Abstract
Introduction:
Adenomyosis is histologically defined by the presence of endometrial glands and stroma in the myometrium. Ultrasound findings of adenomyosis are being redefined to better diagnose adenomyosis pre-operatively.
Methods:
A single-centre retrospective study was performed at a regional hospital. The myometrial–cervical ratio was calculated on pre-operative ultrasounds, and histopathology reviewed for each case. Logistic regression was used to estimate the association between the myometrial–cervical ratio and adenomyosis confirmed on histopathology, and the area under the receiver operating characteristic curve was calculated. Comparisons were performed based on the presence of fibroids on ultrasound.
Results:
Complete data were available for 136 benign hysterectomies between 1 January 2015 and 31 December 2020. When the myometrial–cervical ratio was treated as a continuous variable in a logistic regression of adenomyosis on histopathology, there was no statistical evidence (χ2(1) < 0.01, p = 0.98) of an association. When cases including fibroids in the myometrial–cervical ratio were excluded, there was a non-significant association between myometrial–cervical ratio and adenomyosis on histopathology (odds ratio = 3.435, 95% confidence interval = 0.964, 12.235; χ2(1) = 3.62, p = 0.057); area under the receiver operating characteristic = 0.637 (95% confidence interval = 0.504, 0.770). The optimal myometrial–cervical ratio cutpoint was 1.875 (95% confidence interval = 1.698, 2.051), which achieved 71.43% sensitivity and 60.00% specificity. There was strong statistical evidence (χ2(1) = 9.02, p = 0.003) that the myometrial–cervical ratio outperformed standard pre-operative ultrasound identification of adenomyosis.
Conclusion:
While accuracy remains suboptimal, the myometrial–cervical ratio outperformed standard ultrasound diagnostic features of adenomyosis in a regional setting. The myometrial–cervical ratio may offer a simple imaging measurement for adenomyosis in inexperienced hands when fibroids are absent.
Introduction
Adenomyosis is a pathologic disorder where endometrial glands and stroma are present within the uterine myometrium. 1 While some women with adenomyosis may be asymptomatic, the majority report pelvic pain, dysmenorrhea, abnormal uterine bleeding (AUB) and subfertility.2,3 The reported prevalence of adenomyosis varies widely from 5% to 70%. 4 There are multiple factors that contribute to a precise prevalence remaining unclear. These include a lack of consensus surrounding diagnostic ultrasound features, evaluation of histopathology remaining the gold standard for diagnosis and the similarity of the clinical features of adenomyosis to other pelvic pathologies.4,5
In 2015, a Morphological Uterus Sonographic Assessment (MUSA) was published to aid in the standardisation of ultrasound findings and more recently in 2021 these findings have been redefined in attempt to better diagnose adenomyosis on ultrasound.6,7 Ultrasound findings for adenomyosis remain somewhat subjective with terminology such as ‘globular’, ‘ill-defined’ and ‘diffuse vs localised’. Other findings although more specific have minimal clinical relevance and/or require a high level of ultrasound training to interpret. 7 A recent meta-analysis of transvaginal ultrasound diagnosis of adenomyosis found it has high sensitivity as a non-invasive tool. Transvaginal ultrasound performed best with the use of a combination of MUSA criteria; however, individually the criteria had low sensitivities or limited data. 8
The myometrial–cervical ratio (MCR) has previously been reported as a potential tool in the diagnostic armamentarium for adenomyosis by Mooney et al. 9 and McCaughey et al.; 10 however, its accuracy is suboptimal. This previous study was performed in a metropolitan centre, with onsite specialist gynaecology ultrasound. In regional settings where there is minimal access to specialist gynaecology scanning, it is unknown whether the MCR has a role in pre-operative diagnostics. This study aimed to further assess the accuracy of the MCR in the diagnosis of adenomyosis, and the utility of the MCR as a diagnostic tool in a regional setting without access to specialist gynaecology imaging.
Materials and methods
Study design
We conducted a single-centre retrospective study that evaluated the diagnostic performance of the MCR in a regional health service.
All patients who underwent hysterectomy at a regional health service in Victoria, Australia, between 1 January 2015 and 31 December 2020 were identified by the health information services using the International Classification of Diseases (ICD) coding description ‘hysterectomy’. Cases were excluded based on the following criteria if: the hysterectomy was performed for an obstetric indication; the hysterectomy was performed due to malignancy; malignancy was found in the pathology; and those without available or adequate pre-operative ultrasound images.
Medical records were reviewed by a junior gynaecological registrar and data recorded. Data included participant age, parity, menopausal status, use of exogenous hormones, prior surgery involving uterus or cervix and the indication for hysterectomy. The histopathology reports were reviewed for presence and absence of adenomyosis, fibroids and malignancy. The histopathological assessment of the hysterectomy specimen was used as a reference test as this is the diagnostic gold standard for adenomyosis. 7
The pre-operative ultrasound images were reviewed by the same junior gynaecological registrar (M.S.), following training by a specialist sonologist (K.S.). They were blinded to the medical records and histological findings at the time of reviewing the images. Ultrasound images were performed by general sonographers at various locations in the surrounding regional area and reported by general radiologists. The MCR was calculated from a single standard sagittal view that included both the uterine body and cervix. Two measurements were performed: one at the mid-point in the cervical canal and a second at the greatest anterior posterior diameter of the myometrium at the fundus (Figure 1). Fibroids were included in the calculation of the myometrial component of the MCR.
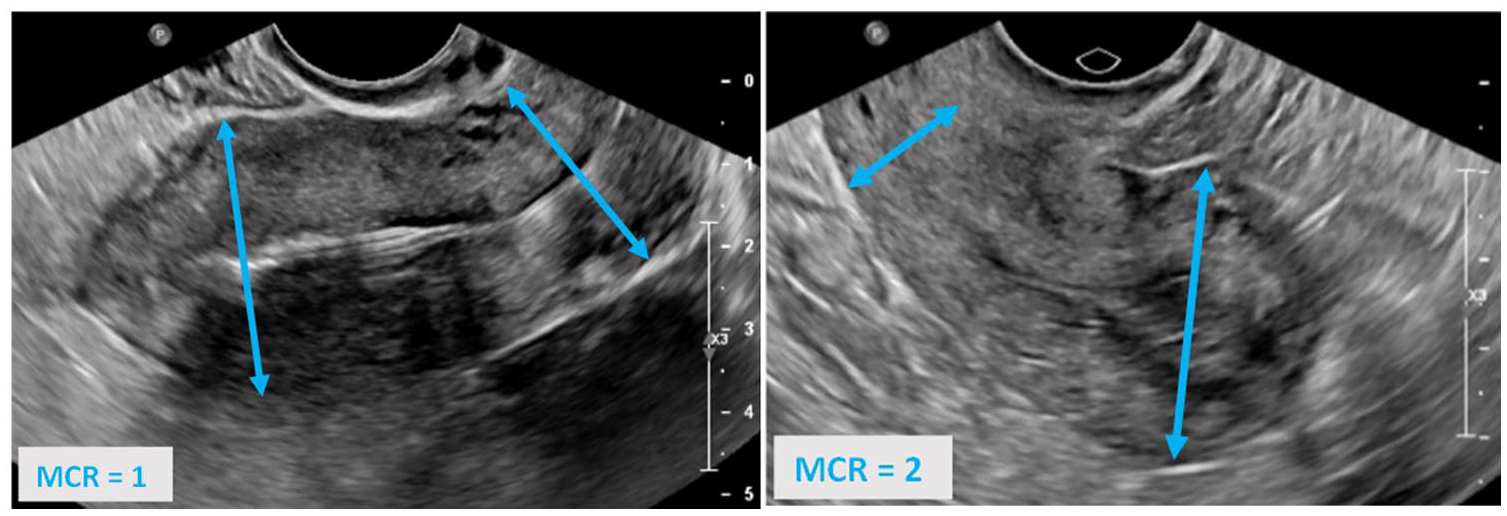
Diagrammatic representation of the myometrial–cervical ratio (MCR) measurement.
The study was approved by the Northeast Health Wangaratta Human Research and Ethics Committee (reference: LNR/73071/NEHW-2021-249166(v1)). Informed consent was not required for this retrospective audit. A file review of time course and outcomes was performed and patient care was not altered in any way during the course of this audit; in addition, all information was de-identified.
Data analysis
Statistical analyses were conducted using Stata/BE v17.0 (StataCorp LLC, College Station, TX, USA). Participant characteristics were compared using the chi-square tests for categorical variables and using independent sample t-tests for continuous variables (or non-parametric rank-sum tests for skewed continuous variables). Logistic regression was used to estimate the association (odds ratios with 95% confidence intervals (CIs)) between the MCR and adenomyosis confirmed on histopathology, with the area under the receiver operating characteristic (AUROC) curve calculated. The optimal MCR cutpoint was chosen to maximise model sensitivity and specificity using the cutpt package. 11 The 95% CIs were determined from 10,000 bootstrapped samples. Performance (as AUROC) was compared to a previously described out-of-sample MCR cutpoint of 1.74 using the algorithm proposed by Mooney et al. 9 and DeLong et al. 12 as implemented in Stata. Comparisons and subgroup analyses were then performed based on the presence of fibroids on ultrasound. Statistical significance for all comparisons was set at p < 0.05.
Results
Between 1 January 2015 and 31 December 2020, 377 hysterectomies were performed. The prevalence of adenomyosis on histopathology, taken from the total sample was 37%. Figure 2 describes the identification of eligible participants. One case was excluded as the hysterectomy was performed for obstetric indication, and 12 excluded due to malignancy. Of the remaining 364 cases, pre-operative pelvic ultrasound images were available for 136 cases; 202 cases were excluded due to pre-operative imaging not being available, and 26 excluded due to available images being inadequate to perform the MCR.
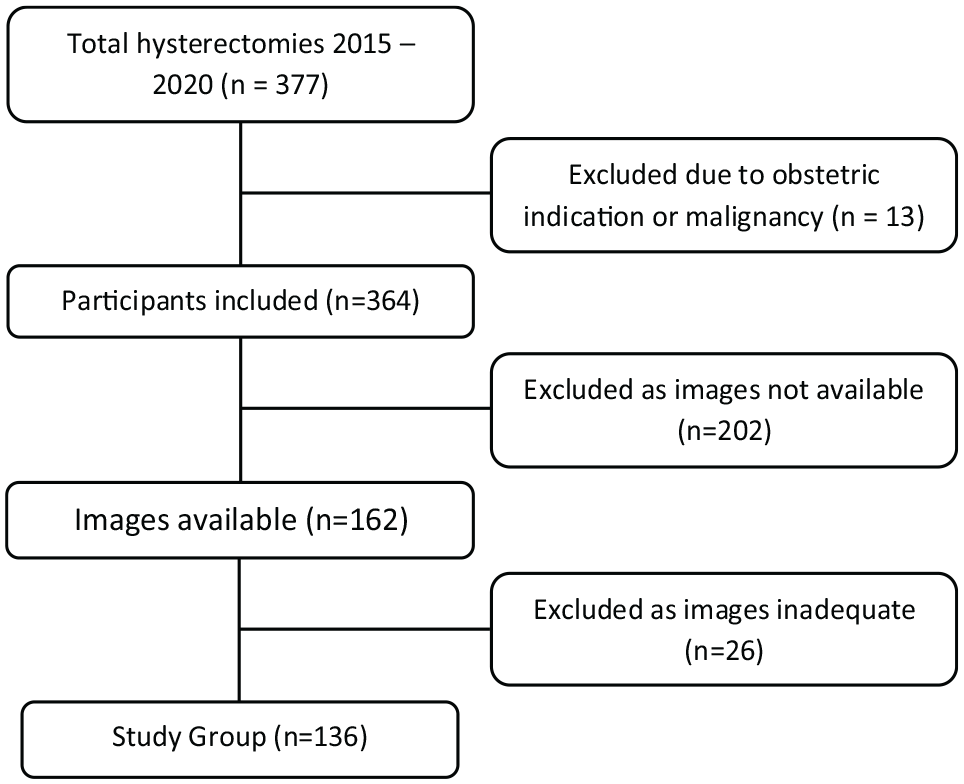
Flowchart of participants.
The demographic and clinical features of the cohort are presented in Table 1. The median time between ultrasound and surgery was 156 days. This was significantly longer in those who had adenomyosis (216 days), compared to those who did not (145 days, p = 0.045). There was no difference in menopausal status, parity, use of exogenous hormones and uterine fibroids, between those who did or did not have adenomyosis. Most hysterectomies were performed for AUB; the second most common indication was pelvic organ prolapse.
Patient demographic and clinical features in relation to presence of histological adenomyosis.
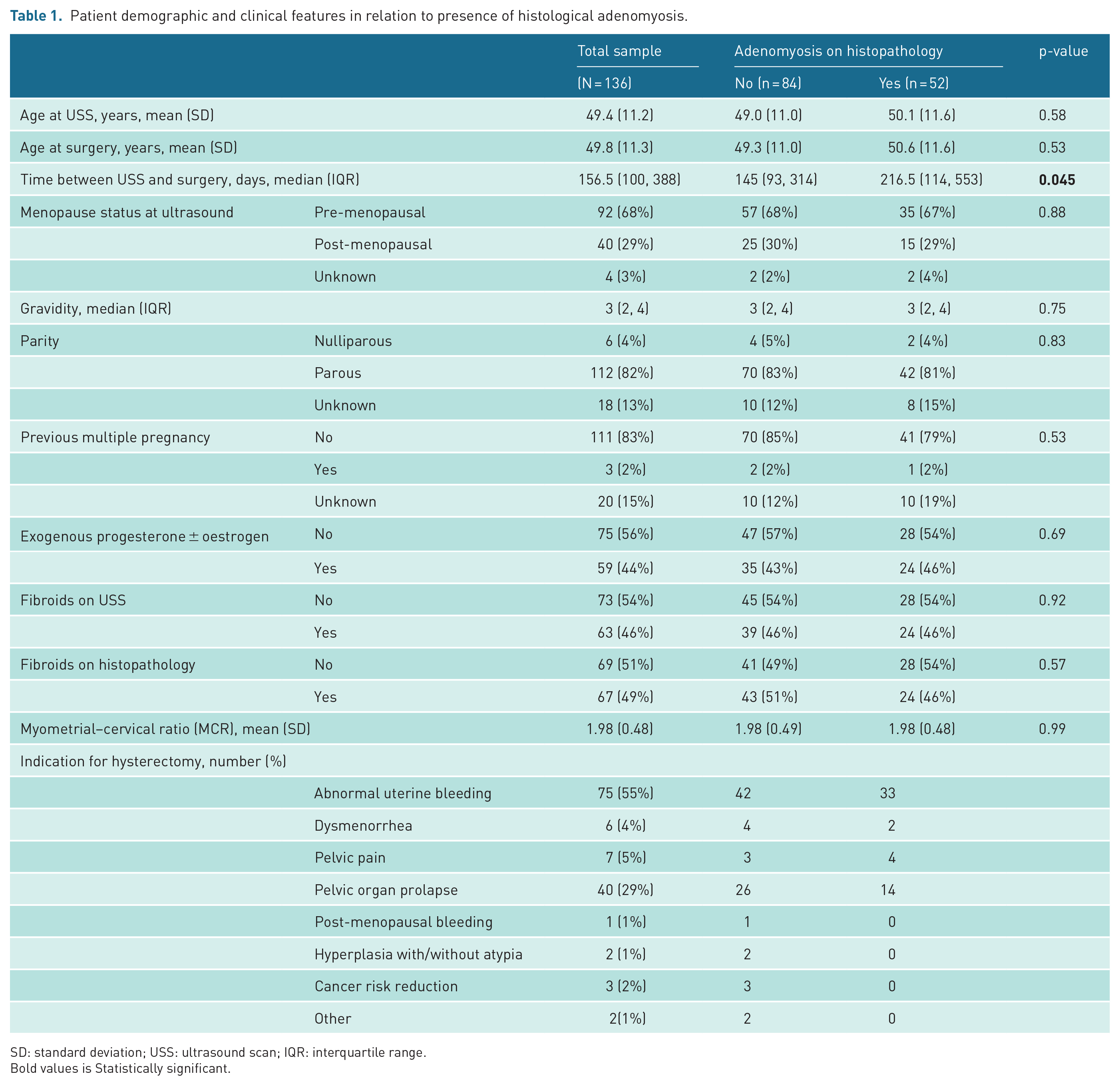
SD: standard deviation; USS: ultrasound scan; IQR: interquartile range.
Bold values is Statistically significant.
There was no statistical evidence (χ2(1) < 0.01, p = 0.98) of an association between the MCR and adenomyosis on histopathology, when the MCR was treated as a continuous variable in a logistic regression of adenomyosis on histopathology (odds ratio (OR) = 0.993, 95% CI = 0.483, 2.041). The AUROC for this model is 0.516 (95% CI = 0.416, 0.616; Figure 3).
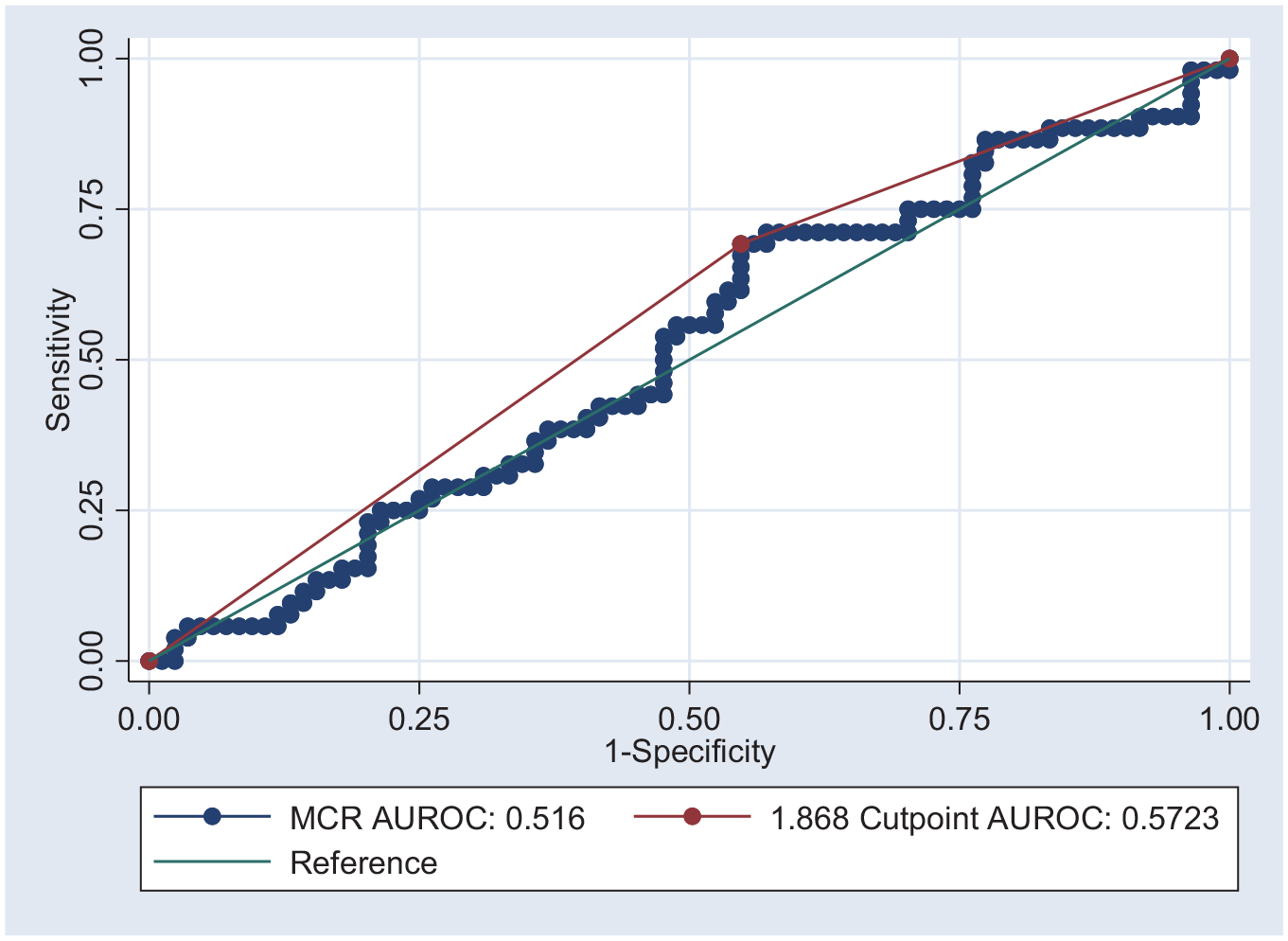
Area under receiver operator curve for MCR using all samples (N = 136).
The optimal cut-off for MCR to maximise sensitivity and specificity for adenomyosis using this sample was 1.868 (95% CI = 1.731, 2.006 from 10,000 bootstrapped samples). Using this cutpoint achieved 69.23% sensitivity and 45.24% specificity, with 54.41% correct classification. Using this MCR cutpoint in logistic regression results in an OR of 1.859 (95% CI = 0.987, 3.853; p = 0.096; AUROC = 0.572).
There were 63 cases where fibroids were described on pre-operative ultrasound and 73 without. There was no difference in the absence of fibroids on ultrasound between those who did or did not have adenomyosis (p = 0.92). A comparison of the performance of MCR for adenomyosis based on the presence of fibroids on ultrasound scan (USS) identified a statistically significant difference (χ2(1) = 6.26, p = 0.012), with MCR performing better in the absence of fibroids on USS (AUROC = 0.641, 95% CI = 0.507, 0.774) compared to when they were present (AUROC = 0.388, 95% CI = 0.241, 0.534). When observations that included fibroids on ultrasound were excluded from the analysis sample (n = 73), there was a trend towards an association between MCR and adenomyosis on histopathology, but this remained statistically insignificant (OR = 3.435, 95% CI = 0.964, 12.235; χ2(1) = 3.62, p = 0.057). The AUROC for this model was 0.637 (95% CI = 0.504, 0.770; Figure 4). The optimal MCR cutpoint in this subgroup was 1.875 (95% CI from 10,000 bootstrapped samples = 1.698, 2.051), which achieved 71.43% sensitivity and 60.00% specificity, with 64.38% of samples correctly classified.
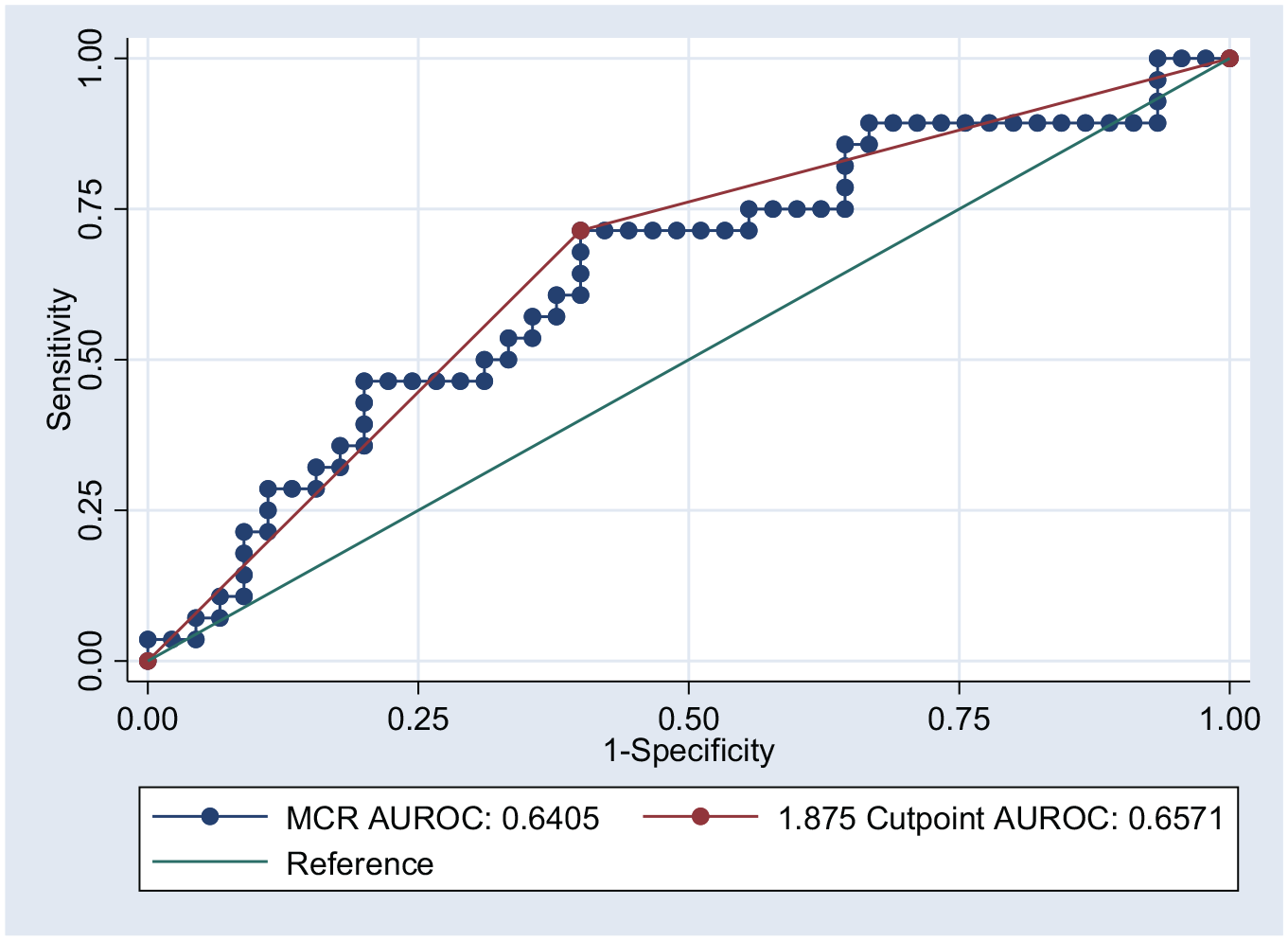
Area under receiver operator curve for MCR excluding samples with fibroids on USS using an MCR cutpoint of 1.875 to optimise sensitivity and specificity (n = 73).
On review of the 136 included participants pre-operative ultrasound reports, only 6 cases were reported to have adenomyosis. Only one of these cases was confirmed histologically, the other five did not have adenomyosis. Standard ultrasound reporting in the regional setting correctly classified 1 on the 52 histologically confirmed cases (1.92%). There was statistical evidence (χ2(1) = 4.18, p = 0.041) that MCR outperformed standard ultrasound identification of adenomyosis (AUROC = 0.480, 95% CI = 0.448, 0.512). When fibroids were excluded, there remained strong statistical evidence (χ2(1) = 9.02, p = 0.003) that MCR outperformed standard ultrasound identification of adenomyosis (AUROC = 0.473, 95% CI = 0.419, 0.528).
Discussion
In this study, the prevalence of histological adenomyosis was 38%. Prevalence is currently calculated using histopathology as the gold standard of diagnosis, and reported rates vary significantly. 4 Many studies, including ours, use histopathology as an index test; however, ultrasound is more commonly used as a first-line method to diagnose and guide clinical management of adenomyosis.8,9,13 In the regional setting, there is limited access to specialist imaging, and unless a hysterectomy performed, adenomyosis is likely to go undiagnosed. Ongoing research into developing imaging tools to aid in diagnosing adenomyosis pre-operatively, could help provide a diagnosis for women experiencing pelvic pain and heavy menstrual bleeding, and in turn further clarify clinical features and effectiveness of non-surgical management options of adenomyosis.
Overall, this study demonstrates that the MCR has lower accuracy in diagnosing adenomyosis pre-operatively, as was shown previously by Mooney et al. 9 and McCaughey et al. 10 However, in the regional setting, where due to resources pre-operative ultrasound diagnosis of adenomyosis is uncommon, the MCR may be a useful adjunct, as it outperformed the traditional non-specialist sonographic measures. In the absence of fibroids, the MCR sensitivity was 71.4% and specificity 60.0% using the cutpoint of 1.875. Using the MCR in the absence of fibroids the diagnosis adenomyosis was correct in 64.38% of cases, which is superior to reporting by the region’s general sonographers and radiologists which was accurate in less than 2% of cases.
Current ultrasound criteria used to diagnose adenomyosis have been found to have higher sensitivity and specificity rates when using a combination of multiple sonographic features including myometrial wall asymmetry, myometrial cysts, hypoechoic linear striations, fan shadowing, lines and buds, vascularity and heterogeneous myometrium. However as individual measurements, their sensitivities and specificities are wide ranging, or in other cases, such as fan shadowing, there is limited data to assess them.5,8 The recently revised MUSA guidelines have categorised the finding of a globular uterus as an ‘indirect’ finding of adenomyosis, reflecting the impact on the myometrium on the presence of adenomyosis. 6 Similar to the MCR, the finding of a globular uterus can be affected by the presence of fibroids; however, where a globular uterus is subjective, the MCR is a more specific measurement.
Detection rate of adenomyosis is significantly higher when the ultrasound is performed by a specialised gynaecological sonographer. 14 This study shows, however, that the MCR can be successfully applied by a gynaecology trainee, with similar outcomes to when used by a specialist sonologist. 9 Given the lack of specialised ultrasound services in regional settings, the MCR may be a useful tool to improve adenomyosis detection.
This study has several limitations. The study population was compiled from women who had a hysterectomy, creating a selection bias by excluding any women who may have adenomyosis but not had a hysterectomy. Due to the retrospective design of the study, there is potential bias in the collection of data from records that may have missing information or been interpreted incorrectly. Many of these cases were excluded as pre-operative ultrasound images were not available, which reduced the sample size and may be a source of bias.
In the absence of fibroids, the MCR is a simple tool with possible clinical utility in the regional setting. While it has been shown that the MCR is inadequate as a solo diagnostic test, there is potential for this measurement to be a useful adjunct if specialist USS is unavailable.
Footnotes
Acknowledgements
The authors acknowledge the Obstetrics and Gynaecology Department of Northeast Health Wangaratta.
Contributors
Megan Sewell contributed to the acquisition of data, drafting of article and final approval of the article. Samantha Mooney contributed to the conception and design, revising article critically and final approval of the article. Erin Cvejic contributed to the analysis and interpretation of data, revising article and final approval of the article. Kate Stone contributed to the conception and design, lead on research project, revising article critically and final approval of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical approval
This study was approved by the Northeast Health Wangaratta Human Research Ethics Committee (reference no. LNR/73071/NEHW-2021-249166(v1) and date of approval: 10 February 2021).
Guarantor
MS is the guarantor of this article.
