Abstract
Introduction:
Saving Babies’ Lives Care Bundle Version 2 highlights the importance of correct identification and reporting of echogenic bowel to improve maternal and newborn outcomes. Yet there is no national consensus to guide sonographers in identifying and reporting fetal echogenic bowel. This two-phase study aims to develop a national consensus to guide sonographers on the identification, classification and reporting of fetal echogenic bowel during the Fetal Anomaly Screening Programme (FASP) second trimester anomaly scan. Phase 1 results are presented capturing the national current practice of sonographers in its identification.
Methods:
An online questionnaire survey was deployed to capture numerical and free text data. Data analysis was by descriptive statistics. Participants were recruited via social media and through professional networks and organisations.
Results:
A total of 95 participants completed the questionnaire during an 11-week period. Common practice across England included sonographers using a subjective method for identifying fetal echogenic bowel and making comparisons to fetal bone. However, there was wide variance in the fetal bone used and the transducer frequency typically used to assess bowel echogenicity. Confirmation of echogenic bowel was made at the 20-week scan in 58% of cases, 32% following fetal medicine department review with the remaining 10% unsure when confirmation occurred.
Conclusion:
While there is common practice in identifying and report echogenic fetal bowel in some areas, there remains disparity within sonographer practice in England’s national screening service. This study allowed baseline data to be collated, providing the first steps towards development of guidance for sonographers in identifying and reporting this appearance.
Introduction
Fetal echogenic bowel was first described in the late 1980s and early 1990s as a normal variant found during obstetric ultrasound scanning.1,2 Reports of associations with chromosomal syndromes, however, began to emerge in the mid-to-late 1990s resulting in the finding being reclassified as a ‘marker’ for chromosomal syndromes.3 –5 A decade later, the term ‘marker’ was removed, and fetal echogenic bowel became a non-specific second trimester finding warranting onward referral for further evaluation. 6 Today, echogenic bowel is included as a high-risk factor in the Saving Babies’ Lives Care Bundle Version 2. 7
The prevalence proposed in the literature varies widely ranging from 2% to18% of all pregnancies,8,9 and it is therefore unsurprising that traditionally opinions have varied as to its significance as an isolated finding. The assessment methods for describing echogenic bowel also vary in the current evidence base and include both subjective and objective methods. The fetal structure to which the brightness of bowel is compared also varies in the available literature.
The Fetal Anomaly Screening Programme (FASP)’s national standards and guidance in relation to the reporting of fetal echogenic bowel 10 state echogenic bowel with density equivalent to fetal bone should be reported. There is no specific clarity, however, in relation to which fetal bone the comparison should be made or how the finding should be reported, for example, using a subjective method such as moderately echogenic or an objective method such as a grading system.
With the introduction of the Saving Babies’ Lives Care Bundle Version 2, 7 the correct identification and reporting of echogenic bowel is imperative as it can contribute to the reduction of stillbirth, morbidity, injury and preterm delivery as it is identified by the authors of that document as a high-risk factor. The non-identification of echogenic bowel at the 20-week scan can therefore have potentially serious implications for both mother and child.
A national consensus is required to guide sonographers in identifying and reporting fetal echogenic bowel. This two-phase study aims to develop a national (England) consensus to guide sonographers on the identification, classification and reporting of fetal echogenic bowel during the FASP second trimester anomaly scan. The Phase 1 results presented in this article capture the national current practice of sonographers defining echogenic fetal bowel.
Methods
Ethical approval for the study was from Sheffield Hallam University (ER36147075).
Phase 1 of the study captured sonographers’ current practice across England. England only is included and not the whole United Kingdom as the current FASP screening programme is applicable to England only. This allowed a baseline to be established as well as identification of the national consensus.
An online questionnaire survey including multiple choice and open-ended questions (Appendix 1) was piloted and then deployed to capture numerical and free text data. The choices included were informed by current relevant literature and participants consented through the platform. Responses were anonymous unless participants wished to add their contact details to participate in Phase 2.
All sonographers practising within English obstetric services were invited to participate from 5 November 2021 to 20 January 2022. Participants were recruited via social media and through professional networks and organisations, for example, the British Medical Ultrasound Society (BMUS) website. Quantitative data analysis in the form of descriptive statistics was conducted.
Results
A total of 95 participants completed the questionnaire with the majority identifying as sonographers (59%, n = 56), 28% (n = 26) advanced practitioners, 5% (n = 5) specifying ‘other’, 4% (n = 4) doctors and 4% (n = 4) midwives. Participants were from a wide geographical area.
All participants stated they identified fetal echogenic bowel by eye rather than using machine computerised software and the majority (n = 93, 98%) did this by comparing the brightness to other fetal structures. The remaining two participants did not provide any further detail as to how they identified echogenic bowel. All 95 participants stated they made the comparison to fetal bone with 14 (13%) also indicating they made comparisons to other fetal structures such as liver and lung.
The most common bone used for comparison was the femur bone (n = 54, 57%) followed by the pelvic bones (n = 35, 37%), participants also reported comparing to the spine (n = 16, 17%), ribs (n = 10, 10%) any long bones (n = 2, 2%) and any bone (n = 10, 10%), some participants listed more than one fetal bone structure in their responses.
Only seven (7%) participants used a grading system which consisted of three categories: mild, moderate or severe. When asked what frequency transducer participants typically used to optimally assess bowel echogenicity, some participants opted not to provide a response (n = 17), and some gave multiple values. The ranges stated differed too as some participants indicated the transducers were broadband or multifrequency. This made categorising the frequencies challenging; the range, however, was between 1 and 14 MHz from the 62 participants stating a figure. The most common response from these 62 participants was under 10 MHz (n = 49, 79%), with 13 (21%) using 10 MHz and over.
Further breakdown (Figure 1) identified the most commonly used transducer frequency was 2–9 MHz (n = 15, 24%), followed by 3–6 MHz (n = 9, 14%) and 1–5 MHz (n = 7, 11%). Several of the 80 participants also included comments with 11% (n = 9) stating they would use the highest frequency transducer available while conversely, 3% (n = 2) would select the lowest frequency transducer available. Dependency on the maternal body habitus and/or body mass index (BMI) was highlighted by 11% (n = 9) of participants despite not being asked specifically about this, and concerningly, 5% (n = 4) of participants indicated they were not sure what frequency they used.
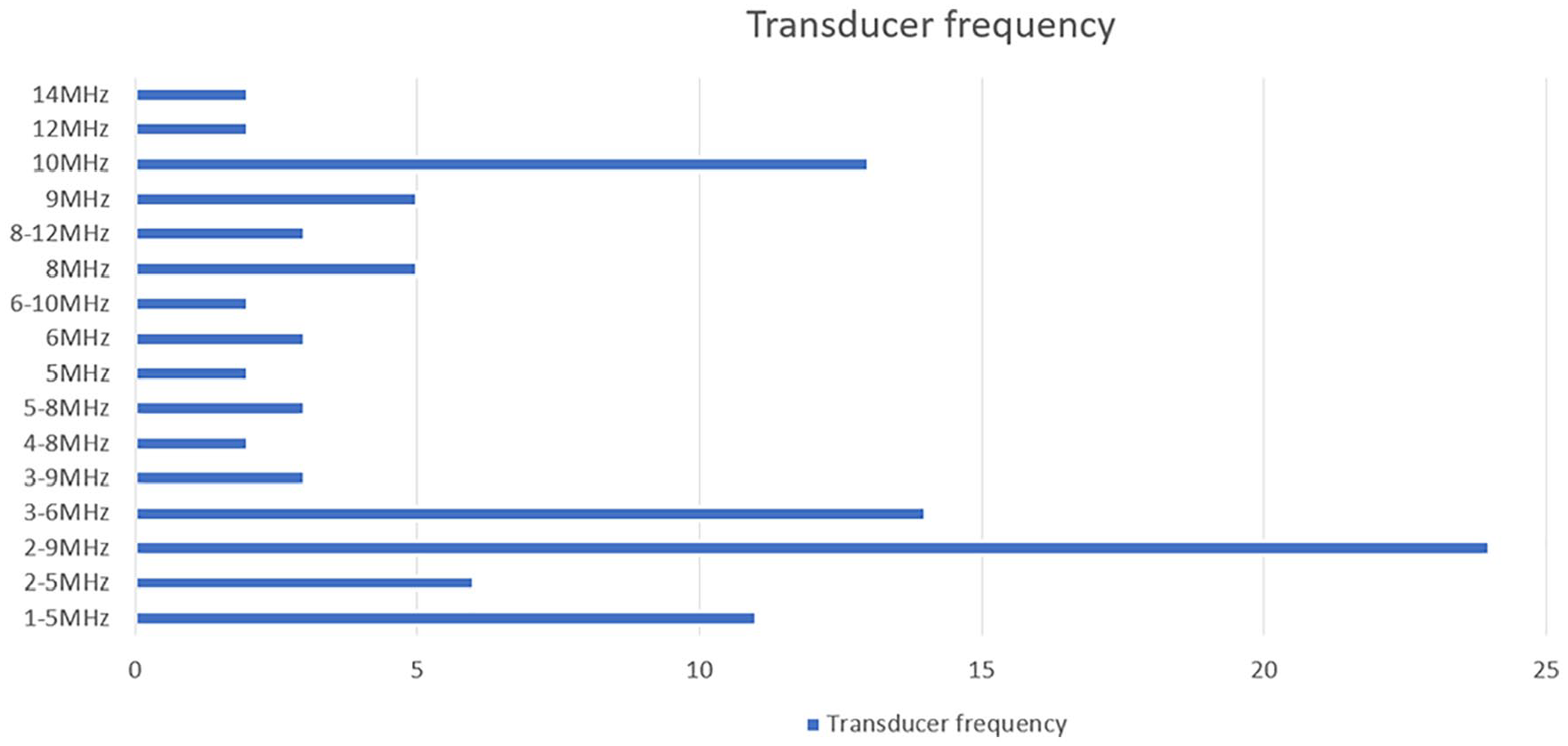
Further breakdown of transducer frequency used to optimally assess fetal bowel brightness.
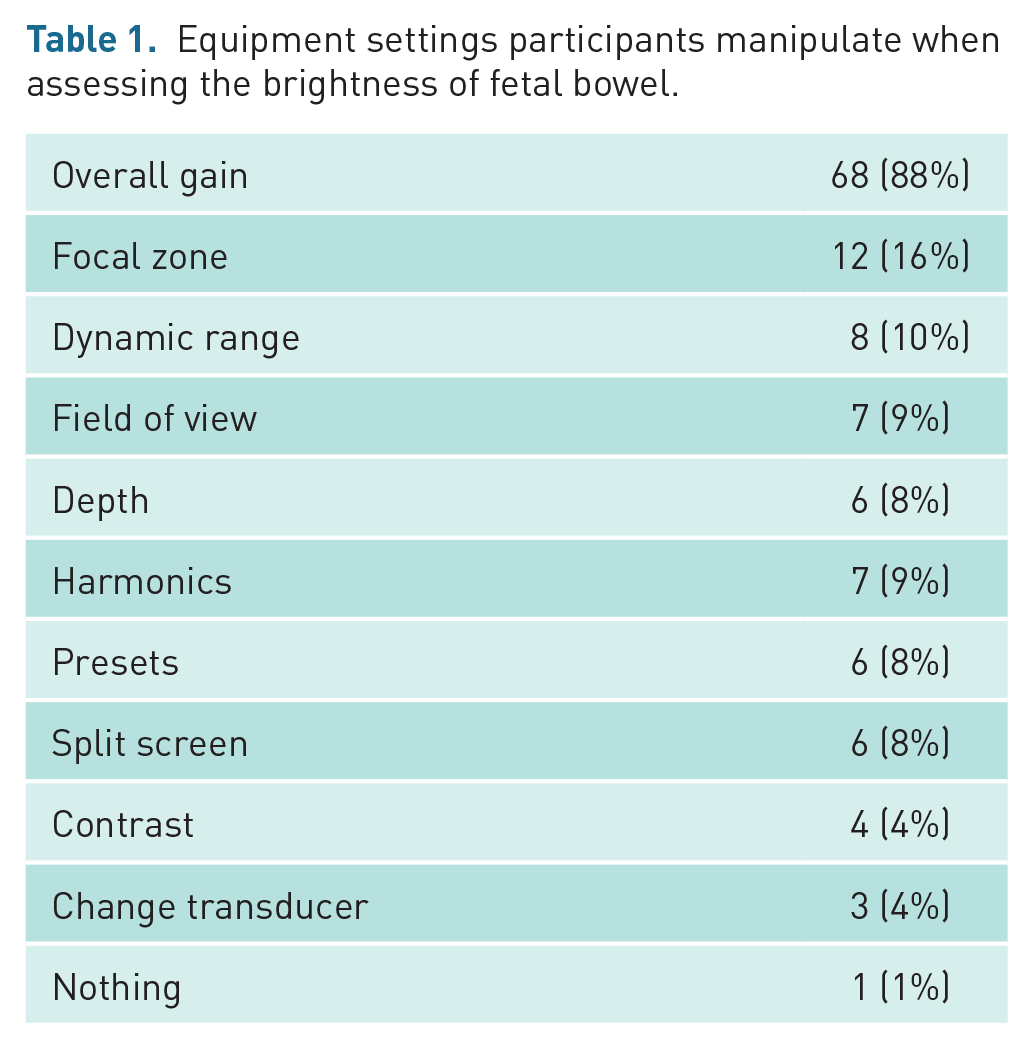
When asked what equipment settings participants (n = 77) would change when assessing the brightness of fetal bowel, some gave multiple responses. The majority (n = 68, 88%) stated the overall gain with other settings including the focal zone, field of view, depth, harmonics, contrast, dynamic range, and equipment presets (Table 1).
Equipment settings participants manipulate when assessing the brightness of fetal bowel.
Of the 78 participants responding to this question, echogenic bowel was confirmed by the local fetal medicine units in 45 (58%) of the participants departments and at the 20-week scan in 25 (32%) of the participants departments with the remaining 8 (10%) participants stating they were unsure when confirmation occurred.
Discussion
This study highlights common practice across some aspects of identification of echogenic fetal bowel as well as some differing practice mirroring the variation in the existing evidence base.
The current published literature presents a variety of methods for assessing and describing echogenic fetal bowel which includes both subjective and objective methods. The results from this study revealed the current practice across England was to use a subjective method (98% of participants) which relied on the sonographer making a comparison of the level or degree of echogenicity in relation to the other fetal structures. This has been reported typically to include a comparison to the fetal bone3,4,10 which was also the case in this study (100% of participants). However, 13% of participants also stated they made comparisons to other fetal structures, such as liver and lung, concurring with previous studies.11 –14 These participants did not offer any additional detail on how this occurred in practice.
The most common bone used for comparison in this study was the femur bone followed by the pelvic bones with responses including pelvis, pelvic bones, ileum, and iliac crest. Participants also reported comparing to a variety of other fetal bony structures including the spine, ribs, long bones, and any bone. This variation is unsurprising given the lack of guidance for sonographers; current national guidance advocates the reporting of echogenic bowel which has a density equivalent to fetal bone 10 without specifying which fetal bone the comparison should be made to. Other publications do offer additional detail in relation to which fetal bone the comparison should be made, for example, Chudleigh et al. 15 advocate the comparing to the iliac crests, although the authors are silent on whether single or multiple transverse, coronal and/or sagittal sections should be used. There is little/no published literature exploring whether the actual bone used for the comparison is relevant, or whether or not adjusting specific controls affects the echogenicity of the bowel and the comparator bone equally.
To decrease the inter observer variation, grading systems have been advocated in the past to quantify the degree of echogenicity of fetal bowel12,16 (Table 2).
Grading systems for echogenic fetal bowel.
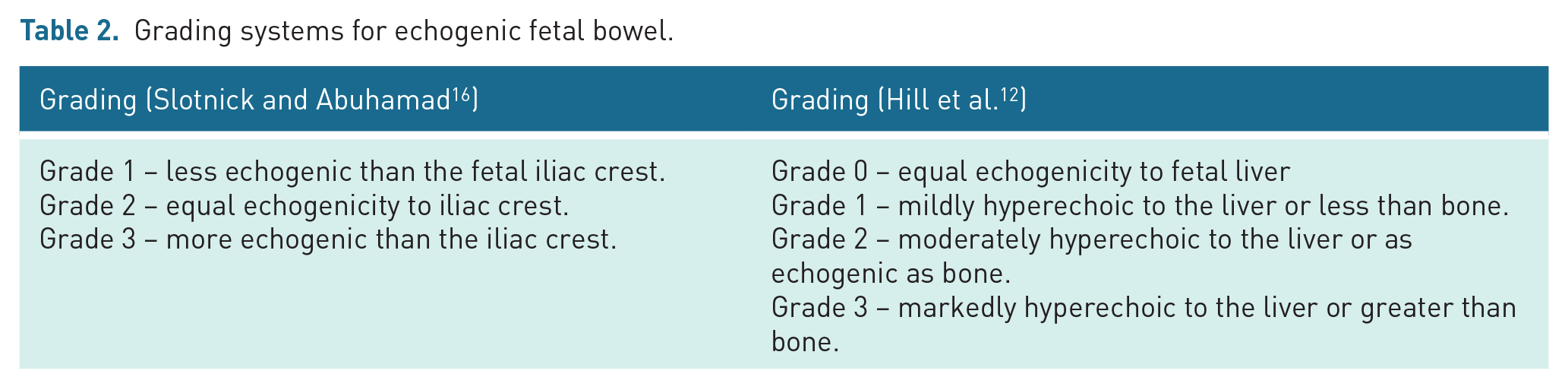
However, there is still subjective reliance on the sonographer to compare the bowel echogenicity to other fetal structures reducing the objectivity of this method of assessment. Only 7% of participants in this study used a grading system which consisted of three categories: mild, moderate or severe. While this appears to align with the Hill et al.’s 12 study, the terms used differ slightly.
Objective methods have been developed using quantitative computerised grey scale analysis17,18 to overcome the equipment gain and/or settings and inter-patient variability as well as the use of maximum pixel density of echogenic bowel compared to the density of the fetal liver. 19 Other authors have attempted to use quantitative assessment methods to demonstrate the effect of different equipment settings on the level of echogenicity of the fetal bowel. Lee and Cho 20 concluded in their study that the use of harmonics, an equipment image processing technique, resulted in the fetal bowel appearing to have an increased level of echogenicity. Leibovitz et al. 21 concurred with this finding that the use of harmonic imaging in early second trimester scans may significantly increase the brightness of the fetal bowel. Only 9% of participants in this study recognised the effect of harmonics on identification of echogenic bowel suggesting this is not widely implemented practice.
The frequency of the ultrasound transducer used for scanning was also found to impact on the level of brightness or echogenicity of fetal bowel with higher frequency transducers producing false positive results. 22 This study revealed a wide variation in the frequency of transducers used to optimally identify echogenic fetal bowel with 7% advocating the use of high-frequency transducers (greater than 10 MHz). There were only 19% of participants using transducers in the 1–5 MHz range to make the diagnosis and a small number (3%) stating the lowest frequency available.
There were a number of participants who did not know what frequency transducer they used, and this seeming lack of awareness was also seen in relation to whether the diagnosis of echogenic bowel was made at the 20-week scan or following review by a fetal medicine consultant. Just over half of the participants in this study confirmed diagnosis of echogenic bowel at the 20-week ultrasound scan themselves whereas just over a third of cases were not confirmed until after review by a fetal medicine consultant.
However, reassuringly, a significant number of the participants in this study indicated they would adjust the overall gain to make the diagnosis. In addition, there were several other equipment settings participants would manipulate to gain optimal image quality, including to account for different patient body habitus.
The lack of clarity, even in national guidance, as to how sonographers should identify and define echogenic bowel appears to have led to inevitable variations in practice and reliance on professional autonomy. However, it is worthy of note that sonographers are legally accountable for their professional actions and in the absence of clearly defined guidance, they are at increased risk of being subject to litigation on account of their professional actions. A key component in medicolegal cases is to determine whether a sonographer’s actions were justified, and they acted in accordance with a responsible body of sonographers. This would therefore apply to their diagnosing of fetal echogenic bowel, for example. There is an urgent need for national guidance to support sonographers in the identification, diagnosis and reporting of echogenic fetal bowel.
This phase of the project has allowed an exploration of the evidence base for best and suggested practice and aligned it to current practice. From these findings and the current published literature, the following recommendations are made to ensure a consistent approach to practice:
Identification by ‘eye’ with optimal equipment settings for that particular patient.
The assessment should be made with the suspected echogenic bowel and the bone to which it is being compared in the same image. This is often not possible if using the femur as the comparator and is important for professional accountability.
We, therefore, advocate the comparison should be made in a single coronal view using both iliac crests as the comparator bone.
Turning down the overall gain until the bowel and/or bone disappear.
The novel findings of the study have highlighted variation in sonographer practice in relation to the identification of fetal echogenic bowel. This is perhaps unsurprising giving the paucity of comprehensive guidance both in the United Kingdom and internationally. The findings of the study have allowed recommendations to be developed to assist sonographers and to promote consistent practice across English obstetric screening at the 20-week scan.
The strengths of the study include capturing the national consensus of sonographer practice across English screening services and developing recommendations for practice which are not currently in existence. This phase of the study creates a foundation for Phase 2, development of a guidance document for sonographers.
It must be acknowledged that there were only 95 participants in the study which may represent a selection of those practicing in the FASP screening services. Public Health England confirmed they have approximately 2000 ultrasound practitioners registered with Down’s syndrome screening Quality Assurance Support Service (DQASS). However, this number also includes some private providers and fetal medicine consultants and does not relate to those performing the 20-week screening scan alone (Public Health England, personal communication). The exact number of sonographers performing 20-week anomaly scans remains unknown.
Conclusion
Phase 1 of this study has provided valuable insight into the current practice of English sonographers participating in this study when identifying echogenic fetal bowel. It has revealed that there is a national consensus on using a subjective method of assessment of the echogenicity of fetal bowel. It has also highlighted that common practice is to reduce the overall gain settings to aid diagnosis, but there is wide variation in terms of the manipulation of other equipment settings.
Furthermore, while there appears to be a national consensus on the comparison of the bowel brightness to fetal bone, there is a disparity on which bone is used and the technique with which to make this comparison. There is also inconsistency in the transducer frequency used increasing the risk for false positive cases. This exacerbates the need for guidance for sonographers to be able to undertake this reliably and consistently. Recommendations have been made in this phase of the project and will be built upon in the next phase of this project.
Footnotes
Appendix 1
Acknowledgements
The authors first thank the participants of this study for their insight and involvement. The authors also thank Emma Tucker, BMUS for compiling the online survey and online poll, Suzanne Beattie-Jones, Queen Charlotte’s & Chelsea Hospital for providing images for the pilot study and Sonia Cumming, Rosie Hospital for highlighting the issue through local survey.
Contributor
T.C. is the contributor for this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethics approval for this study was obtained through Sheffield Hallam University (ER36147075).
Informed consent
Written informed consent was obtained from the patient for publishing her case details and images.
Guarantor
T.J.S. is the guarantor for this article.
