Abstract
Background
The rising costs of pharmaceuticals globally have increased the demand for generic medications as people seek more affordable alternatives to costly branded drugs. Generic medications are a beneficial approach to reducing pharmaceutical expenses without compromising healthcare quality. Yet, the prescribing of generic medications is still nil to date due to various factors not explored well. Therefore, we aimed to assess the knowledge, attitudes, perceptions, and challenges physicians and pharmacists face in Pakistan regarding implementing generic medicines.
Methods
A cross-sectional observational survey was conducted among conveniently available physicians and pharmacists from February to July 2024. Data was collected using a structured validated questionnaire. Participants were invited from randomly selected public and private sector hospitals and pharmacies in Pakistan’s twin cities (Rawalpindi and Islamabad). Data from this study was analyzed using descriptive statistics, Mann–Whitney and Kruskal–Wallis tests, and multivariate binary logistic regression in SPSS version 25.
Results
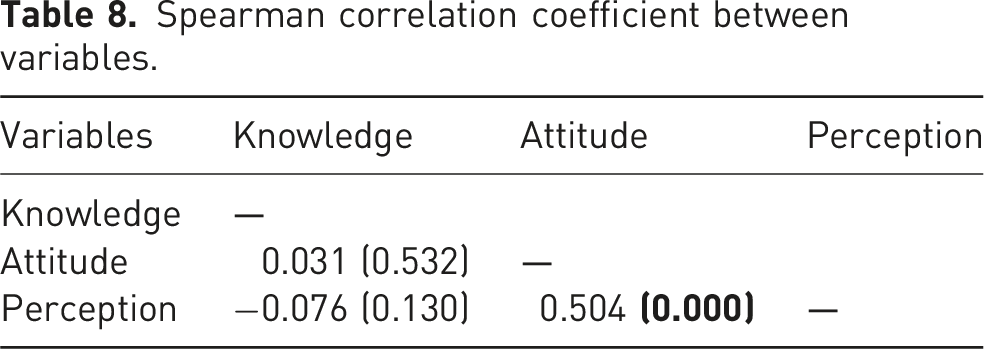
Out of 515 distributed surveys, 415 individuals responded, and 400 surveys were analysed, resulting in an 80.5% response rate. Of the 400 participants, 214 (53.5%) participants were pharmacists, and 186 (46.5%) were physicians. More than half of the participants demonstrated high knowledge, 215 (53.8%), and the majority of them had positive attitudes, 323 (80.7%), as well as perceptions, 249 (62.3%), toward generic medicines. A significant positive correlation existed between participants’ attitudes and perceptions (r = 0.504, p < .05). In multivariate binary logistic regression analysis, age, practice setting, and negative attitude had statistically significant associations with positive attitude.
Conclusion
Health care professionals possess considerable knowledge and exhibit a positive attitude towards generic medications, demonstrating strong support for generic substitution. Educational interventions are necessary to enhance generic drug prescribing and dispensing practices among healthcare professionals.
Keywords
Introduction
A generic medicine (GM) is described as a pharmaceutical product often meant to be interchangeable with the branded medication. It can only be produced after the expiration of the patent protection for the branded medication. Generic pharmaceuticals are regarded as therapeutically equal to their branded counterparts. 1 Generic medications are accessible once the patent protection for branded pharmaceuticals ends. Upon the expiration of patent protection, generic pharmaceuticals may be produced by entities other than the original innovator business. 2 Innovator products are reference items whose efficacy, safety, and quality have been established after rigorous clinical trials and are sold under patent cover by their innovators. 3 Utilizing GM to replace branded medicines (BM) constitutes a cost-effective approach, as BM is significantly more expensive than GM for treating the same clinical problems. 4 GM is sold at a far lower cost. They are generally 20 to 90% less expensive than their brand counterparts.3,5 Most patients have had out-of-pocket expenses for both GM and BM medicine purchases.6–8
The rise in healthcare expenditure may pose a global issue, with pharmaceutical costs constituting a substantial portion of this spending. Consequently, generic alternatives have widespread appeal globally, particularly in developing nations where poverty significantly limits purchasing power.9,10 Governments in numerous nations have implemented continuous cost-containment measures to ensure equitable access to healthcare. However, the practice of substituting GM has been controversial among healthcare professionals, mainly due to quality, safety, and efficacy issues. 11 GM is lower in quality, efficacy, and safety than branded ones.11,12 To advance GM substitution, it is essential to rectify misconceptions regarding GM quality, efficacy, and safety.2,13 Inaccurate or inadequate understanding and perception of generics restrict a more thorough generic medication. 14 The knowledge and attitudes of healthcare professionals are crucial in eliminating the rejection of generic medication utilization and promoting its implementation. The heightened prescription of lower-cost generic medications may decrease healthcare expenses and reduce out-of-pocket patient costs.
However, evidence around Pakistani physicians’ and pharmacists’ knowledge, attitudes, and perspectives of generic drugs is insufficient. Moreover, no adequate generic medication or brand substitute prescribing strategies have been developed in Pakistan. Numerous studies have been undertaken in many nations globally, examining the expertise of physicians and pharmacists concerning generic medications.3,15,16,17,18,19,20 Over time, the confidence and understanding of patients and prescribers about generic medications have boosted, 21 especially in developed nations; despite all, further efforts are required in low- and middle-income countries (LMICs) including Pakistan. 21 The unacceptability of generics may lead to non-concordance, non-affordability resulting in treatment failure. 22 Therefore, it is essential to investigate changing physicians’ and pharmacists’ knowledge, attitudes, and perspectives regarding generic substitution. This study will assist policymakers in establishing effective medication substitution policy regulations to optimize patient benefits, address potential disinformation and misconceptions regarding generic medicines, and improve the prescribing of generics.
Methodology
Study design and study setting
This research followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist to report the study 23 attached in Supplemental file. A cross-sectional observational study was conducted using a paper-based, self-administered questionnaire between February 2024 and July 2024. Data were collected from physicians and pharmacists working in public and private hospitals and community pharmacies across Rawalpindi and Islamabad, Pakistan (twin cities).
Study population
The study population comprised registered physicians and pharmacists. Inclusion criteria included all registered physicians and pharmacists working full-time or part-time, having at least 12 months of professional practice experience, and providing informed written consent. Exclusion criteria included intern physicians, pharmacists, and individuals who did not provide informed consent.
Sampling techniques and sample size
A convenience sampling technique was employed to collect the data from 400 participants. The sample size was calculated using the Rao Soft calculator 24 based on a 5% margin of error, a 95% confidence level, and an expected response rate of 50%, which indicated a minimum required sample size of 377 participants.
Data collection tool
A structured questionnaire was developed after extensive reviews of relevant literature on generic medicines.3,12,16,20,25,26 Face and content validity of the initial questionnaire items was done by a panel of six members, experts in clinical pharmacy, pharmacy practice, and physicians, to ensure the questionnaire’s relevance, specificity, and comprehensiveness, and final adjustments made based on their feedback. Subsequently, a pilot study was conducted on 20 physicians and pharmacists to validate the questionnaire and review structure, clarity, and comprehension. The overall Cronbach’s alpha value of the questionnaire was 0.895, with a subscale of 0.592 for knowledge, 0.890 for attitude, and 0.864 for perception. The participants involved in pilot testing were excluded from the final analysis.
The questionnaire was divided into five sections. The first section collected participants’ demographic characteristics; the second assessed participants’ knowledge about generic medicines; the third evaluated attitudes toward generic medicines; the fourth measured participants’ perceptions of generic medicines; and the fifth identified the challenges/barriers to the prescribing and dispensing of generic medicines.
The knowledge section was assessed using 10-item statements, with respondents indicating their responses as ‘yes,’ ‘no,’ or ‘don’t know’ answers. ‘Yes’ answers scored 1 point, while ‘No’ and ‘Don’t know’ responses were given 0. The total score ranges from 0 to 10; a higher score means high knowledge. The scores were divided into two groups based on the midpoint of the total score: low knowledge (scores of 0–5) and high knowledge (scores of 6–10). The attitude and perception sections were evaluated using 10 items each. A 5-point Likert scale ranging from ‘strongly agree’ to ‘strongly disagree’ was employed to assess participants’ attitudes and perceptions. The total score for both sections range from 1 to 50; a higher score means a positive attitude and perception. The scores were divided into two groups based on the midpoint of the total score: negative attitudes and perceptions (scores of 1–25) and positive attitudes and perceptions (scores of 26–50). The fifth section examined the challenges and barriers faced by physicians and pharmacists in Pakistan when implementing generic medicines.
Data collection procedure
We respectfully approached physicians and pharmacists during their breaks and afternoons, when patient numbers were typically low. The researcher provided complete information to the study participants about the study’s objective, voluntary participation, data anonymity, and the estimated time required to complete the survey. The participants signed the informed written consent before filling out the questionnaire, which typically takes 15 to 20 min to complete.
Statistical analysis
The data obtained, coded, entered, and analyzed for this study was conducted using the Statistical Package for the Social Sciences (SPSS) version 25. Descriptive statistics, including the median with interquartile range, were used for continuous variables, while frequency and percentage were employed for categorical variables. The association between groups was assessed using Mann-Whitney and Kruskal-Wallis tests and multivariate binary logistic regression. The study further analysed the participants’ knowledge of generic medicines based on their sociodemographic characteristics. Due to the non-normality of the data (i.e., the knowledge score), the Mann–Whitney U and Kruskal–Wallis non-parametric tests were used. Spearman correlation coefficient was used to identify the relationships between study variables. P values <0.05 were considered statistically significant.
Ethical considerations
The study adhered to the principles outlined in the Declaration of Helsinki 27 and received approval from the Research Ethics Committee of the Riphah Institute of Pharmaceutical Sciences, Riphah International University, Islamabad, Pakistan (Ref. No. REC/RIPS/2023/30). All participants were provided informed consent before participating and explaining the study’s objectives and procedures. Participants signed the consent form to confirm their voluntary participation before enrolling in the study.
Results
Sociodemographic characteristics of the participants.

Knowledge of the participants about generic medicine
Knowledge of the participants about generic medicine.

Note: T: True, F: False.
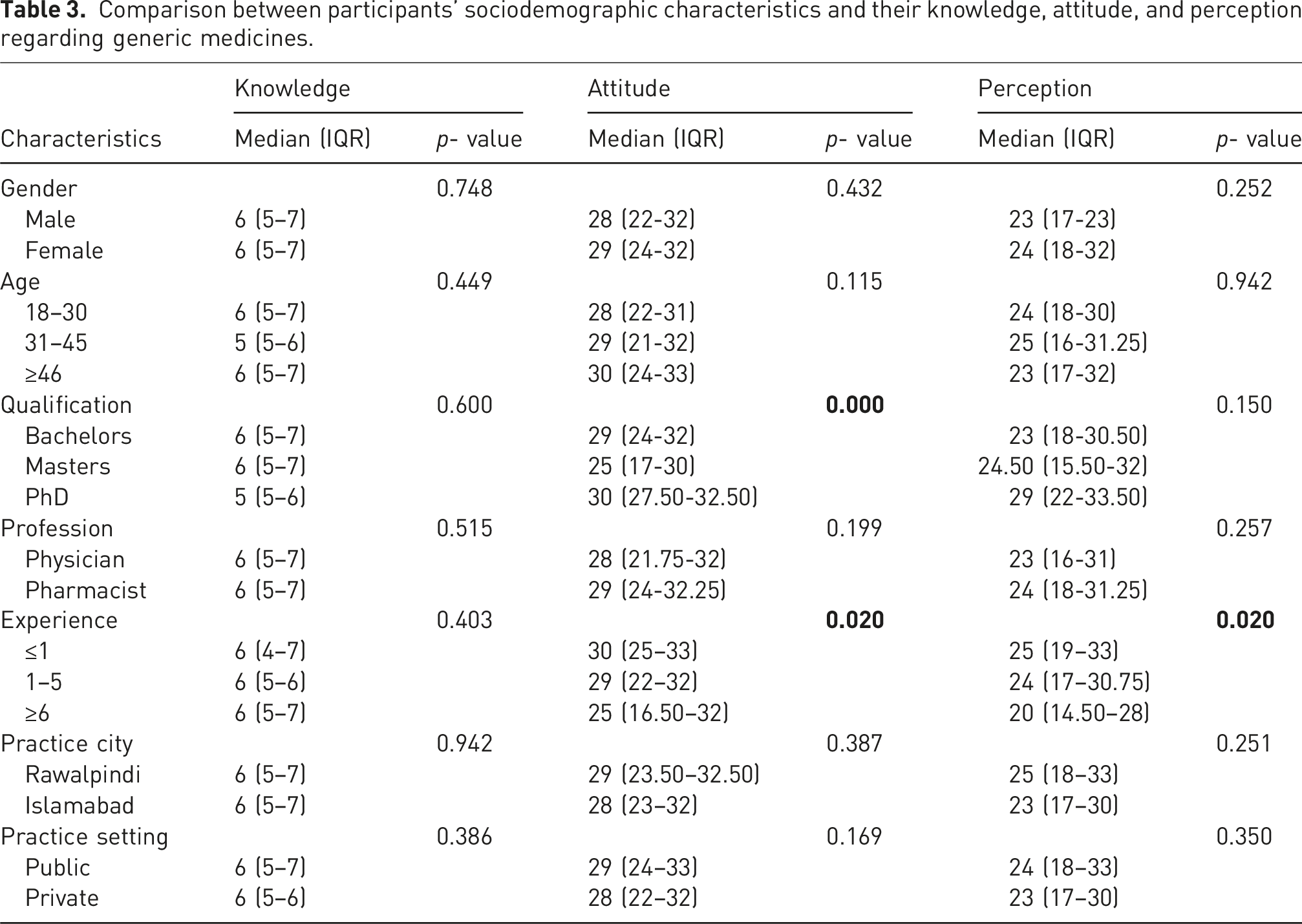
Comparison between participants’ sociodemographic characteristics and their knowledge, attitude, and perception regarding generic medicines.
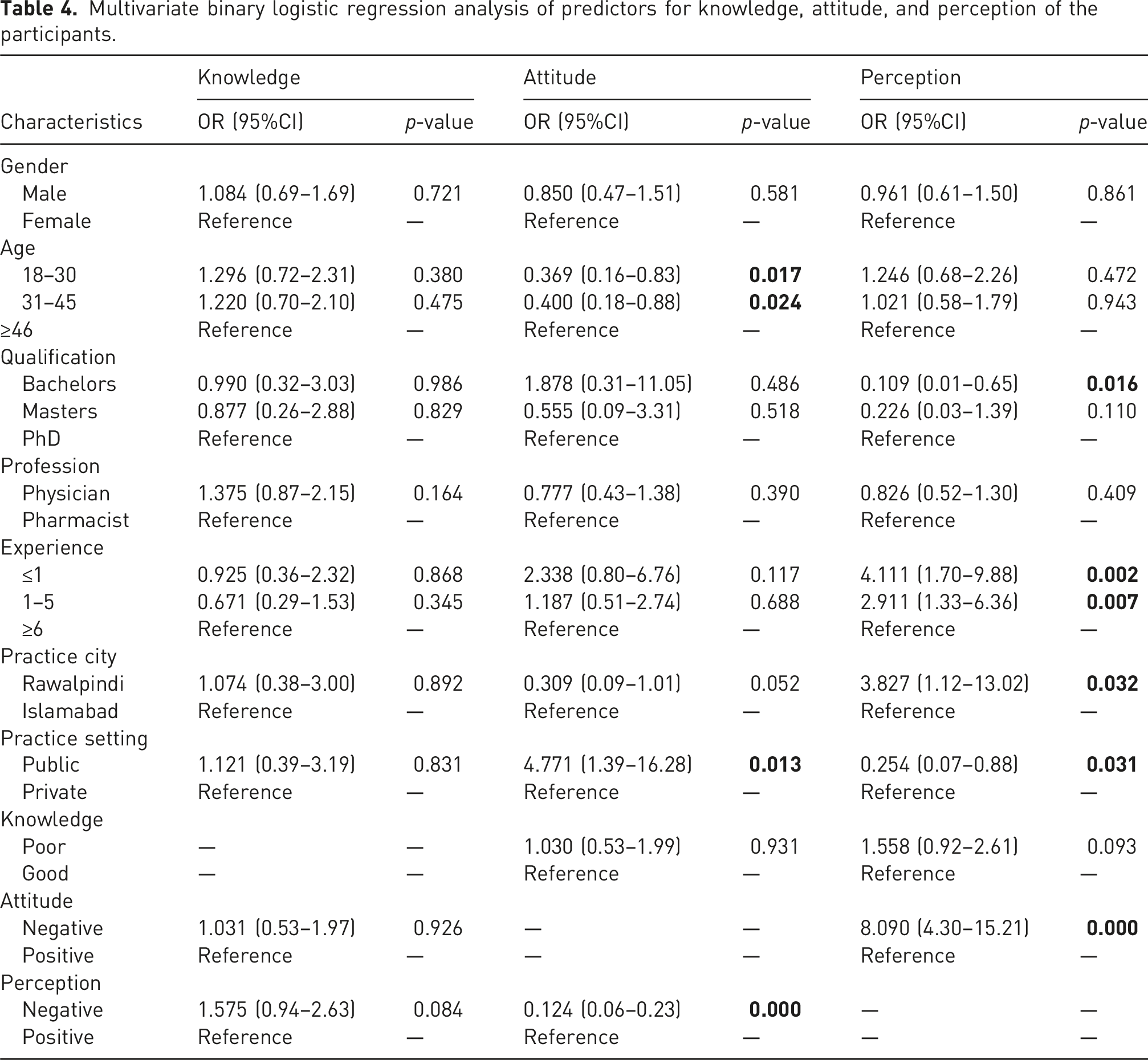
Multivariate binary logistic regression analysis of predictors for knowledge, attitude, and perception of the participants.
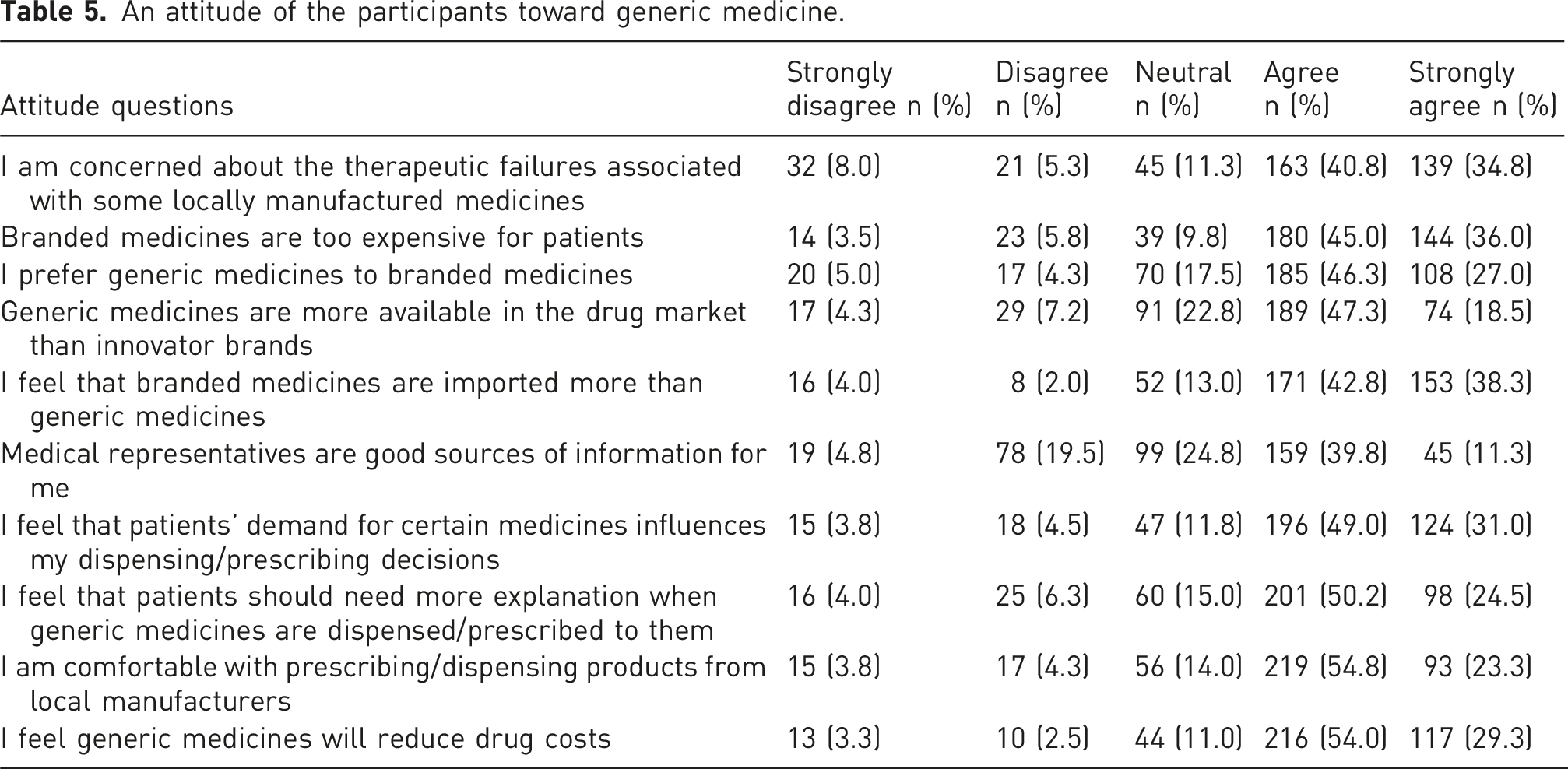
The attitude of the participants toward generic medicine
An attitude of the participants toward generic medicine.
The study analysed the participants’ attitudes toward generic medicines based on their sociodemographic characteristics. Those participants with PhD qualifications had significantly higher attitude scores than those with bachelor’s and master’s degrees [median (IQR): 30 (27.5–32.5) versus 29 (24–32) and 25 (17–30)]. Similarly, participants with <1 year of practice experience had significantly higher attitude scores than those with 1–5 years and more than 6 years of practice experience [median (IQR): 30 (25–33) versus 29 (22–32) and 25 (16.50–32)] (Table 3).
In multivariate binary logistic regression analysis, age had statistically significant associations with attitude; those aged 18–30 (OR = 0.369, 95% CI: 0.16–0.83, p = .017) and 31–45 (OR = 0.400, 95% CI: 0.18–0.88, p = .024) were less likely to have a positive attitude compared to those aged 46 and above. Similarly, the practice setting of participants had statistically significant associations with attitude, with public sector participants being more likely to have a positive attitude (OR = 4.771, 95% CI: 1.39–16.28, p = .013) compared to those in the private sector. Additionally, attitude had significantly significant associations with perception, as those with a negative attitude were substantially more likely to have a negative perception (OR = 8.090, 95% CI: 4.30–15.21, p < .001) (Table 4).
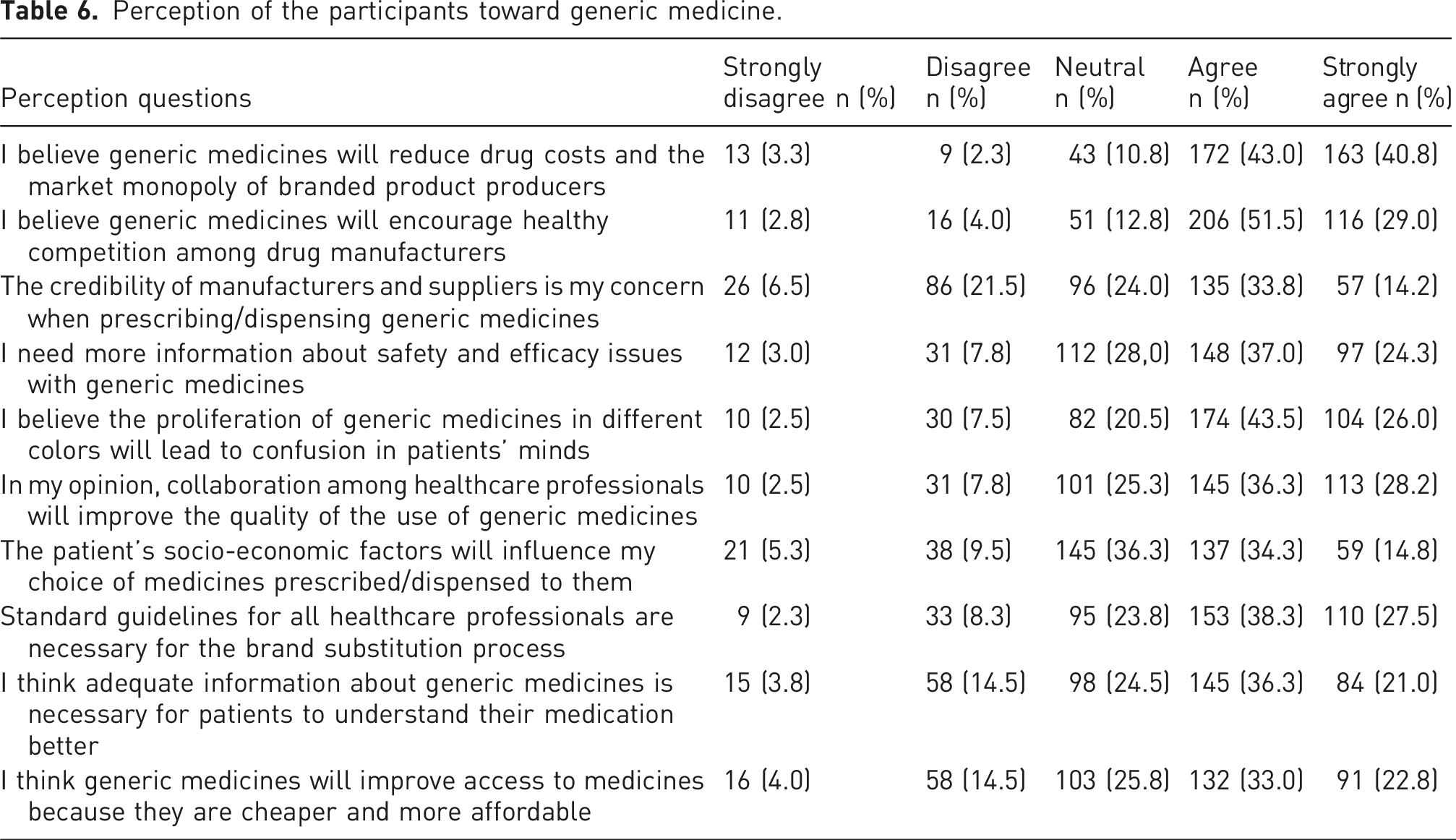
Perception of the participants toward generic medicine
Perception of the participants toward generic medicine.
The study analysed the participants’ perception of generic medicines based on their sociodemographic characteristics. Those participants with <1 year of practice experience had significantly higher perception scores than those with 1–5 years and more than 6 years of practice experience [median (IQR): 25 (19–33) versus 24 (17–30.75) and 20 (14.50–28)]. No other participants’ sociodemographic characteristics had a statistically significant effect on the perception score (Table 3). In multivariate binary logistic regression analysis, education had statistically significant associations with perception, with those holding a bachelor’s degree less likely to have a positive perception (OR = 0.109, 95% CI: 0.01–0.65, p = .016) compared to PhD holders. Moreover, experience also had statistically significant associations with perception, with participants having 1–5 years (OR = 2.911, 95% CI: 1.33–6.36, p = .007) and ≤1 year (OR = 4.111, 95% CI: 1.70–9.88, p = .002) of experience being more likely to have a negative perception compared to those with ≥6 years. Additionally, the practice city also had statistically significant associations with perception, with those practicing in Rawalpindi more likely to have a positive perception (OR = 3.827, 95% CI: 1.12–13.02, p = .032) than those practicing in Islamabad (Table 4).
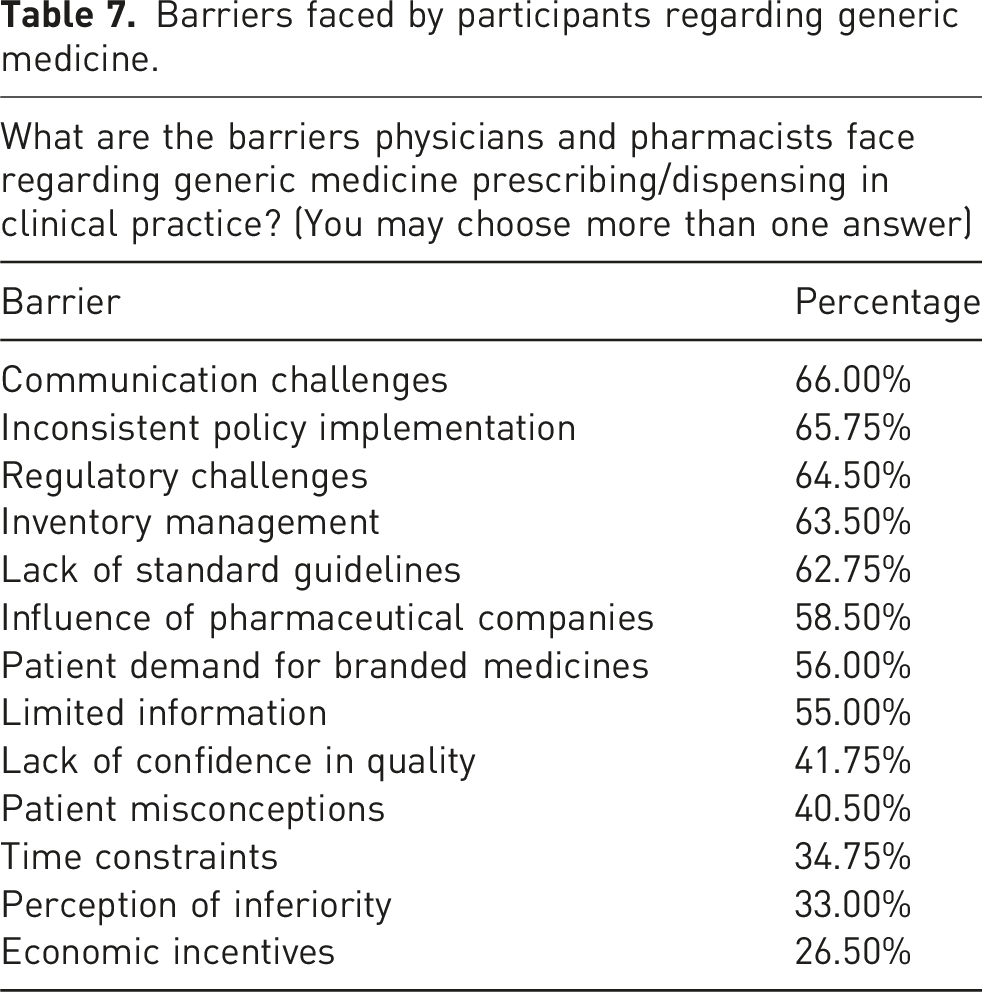
Barriers faced by participants regarding generic medicine
Barriers faced by participants regarding generic medicine.
Correlation coefficient between variables
Spearman correlation coefficient between variables.
Discussion
This study highlighted favorable results concerning the knowledge and acceptance of generic medications among healthcare professionals in Pakistan. Specifically, the majority of participants demonstrated a satisfactory level of knowledge in recognizing the therapeutic equivalence of generic and branded medications, as well as in understanding that generics are not contributing to increased side effects. Additionally, participants exhibited an overall favorable attitude, recognizing the capacity of generic medications to lower healthcare expenses. In terms of perception, a significant number of respondents supported the role of generics in promoting market competition and reducing the control of branded drug brands. Overall, the results indicate that medical professionals have a positive attitude regarding the increased utilization and acknowledgment of generic medications in healthcare settings.
Furthermore, a significant positive correlation was observed between participants’ attitudes and perceptions toward generic medicine. Despite variations in professionals’ knowledge, attitudes, and practices regarding generic drug replacement among countries due to cultural and policy differences, a strong understanding and positive attitude toward generic drugs could enhance the practice of generic prescribing. 28
This finding is higher than those of a comparable study conducted in New Zealand (70%), 29 and Qatar (38%), 16 yet aligns closely with results from studies in Ethiopia (77.1%),14,25 and Palestine (76.5%). 30 These comparable outcomes in the mentioned countries may be attributed to better educational qualifications and valuable work experience. The current study indicated that most participants (85.5%) reported that generic and brand medications contained equivalent quantities of active components. This result aligns with research conducted in Ethiopia (89%).14,25 Additionally, 73.5% of participants claimed that generic medications should be in the same dose form as their brand-name counterparts. This corresponds with research conducted in Ethiopia (84.4%).14,25
Participants exhibited a positive attitude towards generic drugs in the study area. This positive attitude could possibly be attributed to the cost-effectiveness of the generic medications. The majority of participants in this study expressed concerns regarding therapeutic failures related to some locally produced medicines. Consequently, the quality of both locally manufactured and imported generic medications must be rigorously inspected by regulatory authorities to guarantee compliance with established quality standards. This could improve the confidence of healthcare practitioners regarding the generic replacements available in Pakistan. 31 The majority of participants in this study recognized that branded medications were excessively expensive for patients. This finding is comparable to the research conducted in India. 32
In this study, most respondents preferred generic drugs to branded ones. The interaction between pharmaceutical marketing presenters, physicians, and pharmacists has generated significant global discourse. 20 In our study, more than half of respondents believed medical representatives were trustworthy source of information regarding generic medicines. This is similar to the study by Joseph et al. among physicians conducted in Nigeria. 20 The majority of the study participants believed that patients should be given additional information when generic medicines are prescribed/dispensed to them. Physicians have a crucial role in prescribing generic medications. Pharmacists are at the forefront of delivering medications based on medical prescriptions and are the final healthcare experts determining medication selection. Therefore, establishing communication between physicians and pharmacists is essential in improving generic substitution. 19
Sixty-nine percent of respondents believed that GM packaged in various colors could lead to confusion among patients. This finding is consistent with the research conducted in Nigeria. 33 This may be why more than half of the participants agreed with this study: it is necessary to educate patients on the medicines being administered to them or give them adequate information about generic medicines to better understand their medication. Replacing branded medications with their generic counterparts is regarded as a way to lower healthcare costs for patients. 3 More than half of the participants in the present study concurred that generic medications enhance treatment access due to their widespread availability and reduced cost. This ratio is rather low when compared to findings from similar studies conducted in Nigeria, Qatar, Poland, and Saudi Arabia, where health professionals expressed more favorable views regarding the role of generics in improving access.16,20,33,34,35 However, the overall use of generics has greatly increased over the past two decades due to their reduced cost, offering an economical alternative to branded medications. Consequently, promoting the use of generic medications in developing countries may lead to considerable reductions in healthcare costs.36,37
The implication of findings and future research
A clear and standardized generic substitution approach must be implemented immediately in clinical settings. Professional groups need to guide health authorities on a standardized approach for the utilization of generic medications. Overall, numerous laws and regulations concerning prescribing, dispensing, patients/consumers, and healthcare organizations should be considered to promote effective generic substitution. These findings possess significant implications for the continued development of generic replacement.
Limitations of the study
This research is exposed to numerous limitations. First, respondents were given sufficient time to complete the survey in order to reduce the possibility of response bias induced by the self-administered nature of the questionnaire and self-reporting tendencies. Secondly, by limiting participation to individuals in proximity, convenience sampling introduced survey bias, excluding senior physicians. Third, the involvement of many healthcare experts from the same institution may have led to inconsistency in responses; however, this was minimized by thoroughly training data collectors to ensure consistency across facilities.
Conclusion
The survey indicated that considerably over half of the participants exhibited a strong understanding of generic medications, with significant awareness of bioequivalence, efficacy, and adverse effects. Nonetheless, there were still unanswered questions about safety regulations and the idea of generic replacement. However, their responses revealed a positive attitude and perception toward them. Despite this, several barriers were identified, including inconsistent policy implementation, regulatory challenges, inventory management, lack of standard guidelines, influence of pharmaceutical companies, communication challenges, patient demand for branded medicines, limited information, and lack of confidence in quality. These misconceptions may restrict the utilization of generic medications in Pakistan and, thus, must be rectified. It is imperative to equip healthcare professionals with comprehensive information regarding the efficacy, safety, and quality of generic medicine and the benefits of their utilization, through training programs and educational initiatives in various clinical environments and to encourage generic medication substitution. Moreover, physicians and pharmacists must engage proactively in patient counselling and education, adopting a distinctly favourable perspective on generics.
Supplemental Material
Supplemental Material - Assessment of physicians’ and pharmacists’ knowledge, attitudes, perceptions, and barriers concerning generic medicine in Pakistan: A cross-sectional study
Supplemental Material for Assessment of physicians’ and pharmacists’ knowledge, attitudes, perceptions, and barriers concerning generic medicine in Pakistan: A cross-sectional study by Ayesha Azam, Muhammad Amir Hamza, Rehana Sarwat, Abdullah Abdul Latif Akhter, Bushra Batool Zahra, Bilal Awan, Ali Ahmed in Journal of Generic Medicines
Footnotes
Acknowledgment
The authors express gratitude to all the physicians and pharmacists who contributed to the study.
Ethical considerations
The research was authorized by the Riphah Institute of Pharmaceutical Sciences’ Research Ethics Committee at Riphah International University in Islamabad, Pakistan (Ref. No. REC/RIPS/2023/30).
Consent to participate
All participants were provided with written consent prior to their involvement, which included an explanation of the study’s objectives and procedures. Prior to enrolling in the study, participants confirmed their voluntary involvement by signing the consent form.
Author contributions
Conceptualization: Ali Ahmed, Ayesha Azam.
Data curation: Ayesha Azam.
Formal analysis: Muhammad Amir Hamza, Ayesha Azam.
Investigation: Abdullah Abdul Latif Akhter, Bilal Awan, Ayesha Azam.
Methodology: Ayesha Azam.
Project administration: Rehana Sarwat, Bushra Batool Zahra.
Resources: Bilal Awan, Ayesha Azam, Abdullah Abdul Latif Akhter.
Software: Muhammad Amir Hamza, Ayesha Azam.
Supervision: Ali Ahmed.
Validation: Ali Ahmed, Ayesha Azam.
Visualization: Ayesha Azam, Rehana Sarwat, Muhammad Amir Hamza.
Writing-original draft: Ayesha Azam.
Writing-review & editing: Ayesha Azam, Muhammad Amir Hamza, Rehana Sarwat, Abdullah Abdul Latif Akhter, Bushra Batool Zahra, Bilal Awan, Ali Ahmed.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data are accessible upon reasonable request. The data collected and/or examined in this study are not publicly accessible; however, they can be obtained from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Author biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
