Abstract
Following the 2018 patent expiration of abiraterone, a hormonal therapy for prostate cancer, the drug experienced a substantial influx of generic manufacturers and a marked decline in gross list prices. Using Medicare Part D reimbursement (gross pricing) data from 2019 to 2023, we analyzed trends in both pricing and market share post-generic entry. Among 19 manufacturers that entered the market, generic versions quickly dominated market share. However, despite the availability of significantly lower-cost options, Medicare Part D continued to reimburse for versions with gross prices 10–40 times higher than the cheapest available alternatives based on aggregate manufacturer-level averages. This case study illustrates how, even after generic entry, medication pricing can remain uncompetitive, suggesting inefficiencies in reimbursement dynamics. Policy tools such as reference pricing may offer a pathway to mitigate excessive spending and promote value-based purchasing in Medicare Part D.
Introduction
In October 2018, the U.S. District Court invalidated Johnson & Johnson’s contested use of ‘438’ patent, which had extended market exclusivity for abiraterone acetate (ABI) through its claimed combination with prednisone. The court deemed the combination “obvious,” thereby clearing the path for generic entry of a drug that had accounted for billions of dollars in cumulative spending by public and private payers. 1
Early evaluations of abiraterone’s generic entry revealed surprising findings. In a 2022 JAMA Network Open study, Gupta et al. reported that even after the arrival of multiple generics, patient out-of-pocket costs remained high; and in some cases, select generics were priced higher than the branded version. 2 Additional work by Cortese et al. estimated that Medicare could save hundreds of millions of dollars by sourcing abiraterone from the Mark Cuban Cost Plus Drug Company, which listed the medication at substantially lower prices than those currently reimbursed under Medicare Part D. 3
These findings raise critical questions about how generic competition translates or fails to translate into price savings for the Medicare program and for patients. In this analysis, we use Medicare Part D spending data to examine abiraterone pricing and market share trends from 2019 through 2023, highlighting persistent inefficiencies in the generic drug market.
Methods
We analyzed publicly available data from the Medicare Part D Spending by Drug file from 2019 to 2023 to examine trends in pricing based on Medicare reimbursement and market share of abiraterone stratified by generic versus brand name and by manufacturer following generic market entry. 4 Our analysis focused on conventional formulations of abiraterone acetate, excluding the submicronized formulation (Yonsa) and combination products such as niraparib/abiraterone, in order to maintain consistency in clinical indication.
We extracted annual data on total reimbursement, pricing, and number of units for generic versus brand name versions, and for each manufacturer. Market share was determined based on the total Medicare reimbursement per calendar year. Because the Medicare Part D Spending by Drug dataset aggregates claims across National Drug Codes (NDCs), strengths, and dosage forms, all pricing estimates represent claim-weighted averages rather than reflecting NDC-specific prices. All spending and pricing measures reflect gross drug costs and do not account for manufacturer rebates.
All analyses were descriptive and conducted using Microsoft Excel. This study used de-identified, publicly available data and was therefore exempt from institutional review board approval.
Results
By 2023, 19 generic manufacturers had entered the abiraterone market, with generics successfully capturing the majority of market share. From 2019 to 2023, the generic share of total Medicare Part D reimbursement for abiraterone rose from approximately 27% to 96% (Figure 1(a)). Over the same period, the weighted average price per dosage unit declined from about $85 in 2019 to $28 in 2023, reflecting a strong association between increased generic competition and declining average prices (Figure 1(b)). In contrast, the brand-name product, Zytiga, maintained a stable price of approximately $130 per dosage unit throughout the study period; its declining contribution to overall spending was driven largely by reduced prescribing volume. Generic and brand name (Zytiga) abiraterone pricing and market share. (a) Total reimbursement change (2019–2023) for generic vs brand-name abiraterone from Medicare Part D spending by drug. (b) Price change (2019–2023) for generic abiraterone, brand-name abiraterone, and weighted average price change. Weighted average price was calculated by dividing (generic reimbursement + Zytiga reimbursement) by (generic units sold + Zytiga units sold). Data were sourced from centers for Medicare and Medicaid Services (CMS) from the Medicare Part D spending by drug file for 2023.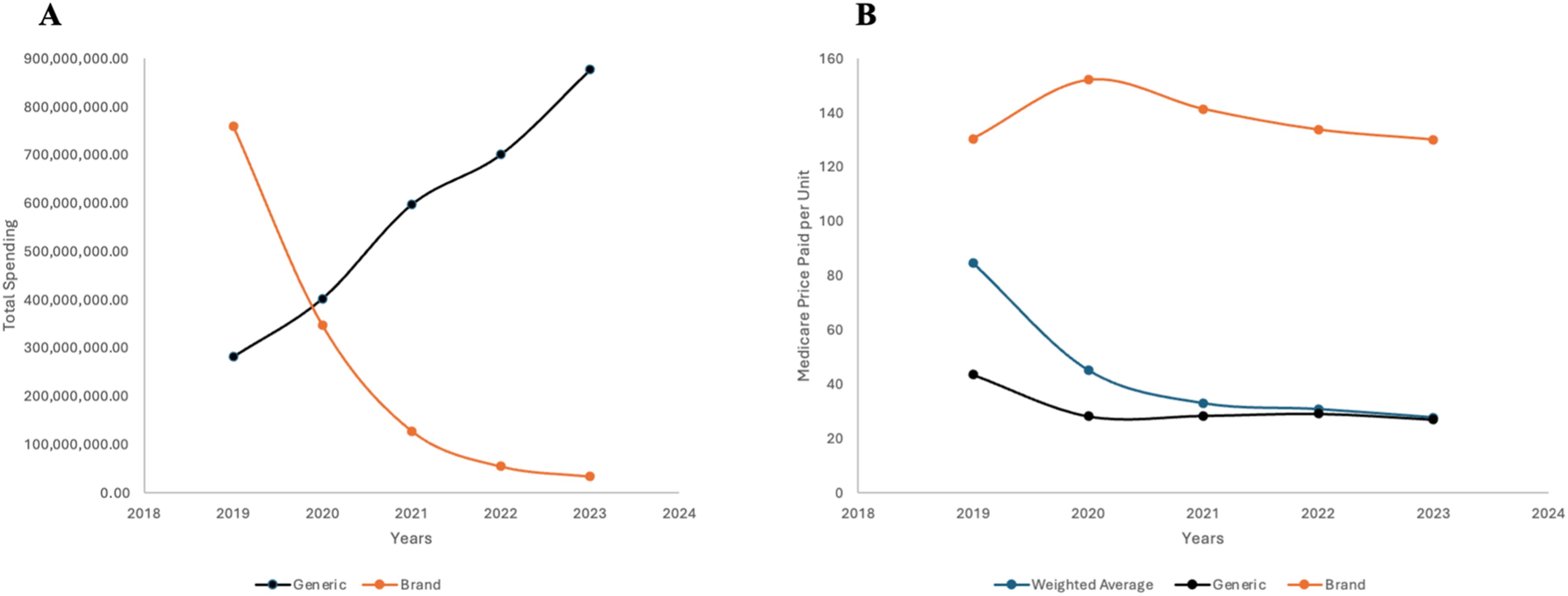
Despite these favorable aggregate trends, a closer examination reveals that the benefits of generic competition have not been equitably distributed. While the market includes generic versions priced as low as $2 per dosage unit, the majority of Medicare claims were reimbursed at significantly higher rates, often 10–40 times more based on aggregate manufacturer-level averages. In 2023, four low-cost manufacturers (Westward/Hikma, Major Pharmaceuticals, CivicaScript, and AHP) offered abiraterone for under $5 per dosage unit but collectively accounted for only 0.5% of total Medicare spending and roughly 10% of all units dispensed (Figure 2). Abiraterone manufacturer’s reimbursement and number of units purchased in 2023. Figure displays percent of dosage units sold by each manufacturer and percent of total reimbursement paid by Medicare to each manufacturer. Data were sourced from centers for Medicare and Medicaid Services (CMS) from the Medicare Part D spending by drug file for 2023.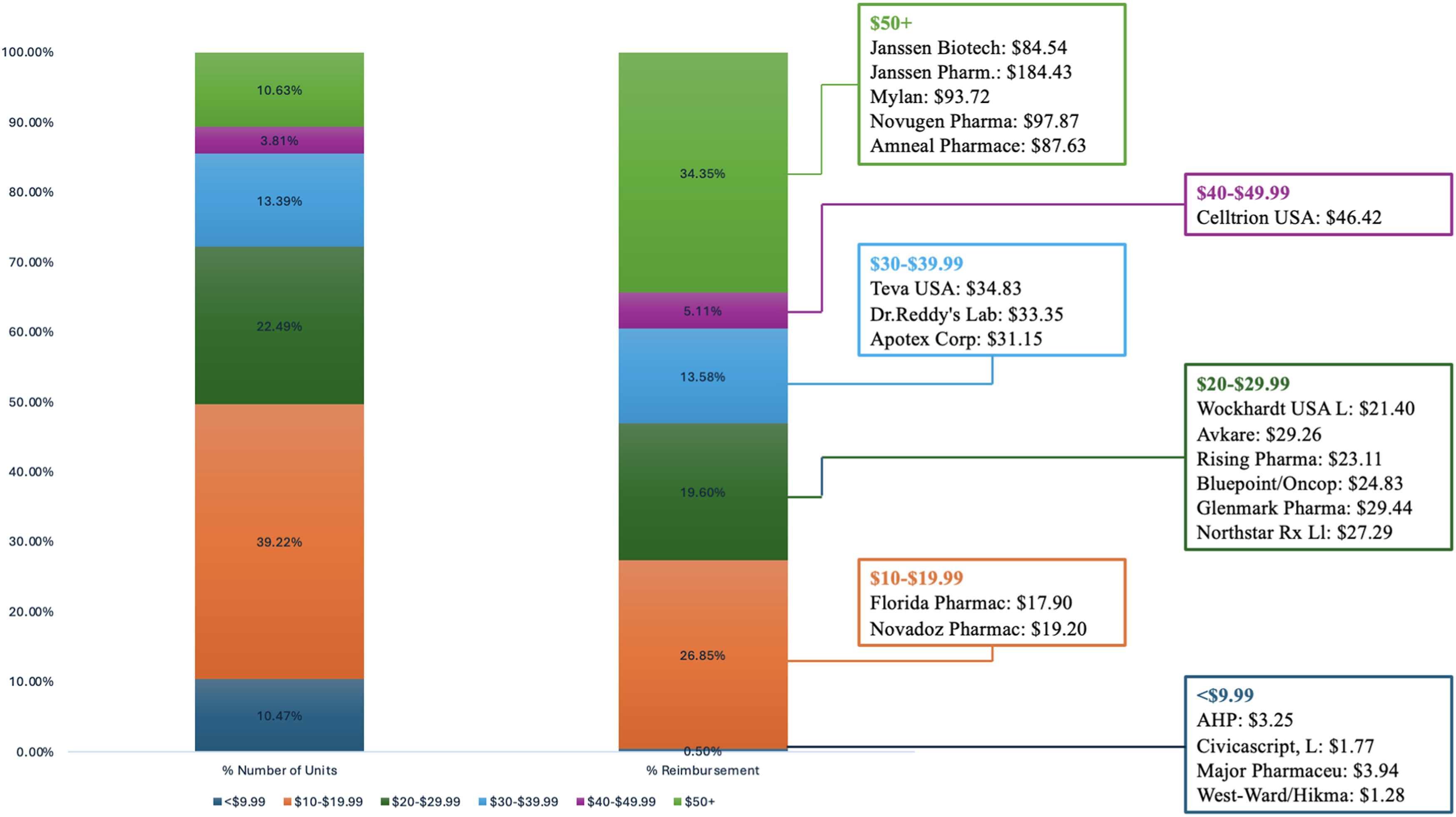
Across all manufacturers, gross Medicare reimbursement for abiraterone in 2023 ranged from approximately $2 per dosage unit for low-cost generics to $98 per dosage unit for high-cost generics, with the brand-name product reimbursed at $184 per dosage unit. Specific manufacturer-level data are provided in Supplemental Table 1.
Discussion
Despite the presence of 19 generic manufacturers and sharply declining average prices, our findings reveal that Medicare continues to reimburse abiraterone at gross prices significantly above the lowest-cost options. In 2023, the weighted average gross price remained elevated at $28 per dosage unit, largely because the lowest-cost generics, priced as low as $2, captured only a fraction of total market share. As a result, Medicare continues to incur higher gross costs than would be expected if lower-cost generics were used more widely.
This inefficiency reflects a broader structural issue: while market competition creates the potential for lower prices, current U.S. reimbursement and purchasing practices offer little incentive to steer prescriptions toward the most affordable generic alternatives. In contrast, countries such as the United Kingdom have achieved far lower prices for abiraterone at approximately $1.90 per tablet through centralized negotiation and procurement strategies. 5
If Medicare had theoretically reimbursed abiraterone at the lowest generic price available in 2023, potential savings could be substantial; however, these estimates do not account for confidential rebates. The persistence of high reimbursement levels despite broad generic entry may reflect opaque intermediary incentives rather than acquisition cost differences due to the influence of Pharmacy Benefit Managers (PBMs) and formulary design. For context, the National Average Drug Acquisition Cost (NADAC) for abiraterone acetate 250 mg in October 2025 was only $1.08 per tablet, providing a rough benchmark for acquisition costs that is far below typical Medicare reimbursement levels. 6
The persistence of high reimbursement levels may be influenced by factors such as PBM contracting and rebate agreements described in prior Federal Trade Commission (FTC) analyses. A 2024 FTC interim report on Abiraterone and Imatinib documented that pharmacies affiliated with the three largest PBMs in the US (CVS Caremark, Express Scripts, and OptumRx) retained $685 million for abiraterone acetate and charged 20–40 times NADAC. 7 Prior investigations have indicated that higher list prices may translate into larger rebate amounts for PBMs, allowing them to retain greater revenue.
Part D plans also commonly use tiered formularies intended to steer towards preferred drugs, yet preferred status does not necessarily correlate with the lowest-cost drug. Clinicians often are not aware of these pricing differences and electronic prescribing defaults can heavily influence purchasing decisions. 8 Improving NDC-level pricing visibility at the point of prescribing can help clinicians support value-based prescribing practices.
This systematic misalignment of incentives may translate to higher out-of-pocket spending for beneficiaries, where the most burden falls on low-income beneficiaries. Part D plans frequently apply a co-insurance of 25% until beneficiaries reach their out-of-pocket maximum. 9
While these findings highlight important economic inefficiencies at the manufacturer-level, this analysis has several limitations. Our analysis uses publicly available Part D reimbursement and manufacturer-level data but does not observe confidential rebate agreements or plan-specific formulary variation, making it difficult to elucidate the mechanism of high-priced generics. Additionally, the dataset aggregates claims across all plans and does not reflect differences in formulary placement and cost-sharing structures that may influence which NDCs are dispensed. Because the analysis is descriptive, causal inferences about PBM behavior and formulary design cannot be reliably made.
These findings highlight a persistent disconnect between the theoretical benefits of generic entry and real-world outcomes for Medicare. Policy reforms such as reference pricing, which pegs reimbursement to a benchmark price across therapeutically equivalent options, may help align incentives and promote the use of cost-effective generics. Additionally, formulary redesign to automatically place the lowest-cost NDC generics on preferred tiers could align plan incentives with affordability goals. Further, greater transparency in manufacturer-level pricing and PBM rebate agreements would further enable payers and policymakers to identify misaligned economic incentives through future research.
More broadly, targeted interventions are needed to ensure that competition leads not only to availability, but also to affordability at scale.
Conclusion
The case of abiraterone illustrates how generic entry alone does not guarantee lower gross spending in Medicare. Although numerous low-cost generic alternatives are available, structural barriers within the U.S. drug pricing and reimbursement system may limit their adoption. Our analysis shows that Medicare could theoretically save taxpayer dollars if reimbursement were aligned with the lowest available generic prices. To fully realize the cost-saving potential of generic competition, federal policymakers should consider mechanisms such as reference pricing, formulary redesign, and transparency requirements that encourage uptake of the most affordable therapeutic options. Without such reforms, the promise of affordable access to essential medications will remain only partially fulfilled.
Supplemental material
Supplemental material - Competition isn’t enough: Medicare still overpays for abiraterone
Supplemental material for Competition isn’t enough: Medicare still overpays for abiraterone by Parthay H Rokad and Chan Shen in Journal of Generic Medicines
Footnotes
Acknowledgements
Language editing assistance was provided using OpenAI’s ChatGPT to assist with grammar and sentence structure. All researchers have reviewed the manuscript after the use of this tool and take full responsibility over the content of the work.
Ethical considerations
There are no human participants in this article and informed consent is not required.
Author contributions
PHR collected and analyzed the raw data. PHR and CS edited and prepared the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
A spreadsheet listing the raw data on drug costs collected from the Centers for Medicare and Medicaid Services (CMS) will be available upon reasonable request from the corresponding author.
Supplemental material
Supplemental material is available online.
