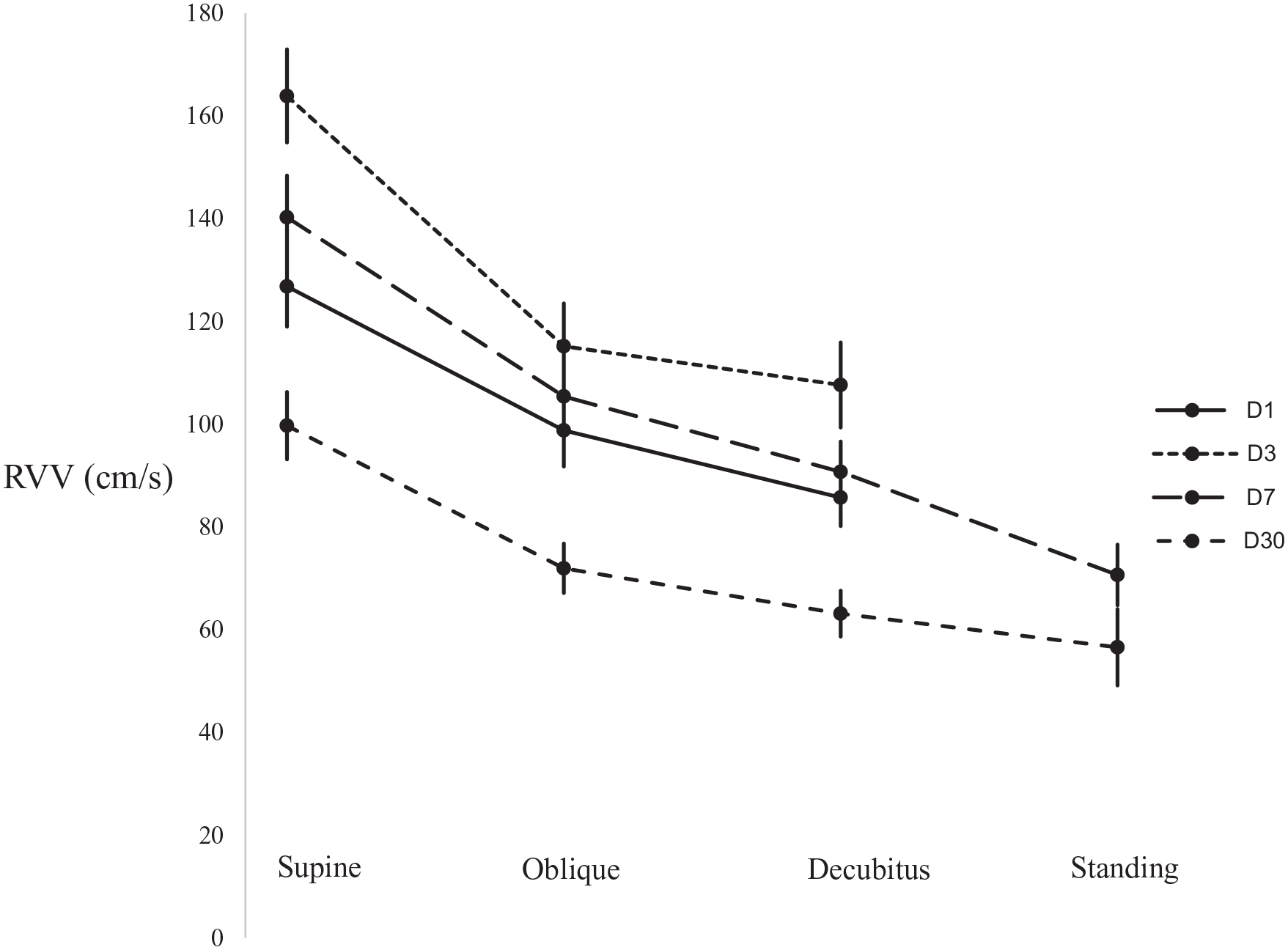
Changes in RVV according to positional change and day of scan.
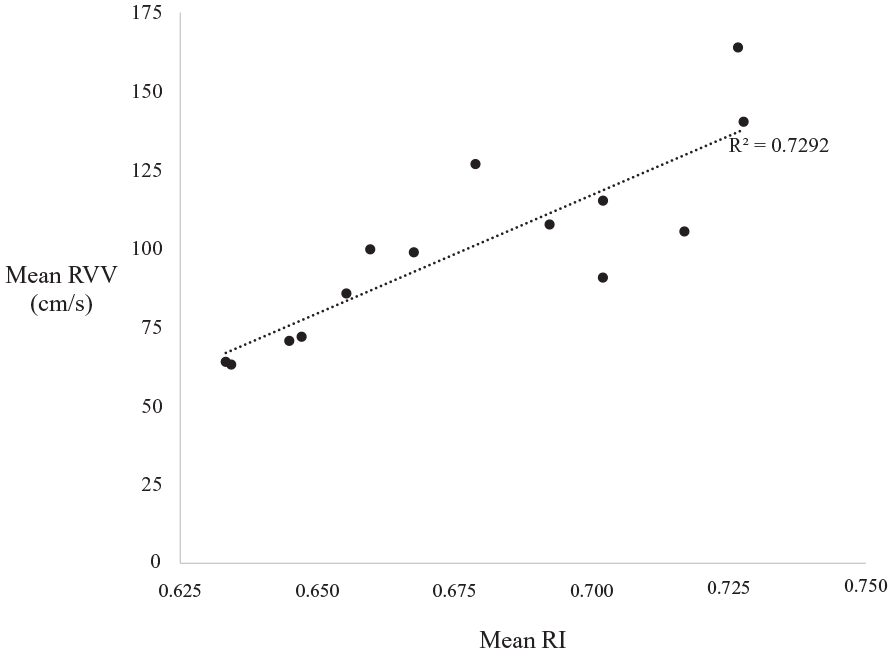
Mean RIs and mean RVVs for different days of scanning and positions.
Discussion
Maternal positional changes resulting in caval compression and reduced venous return are well described in the third trimester of pregnancy 3 with consequent changes in placental perfusion and fetal wellbeing.11,12 Similarly, increased creatinine in a mobile transplanted kidney has been reported. 4 Furthermore, changes in perfusion between supine and standing positions has also been infrequently reported in normally functioning grafts. 5 Thus, the aim of this study was to evaluate the effect of patient position on renal transplant perfusion parameters. Patients with higher weight and BMI have higher rates of DGF 7 but only a slightly greater rate of long-term graft loss suggesting that the effect of obesity is mostly in the peri-operative period. This led us to assess anthropomorphic factors.
Quarto di Palo et al. 13 reported cortical RIs of 0.54 in live donor grafts in healthy recipients with normal creatinine scanned more than 10 days post-implantation, and higher RIs in diabetic (0.58) and hypertensive (0.63) recipients. These values are also consistent with other studies,14–16 with arcuate artery RIs of 0.64, 0.65 and 0.61 in normally functioning grafts scanned at varying times after transplantation. Like Aschwanden et al., 16 we found higher mean supine RIs on days 3 and 7 than on days 1 and 30. RIs < 0.70 in some grafts on day 1, were greater than 0.70 on days 3 and 7. There were higher RIs in patients with DGF (see Table 4).
Despite the variation of the supine RI post-operatively, RIs were improved by positional change with no evidence of regression to the mean. The RIs of grafts with low supine RIs changed significantly only when standing, while the RIs of grafts with higher supine RIs decreased in all positions. We suggest that changing the patient position from supine reduces compression of the transplant renal vein and renal cortex by shifting the weight of overlying structures, including the abdominal wall pannus and omentum, off the kidney and transplant vessels. When patients are standing, although the measurement of the abdominal girth at the ASIS is greater than when supine, the overhanging abdominal pannus can fall forward with less weight resting on the kidney.
Most patients are nursed and managed supine in the early post-operative period. Graft compression is reduced with postural change and therefore nursing patients consistently in the decubitus position for extended periods in the early post-operative period may increase graft perfusion in flow-compromised grafts. This suggestion is also supported by the findings of a systematic review 8 of obesity and renal transplantation, which found that while rates of DGF are substantially increased in obese patients, the increased risk of graft loss in the longer term is small, suggesting that the major effect of obesity is peri-operative. We note that going to sleep in the supine position in the third trimester is now recognised as a modifiable and avoidable risk factor for stillbirth and small birth weight babies, and that this information is now provided widely in the public health arena to pregnant women and those planning childbirth. 17 Similarly, we suggest that this information regarding positional changes in graft perfusion should be provided to clinicians and renal transplant recipients as a modifiable risk factor to improve graft outcomes with patients encouraged to sleep and be nursed in the decubitus position. However, unlike pregnancy where the side of sleeping does not matter, the improvement in graft perfusion occurs with the kidney side up. We also note that the positional changes in RI with patients standing were even greater than in decubitus suggesting that early mobilisation may also be of benefit.
While abdominal girth, girth change, patient weight and weight gain peri-operatively were all significantly associated with higher supine RIs, abdominal girth was the most significant. This suggests that abdominal girth may be a more useful measurement in the pre-operative assessment of transplant recipients, than either of the conventional measures of weight and BMI, noting that girth was also more strongly related to positional change in RI than either weight or BMI.
It has previously been demonstrated that RVVs vary considerably from graft to graft. 18 We also found this, but there was a consistent reduction in mean RVV with change from the supine position. While the RVV of a single scan may not provide useful clinical information for an individual patient, the trend of velocity change does. We have also shown a strong relationship between RVV and RI with grafts with higher RVVs having higher RIs and therefore poorer perfusion.
This study is possibly limited by the unexpected inability or unwillingness of patients to undergo standing scans on day 3, but we suggest that the positional reductions in RIs in standing scans seen on days 7 and 30 would have also occurred on day 3. Similarly, while the patients who did not undergo positional scans generally had higher supine RIs than those that did, we expect that these patients also would have had positional changes in RIs.
Conclusion
We have demonstrated significant peri-operative positional changes in renal graft perfusion. Patient position is a modifiable factor that may improve transplant outcomes, particularly for at-risk grafts, and in recipients with high abdominal girth or substantial peri-operative weight and fluid gain. While further research is advised, patients should be informed of the effect on graft perfusion of lying on their side and side-sleeping, and encouraged to stand early in the peri-operative period. Abdominal girth was more strongly associated with high peri-operative RIs than either weight or BMI and should be included in the pre-operative assessment of potential graft recipients. There are additional implications for post-operative ultrasound scanning protocols and, in particular, documentation and reporting should include the position of the patient.
Footnotes
Contributors
L Thebridge (1,2,3), C Fisher (1,2,3), V Puttaswamy (1,2,3), C Pollock (1,2,3), J Clarke (1,2,3)
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Northern Sydney Local Health District (NSLHD) Health and Research Ethics Committee (HREC): 2020-ETH00443
Permission from patient(s) or subject(s) obtained in writing for publishing their case report
Yes.
Permission obtained in writing from patient or any person whose photo is included for publishing their photographs and images
No. Only de-identified ultrasound images used.
Confirm that you are aware that permission from a previous publisher for reproducing any previously published material will be required should your article be accepted for publication and that you will be responsible for obtaining that permission
Yes, aware. No previously published material is reproduced.
Guarantor
L Thebridge
