Abstract
Introduction:
Liver biopsies are the main method in the diagnosis and treatment of paediatric liver pathologies. Major complication rates of paediatric liver biopsies range from 0% to 6.6% in the literature and minor complication rates range from 0% to 25%. In this study, we aimed to review the complications, indications and results of percutaneous core liver biopsies with paediatric sonography in a tertiary care centre by an interventional radiologist.
Methods:
We retrospectively evaluated the results, indications and complications of paediatric liver biopsies performed in our tertiary health centre between January 2017 and December 2020. Biopsies were performed with a 16G semi-automatic needle in 17 patients (29.8%) and with an 18G semi-automatic needle in 40 patients (70.2%). Biopsies were performed only with local anaesthesia in patients older than 12 years; in younger patients, it was performed under general anaesthesia.
Results:
Fifty-eight liver biopsies were obtained from 57 children (34 males, 23 females). The most common indications were elevated liver enzymes (33 patients), cholestasis (14 patients), and adiposity and metabolic problems (6 patents). The most common pathological diagnoses were chronic hepatitis (33 patients) and steatosis (10 patients). Major complication in the form of symptomatic subcapsular haematoma developed after liver biopsy performed with 18G needle in only one patient (1.8%).
Conclusions:
As previously stated in the literature, percutaneous biopsies performed by interventional radiologists in paediatric patients under the guidance of sonography can be used in diagnosis and treatment; the complication rate is low and it is a safe method.
Introduction
Histopathological analysis is required in both children and adults in terms of definitive diagnosis, grading, determination of prognosis and monitoring of treatment efficacy of congenital or acquired diseases of the liver. The necessary tissue acquisition methods for histopathological analysis are transjugular biopsy, surgical biopsy taken by laparotomy and percutaneous needle biopsy. Percutaneous liver biopsy is a frequently used method in terms of being easily performed on an outpatient basis, being safe and being cost-effective.
Percutaneous liver biopsies can be performed blindly or under ultrasound guidance. Ultrasound allows for optimal needle entry site marking for biopsy and biopsy needle tracking by avoiding bile ducts and blood vessels with real-time imaging. In experienced hands, the procedure is performed relatively easily at the bedside using procedural sedation. However, complications may develop due to the fact that it is an invasive procedure.
Paediatric liver biopsies have been increasingly performed by interventional radiologists in recent years. However, there is a lack of experience in the literature regarding percutaneous liver biopsies performed in the interventional radiology setting in paediatric patients, and reported data are limited. This article describes our experience with the safety and efficacy of percutaneous liver biopsies in 57 paediatric patients under standard real-time sonography guidance in a tertiary care paediatric patient group.
Materials and methods
The study was carried out with the permission of the University Clinical Ethics Committee (date: 2019, Decision No: 2019-11-4). All procedures that were performed adhered to the ethical rules and principles of the Helsinki Declaration.
Medical records of all paediatric patients who underwent ultrasound-guided percutaneous liver biopsy at a tertiary care centre between January 2017 and December 2020 were retrospectively reviewed. During a 4-year period, 58 percutaneous liver biopsies were performed in 57 paediatric patients under ultrasound guidance. Medical records of all patients included demographic information such as age and gender, indication of liver biopsy, laboratory findings, size and type of biopsy needle used, failed biopsies, sedation technique, severity and rate of post-procedure complications and pathological outcome. Laboratory tests, including blood count and coagulation parameters, were performed 1 day before the procedure and between 4 and 6 hours after the procedure in terms of possible bleeding complications. Exclusion criteria are unexplained bleeding history, blood group owners where blood is not available for transfusion, those with suspected vascular tumour such as possible haemangioma, arteriovenous malformation, those with suspected echinococcal cysts in the liver, those with unsuitable blood count and coagulation parameters, those with a tendency to bleed, those with morbid obesity, those intra-abdominal with massive acid, infection in the right pleural space or infradiaphragmatic area.
Complications that may occur due to the procedure were classified as major and minor. Major complications were death, transfusion, bleeding requiring surgical intervention or intensive care, haemobilia, gallbladder perforation, pneumothorax, haemothorax and conditions requiring prolonged hospitalisation for more than 1 day. Symptomatic clinical manifestations such as bleeding, hypotension and tachycardia were defined as a decrease in haemoglobin (Hb) of 2 g/dL or more than 5% in haematocrit within 24 hours. Minor complications were determined as high fever (>38.5°C), vomiting, pain requiring analgesics, asymptomatic subcapsular haematoma and mild leakage from the biopsy site. Local pain not requiring painkillers and fever below 38.5°C were not considered as complications.
Biopsy technique
This study was conducted in accordance with the principles set forth in the Declaration of Helsinki, and informed consent was obtained from the patients’ parents. In the laboratory tests performed before the procedure, biopsy was performed for the patients who fulfilled the condition that Hb, which is used as the safe reference limit values of our institution, was above 7 g/dL, international normalised ratio (INR) was less than 1.5 and the platelet count was 60,000/mm3. The condition of not receiving antiaggregant or anticoagulant treatment was sought in the patients. All procedures were performed in a vascular and interventional radiology unit or operating rooms equipped with procedural and anaesthesia equipment. The procedure was performed by radiologists with experience in interventional procedures, accompanied by real-time ultrasonography (USG). The adequacy of the biopsy specimen was determined by the radiologist as the sufficient amount of tissue required to make a diagnosis. The size of the biopsy needle was at the operator’s discretion. Biopsy was performed under local anaesthesia or general anaesthesia. Local anaesthesia (subcutaneous dose: 10 mg/mL) was administered with ropivacaine in compatible patients older than 12 years; general anaesthesia with midazolam (IV dose: 0.1–0.2 mg/kg, max: 5 mg) was administered to children younger than 12 years of age or incompatible children during the procedure. In all cases, hepatic USG was performed using 3.5 and 12 MHz probes (Toshiba Aplio 500) to determine the safest site and optimal biopsy approach by avoiding intraparenchymal vascular structure and biliary structures. Intercostal, subxiphoid or the shortest and safest orbit was chosen according to diffuse or focal liver disease (Figure 1). If biopsy was to be performed from the intercostal space, the needle was inserted through the intercostal space along the upper part of the costal margin to prevent possible intercostal artery injury. Under real-time ultrasound guidance, while the patient was in full exhalation and apnoea, in the free-hand sonography technique using 16G (17 patients) or 18G (40 patients) semi-automatic biopsy needles, the needle was advanced to the liver capsule and then to the hepatic parenchyma and the biopsy sample was taken. After the biopsy, an average of 5 minutes of compression was applied to accelerate haemostasis manually. A limited ultrasound was performed by the radiologist at the end of the procedure to assess possible acute complications. After the procedure, all children were followed up for 24 hours for haemodynamic status evaluation, a constant pulse oximeter, temperature, respiratory rate and arterial blood pressure measurement and possible complications, starting with 30 minutes and increasing intervals. Pre-procedure haematocrit values were compared with follow-up haematocrit values. Additional USG was performed in addition to blood examination in patients whose haemodynamics were disturbed during the follow-up. If no complications were encountered in the patient, the patient was discharged after informing his family.
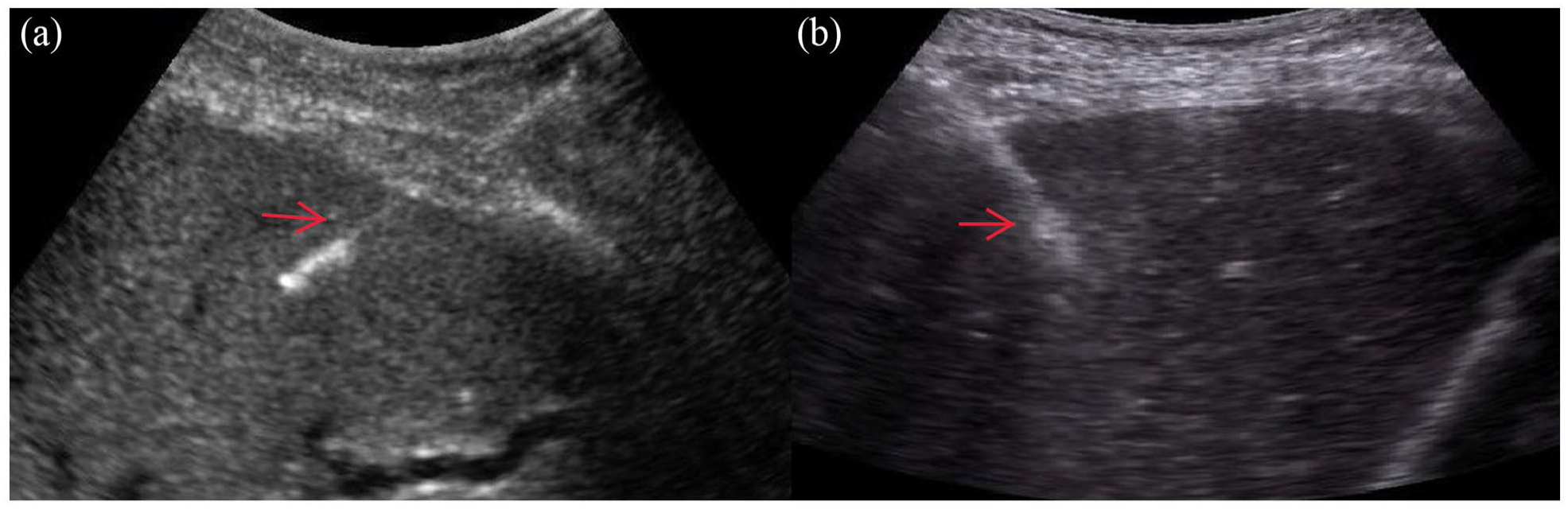
Intraprocedural sonogram shows biopsy needles in the liver (red arrow).
Results
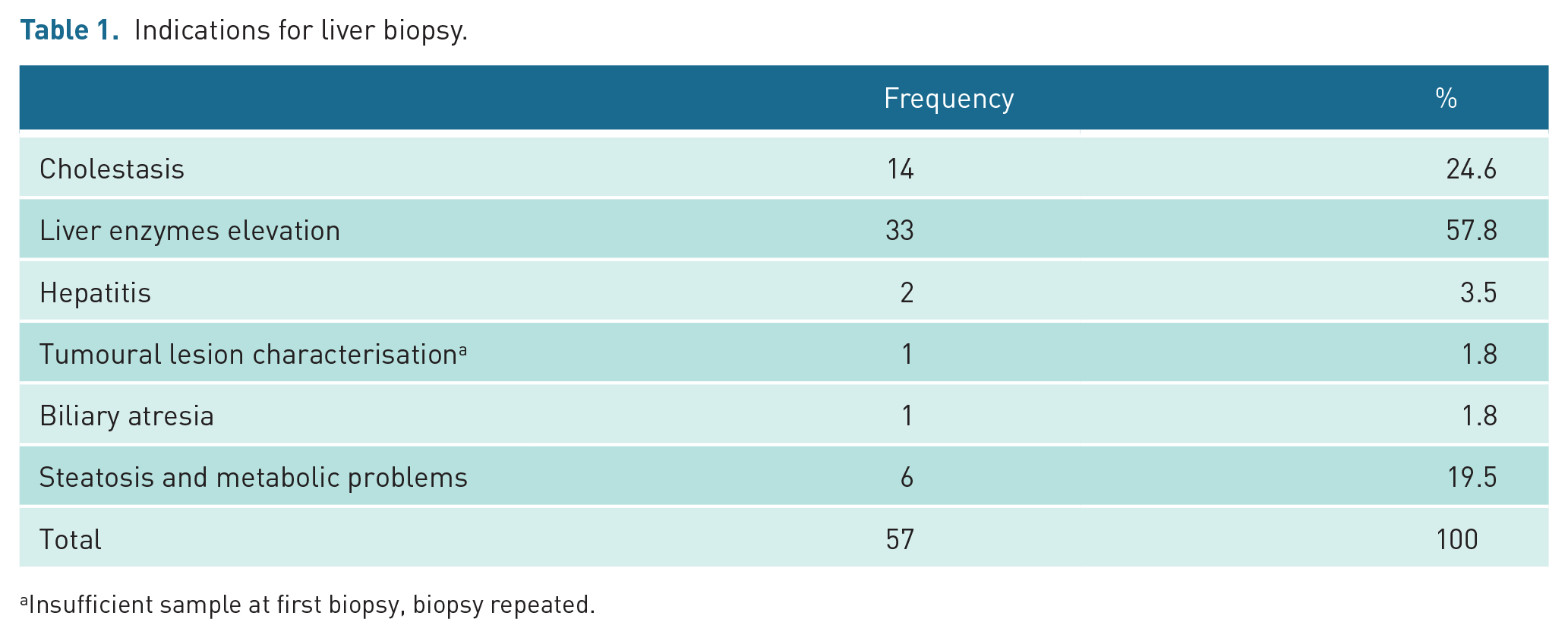
Over a 4-year period, 57 children aged 1–204 months (mean age 9 years) underwent 58 ultrasound-guided percutaneous liver biopsies; of the patients, 34 (59.6%) were male and 23 (40.3%) were female. All biopsies were performed in native liver. Most of these biopsies were performed to evaluate diffuse parenchymal liver diseases (n = 57, 98.2%). Our most common preoperative biopsy indications were unexplained elevation of liver enzymes (n = 33, 57.8%), cholestasis (n = 14, 24.6%), steatosis or a suspected metabolic liver disease (n = 6, 19.5%). Clinical indications for liver biopsies are presented in Table 1. The median INR of the children was 1.09 (<1.5) and the median platelet count was 295 G/L (>60 G/L).
Indications for liver biopsy.
Insufficient sample at first biopsy, biopsy repeated.
Sedation
Biopsy was performed under general anaesthesia in 42 of the children (72.4%), and biopsy was performed in 15 of them under local anaesthesia (26.3%). There was no sedation failure that required transition to general anaesthesia.
Technical
All procedures were performed under real-time sonographic guidance. Most biopsies were performed using 18G (n = 40, 70.1%) semi-automatic needles. The rest were performed using 16G semi-automatic needles (n = 17, 29.8%). Intercostal, subxiphoid or the shortest and safest orbit was chosen according to diffuse or focal liver disease.
Insufficient sample
An 11-year-old girl with a focal lesion underwent a repeat biopsy 5 days later due to inconclusive results due to tissue insufficiency. An 18G needle was used in the first biopsy procedure, and a 16G needle was used in the second application. All remaining patients had enough tissue to make a diagnosis by pathologists. The overall diagnostic yield of liver biopsy was 100% if sufficient sample was taken.
Results
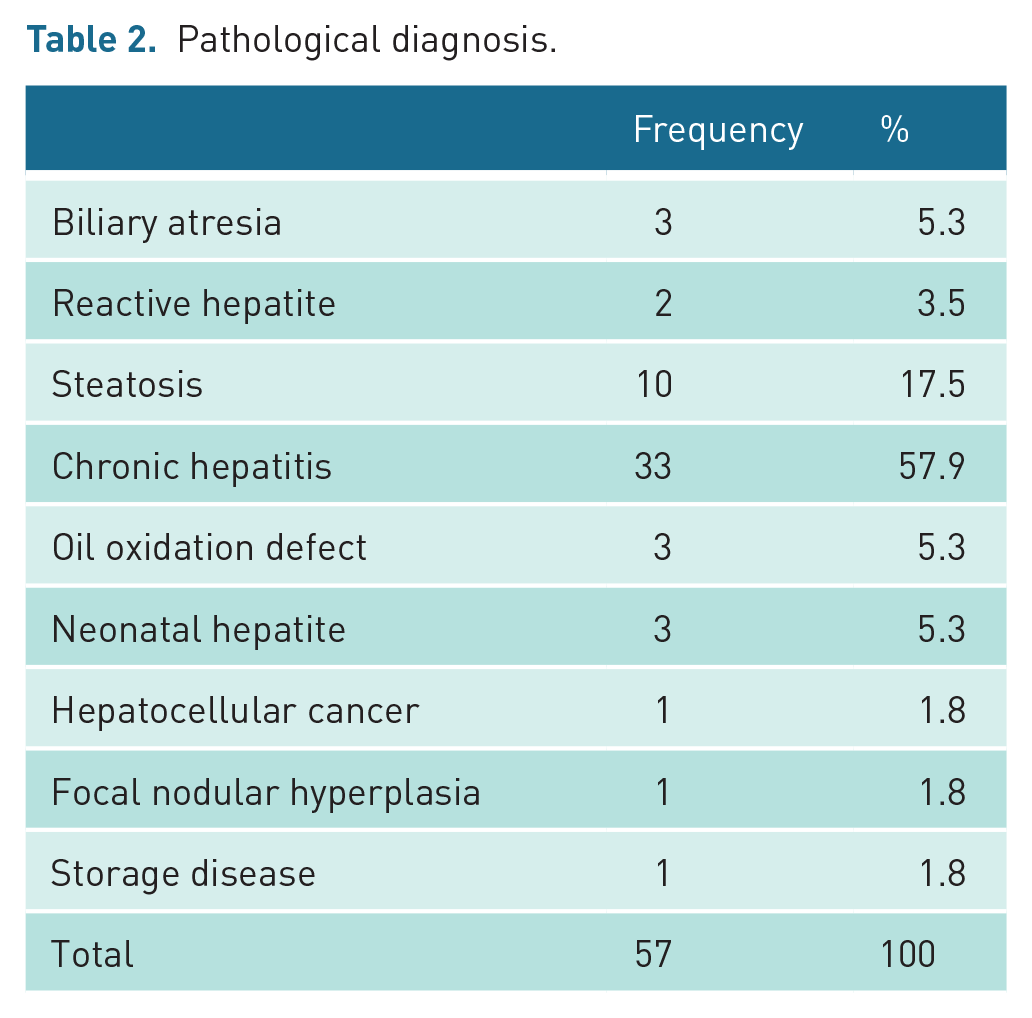
All biopsies were performed by a radiologist. The most frequent pathological diagnosis was chronic hepatitis (n = 33, 57.9%) and steatosis (n = 10, 17.5%) (Table 2). Adequate tissue was obtained in 98.2% of the biopsies. In one patient, a repeat biopsy was performed 5 days later, since sufficient tissue was not obtained. Major complication was seen in a 4-year-old patient (1.8%) who underwent biopsy. This patient presented with tachycardia and hypotension and hemodynamic deterioration at the 6 hours during the 24-hour routine follow-up. In this patient, there was a decrease of 2.1 g/dL in haemoglobin and a 6% decrease in haematocrit in control blood tests performed due to deterioration of hemodynamic. A large subcapsular haemorrhage area was recorded in the bedside USG performed on the patient (Figure 2). The patient was subjected to medical treatment in the hospital for 5 days until the values returned to normal. Complications such as pneumothorax, bowel perforation, bile leakage, sepsis and death reported in the literature were never observed. Minor complications were seen in 4 (7%) of 57 patients who underwent biopsy. Two of these patients had localised pain that improved with paracetamol after the procedure, and the other two had a fever exceeding 38.5°C, which was relieved by cold compression. The patients whose pain improved with paracetamol were 4 and 5 years old. The patients with fever were 2 and 7 years old. It was noted that the patients with pain symptoms were given medication around the fourth and fifth hours. In two patients, fever was detected at the controls around the fourth hour. A definitive pathological diagnosis (100%) was obtained in all 57 patients, but the initial biopsy efficiency was 98.2%. The results of the diagnostic biopsies are presented in Table 2. None of the children who were discharged after 24 hours of observation and met the discharge criteria had any complications that required readmission.
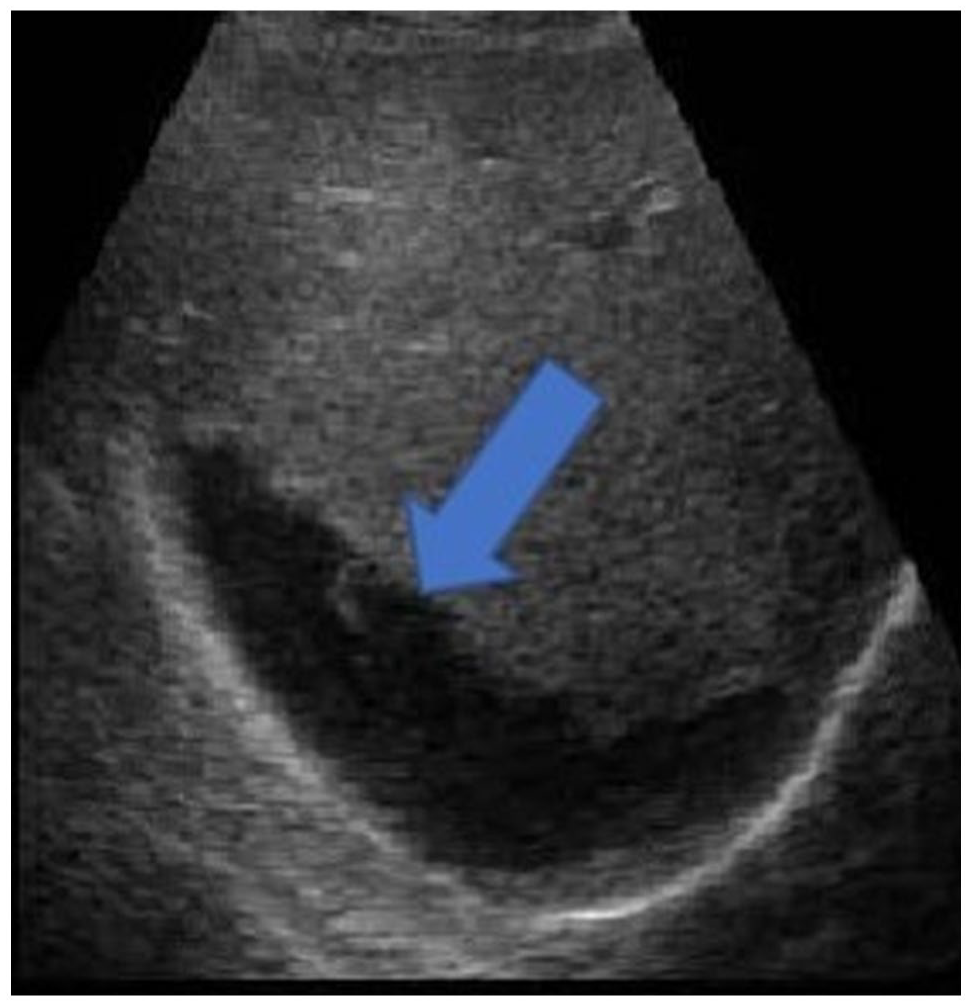
Post-procedure sonogram shows subcapsular haematoma (blue arrow).
Pathological diagnosis.
Discussion
Liver biopsy is indicated for diagnosing liver diseases in children, evaluating prognosis and monitoring treatment. 1 There are many ways to obtain a liver biopsy sample. Among these, sonography-guided or blinded percutaneous approach is commonly used and routine procedures. It has been reported that sonography-guided real-time percutaneous liver biopsy is more efficient, safer, more comfortable and less expensive than blind biopsy.2,3
Recently, an increasing number of percutaneous liver biopsies are now performed by interventional radiologists with imaging guidance. 3 Our retrospective study was performed by radiologists with interventional experience in a tertiary paediatric hospital to evaluate the safety and efficacy of percutaneous liver biopsy.
The most frequently reported major complication of percutaneous liver biopsy is symptomatic bleeding and minor complication is localised pain. Various reports in the literature have an incidence of major complications ranging from 1.4% to 6.6% and minor complications ranging from 0.8% to 11.7% in biopsies performed by a paediatric gastroenterologist.4 –10 Various reports in the literature have an incidence of major complications ranging from 0% to 4.6% and minor complications ranging from 0% to 25% in biopsies performed by an interventional radiologist.11 –16
In this study, the major complication rate was 1.8% and the minor complication rate was 7%. An overall complication rate of 8.8% was noted in this case series, which is consistent with the literature.11 –16 Therefore, our retrospective review shows that liver biopsy by an interventional radiologist is at least as safe as reported in available studies in the literature.
In current reports of biopsies performed by paediatric gastroenterologists, the occurrence of bleeding requiring blood transfusion ranges from 0% to 4.5%. The incidence of bleeding requiring transfusion in biopsies performed by interventional radiologists ranges from 0% to 4.6% and is similar to paediatric gastroenterologists. 13 In a previous report of 469 percutaneous liver biopsies performed by a paediatric gastroenterologist, 0.6% had deaths, 2.8% had bleeding requiring transfusion and some other major complications. 6 No patient died as a result of the procedure in our series. Similarly, no deaths were reported in other studies conducted by interventional radiologists in the literature.11 –16 In our study, there were no major complications other than symptomatic subcapsular bleeding that did not require blood transfusion. The European Society for Paediatric Gastroenterology Hepatology and Nutrition (ESPGHAN) recommends that the platelet count should be 60 × 109/L or higher and the INR should be 1.5 or less prior to percutaneous liver biopsies. 17 In our study, none of the patients, including the patient with symptomatic subcapsular bleeding complication, had abnormal platelet counts or INR values before the procedure. In our series, routine haemogram tests were obtained 4 hours after the procedure and before discharge for new possible bleeding complications. The mean post-procedure 4-hour haematocrit decrease was 2.3%, and the haematocrit decrease was less than 5% in all but one child. In our series, symptomatic subcapsular haemorrhage occurred in only one (1.8%) of the patients after biopsy, which caused a 2.1 g decrease in haemoglobin and a 6% decrease in haematocrit. This patient also had no known haematological disease, and there was no evidence of coagulopathy in pre-biopsy tests or a significantly increased risk of bleeding. In our study, pre-procedural INR and platelet counts did not predict bleeding, and these tests show that more studies are needed to predict the risk of bleeding after liver biopsy in children.
According to Amaral et al. 11 4.5% after percutaneous liver biopsies in patients under 1 year of age bleeding requiring transfusion has been reported. This is the series with the highest major complication rate reported by interventional radiologists. In some similar studies, it has been suggested that the risk for bleeding in paediatric patients increases as the age of the patient decreases.10,11,18,19 The only case with haematoma in our study was 4 years old, but due to our small sample size, we could not evaluate age as a risk factor for bleeding complications.
Previous studies have shown that the frequency of complications increases with the number of core samples obtained, and that one to two core samples is sufficient to make a diagnosis without an increased risk of bleeding.15,20,21 We took only one core sample in our series, except for the patient with insufficient sample. The biopsy of the patient who received insufficient sample was repeated 5 days later. Our histopathological analysis results confirmed that ultrasound-guided percutaneous biopsy resulted in sufficient tissue for pathological evaluation in most cases (98.2%). It is similar to other published reports in the literature ranging from 81% to 99.7%.11,14,16,18 Given this high success rate, it can be concluded that adequate biopsy material in biopsies can be obtained by interventional radiologists with virtual certainty.
A 16G needle is recommended for liver biopsies in adult patients, since it enables the evaluation of more portal tracts. 2 There is no standardisation in this regard in the paediatric group. In a series of 513 paediatric patients who did not have an increased risk of bleeding complications, an accuracy of 99.3% was reported with the use of a 16G needle. 16 In the series of 57 patients in our clinic, the diagnostic efficiency was 100% in biopsies using 16G needle (n = 17), and 97.5% in biopsies using 18G needle (n = 40). Although 18G needles were mostly used in biopsies performed in our clinic, the diagnostic efficiency was high (98.2%).
In a study conducted on paediatric oncology patients, it was reported that there is a 15% higher risk of complications in patients with localised lesions. 22 This reflected the difficulty of attempting to obtain adequate samples from a solitary liver lesion. In our patient with a focal lesion, the diagnosis could not be made due to tissue insufficiency in the biopsy performed with an 18G needle in the first application.
Apart from symptomatic bleeding in the literature, other rare major complications have been reported following percutaneous liver biopsy in children. Other reported complications were sedation-related complications (0.8%‒5.5%), arteriovenous fistula (0.7%), biliary leaks (0.6%‒1.5%), sepsis/infection (0.3%‒1.7%), pneumothorax (0.2%‒0.4%) and ascites leaks (0.2%).5,9,11,23 No other major complications were observed in our study.
The most frequently reported minor complications in the literature are asymptomatic subcapsular haematoma and localised pain.14,19 In our study, there was no patient with asymptomatic subcapsular haemorrhage. However, two of our patients had localised pain relieved by paracetamol and two patients had high fever. In the study conducted by Short et al., 18 it was suggested that there is a significant correlation between the increase in the incidence of minor bleeding in children younger than 3 years old and weighing less than 16 kg. In our study, there was no patient with minor bleeding.
There is no standardisation in terms of observation time after liver biopsy. In our study, an inpatient bed was reserved for outpatient liver biopsies during the entire study period and the patients were followed up for 24 hours. However, the need for this long follow-up period has been questioned in several studies. In some studies, it has been stated that the first 4–8 hours are critical in the development of complications and that patients can be discharged if no complications develop within this period.8,14,16,19,24 In our study, the first symptoms, consistent with the literature, appeared within 6 hours in patients who developed biopsy-related complications.
This study had several limitations. Due to the small number of patients in the study and the relatively heterogeneous mix of children of all ages, it was not possible to identify risk factors for a particular group. In addition, the retrospective nature of the study and the collection of patients in a single centre were other limitations.
In conclusion, sonography-guided percutaneous liver biopsy performed by interventional radiologists is safe because of the low rate of major complications in paediatric age, and it is an effective procedure due to its high diagnostic efficiency.
Footnotes
Contributors
All of the authors declare that they have all participated in the design, execution and analysis of the paper, and that they have approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethics approval
The study was carried out with the permission of the Erzincan Binali Yıldırım University Clinical Ethics Committee (Date: 2019, Decision No: 2019-11-4).
Guarantor
OK: Ozlem Kadirhan.
Concern for publication
The images of the patients are recorded in the computer system as it is necessary during patient follow-up. Since the study is retrospective, written permission was not obtained for publication, but existing images were de-identified and anonymized. We are aware that permission from a previous publisher for reproducing any previously published material will be required should our article be accepted for publication and that we will be responsible for obtaining that permission.
