Abstract
Background:
Intracranial atherosclerotic stenosis is a common cause of ischemic cerebrovascular events and is associated with a high risk of stroke recurrence. This study aimed to assess the diagnostic accuracy of transcranial color-coded duplex sonography for moderate-to-severe middle cerebral artery stenosis in stroke patients.
Methods:
A retrospective analysis was carried out, including 31 patients aged ⩾18 years hospitalized for ischemic cerebrovascular event in whom middle cerebral artery stenosis ⩾30% was identified on computed tomography angiography. Transcranial color-coded duplex sonography findings were compared to the degree of stenosis blindly identified on the computed tomography angiography used as the reference method.
Results:
Overall, 27 patients had M1 stenosis and the other 4 had M2 stenosis. To detect M2 stenosis ⩾ 50% and ⩾ 70%, stenotic to pre-stenotic ratio ⩾ 2 and ⩾ 3 had a sensitivity of 100%, respectively. To detect M1 stenosis ⩾ 70%, peak systolic velocity ⩾ 300 cm/s had a sensitivity of 53.8% and specificity of 85.7% with area under the receiver-operating characteristic curve of 0.753 (95% confidence interval: 0.568–0.938; p
Conclusion:
This study showed that stenotic to pre-stenotic ratio ⩾ 3 was more sensitive than peak systolic velocity ⩾ 300 cm/s to screen M1 stenosis ⩾ 70%. Middle cerebral artery/anterior cerebral artery ratio < 0.7 was a good indirect sign to detect dampened pre-stenotic flow due to M1 stenosis ⩾ 70%.
Keywords
Introduction
Intracranial atherosclerotic stenosis (ICAS) is responsible for ischemic stroke in 8%–10% of cases1,2 and is associated with a high risk of stroke recurrence up to 15% per year.3,4 The prevalence of ICAS is higher in patients with arterial hypertension and diabetes1,5 and is influenced by ethnicity.2,6,7 In the European transcranial Doppler (TCD) ultrasound studies, the prevalence of symptomatic ICAS in Caucasian patients ranges from 2.2% to 12.4%.8 –12
The gold standard for the diagnosis of ICAS is digital subtraction angiography (DSA). 13 However, it is no longer used in routine clinical practice because it is invasive, costly, has limited access, and is associated with stroke risk.14 –17 Transcranial color-coded duplex sonography (TCCS), magnetic resonance angiography (MRA), or computed tomography angiography (CTA) are the imaging modalities used in routine clinical practice in the diagnosis of ICAS. TCCS has advantages, such as being less expensive, rapid, reproducible, and can be performed bedside. In addition, MRA is not suitable for every patient due to the ferromagnetic properties of some implanted devices, such as certain types of pacemakers/implantable cardiac defibrillators and old-generation mechanical heart valves. It is also contraindicated in patients with end-stage renal disease due to the risk of nephrogenic systemic fibrosis related to gadolinium-based contrast media use. 18 CTA also has contraindications due to the use of iodine-based contrast media which is not suitable for patients with allergies or non-dialysis-dependent chronic kidney disease with estimated glomerular filtration rate ⩽ 30 mL/min/1.73 m 2 . 19
Although TCCS is a reliable imaging method to detect ICAS, there is no standardized method for quantification of the degree of stenosis. Some diagnostic criteria are commonly used to quantify ICAS: (1) Baumgartner et al.’s
20
criteria based on TCCS and intra-stenotic peak systolic velocities (PSVs) whose thresholds are fixed to determine the degree of stenosis < or ⩾ 50%, and (2) Zhao et al.’s
21
criteria based on TCD and stenotic to pre-stenotic ratios (SPRs) used to determine the degree of stenosis ⩾ 50% and ⩾ 70%. However, TCCS and TCD have some limitations, such as their high operator-dependent nature and inadequate acoustic bone windows (ABW). The prevalence of inadequate ABW varies according to ethnicity, age, and gender. In Caucasian patients, the prevalence of inadequate ABW ranges between 8% and 20.1%8,10,11,22 while it varies between 16% and 30% in East Asian patients6,23,24 and reaches 47% in African Americans.
25
However, the rate of inadequate ABW could be reduced with the use of ultrasound contrast agents, such as sulfur hexafluoride (
Materials and methods
Study design and participants
We conducted a retrospective study between 1 June 2019 and 31 May 2020. We included consecutive patients aged ⩾ 18 years hospitalized for ischemic stroke or transient ischemic attack (ischemic cerebrovascular event (ICE)) in whom an ICAS ⩾ 30% was identified on the CTA. For patients with two or more ICAS ⩾ 30%, we only retained the symptomatic ICAS. ICAS was considered symptomatic when the stenosis was in the territory of ICE and asymptomatic otherwise. Patients with posterior circulation stenosis were excluded (small samples, see flowchart in Figure 1). Patients with carotid stenosis ⩾ 70% ipsilateral to MCA stenosis were not included. Patients who benefited from intravenous thrombolysis and endovascular thrombectomy were not included. All included patients had benefited from a cardiac exploration (echocardiography and cardiac telemetry for 3–4 days) that permitted the elimination of strokes of embolic origin. The diagnosis of ICAS was made when a patient with ischemic stroke meets at last two of the following neuroradiological patterns: (1) border-zone infarct within the territory of the artery affected by ICAS, (2) scattered cerebral ischemic lesions within the territory of the artery affected by ICAS, and (3) angiographic presence of a narrowing on one or more other arteries of the circle of Willis. 28
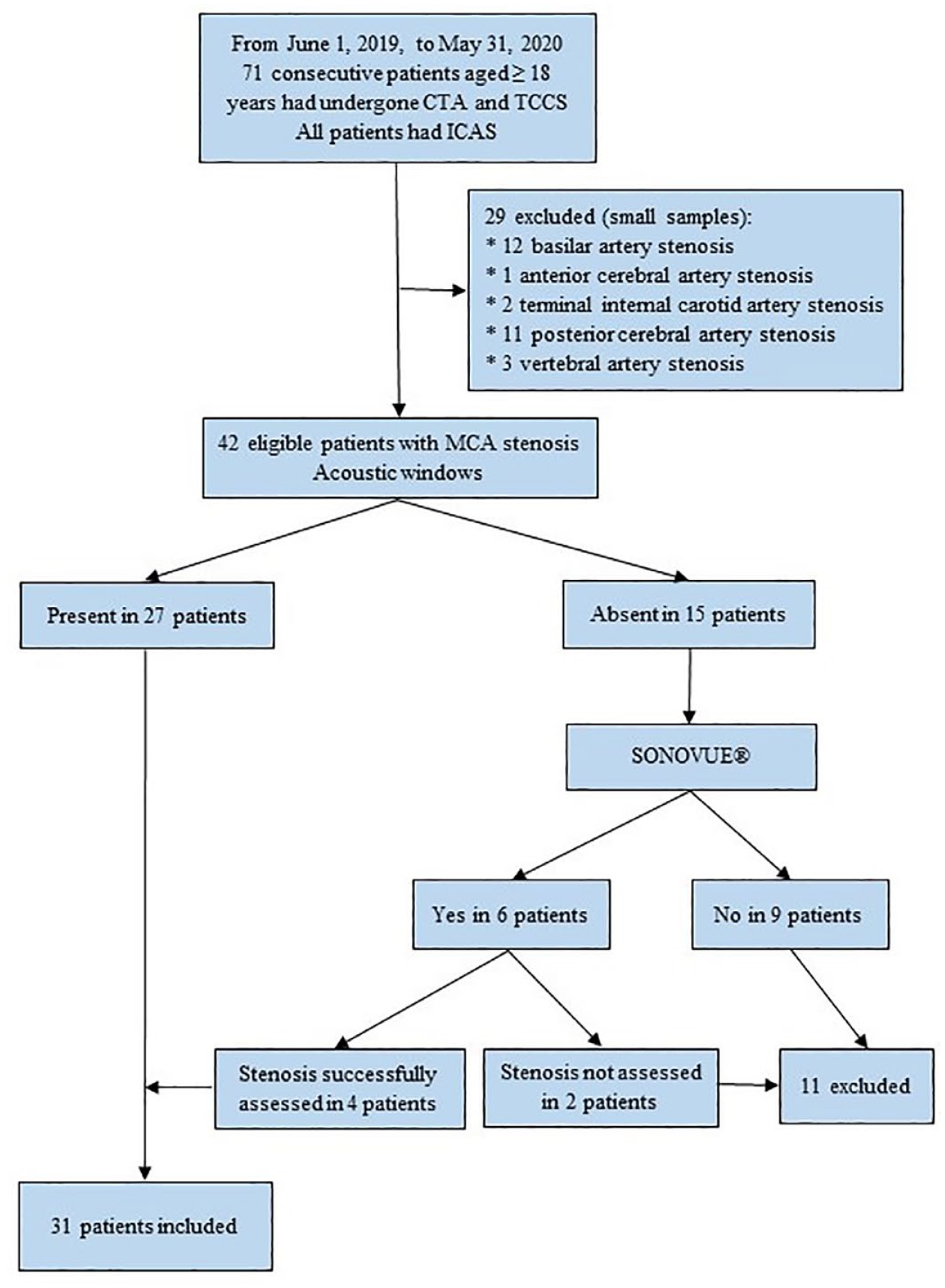
Flowchart of the study. CTA: computed tomography angiography; IQR: interquartile range; ICAS: intracranial atherosclerotic stenosis; MCA: middle cerebral artery; TCCS: transcranial color-coded duplex sonography.
Ultrasound exploration method
All examinations were performed by two neurologists experienced in the use of cerebrovascular ultrasound (MTD and CA). Cervical vessels were examined using a 3–12 MHz linear probe of the Philips Ultrasound System (Philips Affiniti 70G Ultrasound System, 22100 Bothell-Everett Highway, Bothell, WA, USA). Cervical Doppler ultrasound was performed in B-mode planes followed by a color mode sweep from proximal to distal segments of carotid and vertebral arteries. PSVs of both common carotid arteries, internal and external carotid arteries, and vertebral arteries were measured with angle correction (⩽60°). The degree of stenosis of the internal carotid arteries is determined according to the validated criteria. 29 Cervical Doppler ultrasound evaluations were performed to determine patients with carotid stenosis ⩾ 70% ipsilateral to MCA stenosis which were not included in this study. Carotid stenosis ⩾ 70% can hemodynamically influence velocities in MCA stenosis and distort the quantification of the stenosis using velocity criteria.
Intracranial arteries were examined using a 1–5 MHz phased-array probe of the same ultrasound system. The two ultrasonographers (MTD and CA) were blinded to all CTA results and used a consensual transcranial insonation protocol established based on a previously published ultrasound protocol.
30
Patients were examined in a supine position for the MCAs (M1 and M2 segments) and ACAs (A1 segment) through the transtemporal ABW. Some patients with inadequate ABW had benefited from TCCS after the intravenous injection of
To detect an MCA stenosis ⩾ 50%, we used both PSV according to the criteria of Baumgartner et al. 20 and the SPR according to the criteria of Zhao et al. 21 According to Baumgartner et al.’s 20 criteria, for MCA stenosis ⩾ 50%, the cut-off value for PSV was ⩾ 220 cm/s. According to the criteria of Zhao, SPR was calculated by dividing the highest stenotic MFV by the pre-stenotic MFV if the MCA stenosis was located on the distal vessel segment. When the MCA stenosis was located at the origin of the artery, the SPR was calculated by dividing the highest stenotic MFV by the MFV of the contralateral homologous segment at the same depth. SPR ⩾ 2 and ⩾ 3 defined MCA stenosis ⩾ 50% and ⩾ 70%, respectively. 21
MCA/ACA velocity ratio was used to assess the dampened pre-stenotic flow in MCA in this study. The calculation of this ratio comes from our experience in ultrasound practice, but the results have not been previously published. MCA/ACA ratio was calculated by dividing the highest pre-stenotic PSV of the M1 segment by the PSV of the A1 segment of ipsilateral ACA. Ratio < 1 was suggestive of probable dampened pre-stenotic flow in MCA. When stenosis was located at the origin of the M1 or M2 segment or if the A1 segment was hypoplastic or agenesic, the MCA/ACA velocity ratio was not calculated.
CTA
Used as the reference method, CTA was performed on admission or during hospitalization at our Stroke Unit (SU) or peripheral hospital where the patient consulted before being admitted to our SU. Cerebral angiograms were reviewed independently by two of the authors. For each patient, the degree of stenosis was calculated according to the Warfarin–Aspirin for Symptomatic Intracranial Disease (WASID) method:33,34 % stenosis = [1 − (Dstenosis/Dnormal)] × 100. Thus, the patients were grouped into three classes according to the degree of stenosis: 30%–49%, 50%–69%, and ⩾ 70%. When a ‘gap sign’ was observed on the CTA (i.e. the lumen of the vessel could not be visualized at the site of stenosis), the degree of stenosis was defined as ⩾ 80% stenosis. A plaque that induced stenosis < 30% was considered as nonstenotic plaque and therefore the artery was considered a nonstenotic artery. 35
Statistical analysis
For each MCA stenosis, the degree of stenosis blindly identified on the CTA was compared to the TCCS findings to calculate the sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and overall accuracy with corresponding 95% confidence intervals (CIs). Receiver-operator characteristic curve analyses were performed to evaluate the diagnostic accuracy of TCCS for MCA stenosis by determining areas under the receiver-operator characteristic curve of PSV, SPR, and MCA/ACA velocity ratio. Pearson’s correlation was used to test the association between the degree of MCA stenosis measured on CTA with PSV, SPR, and MCA/ACA ratio. PSV and SPR were included in multivariate linear regression analysis to determine independent predictors associated with the degree of MCA stenosis measured on CTA. Statistical analyses were performed with the SPSS statistical software (IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY: IBM Corp). The p-values less than 0.05 were considered statistically significant.
Results
A total of 71 patients with ICAS were enrolled, of which 42 had MCA stenosis. Among these 42 eligible patients with MCA stenosis, 15 had inadequate ABW with a prevalence of 35.7%. However, 6 of these 15 patients underwent TCCS with the injection of
TCCS findings versus CTA.
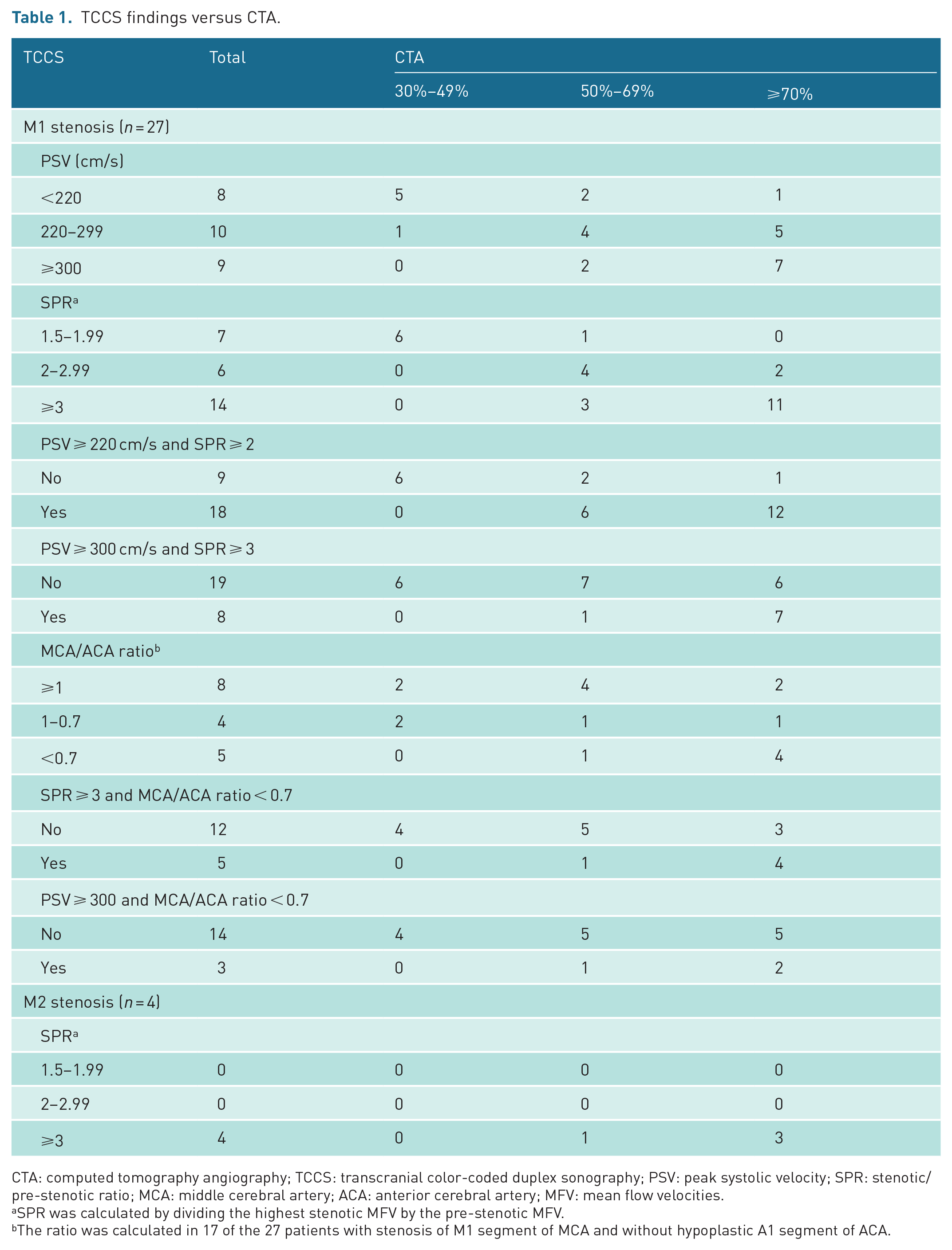
CTA: computed tomography angiography; TCCS: transcranial color-coded duplex sonography; PSV: peak systolic velocity; SPR: stenotic/pre-stenotic ratio; MCA: middle cerebral artery; ACA: anterior cerebral artery; MFV: mean flow velocities.
SPR was calculated by dividing the highest stenotic MFV by the pre-stenotic MFV.
The ratio was calculated in 17 of the 27 patients with stenosis of M1 segment of MCA and without hypoplastic A1 segment of ACA.
To detect M1 stenosis ⩾ 50%, PSV ⩾ 220 cm/s had a sensitivity of 85.7% and specificity of 83.3% with an area under the receiver-operating characteristic curve (AUC) of 0.937 (p
Predictive values and ROC areas for M1 stenosis ⩾ 50% and ⩾ 70%.
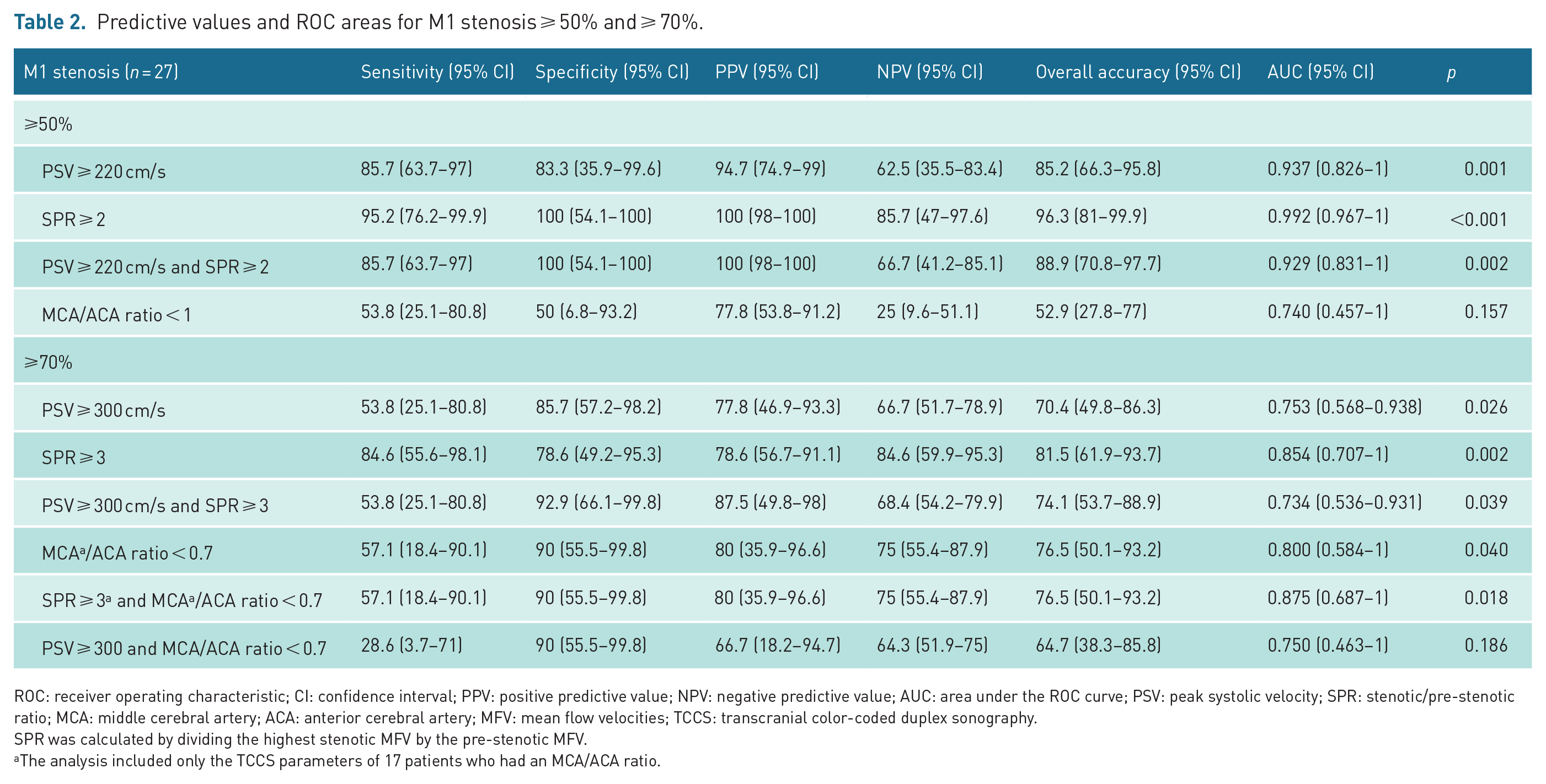
ROC: receiver operating characteristic; CI: confidence interval; PPV: positive predictive value; NPV: negative predictive value; AUC: area under the ROC curve; PSV: peak systolic velocity; SPR: stenotic/pre-stenotic ratio; MCA: middle cerebral artery; ACA: anterior cerebral artery; MFV: mean flow velocities; TCCS: transcranial color-coded duplex sonography.
SPR was calculated by dividing the highest stenotic MFV by the pre-stenotic MFV.
The analysis included only the TCCS parameters of 17 patients who had an MCA/ACA ratio.
To detect M1 stenosis ⩾ 70%, PSV ⩾ 300 cm/s had a sensitivity of 53.8% and specificity of 85.7% with an AUC of 0.753 (p = 0.026). SPR ⩾ 3 had a sensitivity of 84.6% and specificity of 78.6% to detect M1 stenosis ⩾ 70% with an AUC of 0.854 (p
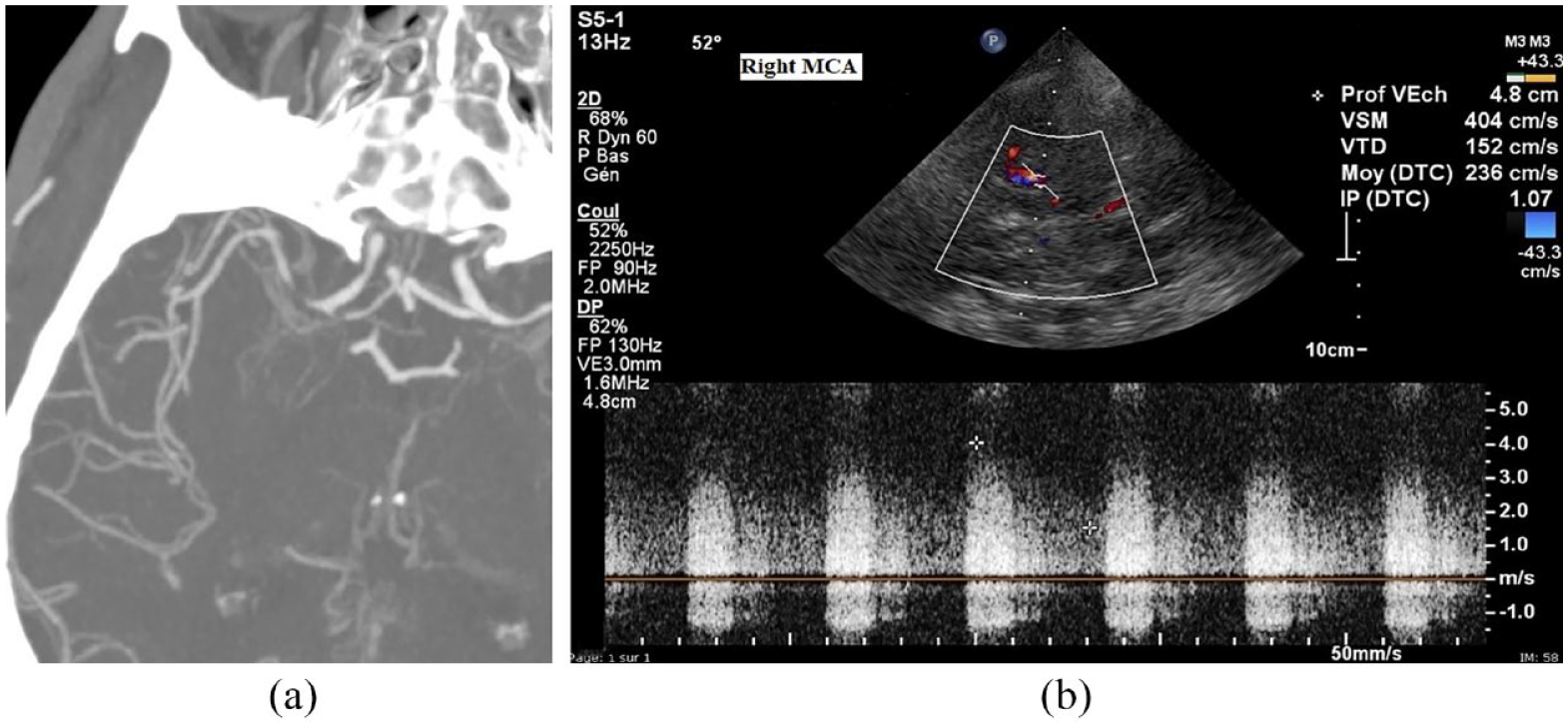
(a) CTA showing stenosis ⩾ 80% of M1 MCA (‘gap sign’ was observed). (b) TCCS showing an approximate PSV of 404 cm/s and MFV of 236 cm/s (calculated by manual tracing because of poor-quality sonogram) in the left M1 MCA, indicating ⩾ 50% MCA stenosis according to Baumgartner’s criteria (PSV ⩾ 220 cm/s) and ⩾ 70% according to our proposed PSV ⩾ 300 cm/s.
In this study, the MCA/ACA velocity ratio has been used as an indirect sign of M1 stenosis. It has been used to assess the dampened pre-stenotic flow in MCA secondary to downstream M1 stenosis. The ratio was calculated in 17 of the 27 patients with MCA stenosis. This ratio has not been calculated in the 10 other patients because either A1 was hypoplastic (n = 4) or the MCA stenosis was at the origin of M1 (n = 2), or the A1 PSV was unavailable (n = 4). Ratio < 0.7 had a sensitivity of 57.1%, specificity of 90%, PPV of 80%, and NPV of 75% to detect dampened pre-stenotic flow in MCA secondary to downstream M1 stenosis ⩾ 70% with an AUC of 0.800 (p
SPR ⩾ 3 alone had a sensitivity of 84.6%, specificity of 78.6%, PPV of 78.6%, NPV of 84.6%, and AUC of 0.854 to detect M1 stenosis ⩾ 70%. When SPR ⩾ 3 was combined with MCA/ACA ratio < 0.7, sensitivity decreased from 84.6% to 57.1% and NPV from 84.6% to 75.0%, but specificity increased from 78.6% to 90%, PPV from 78.6% to 80%, and AUC from 0.854 to 0.875. PSV ⩾ 300 cm/s alone had a sensitivity of 53.8%, specificity of 85.7%, PPV of 77.8%, NPV of 66.7%, and AUC of 0.753 to detect M1 stenosis ⩾ 70%. When PSV ⩾ 300 cm/s was combined with MCA/ACA ratio < 0.7, specificity increased from 85.7% to 90%, but sensitivity decreased from 53.8% to 28.6%, PPV from 77.8% to 66.7%, NPV from 66.7% to 64.3%, and AUC from 0.753 to 0.750. The combination of SPR ⩾ 3 and MCA/ACA ratio < 0.7 was more sensitive with better PPV, NPV, and AUC than the combination of PSV ⩾ 300 cm/s and MCA/ACA ratio < 0.7 for detecting M1 stenosis ⩾ 70% (Table 2).
A linear correlation was present between the degree of M1 stenosis measured on CTA with PSV (r
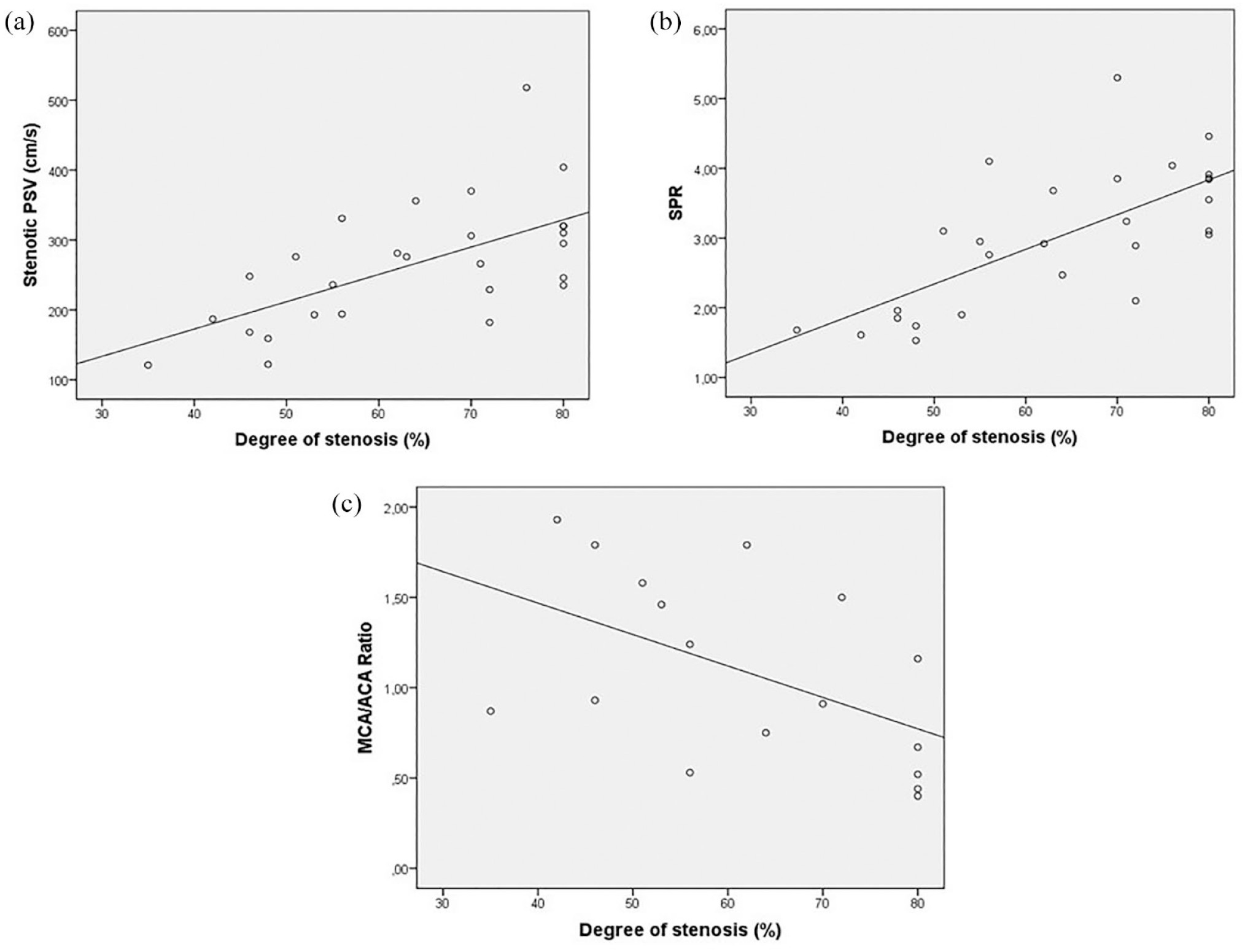
Correlation between CTA and TCCS for MCA stenoses. (a) Correlation between degree of MCA stenosis and PSV (r = 0.618; p = 0.001). (b) Correlation between degree of MCA stenosis and SPR (r = 0.705; p < 0.001). (c) Correlation between degree of MCA stenosis and MCA/ACA ratio (r = −0.517, p = 0.033).
Independent predictors associated with the degree of M1 stenosis (n = 27) by linear regression analysis.

SE: standard error; PSV: peak systolic velocity; SPR: stenotic/pre-stenotic ratio.
Discussion
Despite its limitations, as being operator-dependent and related to patients like inadequate ABW, this study showed that the visualization of severe MCA stenosis (⩾70%) on TCCS was highly correlated with the degree of MCA stenosis measured on the CTA that was chosen as our reference method. This correlation was significant in univariate analysis for the three methods used to detect MCA stenosis (PSV, SPR, and MCA/ACA velocity ratio) and remains significant in multivariate analysis for SPR. Stenotic PSV values measure for the assessment of ICAS was previously described by Baumgartner et al. 20 as sensitive and accurate comparable to DSA in a patient population with 90% of adequate ABW. This study showed that PSV ⩾ 300 cm/s, which was proposed to detect M1 MCA stenosis ⩾ 70%, had a low sensitivity (53.8%), although this was statistically significant. Nevertheless, the study showed that SPR ⩾ 3 (suggested criteria by Zhao et al. 21 ) had a high sensitivity to screen MCA stenosis ⩾ 70%. The study also showed that the TCCS parameters seem to perform better for detecting MCA stenosis ⩾ 50% than stenosis ⩾ 70% (see Table 2), with better AUC, sensitivities, specificities, PPVs, and NPVs. This could be explained by the fact that the PSV and MFV could decline in severe ICAS, which would decrease their sensitivity to detect severe ICAS. This shows the interest to evaluate the pre-stenotic flow and post-stenotic flow pattern of the M1 segment. To evaluate the pre-stenotic flow in this study, MCA/ACA ratio was used to assess this pre-stenotic flow. To our knowledge, this is the first transcranial ultrasonographic study that has used the MCA/ACA ratio to assess the pre-stenotic flow of M1 stenosis. A ratio < 0.7 was statistically significant to detect dampened pre-stenotic flow in MCA secondary to downstream M1 stenosis ⩾ 70%, although this sensitivity was low (57.1%).
Our study showed that to detect M1 stenosis ⩾ 70%, the combination of SPR ⩾ 3 and MCA/ACA ratio < 0.7 was more sensitive with better PPV, NPV, and AUC than the combination of PSV ⩾ 300 cm/s and MCA/ACA ratio < 0.7. Although the combination of SPR ⩾ 3 and MCA/ACA < 0.7 had lower sensitivity than SPR ⩾ 3 alone, specificity, PPV, and AUC were better with the combination (see Table 2).
A study assessing the accuracy of TCCS compared to CTA for detecting M1 MCA stenosis ⩾ 50% found a sensitivity of 54.0%, specificity of 94.0%, PPV of 87.0%, and NPV of 71.0% for PSV ⩾ 220 cm/s. 36 To detect M1 MCA stenosis ⩾ 50%, our findings found better sensitivity (85.7%) and PPV (94.7%) but low specificity (83.3%) and NPV (62.5%) for PSV ⩾ 220 cm/s compared to the results of the aforementioned study. Other studies also show a good correlation between TCCS and CTA for detecting M1 MCA stenosis ⩾ 50%.37,38 CTA was chosen as the reference method for diagnosing ICAS in this study because of its minimally invasive, inexpensive, and easier-to-realize nature compared to the DSA.
TCCS was performed with
Although it is rare that CTA and MRA simultaneously present a contraindication (due to the use of contrast media for the two imaging modalities or ferromagnetic properties of some implanted devices for MRA) and that the TCCS does not formally replace these two imaging methods for the diagnosis of ICAS, TCCS is superior to provide real-time flow direction, detect micro-embolic signals downstream of the stenosis, and evaluate collateralization in the cases of severe stenosis to occlusion of MCA by assessing real-time blood flow of A1 segment of contralateral ACA and ipsilateral posterior cerebral artery. Other advantages, TCCS is a safe, reliable, noninvasive, rapid, reproducible, and inexpensive method to diagnose ICAS that can be performed bedside.
The main novelty of this study was the use of the MCA/ACA ratio combined with SPR or PSV for detecting severe MCA stenosis. MCA/ACA ratio < 0.7 could be used as an additional criterion in clinical practice for MCA stenosis ⩾ 70%. This is based on the combined criteria for grading internal carotid artery stenoses, such as stenotic PSV, post-stenotic PSV, and internal carotid artery-to-common carotid artery velocity ratio. 29
The main limitation of this study was its retrospective nature explaining in part the low use of ultrasound contrast agents (
Conclusion
This study showed that TCCS was a reliable method for detecting M1 MCA stenosis ⩾ 70% as measured on CTA. SPR ⩾ 3 was a more sensitive and accurate ultrasound exploration method to screen M1 MCA stenosis ⩾ 70% compared to PSV ⩾ 300 cm/s. MCA/ACA ratio < 0.7 was a good indirect sign to detect dampened pre-stenotic flow due to M1 stenosis ⩾ 70%. Prospective studies are warranted to confirm the reliability of the MCA/ACA ratio < 0.7 for detecting dampened pre-stenotic flow due to M1 stenosis ⩾ 70%.
Footnotes
Contributors
MT-D designed the study, performed data acquisition and analysis, interpreted the data, wrote the first draft of the manuscript, and revised it critically for important intellectual content. NC performed data acquisition and critically revised the manuscript for important intellectual content. DS performed data acquisition and critically revised the manuscript for important intellectual content. CA designed the study, performed data acquisition and analysis, interpreted the data, wrote the first draft of the manuscript, and revised it critically for important intellectual content. All the authors read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The present study has been performed following the ethical principles depicted in the 1964 Declaration of Helsinki and its later amendments for Medical Research Involving Human Subjects. The local ethics committee of our institution waived ethical approval for this study because it only implied retrospective analysis of anonymized data collected as part of routine care.
Informed Consent
Not applicable.
Guarantor
MT-D.
