Abstract
This study aims to identify how technological, organizational, and environmental factors influence the creation of an Industry 4.0 (I4.0) strategy in the generic pharmaceutical manufacturing industry. In this study, the technology-organization-environment (TOE) framework is used to explore the factors that influence the creation of an I4.0 strategy in generic pharmaceutical manufacturing organizations. A semi-structured interview case study approach was used to gather data from a generic pharmaceutical company using the company’s creation of a digital strategy as the focus of analysis. The study identifies a paradox where Regulatory Compliance serves both as a driving force for I4.0 strategy creation, aligned with stakeholder pressure, and a potential barrier due to complexity and implementation costs. The impact of regulatory changes, such as those in environmental, social, and governance (ESG) reporting, also contributes to the adoption of I4.0 technologies. The importance of organizational executive leader alignment on the broader environmental context and technical benefits for creating an
Introduction
Trends in trade and globalization as well as supply chain disruptions and drug shortages caused by the COVID-19 pandemic have resulted in a renewed focus on building domestic supply chains in the generic pharmaceutical industry. 1 The shift away from low-skill production regions has put tremendous pressure on higher labour cost manufacturing countries to adapt automation to keep up with demand and stay price competitive. 2 Advances in technology has made digital innovation a key component in optimizing business processes and gaining supply chain efficiency to remain competitive. 3 For manufacturing companies, the adoption of the technology used to enable digital innovation is equated with implementing Industry 4.0 (I4.0) technologies and processes. Schwab 4 describes I4.0 as the blurred intersections of digital technology, biological, and physical systems. The implementation of I4.0 technologies have made a positive impact on the performance, costs, quality, delivery, flexibility, and innovativeness of manufacturing organizations. 5 Despite these widely acknowledged benefits that I4.0 offers to businesses, the pharmaceutical industry is mainly operating in a Industry 2.0 framework with some advances into Industry 3.0. 6 However, increasing pressure to reduce the costs of prescription drugs, drug shortages creating requirements to have better visibility into the entire supply chain, and some openness to I4.0 by regulators have put a focus on modernizing the pharmaceutical industry. 7
I4.0 is more than a technology implementation that drives internal benefits. I4.0 requires companies change their processes and organizational structures to create value for customers which requires companies to adapt to the change to remain competitive.
8
For organizations to embark on a I4.0 transformation strategy, they must be aware of what internal and external determinants that influence their need to transform.
9
Determinates are the drivers that favourably influence whether the organization proceeds with the change, or in the case of barriers, unfavourably toward the change.
10
For exploring the determinates of how one leading generic pharmaceutical firm’s approach to Industry 4.0 we adopt the technology-organization-environment (TOE) framework to answer the following research question: What are the technical, organizational, and external environment drivers and barriers for a generic pharmaceutical manufacturing company to develop a I4.0 digital transformation strategy?
Greater understanding of the determinants that lead generic pharma manufacturers to the creation of an I4.0 strategy will enable the industry to create an approach to gain buy-in for strategy creation, moving the industry forward and ultimately benefit consumers, governments and communities with high skilled manufacturing jobs and a reliable supply of affordable quality medication.
In addressing this question an in-depth case study of a generic pharmaceutical manufacturing firm was conducted, using the company’s creation of a digital strategy as the focus of analysis. Specific aspects of the company’s digital strategy were underway and other areas were still under development which created a unique opportunity to gather insights from different executives that were part of the decision-making process to embark on the creation of the initial strategy as well as with those that were still considering the focus areas for digital transformation within their functional areas. Data were collected using semi structured interviews with decision makers and those consulted in or were influencers in the formation of the digital transformation strategy. To build a framework for the analysis we utilized a recent comprehensive literature research into the drivers and barriers of Industry 4.0 technology adoption in the among small and medium sized manufacturing enterprises (SMEs) which have also been slow to adopt I4.0 technologies. 10 By gathering empirical data to study this phenomenon, we respond to the call by 11 using their research as an initial position in order to compare strategies for digital transformation in different countries and industries.
The remainder of this paper is divided into six sections. In the next section we review the current literature to describe the various determinates for creating a I4.0 digital transformation strategy. Section 3 provides justification for the research design, data collection and analysis methods. Section 4 presents the findings from the case study. The discussion section compares the empirical evidence to the existing literature to arrive at the generic pharma specific factors that influence the creation of a I4.0 strategy. The paper is concluded by outlining the study’s contribution to theory, management practice and proposing areas for future research.
Literature review – digital transformation, industry 4.0 and the pharmaceautical industry
Digital transformation drivers and barriers
Given the emergence of digital native companies such as Amazon, Uber, AirBnB to name a few and their disruptive and transformative impact on the industries which they operate its perhaps not surprising that there has been a dramatic increase in the research on Digital Transformation (DX) in the past 10 years. 12 DX is more than simple “digitization” or the conversion of analogue information and processes into digital form using IT systems. 13 However, in a review of the current state of the literature on digital transformation 12 noted that there is no established definition of DX, although all definitions have some components of technology and customers impact while some go further to include more macro level components of the economy, government, and human society.
Reflecting these themes in the DX definitions,
12
literature analysis revealed three distinct clusters in the DX research streams: digital business transformation, technology as a driver, and institutional and societal implications. They found that digital business transformation has business process, technology, and organizational change components. They describe the technology advantages of DX by integrating technology into business strategies and operations that can lead to a competitive advantage (Figure 1). Advantages of introducing technology into business strategies.
12
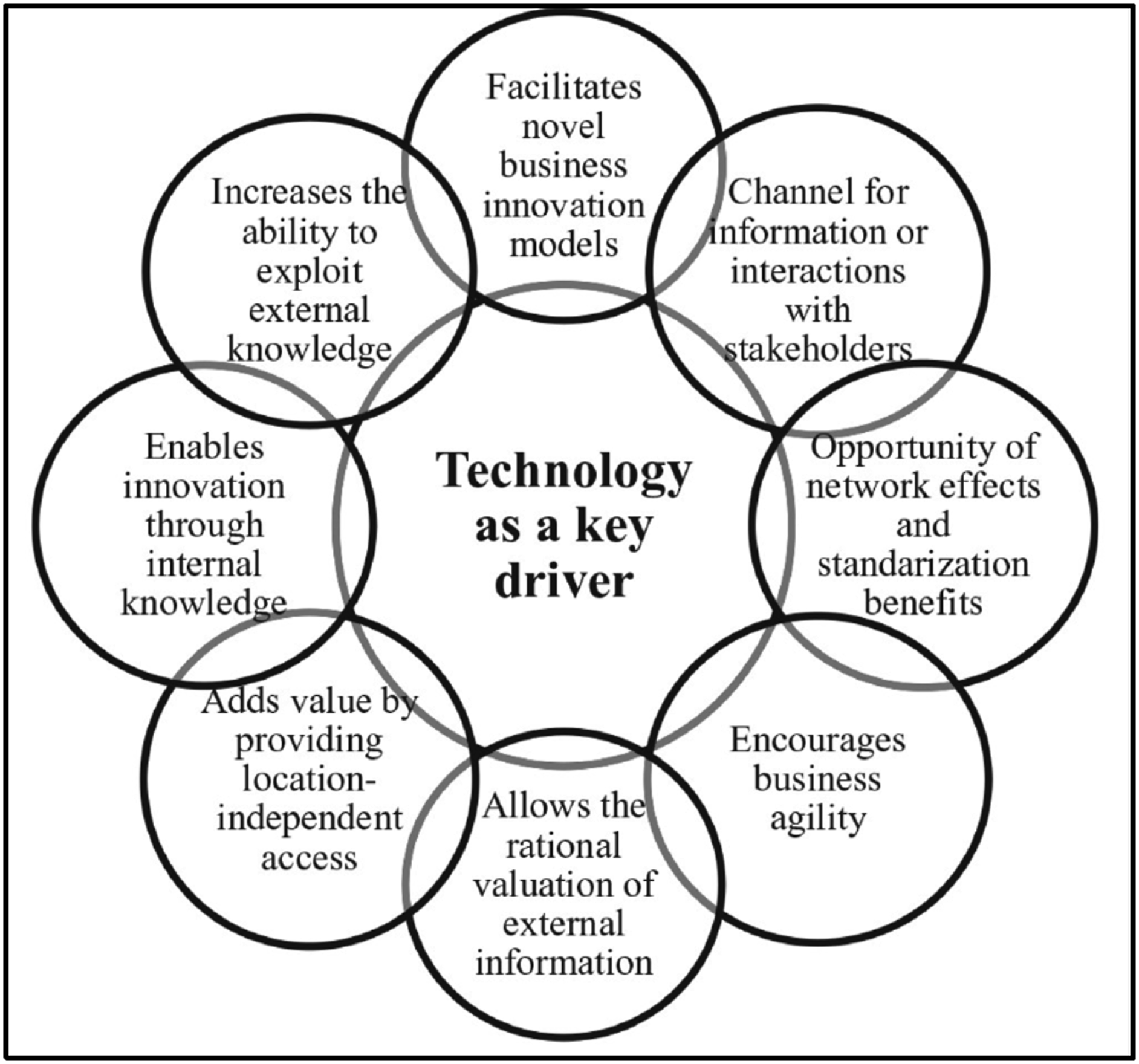
In their literature review that explores why organizations undergo digital transformation, 14 identified four drivers for digital transformation; Customer behaviour and expectations, Digital shifts in the industry, Changing competitive landscape, and Regulative changes. They differentiate drivers as external or internal triggers for DX from objectives being the outcomes or expectations from the DX. They also categorize the dimension of success factors as those attributes that are necessary for an organization to accomplish the digital transformation. They also noted that “we were surprised not to find more emphasis on change management in relation to the digital transformations studied in the reviewed articles.”
Nadkarni and Prügl 15 define digital transformation as organizational change triggered by digital technologies. Through this lens they created digital transformation maps which categorized and describe the existing body of DX research. They revealed nine core themes in the literature as most “influential and unique antecedents and consequences” of DX that they grouped into technology and actor aggregate dimensions.
They observe that the current literature is highly Information Systems (IS) centric, that most of the DX research has been conducted on industries that are already-digitized established industries such as retail or media, a gap in the research that is an opportunity for further research.
Digital transformation in manufacturing - industry 4.0 drivers and barriers
Digital transformation in manufacturing is different than in other industries. DX in manufacturing is about “generating new value from the interconnection of physical and digital assets through data.” 16 Hanelt et al. 17 point out that the key difference between industries that can completely digitize their products and those that need to rely on physical elements as a core element, is the “inevitable need to deal with the tensions that result from interweaving physical and digital layers into business models that originate from a pure physical world.”
Industry 4.0 (I4.0) originated in Germany and despite minor differences is used synonymously with the terminologies of Smart Manufacturing terminology used in the USA as well as the Smart Factory predominately used in Asian countries. 18 Schwab 4 describes I4.0, the fourth industrial revolution, as the blurred intersections of digital technology, biological, and physical systems. I4.0 is viewed as digitalization of the entire manufacturing company value chain with a focus on meeting individualized customer requirements. 19 The U.S. National Institute of Standards and Technology (NIST) defines smart manufacturing as “fully-integrated, collaborative manufacturing systems that respond in real time to meet changing demands and conditions in the factory, in the supply network, and in customer needs.” 20 For the purposes of this study I4.0 is considered equivalent to DX in manufacturing companies.
Theoretical framework
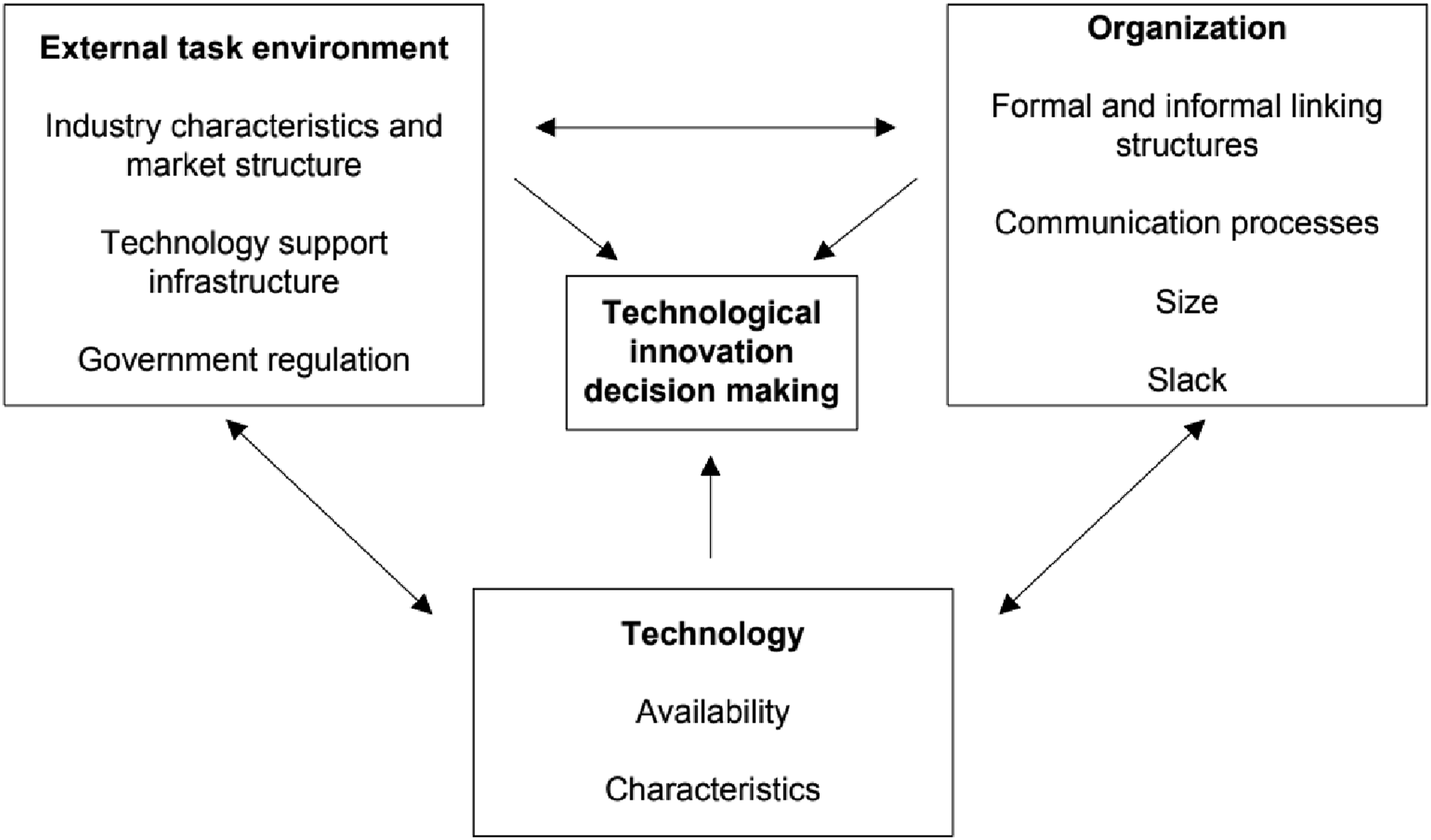
The overarching reach of I4.0 as well as the research question focus on technology innovation adoption is suited to the theoretical study lens of the technology-organization-environment (TOE) framework. 21 The TOE framework is an organization-level theoretical framework that can be used to explain the how the different organizational contexts of new and existing technologies, aspects of the organization itself and the external environment which it operates, their interplay and their role in the adoption and implementation of technology innovation. 22
A portion of the technology context of the framework describes the concept of “competence-destroying” innovations that aligns well with the disruptive nature of I4.0. According to Baker 22 organizations must carefully consider these technologies as “some innovations will have a dramatic impact on the firm and the industry in which it competes.” The organizational context refers to the about the firm such as scope, size, and managerial structure. 21 In the context of organizational factors influencing innovation, the presence of informal “linking agents” working in decentralized structures are important factors. 22 The environmental context includes the regulatory environment which is particularly relevant to the pharmaceutical industry. Government regulation can have positive or negative influencing effect on innovation. Governments impose new regulations on industry that require technology investments to comply or they can require testing, training and documentation requirements that can discourage innovation. 22
Vogelsang et al. 23 described the success factors for DX in manufacturing in the three major dimensions that align with the TOE framework in which DX takes place. Their research highlights the focus on external stakeholders such as customers and suppliers, the internal organization cultural change that is necessary, and the choice of technology being essential but not enough to gain benefits from DX. Ghobakhloo et al. 10 also utilized the TOE framework to systematically categorize and review factors that might impact SMEs behaviour in adopting Industry 4.0 technologies. Using the TOE, we next summarize the research on the various barriers and drivers for DX strategy creation.
There are different definitions of drivers and barriers in found in the systematic literature reviews (SLR) of digital transformation.24,25 For the purposes of the literature review, we take the perspective of Ghobakhloo et al. 10 that drivers being factors that favourably influence whether the organization proceeds with the change and barriers being defined as factors that unfavourably influence an organization toward the change.
Technology determinates
I4.0 involves implementing emerging technologies including cloud computing, Internet of Things (IoT), Additive Manufacturing, Augmented Reality, Artificial intelligence, Cyber-Physical Systems, Blockchain, as well as more foundational systems such as Enterprise Resources Planning, Manufacturing Execution Systems, and Industrial Control SCADA systems. 5 The implementation of digital systems is a prerequisite for achieving the benefits of I4.0.
Khin and Kee 25 developed an integrated conceptual model that identified driving factors that could trigger firms to consider implementing I4.0 technologies; facilitating factors could push them to go ahead with implementation as well as impeding factors should be evaluated to ensure that the benefits of I4.0 adoption outweigh challenges and that the latter can be addressed. They identified operational benefits, market opportunities, competitiveness, customer requirements, labour problems and quality image as the main driving factors towards I4.0. Research by Ghobakhloo et al. 10 identified the perceived benefits of operational efficiency, enhanced operational reliability, improved financial performance and improved marketing performance as the main drivers for SME to move favourably towards I4.0. Mahmood et al. 26 identified and ranked based on frequency in the DX literature the benefit of digital transformation as follows: value creation, operational efficiency, customer relationship/engagement, new business models, and competitive advantage.
In a review of the literature on SME I4.0 adoption Ghobakhloo et al. 10 found that despite the decline of costs for the technology, in the majority of cases reviewed, financing the direct and indirect costs of I4.0 technology acquisition and implementation was the major barrier for I4.0 adoption. Their literature review also found that the complexity of the technology, compatibility and cybersecurity risks were additional key barriers in the adoption of I4.0. These finding seem to align well with other research outside of the SME industries. Khin and Kee 25 similarly found that lack of funding and technical challenges as impeding factors for I4.0 in Malaysian manufacturing companies. Others also found that the high investments in I4.0 to be a barrier for some companies, 24 although this may be more a country specific and company size factor.27,28 Vogelsang et al. 23 research identified reliability, adaptability, and availability of the technology being critical to the successful implementation of I4.0. They also highlight characteristics of the data being critical for I4.0 success such as its relevance, completeness, and timely availability.
Organizational determinants
Jones et al. 3 found that the barriers to DX implementations in manufacturing reported in the literature relate heavily to technology. However, they further define DX in manufacturing as a “wickedly complex problem” that “is less about adding technology to the production line and more about changing all aspects of the business from processes to culture starting with the pervasive mindsets held by individuals and as a collective organization.”
Bellantuono et al. 8 reviewed the existing DX frameworks through the lens of organizational change management models. They found that existing DX frameworks and roadmaps for I4.0 transition do not consider the lessons learnt in the field of change management. From their organizational change perspective clearly defining how the organization intends to create value in the era of I4.0 is the most critical driver for DX. This is in line with Khin and Kee 25 findings on driving factors for I4.0 that firms “need to be aware and convinced of various benefits and opportunities associated with I4.0 and possible expectations of customers and competitors’ threats as these could drive them toward I4.0 adoption.” These findings point to having a well-articulated strategic vision that is incorporates a DX strategy as the most critical factor for digital transformation success or failure by numerous researchers.26,27
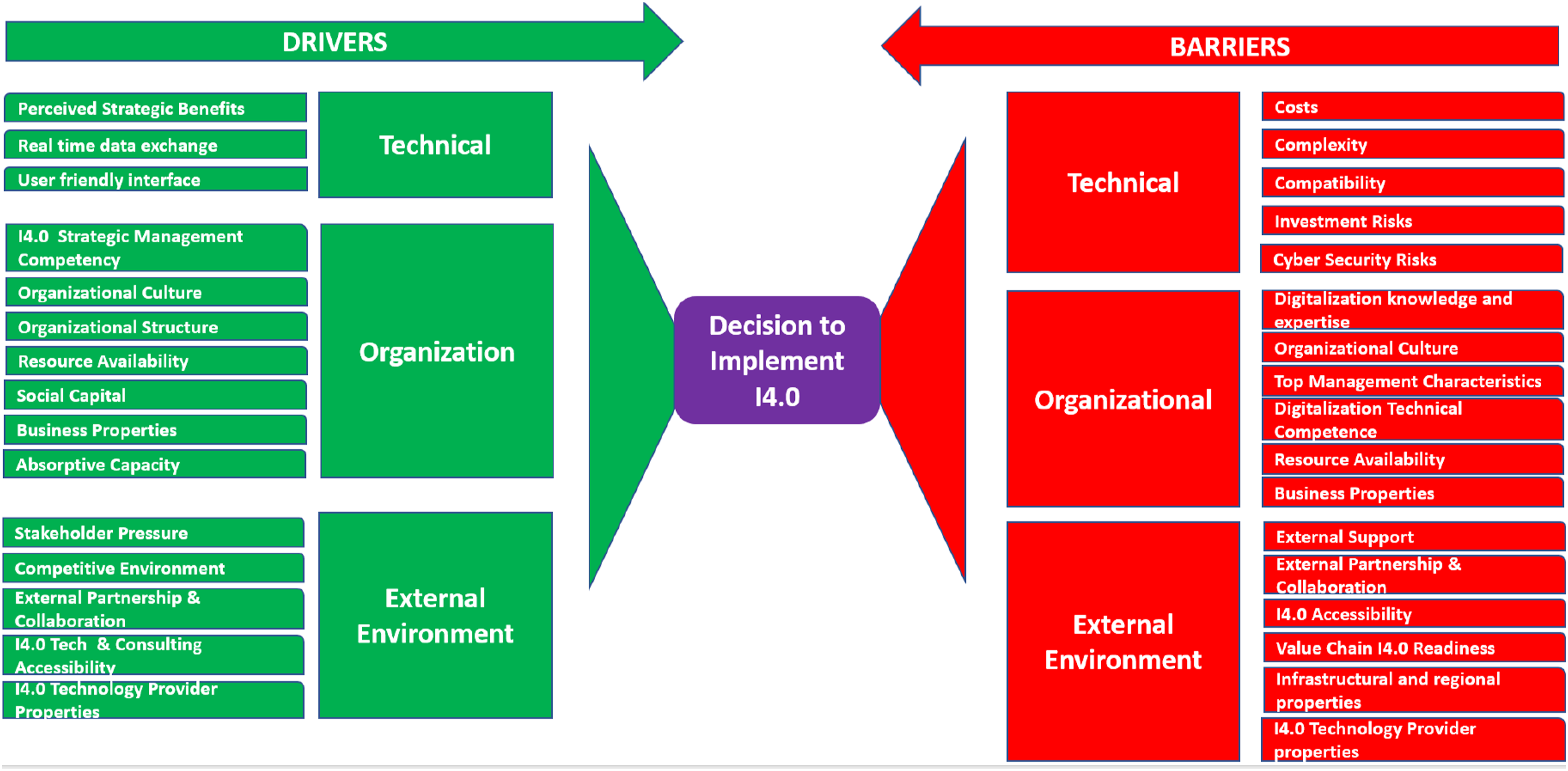
Kane 29 emphasize that “companies will effectively navigate the challenges posed by digital disruption if they look at them as organizational and managerial problems, rather than technical ones.” This perspective is supported by Ghobakhloo et al. 10 that found several organizational barriers to I4.0 in SMEs that aggregated themes in the literature into organizational determinate clusters including the lack of digitalization knowledge and expertise, I4.0 strategic management competency, organizational culture, organizational structure, top management characteristics, resource availability, digitalization technical competency, and properties of the business. The impeding factors for I4.0 found by Khin and Kee 25 lack of knowledge, getting the right people, and changing people’s mindset could also be considered as organizational barriers that may be important considerations when deciding to initiate a I4.0 strategy. In a broad look at supply chain DX literature Lammers et al. 24 also found that knowledge and skills as well as organizational factors such a weak strategies and the lack of time are barriers to I4.0 implementation.
Environmental determinants
I4.0 involves vertical integration of systems within the organization from shop floor sensor level to business level with systems such as Enterprise Resource Planning (ERP) as well as horizontally integrating to external systems across the value chain partners from supply to after-sales services. 30 Both types of integration create challenges, however the horizonal integration has external barriers outside of a firms direct control such as the lack of standards as well as inconsistent laws around data privacy and security. 23
In addition to the systems integration lens, there are external factors that might affect the technology adoption of the company such as company size, manufacturing complexity, and overall attitude towards I4.0. 31 Ghobakhloo 32 identified environmental determinants relating to the business environment such as the competitive environment, stakeholder pressure, external financial support, regional infrastructure and accessibility to I4.0 technology and expertise. Lammers et al. 24 found that the regulatory policies and regulations can be an external barrier or a driver, noting that “governments’ actions can also be a driver for digital transformation in the form of support to the deployment of innovation through incentives that facilitate the adoption of digital technologies.” 11 highlight customer demands for quality assurance in terms of expectations of traceability of components that go into the product as well as through the whole production process as a driver for DX.
Generic pharmaceutical industry determinants
The Pharmaceutical market in North America continues to see robust growth. In Canada revenues were up 6.6% to $35.2 Billion with the generic companies having 73.5% of the market share in terms of prescriptions dispensed. 33 In the US, the total spending in 2021 was approximately $574 Billion on pharmaceuticals, with an 88.9% share of prescriptions dispensed being filled by generic manufacturers. 34
Despite the continued industry growth and profitability, there are several drivers for the generic pharmaceutical industry to invest in I4.0. The pharmaceutical market is facing profit margin pressure challenges due to pricing pressures and rising costs due to inflation related increasing costs for labor, raw materials, and transportation while at the same time being asked to improve the reliability and quality of supply. 2 The COVID-19 pandemic revealed weakness in the pharmaceutical supply chain. Drug shortages due to regional lockdowns, export restrictions and overall supply chain disruptions showed how the dependency on China sourced raw materials creates vulnerability in the supply of medicine to North America. 35 Also, the optimized large batch production at Indian pharmaceutical manufacturers lowers costs and maximizes profitability but sacrifices adaptability, threatens quality, and “reduces the ability to absorb any shocks in times of reduced supply or spikes in demand.” 1 There are also demands from regulators and consumers to have complete traceability of the finished products to deter counterfeit drugs. 36 Regulators also want transparency to the various ingredients used their production to ensure quality and reliable supply. 1 The reaction to the drug shortages and dependence on offshore manufacturing has created pressure on the US and Canadian governments to make significant investments to repatriate pharmaceutical manufacturing and created an opportunity for manufacturing companies to make investments in I4.0 as a focus of operations strategy to overcome these current and future industry challenges.35,37
Despite the benefits of automation associated with I4.0 for the pharmaceutical industry including being able to insulate themselves from increasing costs pressures as well as potentially dramatically improve quality, the pharmaceutical industry has been slow to adopt technology.2,6,7 According to Arden et al. 6 the pharmaceutical industry is mainly operating in an Industry 2.0 (I2.0) framework with some advances into Industry 3.0 (I3.0). They describe I2.0 in the context of pharma as using electronic machine-based operations allowing for large scale production and importantly for the pharmaceutical industry a driver for implementation as they provide the potential for “more monitoring of processes and quality.” I3.0 technologies are described as a driver for pharma manufacturing organizations as the production control systems reduce the need for human operators on the manufacturing floor and facilitate better tracking of parameters and metrics associated with production. However, the industry has been slow to implement other technology components of I3.0 such as continuous manufacturing and inline testing despite their potential to reduce costs, improve productivity and reduce human errors. Arden observes that the barriers to adopting newer technologies are “the lack of precedent in the industry, the costs associated with development, and perceived regulatory uncertainties leave many firms keen on a ‘first to be second’ approach by observing how competitors approach new manufacturing technologies and how regulators respond.” They conclude that new policies that incorporate I4.0 as well as international regulatory convergence will encourage the industry to adopt new manufacturing technologies. This is in line with Manzano 38 that reviewed the adoption in technology in the pharmaceutical industry and recommended that “the pharmaceutical industry work more closely with all the actors involved in the community, including regulators and suppliers, to speed up the regulatory framework of these technologies, as the pharmaceutical industry can play a critical role in this process.”
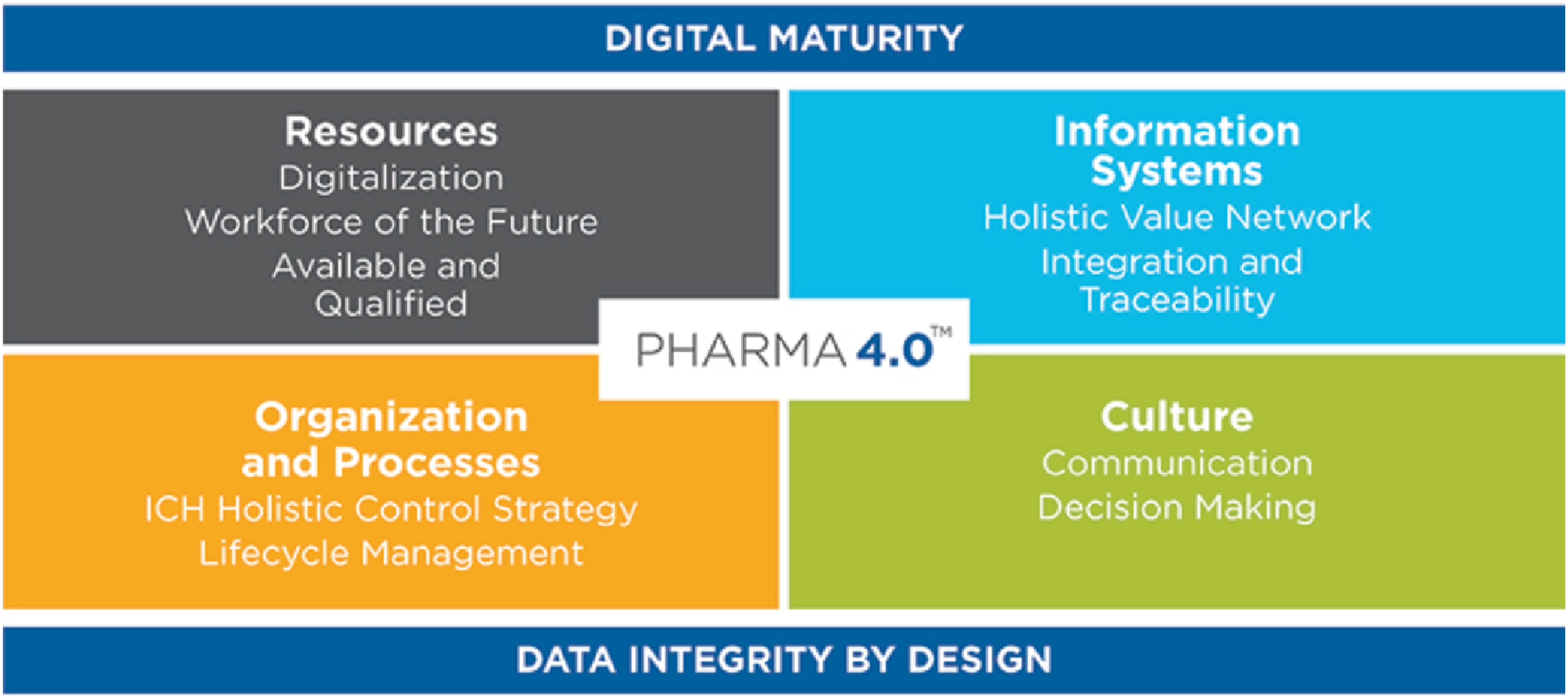
The pharmaceutical manufacturing industry has built on the concepts of I4.0 into an industry specific operating model, Pharma 4.0. Introduced by an industry association, the International Society for Pharmaceutical Engineering (ISPE), Pharma 4.0 takes a manufacturing perspective to the implementation of digital technologies that incorporates industry specific quality systems regulations within a pharmaceutical enterprise. 39 The Pharma 4.0 operating model is divided into four discrete elements of resources, information systems, culture and organization and processes, with the enablers of data integrity by design and digital maturity. 40
The Pharma 4.0 operating model components can be viewed as both drivers and barriers for adoption. The components of Pharma 4.0 are common in the I4.0 and DX literature, the difference being the emphasis on pharmaceutical industry specific quality systems within each element and the focus on data and analytics to improve quality control, optimize production, and reduce costs. Pharma 4.0 also highlights the importance of data integrity as a requirement for reliable traceability and transparency which are regulatory expectations of manufactures in the pharmaceutical industry. Overall, the adoption of the operating model is a driver for investments in I4.0. However, Pharma 4.0 digital maturity model that recognizes that manufacturers are at different levels of maturity in all of the operating model four elements, and that movement from level 1 to move toward Pharma 4.0 has many challenges that requires “the entrepreneurial courage to start and the guidance to change with a controlled road map”. 41
Methodology
Research design
An interpretivist perspective was taken to explore the factors that influence generic pharmaceutical firms to decide to create a digital transformation strategy. A case study method was selected as grounded theory methods are well suited to understanding individual, social and organizational processes. 42 A single case design was selected because it provides for a more in-depth level of investigation than multiple cases. 43 The emic perspective as lens for synthesizing and interpreting study findings was chosen as the author’s participation in the DX program provided in depth knowledge of the organization as well as access to the key decision makers and influencers in the creation of the digital transformation strategy. 42
A deductive directed theoretical content analysis approach was used based on the TOE framework themes, determinate clusters, determinates and sub-determinates identified by a SLR of I4.0 drivers and barriers in SME conducted by Ghobakhloo et al. 10 This framework was selected due to the volume of contemporaneous literature reviewed and the similarly slow adoption of I4.0 between pharma and SME manufacturers. The pre-existing theoretical framework was used as a starting point that was augmented with unanticipated findings that may be company or generic pharma industry specific.
Sampling and data collection
The sampling logic was to collect data with the executives and influencers involved in the creation of the DX strategy. All participants were from the head office location in Toronto, Ontario, Canada. Data was collected from the President, Senior Vice President, Vice President and Director levels across R&D, Regulatory Affairs, Manufacturing, Supply Chain, Engineering, Quality, Sales, Human Resources, Finance, Communications, and Information Technology. The informants interviewed ranged in pharmaceutical industry experience from 10 to over 40 years. The purposeful gathering data from these different perspectives presented a distinct lens on the same phenomenon, limiting bias associated with a single case, while providing deep insights on how technology, the organization and external environmental factors influences the decision making around the DX strategy.42,44
TOE contexts and related interview questions (prepared by author).
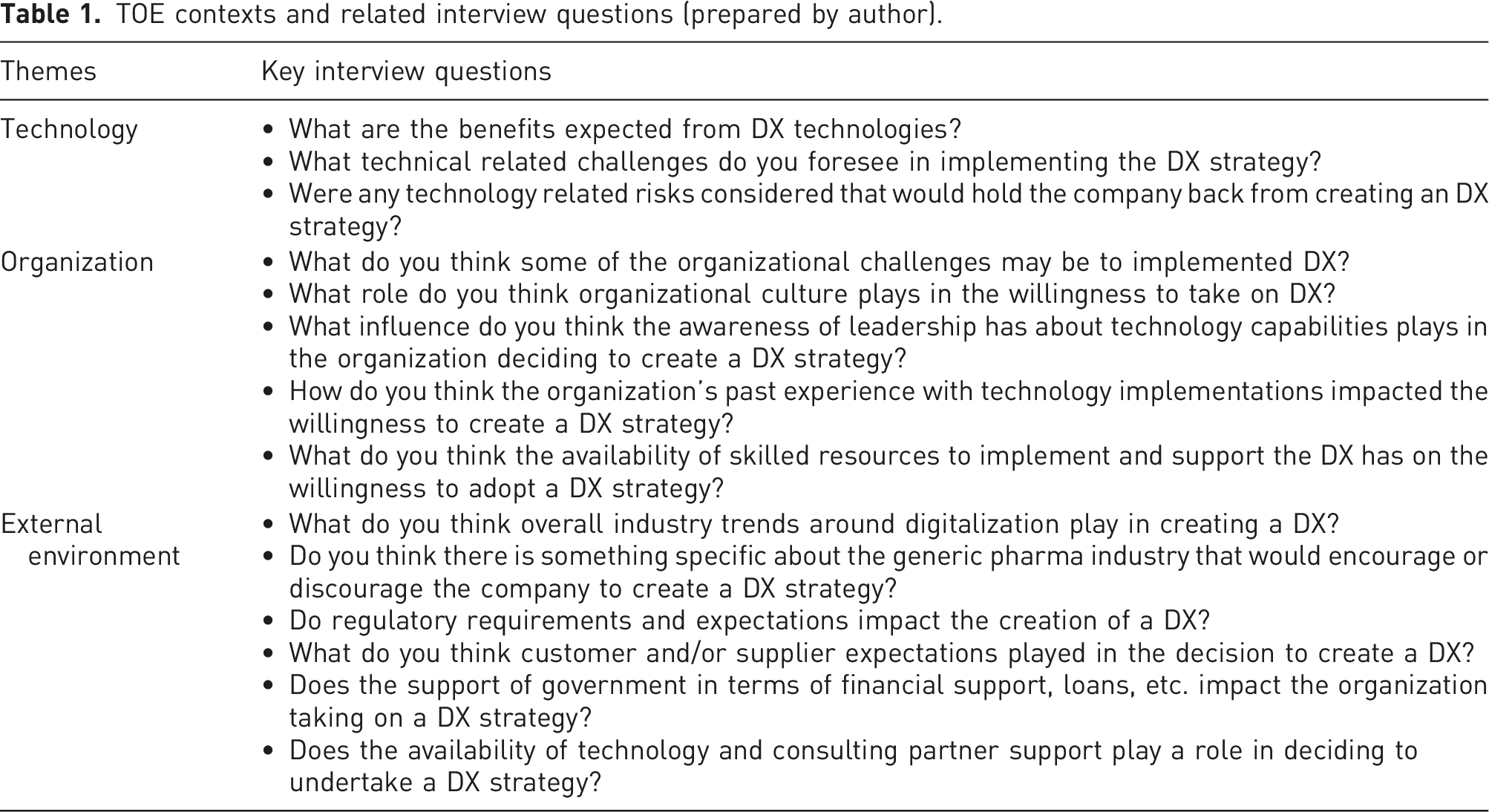
The interviews were conducted by video call using Microsoft Teams and lasted between 45 min to an hour. The interviews were transcribed using the Teams transcription feature then saved to a Word document format.
Profile of participating firm
PharmaCo has created and begun executing a “Smart Factory” DX strategy. The strategy is communicated across the organization as being critical for remaining competitive as well as keeping pace with regulatory expectations and improving customer service.
PharmaCo is a multinational generic pharmaceutical manufacturer with 7500+ employees. There primary markets for sales are the U.S. and Canada. Sales in the U.S. for generic pharmaceutical industry is concentrated on three main national wholesaler customers with pricing being set by the competitive market. The Canadian market has more diverse customers with wholesalers, banners, and independent pharmacies; however, the pricing is controlled by the provincial governments. The Canadian government does not provide tax or other incentives to the domestic pharmaceutical manufacturing industry. 61
PharmaCo has manufacturing sites in Mexico and India, however most products are manufactured in the high labour cost Canadian and U.S. regions. They internally manufacture over three hundred product lines, over five thousand SKU’s. PharmaCo has a relatively average older employee age of 45 years, many employees with greater than 10 years of service. PharmaCo must comply with Health Canada and the U.S. FDA regulations to retain a license to operate and sell products. They are regularly required to submit to Good Manufacturing Practice (GMP) inspection by regulatory authorities as well as customers. Most of PharmaCo’s products are manufactured using the same assets and very similar manufacturing processes. PharmaCo’s profitability is driven by new product launches. The new products approval process requires PharmaCo to submit process steps and ingredients to the regulatory authorities as well as documentation that “validate” process reproducibility and control. Post approval changes to the manufacturing process, equipment used, or ingredients must be re-validated and approved by the regulatory agencies prior to manufacturing commercial saleable products. The requirement for re-validation and the significant capital investments required for new manufacturing equipment have resulted in a mix of modern and legacy equipment throughout the plants.
Data analysis
NVivo was used for data analysis because its features for character-based coding, the ability to define parent child coding levels and automatically aggregate coding from the children to the parent to enable data analysis, as well as the ability to query coding across cases for ensuring coding consistently. All interview transcripts were imported into NVivo from the Teams transcript Word document format with no modifications made by the researcher to the transcript documents. The thematic content analysis coding scheme in Figure 2 was used. The coding scheme is based on the TOE framework themes, determinate clusters, determinates and sub-determinates identified by a SLR of I4.0 drivers and barriers in SME conducted by Ghobakhloo et al.
10
When passages of text did not easily fit the coding scheme, a new coding category was created and assigned to an existing parent code. The coding template was revised in an openminded and iterative fashion until the final version template that provided an extensive explanation of the cases was established.
44
In addition to being coded against the TOE themes, to facilitate the analysis of which Drivers and Barriers were most relevant to the decision-making processes, the passages were coded into strategy influence categories according to the definitions in Table 2 that emphasize the direct influence on the strategy creation. Having completed an initial analysis of the data, the coding process was repeated by the researcher to ensure completeness and consistency Figures 3–5. Digital transformation high-level thematic map emerging from the analysis of the literature.
15
Strategic influence categories and definitions (prepared by author). Technology, organization, and environment framework (Tornatzky et al., 1990). From industry 4.0 to pharma 4.0™ operating model.
40
Determinates of SME industry 4.0 adoption
10
(prepared by author).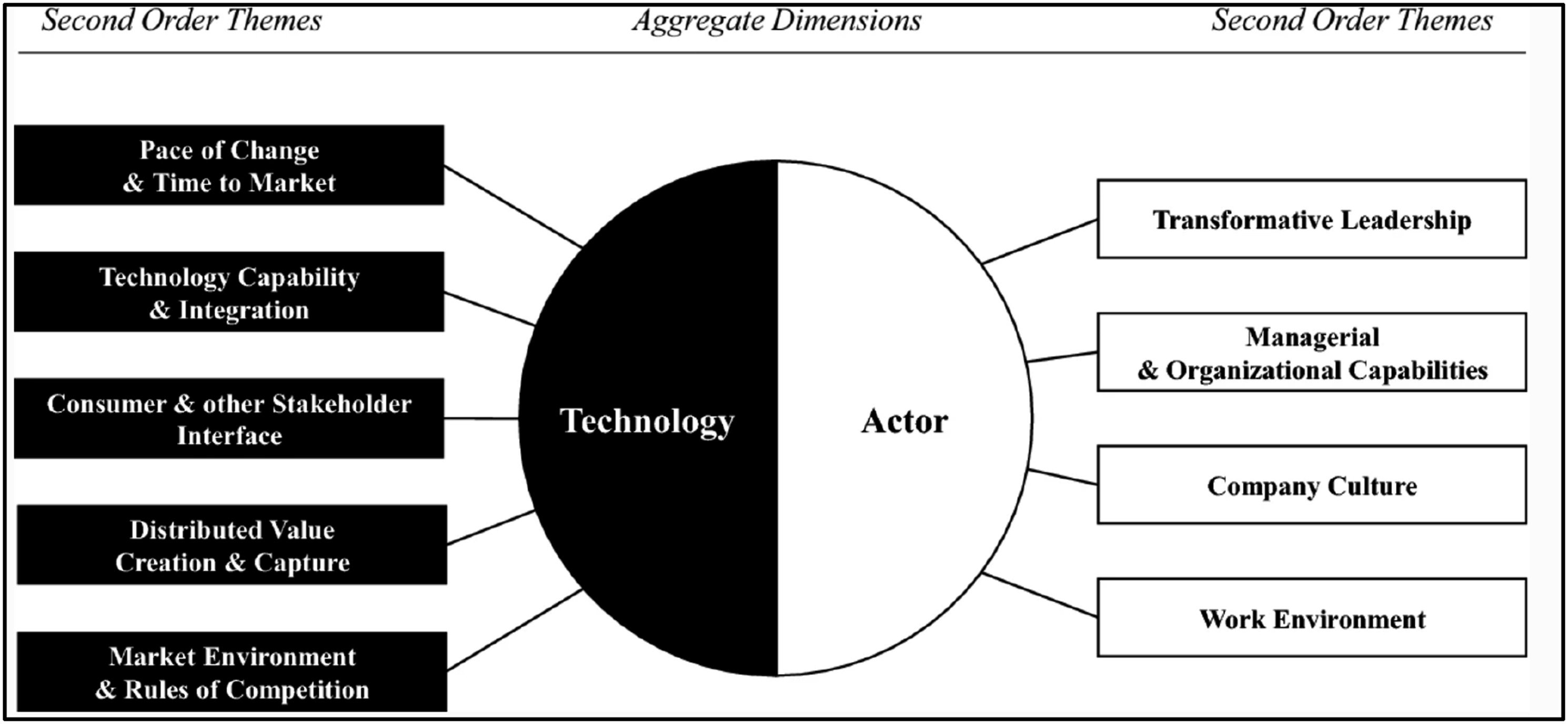

Findings
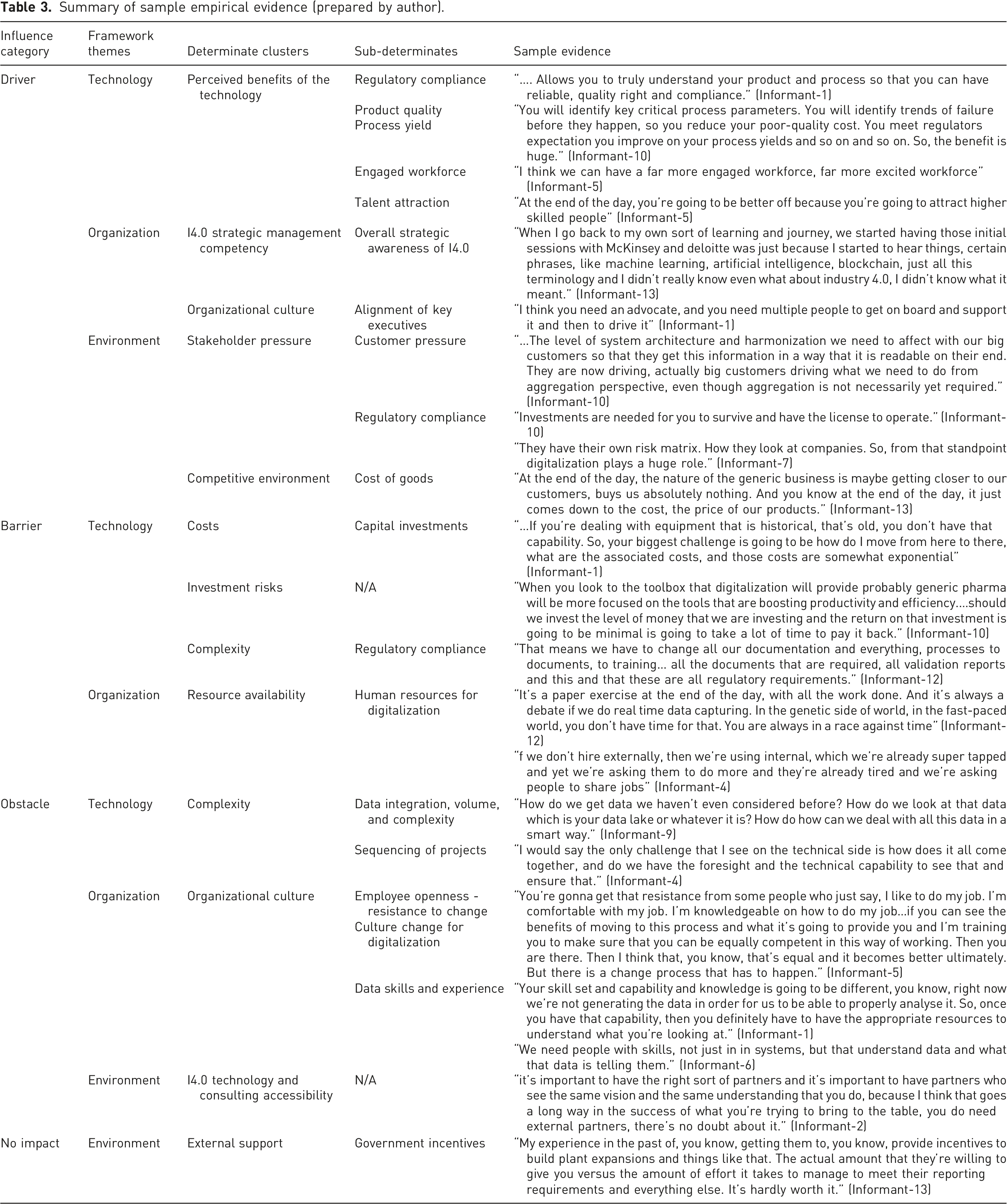
Summary of sample empirical evidence (prepared by author).
Drivers & facilitators to a I4.0 strategy
All informants mentioned Regulatory Compliance as a driver for DX strategy creation. However, the lens in which they viewed the driver differed. Some considered this driver as an outcome of Perceived Benefits of the Technology implementation itself; the technology will make the processes more compliant with regulations. Several others explained the Regulatory Compliance driver, as being more of a result of external Environmental influence of Stakeholder Pressure, as the most important reason to invest in I4.0. The examples given were compliance with data integrity regulations, compliance with the U.S. Drug Supply Chain Security Act (DSCSA) or potential future implementation by the FDA of the Quality Management Maturity (QMM) rating system of manufacturers.
Most informants felt that Customer Pressure was not a driver, the exception being notable, when the customers had to comply with regulatory requirements, as was the case with U.S. DSCSA, that required PharmaCo and their U.S. customers to comply within a certain timeframe. The U.S. wholesaler customers were pressuring PharmaCo to integrate and provide data to pilot the technology and processes well before the deadlines to comply with the legislations.
All informants were asked about the Perceived Benefits of the Technology. The most mentioned benefits that directly influenced the DX strategy creation were Operational Efficiency, Product Quality, Information Flow and Visibility, and Financial Performance. There was consensus that that data generated from the technology and having skilled people to turn the data into actionable information, would be key to realizing the benefits. While the importance of skilled people was recognized by all informants, the view was that these skills could be developed or attracted from other industries.
The Perceived Benefits of the Technology sub-themes of Engaged Workforce and Talent Attraction also emerged as drivers from some of the informants. The informants felt that having state of the art technology would be a key in to retain existing employees as well as attract the talent required for digitalization in a very competitive job market.
The Organizational driver of Overall strategic awareness of I4.0 was identified as being a foundational driver. Most felt that without awareness of the I4.0 benefits then there would be no motivation to develop a DX strategy. This also points to the related Environmental factor of External Partnerships & Collaboration, such as consulting companies and industry groups, which was found to be an important facilitator in the awareness process, but not a driver of strategy creation in and of itself.
The theme of having Alignment of key executives that had a common understanding of the importance of I4.0 emerged during the interviews but was not part of the coding scheme. Several of the informants emphasized that the willingness to develop and commit to the DX strategy would not have been possible without the shared executive alignment. Related themes of Technology Leadership in the C Suite and additional newly identified sub-theme of having a Trusted Leader to drive the DX strategy also emerged from the interviews, however while most informants felt these are important, they were not a deciding factor in developing the strategy but more as a facilitating or important implementation factors.
The Environmental factor of the Competitive Environment, specifically price competitiveness or conversely the Cost of Goods, was also determined to be a driver. The informants felt that the North American generic pharma industry does not have a differentiator other that cost for its products and implementation of I4.0 technologies would reduce costs. Despite the focus on cost, the potential for I4.0 to reduce costs and the main barrier of capital investments identified during the interviews, the Environmental driver of External Support from government in terms of subsidies were not a factor that any of the informants identified as an influence on creating the strategy. However, some felt that the recent incentives offered by the US government for initiatives to develop domestic pharma manufacturing using innovative technologies that enable continuous manufacturing would be an incentive to that will influence strategy and investment going forward.
Barriers and obstacles to a I4.0 strategy
There was consensus across informants that sub-themes of Capital Investments, Initial Investment, and Specialized Skills aggregated as Costs were a barrier to I4.0 strategy. There was recognition that there is significant investment required to replace legacy manufacturing equipment, in consulting costs as well as in the project team itself. Some informants noted that investment in I4.0 in the generic pharma industry is not as significant as in brand pharma. They felt this is due to the significant investments required to continuously develop and deliver new products in order maintain profit margins versus long term strategic investments that may not deliver returns.
In addition to costs the aspect of Technology context of Complexity related to Regulatory Compliance that requires validation of changes to any processes or technology involved with the manufacture or testing of products, submission of data and documentation for regulatory approval, as well as changes to all the internal documentation, training, etc. was identified as a barrier for a manufacturing site with a large product portfolio in which the same equipment used for almost every product. The related Organizational context of Resource Availability was highlighted by some of the informants as being a barrier to creating a DX strategy. The time pressure to deliver new products as well as work on the many other initiatives within the company at any given time with limited resources creates an environment not conducive to creating long term strategies.
There was a common view that once a decision on the strategy was made, and there was commitment that the program would be funded at some level, the other factors were obstacles that may elongate the timelines but could be overcome with planning during the implementation. For example, the Organizational Culture factors such Employee Openness - resistance to change, the Culture Change for digitalization, and others were acknowledged as being critical to the successful implementation of I4.0, but not a barrier that influenced the decision to proceed with the DX strategy. The sub-themes associated with Top Management Characteristics such as Management digitalization awareness and Technology acceptance of management and operations supervisors were also considered obstacles that could be overcome with an Organizational Change Management strategy. Similarly, the Organizational context of Digitalization knowledge and expertise and the associated subthemes such as Data skills and experience was mentioned as an obstacle to achieving the benefits from I4.0, but not a barrier to creating the strategy.
There was acknowledgement by most informants that the implementation of I4.0 technologies is complex. The Complexity sub-themes of data integration, data complexity, data volumes and the sequencing of projects were frequently raised but not considered as factors that would prevent the initiation of the DX strategy. Cyber Security related risks were also considered obstacles that would need to be mitigated by those that mentioned this factor.
The external Environmental context such as accessibility of I4.0 Technology and Consulting in the context of PharmaCo was not a barrier since it was clear based on contacts from potential many consulting partners during the formation of the strategy that in North America there are an abundance of external support options available and that many of the technology components appeared to have matured to the point that it was commercially available.
Discussion and contribution
This case study aimed to better understand the process through which generic pharmaceutical companies decide to create an Industry 4.0 strategy. In this section, we discuss our empirical findings in relation to the prior literature to answer the research question; What are the technical, organizational, and external environment drivers and barriers for a generic pharmaceutical manufacturing company to develop a I4.0 digital transformation strategy?
The findings of the research are in line with the TOE frameworks proposition that the contexts interact with each other to influence innovation decisions. Specific to the generic pharmaceutical industry it is theorised that the impact of the Environment context factor of Regulatory Compliance has a primary impact on other environmental, organizational and technology influences to drive I4.0 strategy creation (Figure 6). However, the nature of the regulation will create a difference in the focus of the strategy. Regulations focused only on manufacturer compliance, such as demonstrating data integrity, will drive investments that are internally focused on digitalization, process control and optimization, whereas regulations focused on the entire supply chain will gain the organizational cross functional executive alignment required drive the creation of a digital transformation strategy. Influencing factors on I4.0 strategy creation decision (prepared by author).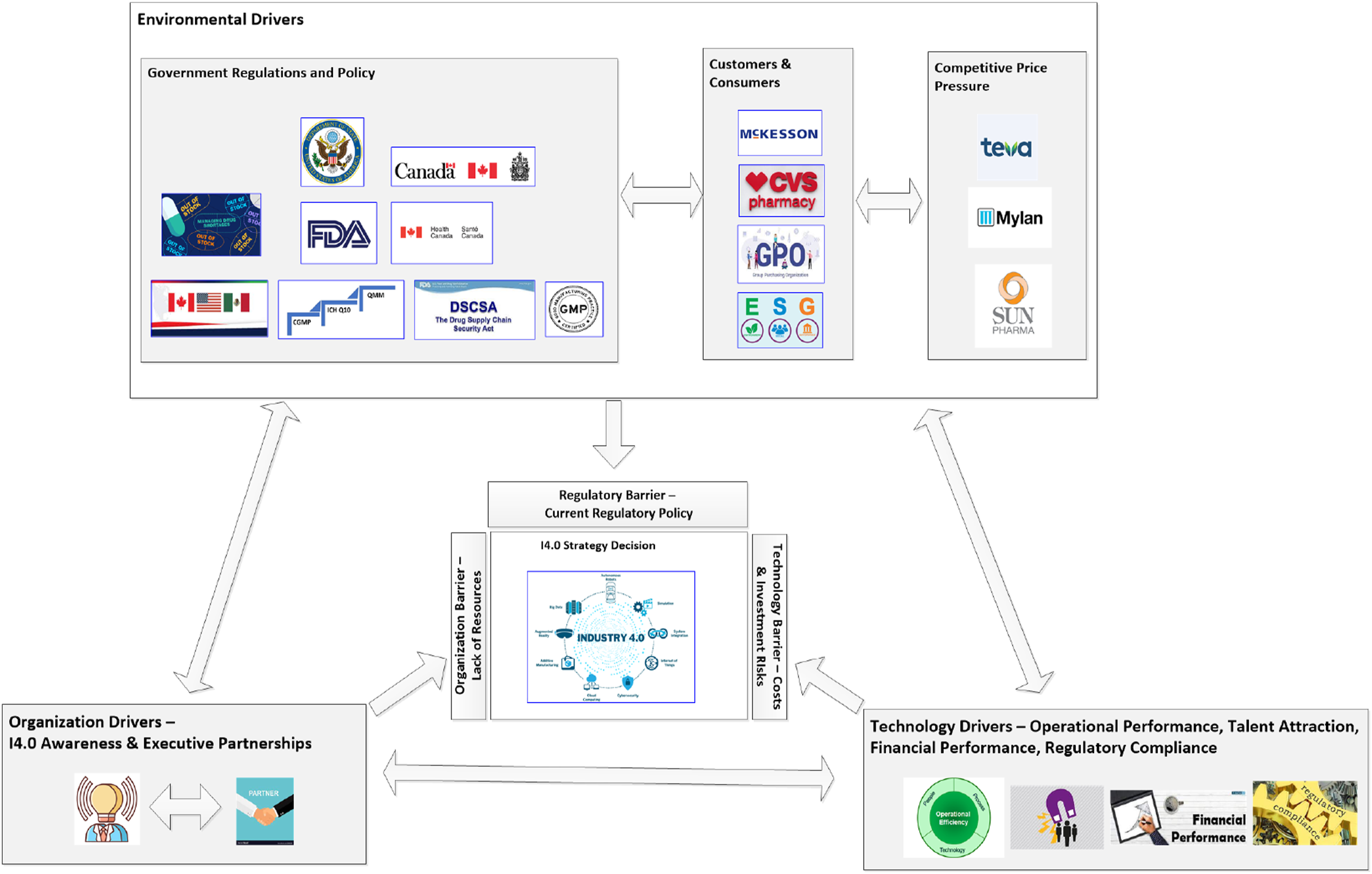
Regulatory Compliance was found to be major driving factor for the creation of a I4.0 strategy which emerged as sub-theme of Stakeholder Pressure aligned to the external Environment context of the TOE framework. Additional Environment drivers of Customer Pressure and Competitive Environment also influenced the need to create a digital transformation strategy. Government regulation, customer pressure, and the competitive environment were previously found in research as drivers for DX in pharma45,46 and other industries.10,15,24,32,47 However, the example given during the interviews of compliance with the U.S. Drug Supply Chain Security Act (also known as Track and Trace or Serialization), provides insight into the primary influence of regulatory compliance interactions and inter-relationships with the factors of Customer Pressure and the Competitive Environment. Compliance with components of the act have gradually been coming into effect over the last 10 years, with the final more granular tracing requirements at the package level coming into place by fall 2023. 48 DSCSA requires companies throughout the U.S. pharmaceutical supply chain to make investments in I4.0 technologies such as vision systems, serialization systems, as well as the development of online platforms that enable connections that enable the transfer of data to upstream and downstream partners in the supply chain. 49 The deadlines to comply with the regulations as well customer pressure on their downstream partners to be ready with the technologies, make changes to their processes to facilitate the implementation of processes and technologies downstream creates some degree of urgency and required a strategy to procure and implement the technology as well as develop the required supporting capabilities. 50 Similar impacts of regulation driving change in industry can be found in studies on environmental, social and governance (ESG). According to Deloitte, the transparency required in the form of sustainability regulatory reporting “can be leveraged to encourage behaviour change toward stated policy goals.” 51 The ESG regulatory and financial reporting requirements have created the awareness and pressure on manufacturing organizations to make changes that leverage I4.0 technologies.52,53
The finding that the Perceived Benefit of I4.0 Technology to ensure Regulatory Compliance is an important driver in I.40 strategy creation for the generic pharmaceutical industry. There is an expectation from regulatory authorities that firms to demonstrate data integrity and process control via the digitalization of manufacturing and laboratory processes. 54 However, there was a counter finding that in the generic pharmaceutical industry Regulatory Compliance requirements can be a barrier to creating a I4.0 due to the complexity and costs involved with implementation and re-validation of processes. These paradoxical findings align with Arden et al. 6 that note “most regulations were developed in an Industry 2.0 paradigm of traditional batch manufacturing” and that “changes in the regulatory structure to enable new technologies” are needed to encourage I4.0 adoption As per the definitions of digital transformation found in the SLR by Kraus et al. 12 ; to transform the industry, changes that take the view of the customer and consumers as well as overall industry efficiency and quality into account are needed to drive I4.0 strategy. Aligned with intention to drive industry transformation, the FDA has recently proposed framework to objectively rate the Quality Management Maturity (QMM) of pharmaceutical manufacturing to create transparency that will align both the customer and manufacturers by rewarding firms that invest in quality systems. 55 According to FDA, “In the absence of the transparency generated by ratings of QMM, there is risk that price competition and cost minimization will continue to be key market drivers, especially for generic drugs, without direct reward for manufacturers who actively invest to avoid future shortages.” The proposed preferential regulatory treatment for manufacturers that invest in I4.0 to demonstrate “continual improvement” and “capability robustness” could result in a competitive advantage that will drive generic pharmaceutical manufacturers to create a I4.0 strategy. The proposed QMM regulatory approach aligns with the TOE framework and what Hanelt et al. 56 found in their SLR of DX from the change and innovation theoretical lens in that DX changes are “a confluence of organizational, technological and environmental forces.” They describe the forces as digital business ecosystems, which define business environments as a network of “interdependencies specifically generated through digital technologies.” Regulatory changes that focus on creating a business ecosystem between FDA, the customers, consumers and manufacturers will provide the incentive to for the generic pharmaceutical industry to create an I4.0 strategy.
The Organizational driver of Overall strategic awareness of Industry 4.0 is consistent with other research findings on DX. 57 The theme of having the Alignment of key executives as a key driver however was not explicitly mentioned as a driver in the SLRs of I4.0 or DX. Perhaps this is not surprising given the focus of DX research has been IS centric 15 and Bellantuono et al. 8 findings from a SLR of digital transformation models (DMT), that most of the DTMs do not include “key activities proposed by change management models”. When viewed from the organization change management lens both of these study findings are in line with what Kotter 58 identified as “Establishing a sense of urgency” and “Creating a Guiding Coalition.” According to Kotter, “the organization realizes the need for change and leadership must be able to explain to other members the urgent need to introduce changes; this first step is critical for increasing organization awareness of the need for change.” In a proposed framework for Digital Transformation readiness Gurbaxani and Dunkle 59 identified an element of Strategic Vision being “Senior executive team has a clear understanding of digital technology capabilities and how they will support business objectives.” They also indirectly acknowledge the importance of Alignment of key executives by highlighting in the Strategic Alignment dimension of their proposed DX framework the alignment of digital and business units, and its importance to the commitment of long-term funding and resources assignment to DX. To overcome the barriers of Complexity and Resources Availability to embark on creating a I4.0 strategy, cross functional alignment of the key executives on strategic initiatives is according to Kotter and Whitehead 60 needed to get the “buy-in” to support the creation of the strategy, and the support needed to overcome the main barrier identified for creating a I4.0 strategy; the capital investment required.
Comparison of primary determinants between PharmaCo and SMEs manufacturers.

Conclusions
The research findings highlight the unique drivers and barriers for the generic pharmaceutical manufacturing industry to adopt an I4.0 strategy. The findings also reveal how the technical, organization and environment contexts interact with each other their importance in influencing the creation of an I4.0 strategy.
The research has identified Regulatory Compliance as a key driver for digital transformation in the generic pharmaceutical industry. Unlike some other industries, if regulatory changes are proposed that align with the spirit of DX through the lens of how the technology will impact the customer as well as the entire supply chain, then regulatory compliance can drive I4.0 transformation, as the example of Serialization illustrates. However, regulations that focus only on the manufacturing firm’s technology and organizations processes, will result in investments to ensure regulatory compliance as well as reduce costs, but will not get the cross functional executive buy-in to create a transformational I4.0 strategy in the spirit of DX. Having a coalition of executives that understand the environmental context of regulatory compliance, the potential impact on customers and the opportunity to gain competitive advantage and well as the technical benefits that can results from an I4.0 strategy execution is key to overcoming the major barrier of funding that can prevent organizations from even considering I4.0 investments.
The findings of the research are limited by the scope of being a single company operating in a specific geographic region with a limited number of informants. Also, the research was conducted by a solo researcher, which was close to the content and the strategy development which may have biased the coding of the transcripts and view of the findings. The questions asked of the informants were intended to provoke discussion on the various determinants of DX strategy however they may have biased the answers or led away from other factors that did not align with the TOE framework. Despite the presented limitations, the recency of the companies PharmaCo’s I4.0 strategy creation and open participation of key executives responsible for the creation of the I4.0 strategy in the research make the finding insightful and relevant for both researchers and practitioners to better understand the determinants for Industry 4.0 strategy creation.
These findings can be applied to a broader range of industrial settings and organizations. The results provide an expanded view for researchers to consider in the exploration of the determinants for I4.0 strategy creation, particularly in regulated industries. Further research is needed to confirm the weighted influence of regulatory compliance interactions and intersections among the technology, organization, and environment contexts as well as their determinants to further expand on how generic pharmaceutical companies or manufacturers in similarly regulated industries should consider them as they embark on creating their Industry 4.0 digital transformation strategy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
