Abstract
2008 can be considered a watershed year for generic medicine in India, as it saw the launch of the Jan Aushadi scheme, to provide cost-effective quality medicines to its populace, through special stores called Jan Aushadi stores. With more than 8800 stores, the scheme is touted as one of the world’s largest pharmaceutical retail chains. Since then, many generic medicine start-ups have also launched operations in the country- Zeno Health, One India Pharmacy, Generic Aadhaar and StayHappi Generics. These supply side developments will however have to be complemented with commensurate demand side responses for it make a sustainable impact towards the push for affordable and accessible healthcare for all (Sustainable Development Goal 3). Many studies have established that, on the demand side, owing to the lack of requisite medical knowledge on part of the patients, there exists a strong agency relationship, where patients rely heavily on their physicians (prescribers) to make decisions on choice of treatments. Though promotion of generic medicines requires a coordinated effort from many stakeholders, analysis reveals that the prescribers, and hence their perception of generics, plays a pivotal role in their adoption. This study undertakes a bibliometric analysis of the top 100 most-cited research papers published between 2012 and 2022 on the perception of prescribers towards generic medicine from SCOPUS database. Suitable keywords have been incorporated to reach the relevant publications and VOSviewer (version 1.6.10) has been used for analysis. The findings of the study recommend further research on interventions aimed at improving prescriber’s perception towards generic medicines to enhance their adoption and promote cost effective healthcare delivery.
Introduction
The pharmaceutical industry is known for discovering, developing, manufacturing, and marketing pharmaceutical drugs. 1 Prior to these products' mass release in the markets, several laws and regulations (both global and national) control the patenting, testing, safety, efficacy, and marketing of medications. As per Patent Law, Market exclusivity for a new molecule can be legally secured by a company for up to 20 years from the date of patent filing. However, the time it takes to develop a new drug and have it approved by the authorities is awfully long. 2 This leaves lesser time for the new drug to spread, while recovering development costs and skimming market profits. Almost all brand-name medications see a sharp decrease in sales and market share after their patents expire due to the quick development of less priced generic counterparts. As a result of this alleged “patent cliff", businesses are supposedly forced to continuously create and market new medications in order to remain profitable. 3
All responsible governments around the world have broadly accepted health equity as a policy goal. “Equal access to available care for equal need, equal utilization for equal need, and equal quality of care for all" are the three components of equity. 4 Making sure that everyone has the opportunity to be as healthy as possible is the goal of health equity, to put it simply. As drugs continue to make up a significant amount of out-of-pocket (OOP) costs and frequently put the majority of households in a state of financial crisis, access to inexpensive medicines becomes a vital factor in achieving health equity. In India, the cost of medications makes up 50% to 80% of the total cost of care, according to estimates from NITI Aayog. With private healthcare facilities providing more than 2/3rd of outpatient care and more than half of the inpatient (hospitalization) care, households are exposed to the private sector while purchasing medications. 5 Since these unforeseen costs are a substantial contributor to the poverty that low- and middle-income households experience (Saksena et al., 2010), they are referred to as catastrophic expenditures. 6
In order to achieve improved health outcomes, generic medication offers substantial cost savings and better resource utilization as generic drugs are priced about 9% to 89% less than their branded equivalents. 7 In 17 poor nations, switching from innovator brands to generic medicines resulted in an average cost reduction of 60%. 7
The World Health Organization (WHO), defined generic medicine as “a pharmaceutical product, usually intended to be interchangeable with an innovator product that is manufactured without a license from the innovator company and marketed after the expiry date of the patent or other exclusive rights”. 8 Moreover, generic medicine is undifferentiated from its corresponding innovator medicine in terms of its therapeutic efficacy, safety, quality, strength, dosage, and dosage form, and has the same intended use as that of the innovator medicine. Although, it is generally acknowledged, that some pharma companies do market their innovator drug product as a generic equivalent, with a different brand name, with the primary intention of maintaining market share, the term “generic” or “generics” will be used in this paper to indicate a medication that is being marketed as a bioequivalent therapeutic agent that has not been developed by the innovator (manufacturer) pharma company.
An appropriate prescription implies the maximization of treatment effectiveness and the minimization of the risks and costs associated with the treatment, all while respecting the patients’ choices. 9 The formative experiences of physicians in medical school and residency are imperative in establishing patterns of prescription. 10 Further, their experience during their practice years further adds to those established patterns. Prescribing decisions of physicians can also be influenced by the policies or programs (direct methods) like formularies, prescribing restrictions and prescription guidelines. And also, by more indirect means affecting the prescribers’ cognition like influences from advertisements, medical sales representatives and continuous learning programs. 11
A substantial body of literature regarding physician prescription behavior and generic drug substitution has been published along different perspectives and various regions across the world. Analyzing these studies is crucial to identify and assess their impact, as well as uncover various themes or patterns associated with them. Bibliometric analysis is one such methodology to evaluate impact and uncover patterns. 12
Methodology
Bibliometrics is a statistical method to carry out a quantitative analysis of the research papers concerned, with respect to one special topic.
13
Bibliometrics was first used in 1969, by Pritchard, and has since gained wide popularity as an aid for quantitative analysis of the literature available, helping in deeper insights and better understanding. The appeal of bibliometric analysis in research is not a passing trend,
14
rather is useful in (1) processing massive amounts of scientific data and (2) creating high research impact
Skillfully conducted bibliometric studies have the potential to establish robust groundwork for advancing a field in novel and impactful ways,
15
as they allow scholars to (1) gain an exhaustive overview (2) delineate knowledge gaps (3) spawn original research questions and (4) communicate their intended contributions to the field
In April 2023, a comprehensive electronic search was carried out to locate the published articles on generic drug prescription by physicians, using Scopus database. The search in question included original articles published in the English language. The search terms were identified based on extensive literature review in the research area. The search term used to identify the matching article publication included “doctor” OR “physician” OR “prescriber” occurring along with “generics” OR “generic medicine” and along with “prescription” OR “drug prescription” including the keywords “perception” OR “attitude”, were used as the keyword search queries in the title, abstract and keywords. The details of the research publications that matched the criteria included the language, year of publication, journal, title, author, co-authors, affiliation, keywords, abstract, and counts of citations, which were converted into CSV format. The retrieval took place on 15th April, 2023. The analysis of the themes included co-authorship, cooccurrence, citation, bibliographic coupling and co-citation, which was done using VOSviewer (version 1.6.10). The two common weight attributes of “Links attribute” and “Total link strength attribute” have been used. The articles were arranged in descending order based on the number of citations, with priority given to more recently published articles in the case of a tie in citation count. Incase more than one affiliation for the first author was encountered, then only the first affiliation of the author was recorded.
Search query –
TITLE-ABS-KEY (( “doctor” OR “physician” OR “prescriber”) AND (“generics” OR “generic medicine”) AND (“prescription" OR “drug prescription”) AND (“perception” OR “attitude”) )
Based the search query mentioned above, a total of 460 publications in Scopus database were idenfied. These publications were dated between 1968 and 2023, with a majority of the research occurring during the years 2002-2022. We have refined the query further to get the required publication data for a deeper and much focused analysis. Below flow diagram explains the process followed to get the final results. The top 60 cited articles, according to the number of citations in descending order, have been listed in Annexure 1.
Final/refined search query –
TITLE-ABS-KEY (( “doctor” OR “physician" OR “prescriber”) AND (“generics” OR “generic medicine”) AND (“prescription” OR “drug prescription”) AND (“perception” OR “attitude”) ) AND (EXCLUDE ( LANGUAGE, “French”) OR EXCLUDE (LANGUAGE, “Spanish”) OR EXCLUDE (LANGUAGE, “German”) OR EXCLUDE (LANGUAGE, “Dutch”) OR EXCLUDE (LANGUAGE, “Italian”) OR EXCLUDE (LANGUAGE, “Norwegian”) OR EXCLUDE (LANGUAGE, “Portuguese”) OR EXCLUDE (LANGUAGE, “Swedish”) OR EXCLUDE (LANGUAGE, “Croatian”) OR EXCLUDE (LANGUAGE, “Korean”) OR EXCLUDE (LANGUAGE, “Slovak”) ) AND (LIMIT-TO ( PUBYEAR, 2022) OR LIMIT-TO (PUBYEAR, 2021) OR LIMIT-TO (PUBYEAR, 2020) OR LIMIT-TO (PUBYEAR, 2019) OR LIMIT-TO (PUBYEAR, 2018) OR LIMIT-TO (PUBYEAR, 2017) OR LIMIT-TO (PUBYEAR, 2016) OR LIMIT-TO (PUBYEAR, 2015) OR LIMIT-TO (PUBYEAR, 2014) OR LIMIT-TO (PUBYEAR, 2013) OR LIMIT-TO (PUBYEAR, 2012) ) AND (LIMIT-TO ( PUBSTAGE, “final”) ) AND (LIMIT-TO ( DOCTYPE, “ar”) ) AND (EXCLUDE ( SUBJAREA, “ENGI”) )
Results
Bibliometric analysis of publication output
The top cited article, among the articles on generic drug prescription by physicians, was found to be cited 84 times. The top four cited article titles were – “Contact between doctors and the pharmaceutical industry, their perceptions, and the effects on prescribing habits” (citations - 84), “Reforms and initiatives in Scotland in recent years to encourage the prescribing of generic drugs, their influence and implications for other countries” (citations - 74), “Association between academic medical center pharmaceutical detailing policies and physician prescribing” (citations - 61) and “Underuse of generic medicines in Portugal: An empirical study on the perceptions and attitudes of patients and pharmacists” (citations - 61).
The mean and median for the selected articles were also calculated and were found to be 13.10 and 6, respectively. In the top-cited list of articles, 27 articles had a citation count of 25 or greater.
Bibliometric analysis of the keywords
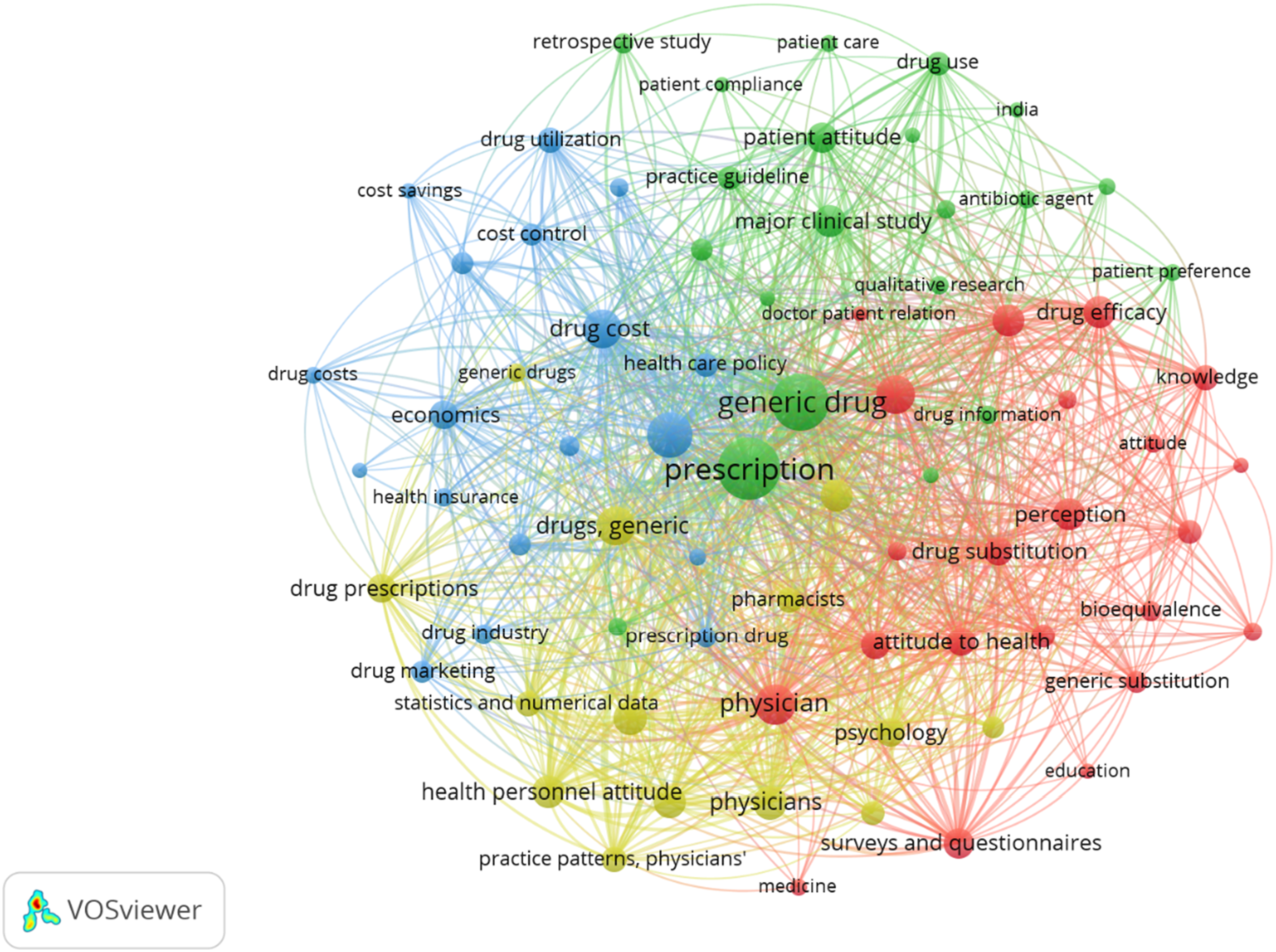
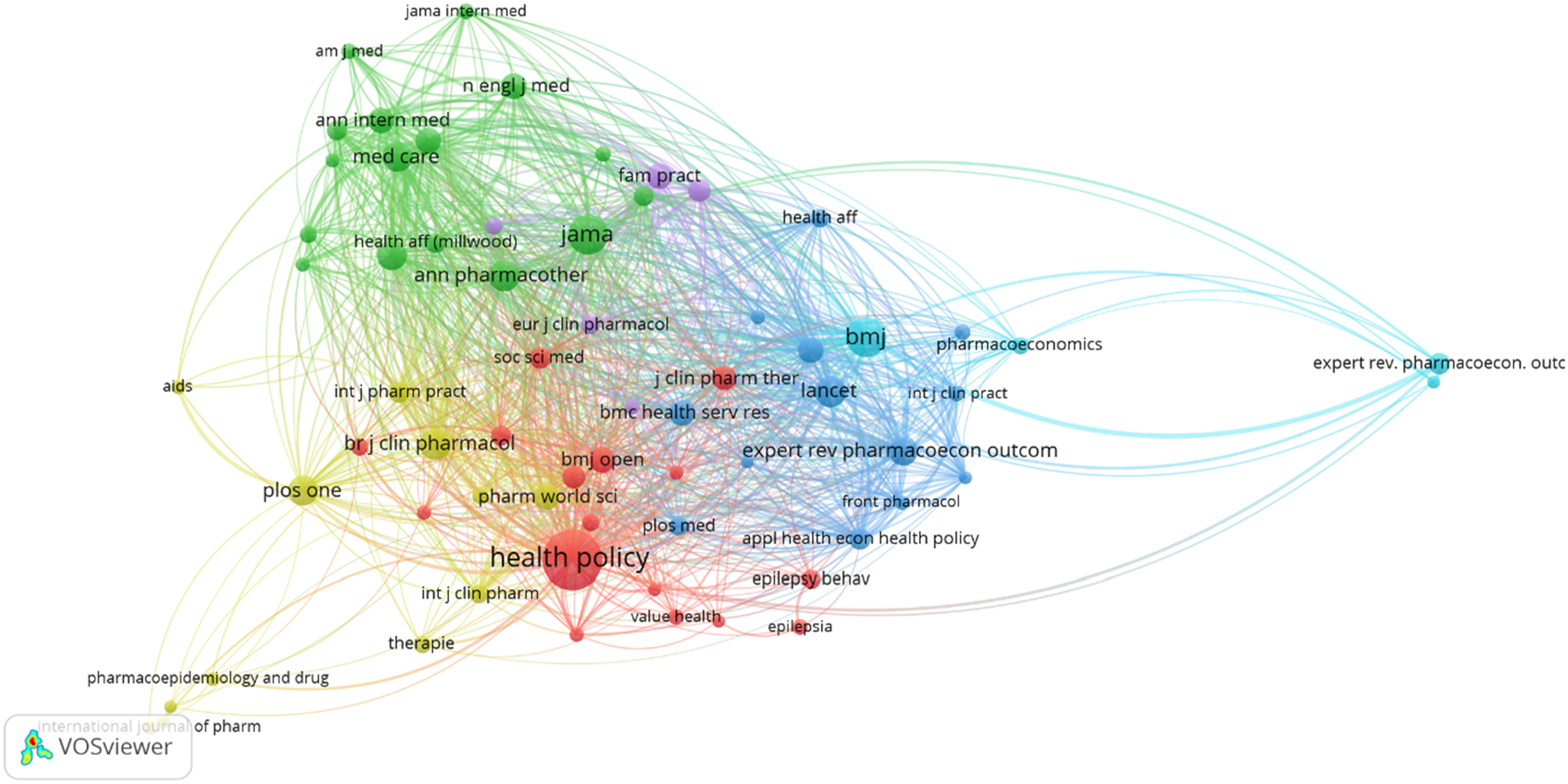
Keywords that occurred for more than 10 times in the Scopus database were empaneled in the final analysis. of the 1852 keywords, 95 of them met the threshold. Few keywords that were not indicative of the research area were removed, such as human, male, female, questionnaire, etc. the keywords that appeared most were “prescription” (occurrence - 151, total link strength - 2644), “generic drug” (occurrence – 126, total link strength - 2374), and “physician attitude” (occurrences – 79, total link strength - 1423) which are strongly connected.
The identified keywords were grouped in four clusters with 75 items in total, including 2370 links with total link strength of 12746. below is the network map or network visualization map (Figure 1) and the density map (Figure 2) of the identified keywords Flow diagram of the Number of Publications of the identified research area. Network map of all the identified keywords.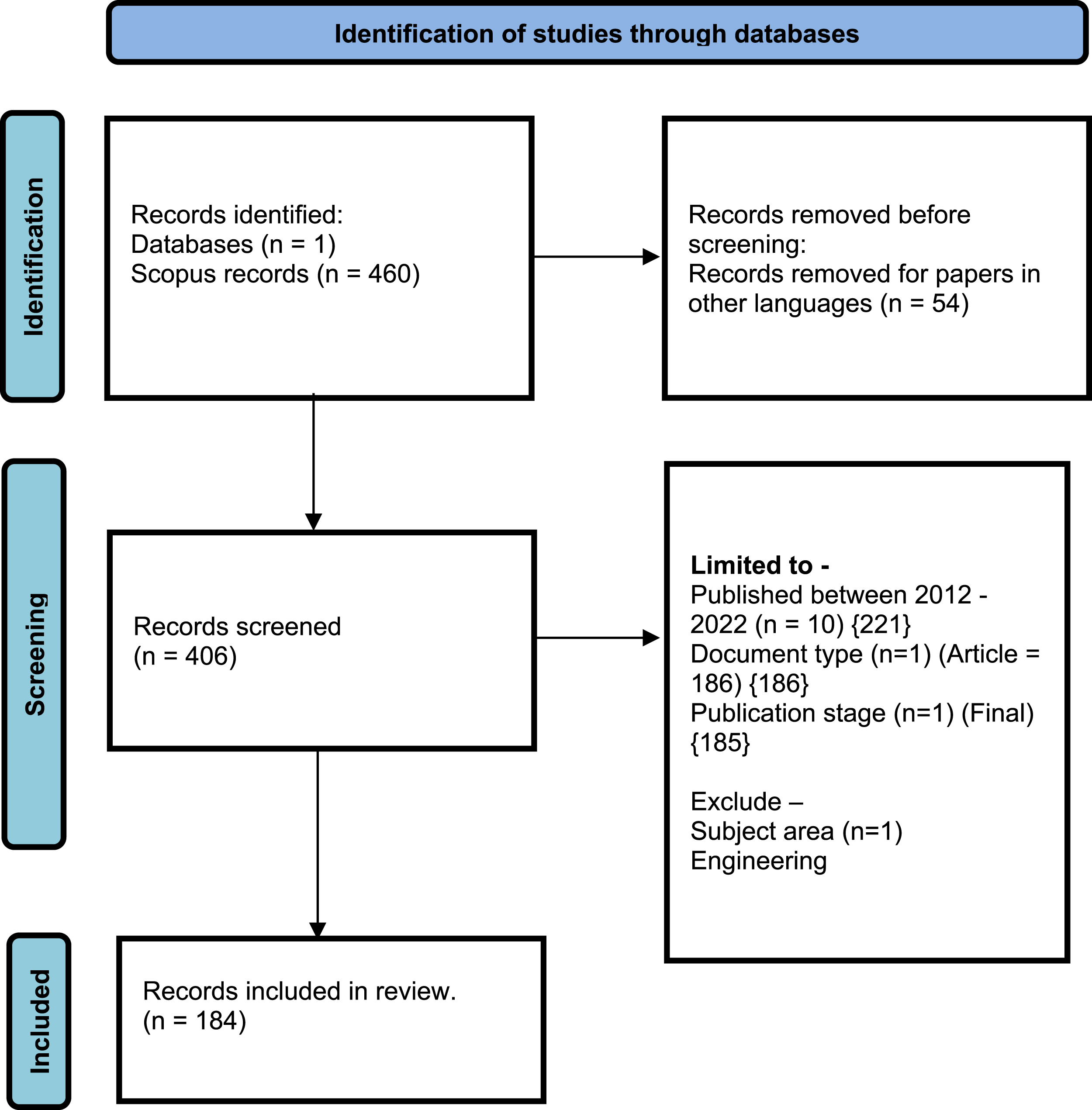
Upon analysis of the identified clusters, it was found that there is a very strong relationship in two clusters, with 126 occurrences, 74 links and total link strength 1465. The density map and network map of the same are presented in Figure 3 and Figure 4 respectively. There is strong relationship between the keywords generics and prescription, and their related factors such as drug costs, physician, drug substitution, cost savings and patient attitude. Density map of all the identified keywords. Network map of the two identified clusters.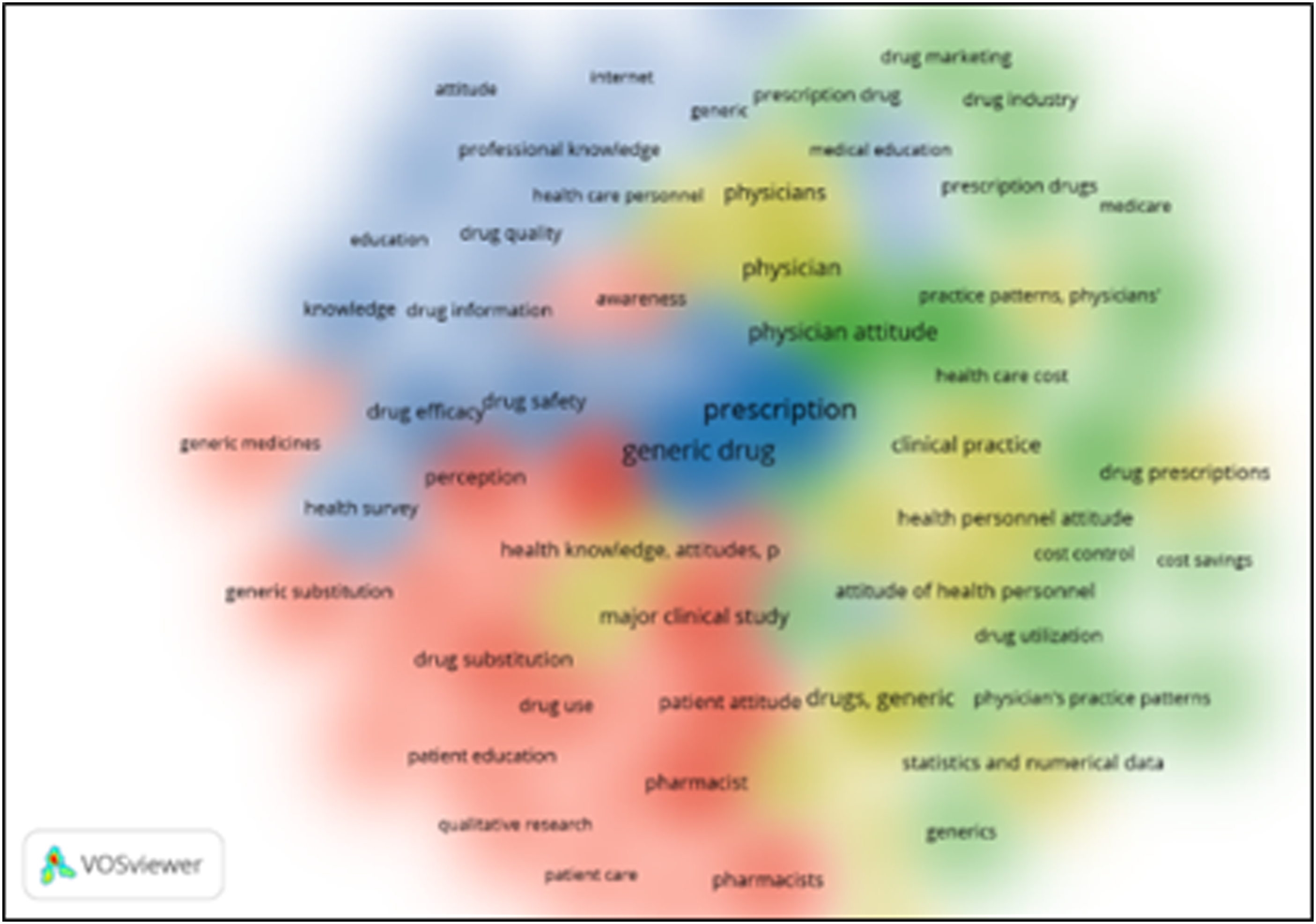
Three other clusters analyzed (Figure 5) also exhibit strong relationship (occurrences 58, links 62, total link strength 637) Network map of all the three identified clusters.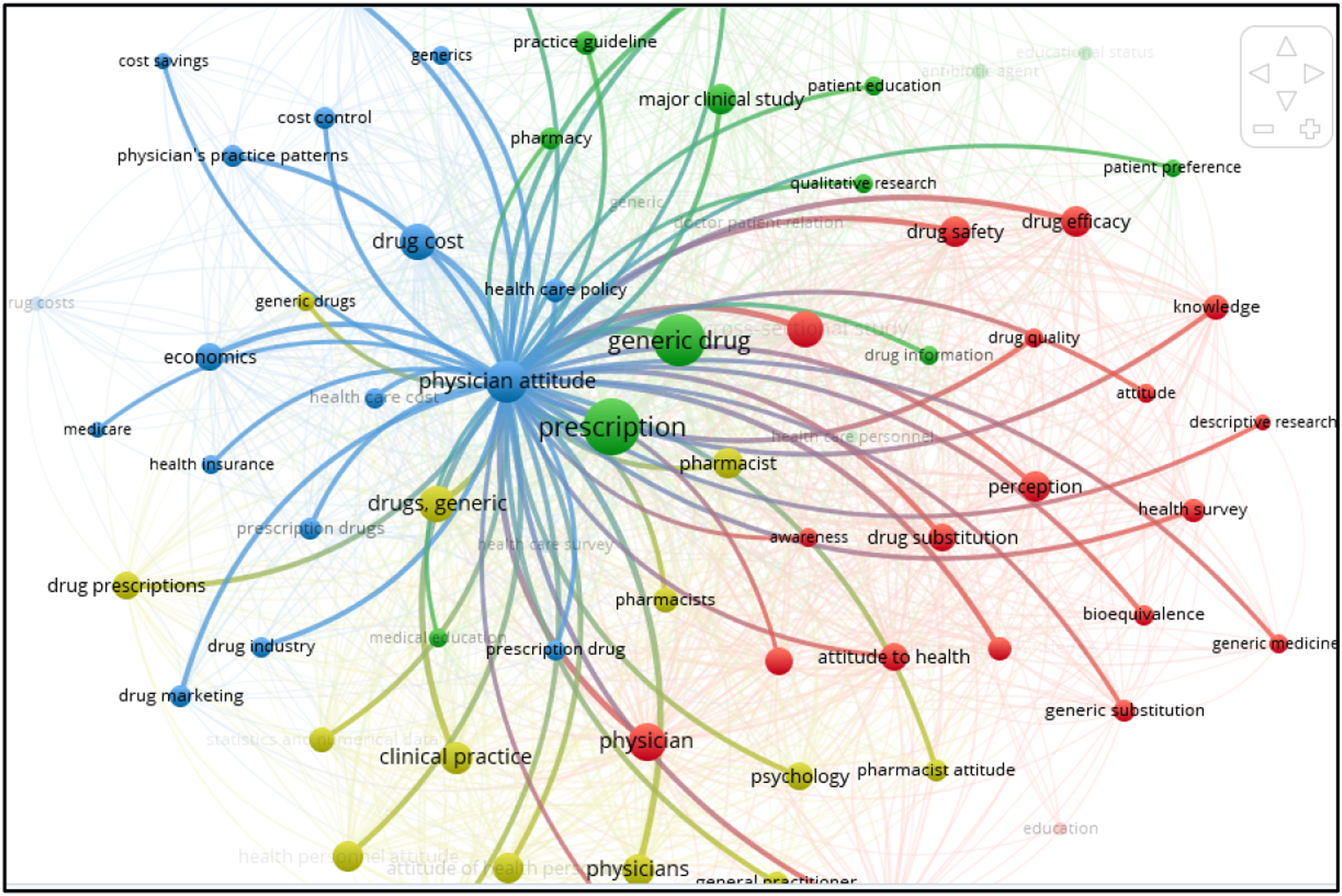
Analysis of one of the clusters (occurrences 39, links 72, total link strength 463) reveals that the perception of generic drugs is closely related to the prescription, awareness, doctor-patient relation, drug cost, health policy and patient preference (Figure 6). Network map of analyzed cluster 1.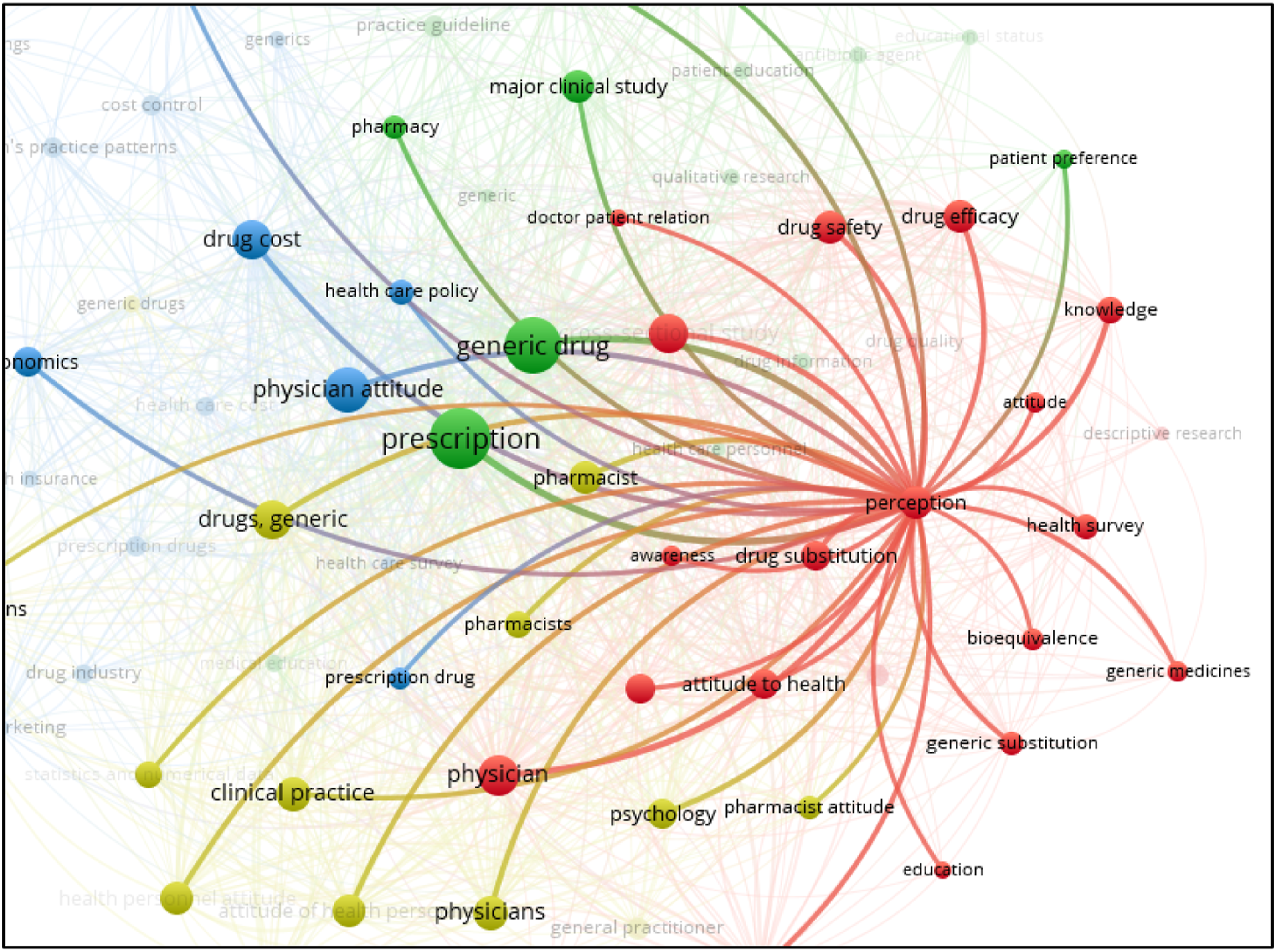
Furthermore, analysis of the cluster (2 clusters, occurrences 11, links 55, total link strength 173) reveals that the patient perception of generic drugs is heavily influenced by perception, physician attitude, patient attitude, drug substitution, and drug efficacy (Figure 7). Network map of analyzed cluster 2.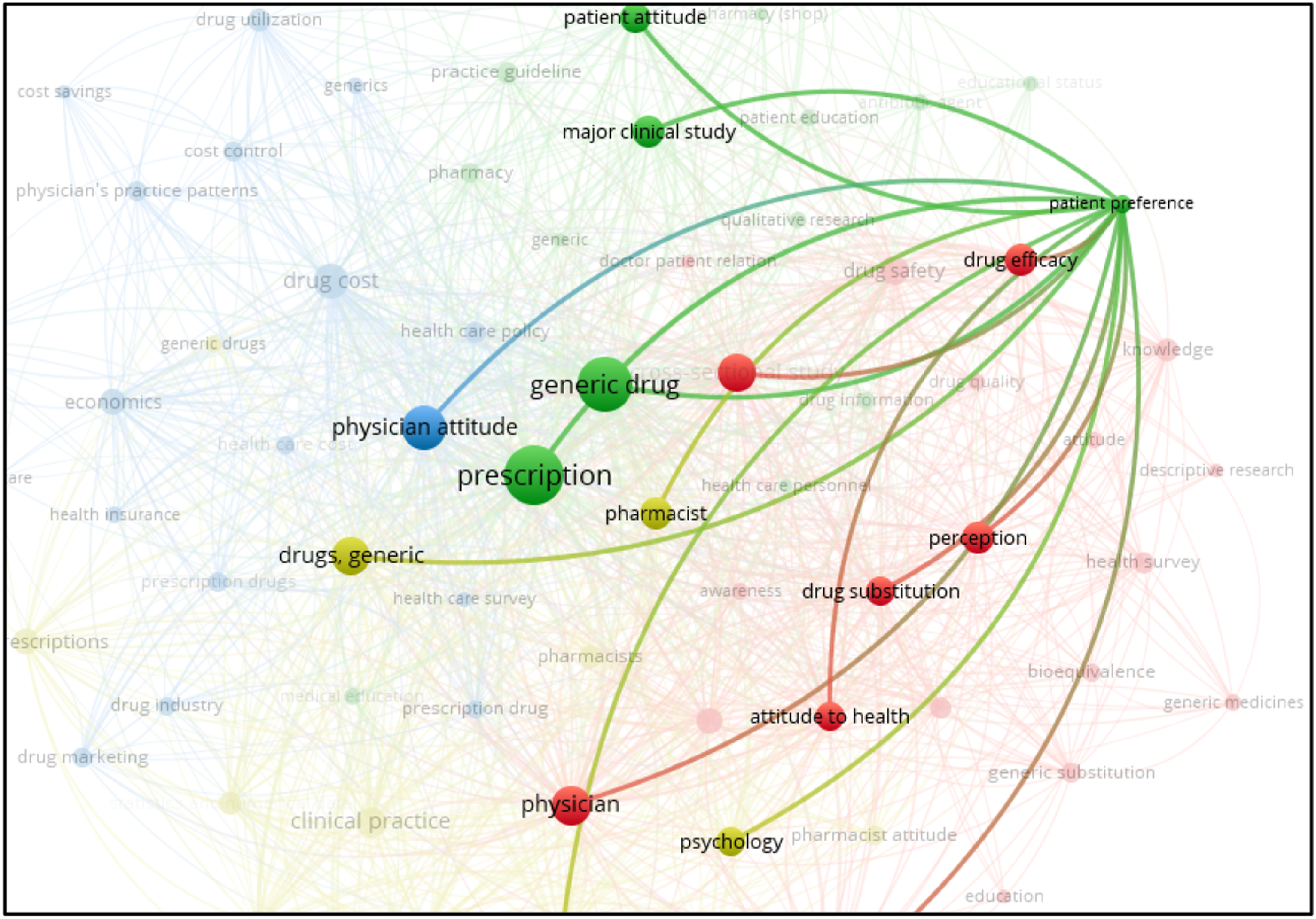
Bibliometric analysis of the citations and publications
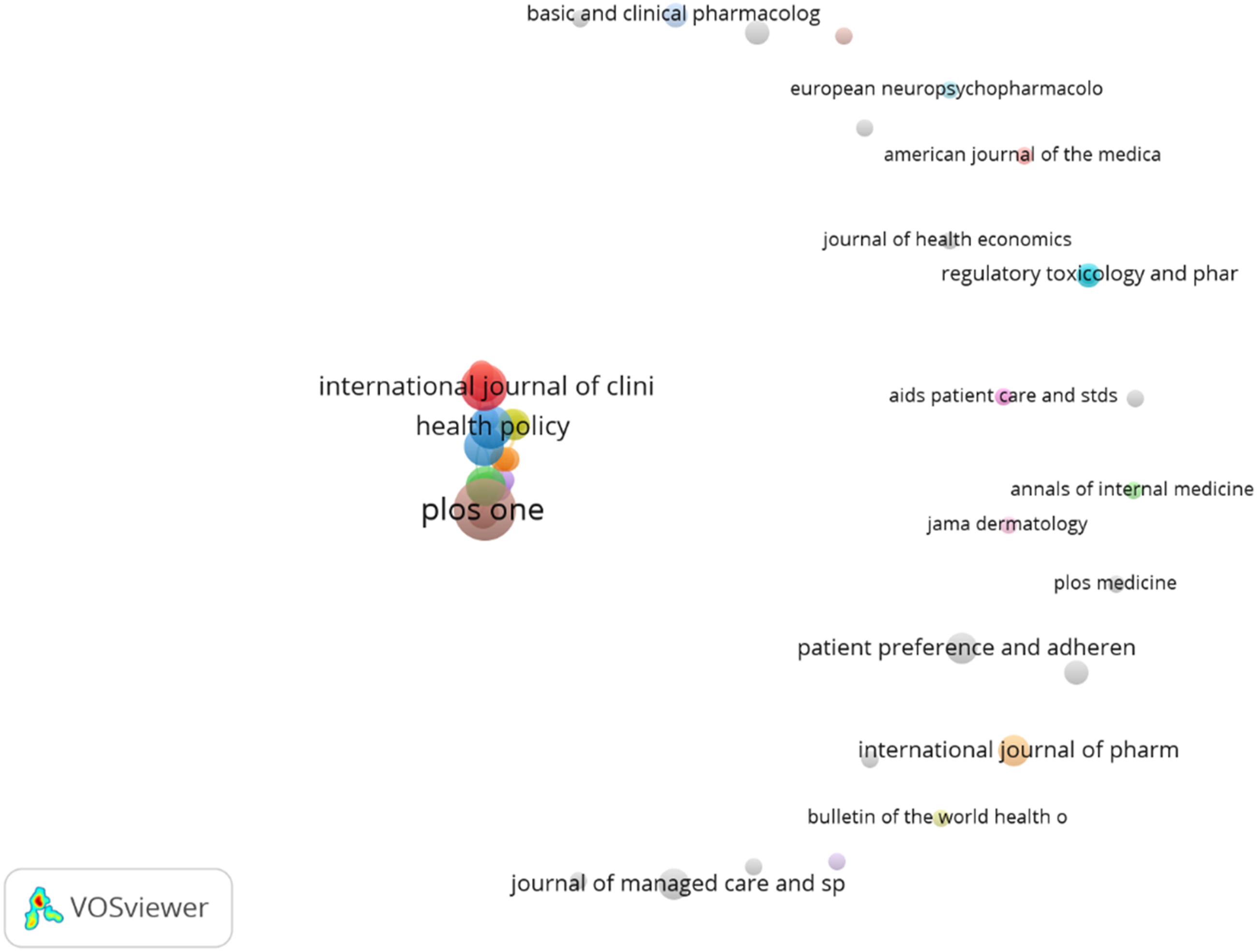
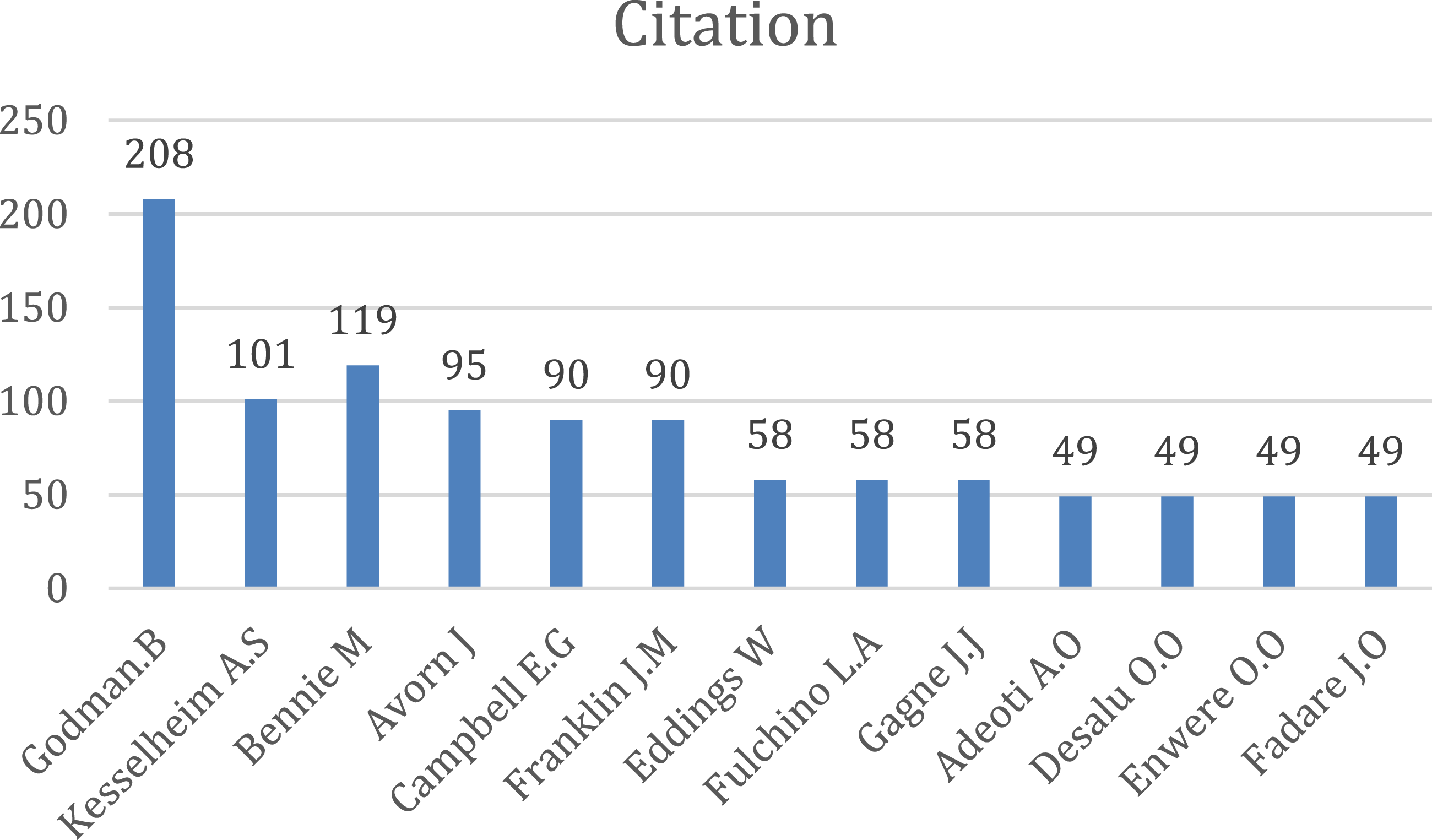
Citation of sources with a minimum of 10 citations of a source were analyzed. Figures 8–15 Out of the 113 sources, 45 met the specified threshold criteria. Upon further analysis, we have identified a cluster of the cited sources. Bar graph of source citations. Network map of sources. Density map of sources. Network map of identified sources cluster. Network map of identified authors. Network map of identified authors’ cluster. Density map of identified authors’ cluster. Network map of countries of origin of the publications.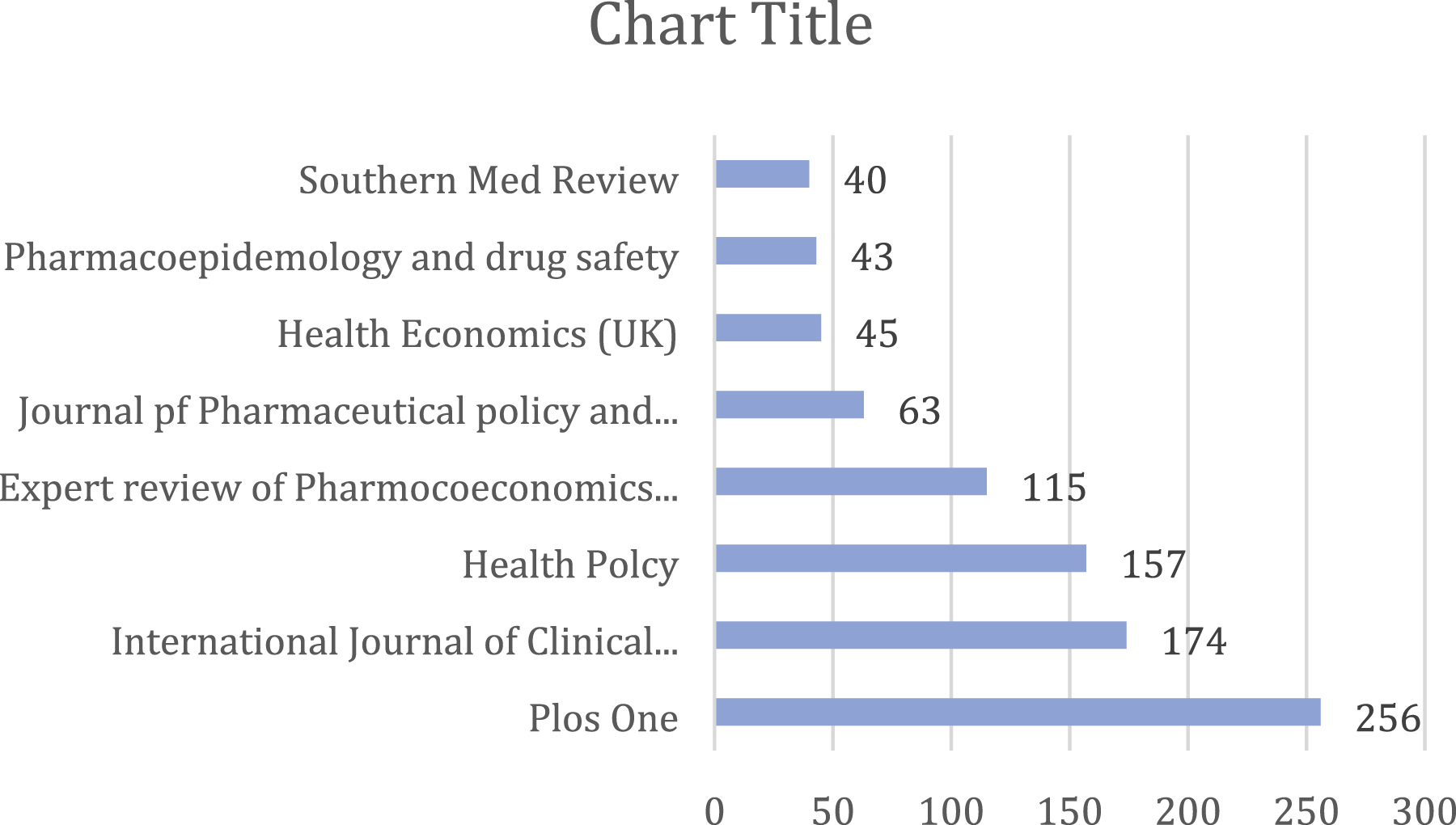

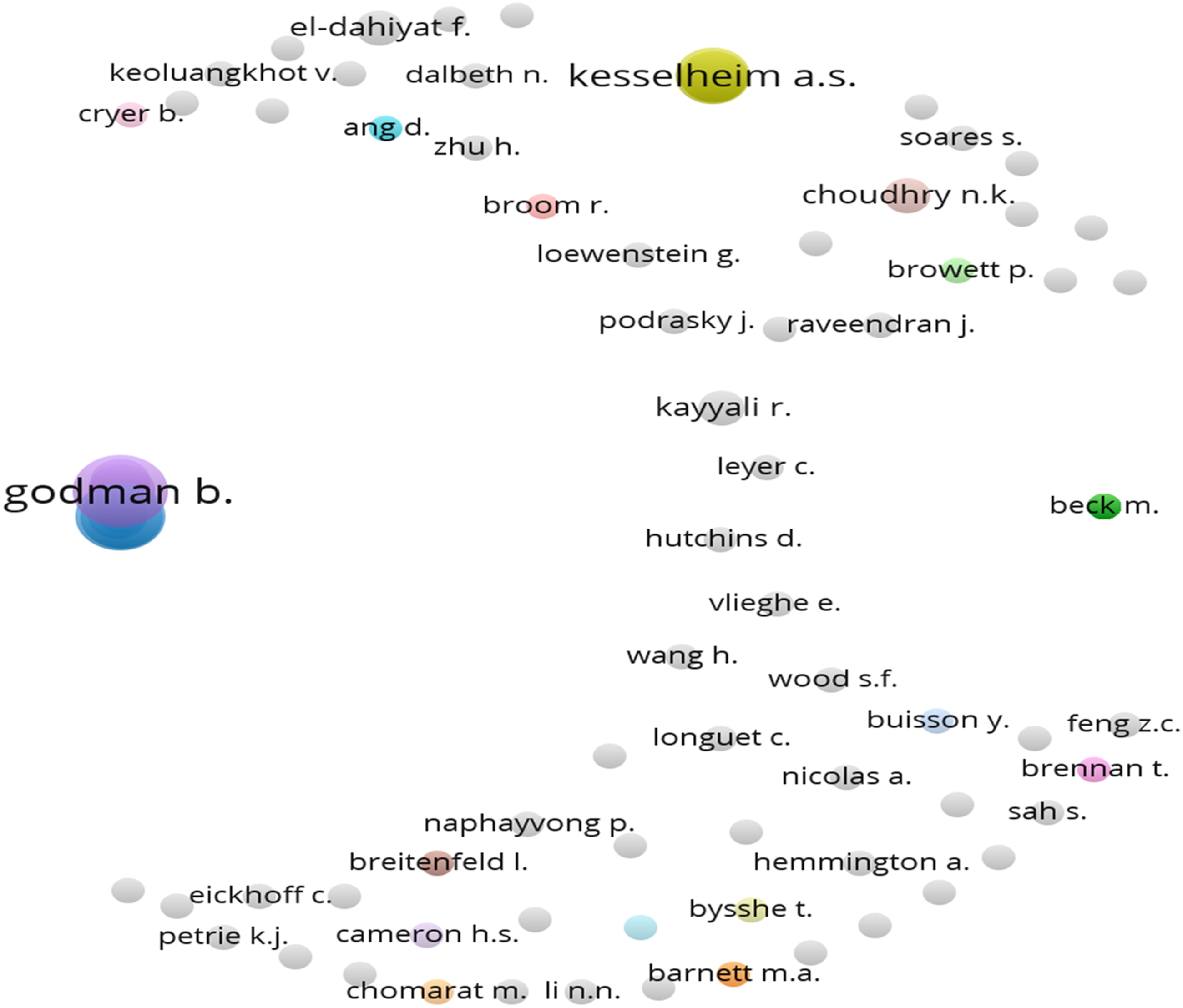

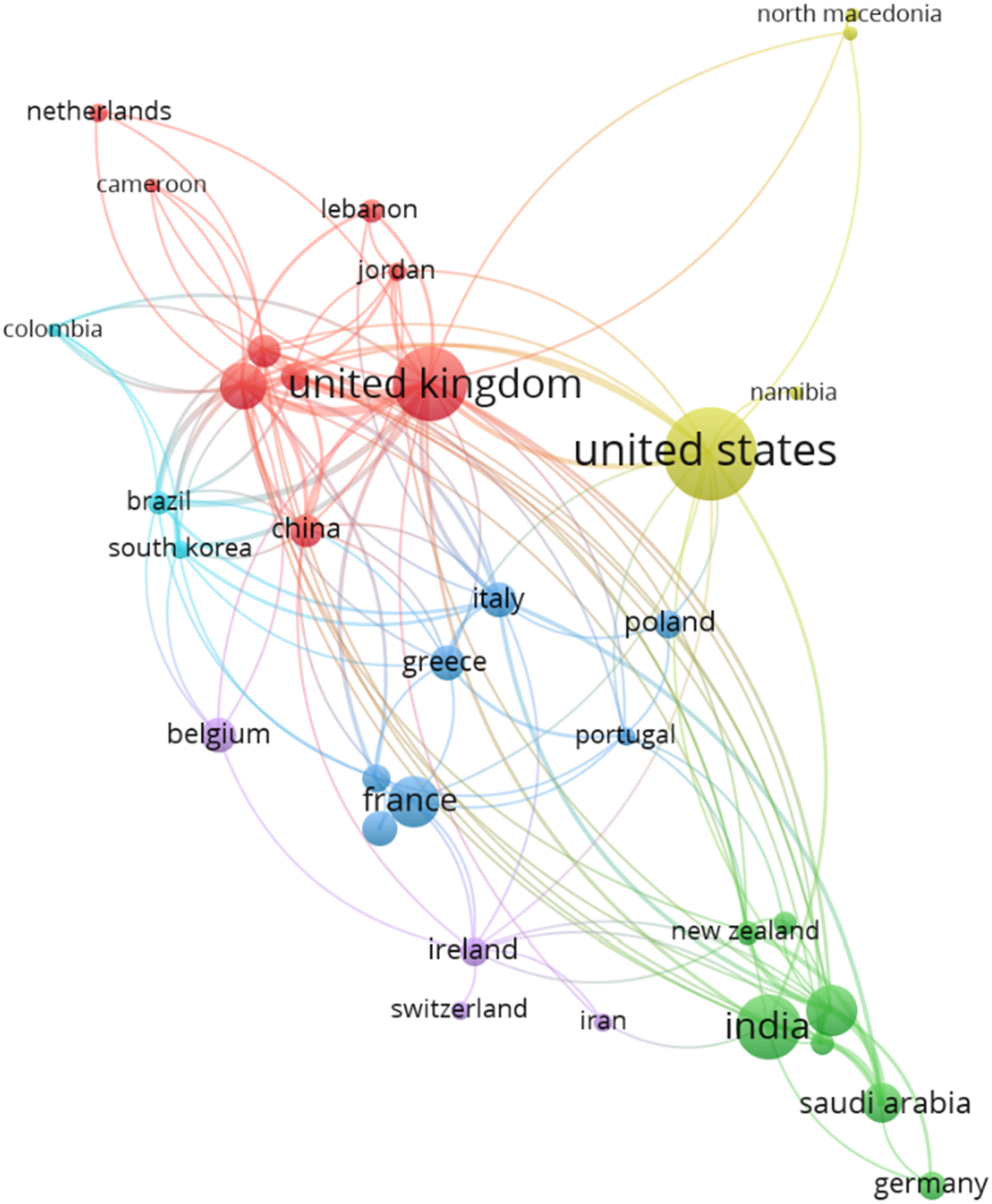
Number of citations per author was studied. For this, we had considered the minimum number of citations of an author to be 35. Out of the 834 authors, only 131 met the threshold criteria specified.
Country and institute of origin
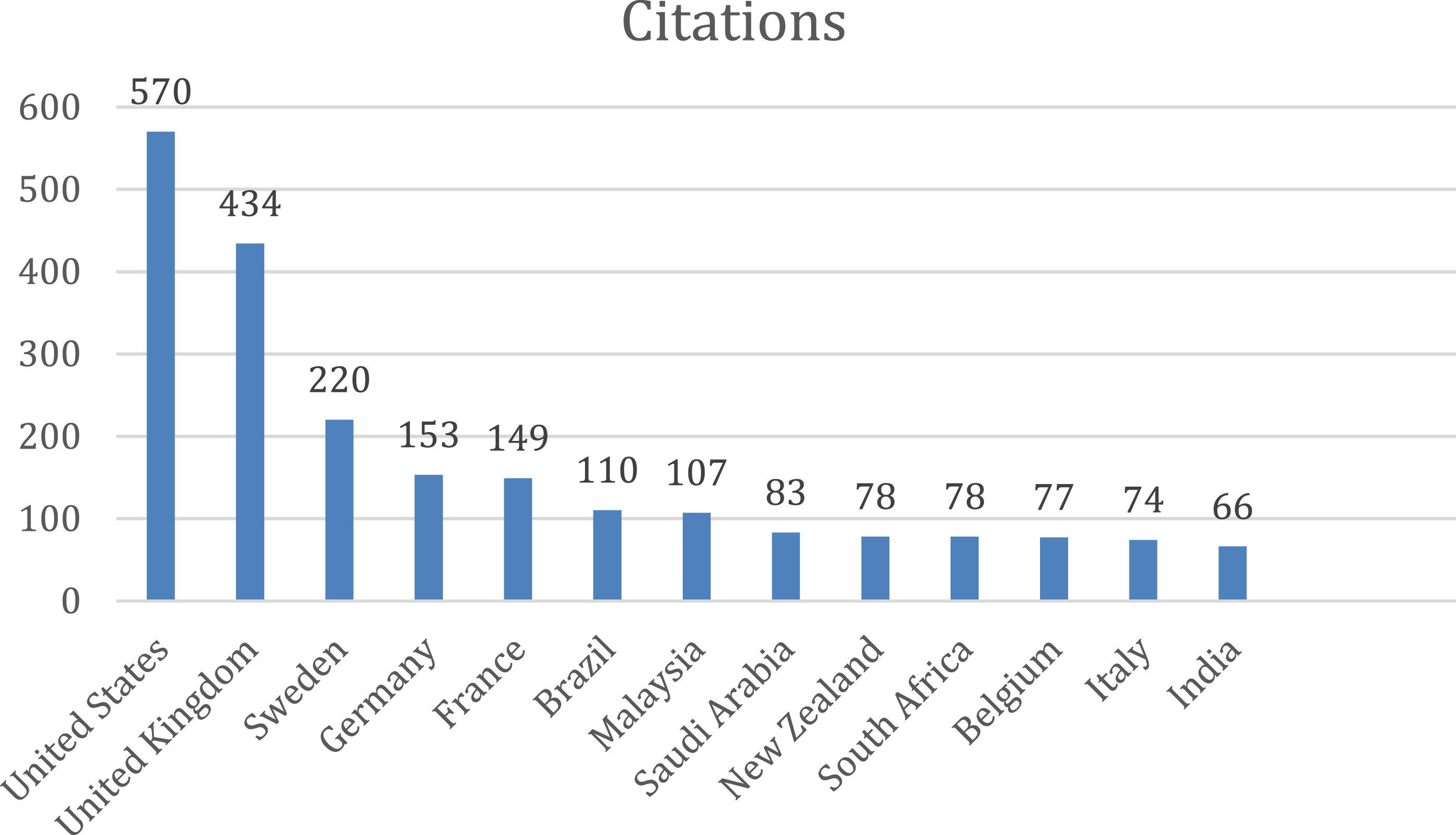
The top-cited articles (with citations of a country more than 10) originated from 39 countries of the total 63 countries. Most of the publications were found to be from the United States of America (document 42, citations 570, total link strength 11), followed by United Kingdom (document 26, citations 434, total link strength 41) and by Sweden (document 11, citations 220, total link strength 23). The below bar chart shows the country wise publications (Figure 16) and citations (Figure 17). Bar graph of top countries in terms of publications. Bar graph of top countries in terms of citations.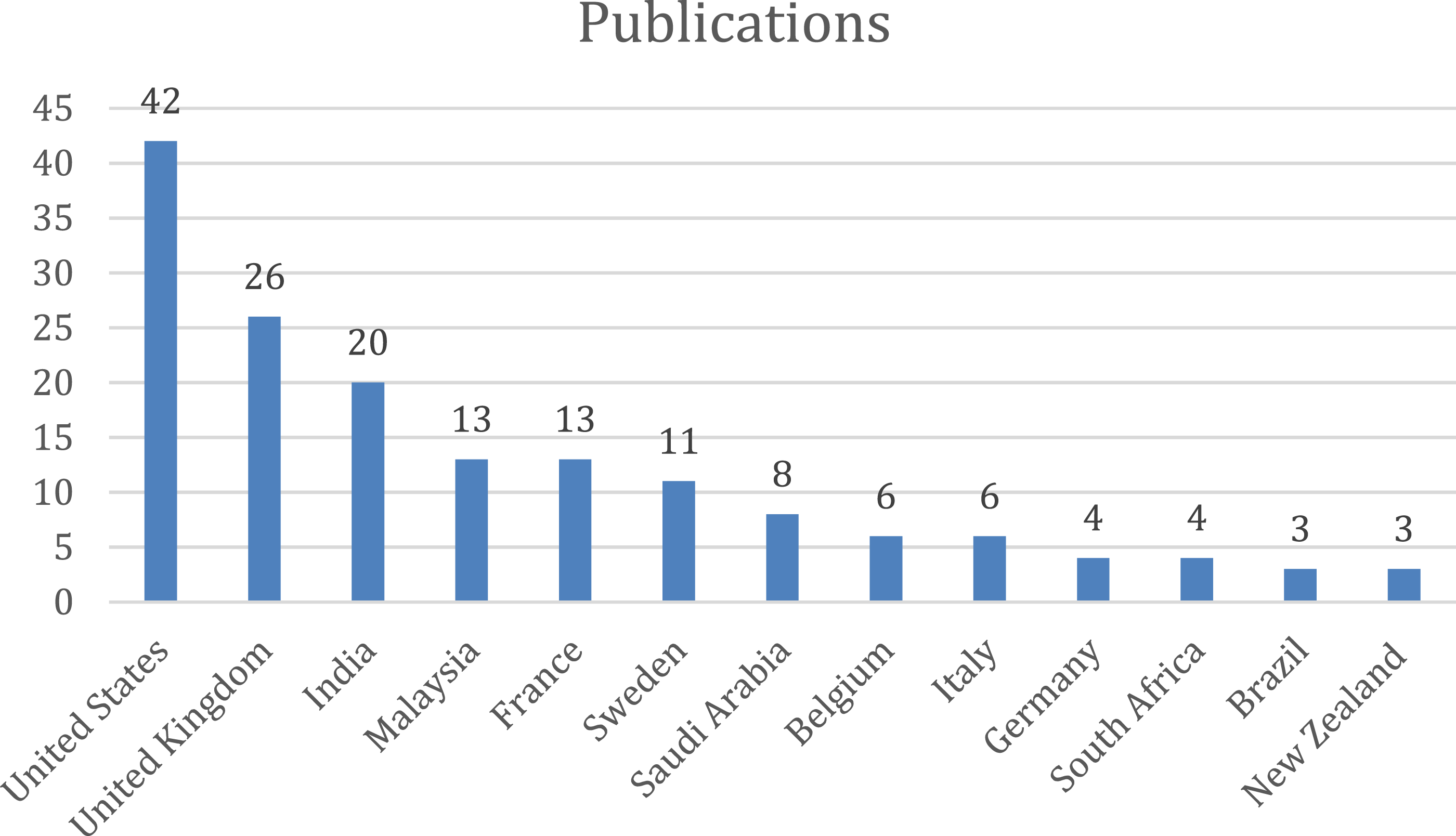
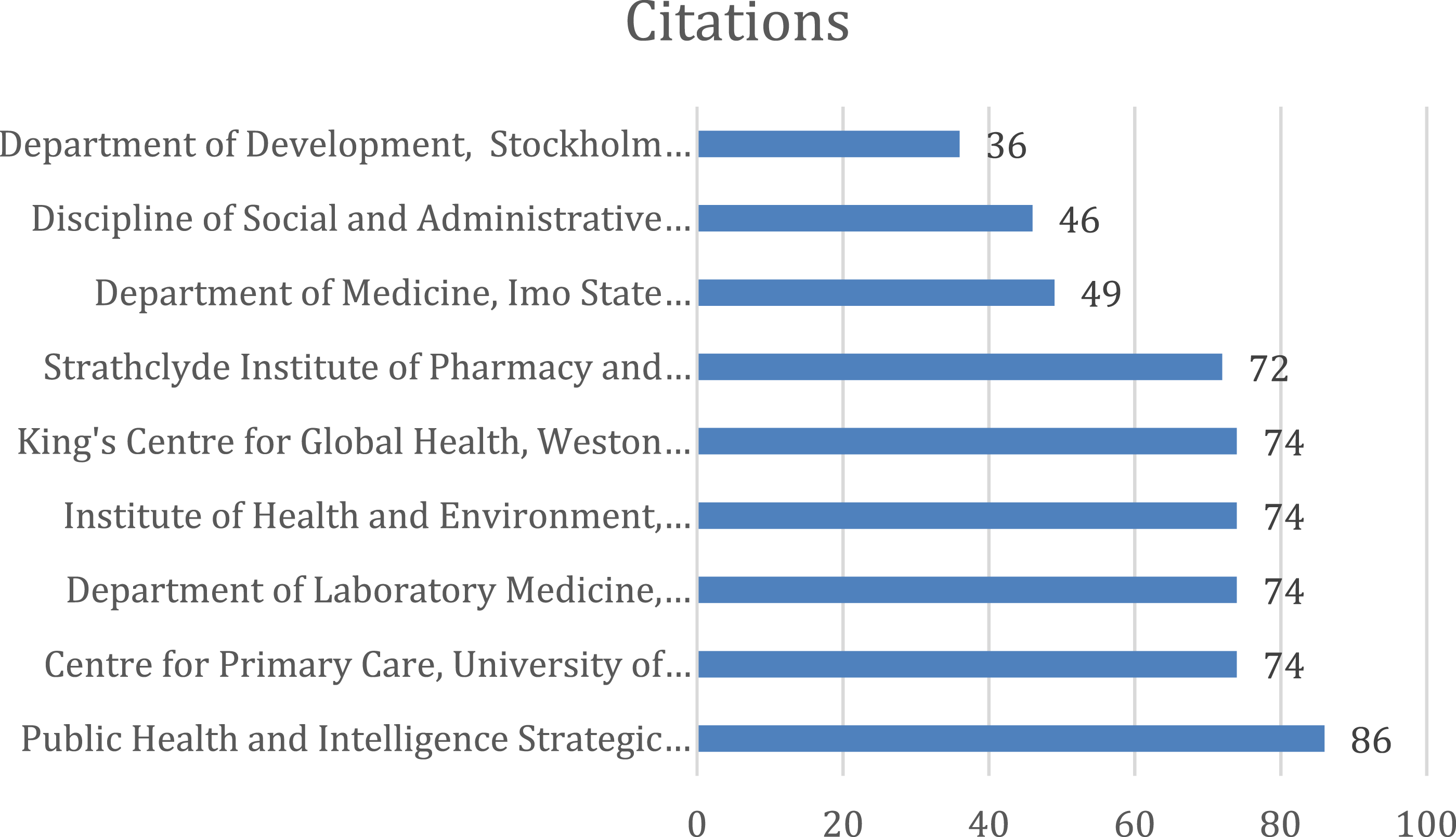
The leading institutes contributing to the top 100 cited articles appear in Figure 18. The minimum number of citations from an organization was set to 30 and 104 met the criteria (total of 602). Strathclyde Institute of Pharmacy and Biomedical Sciences, University of Strathclyde produced the highest number of publications (n = 5) followed by Discipline of Social and Administrative Pharmacy, Universiti Sains Malaysia (n = 3). Figures 19–25 Department of Medicine, Imo State University, Department of Development, Stockholm County Council, and Public Health and Intelligence Strategic Business Unit, NHS National Services Scotland are also among the prominent contributors (n = 2). Network map of Institutes/organizations of origin of the publications. Network map of Institutes/organizations cluster of origin of the publications. Network map of Institutes/organizations cluster of origin of the publications. Bar graph of publications of Institutes/organizations. Bar graph of citations of Institutes/organizations. Network map of Institutes/organizations cluster of origin of the publications. Bar graph of coauthored publications. Bar graph of coauthored publication citations.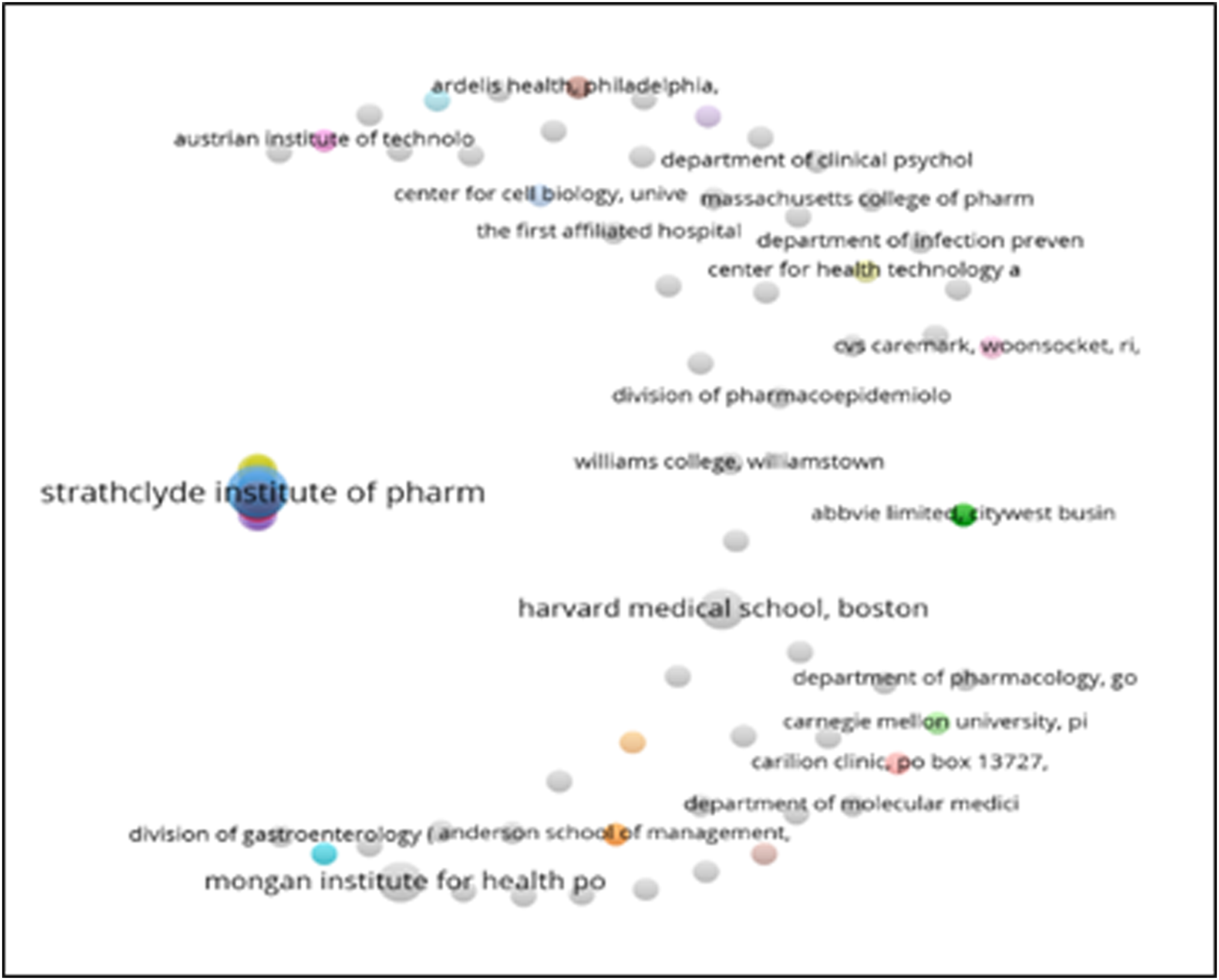

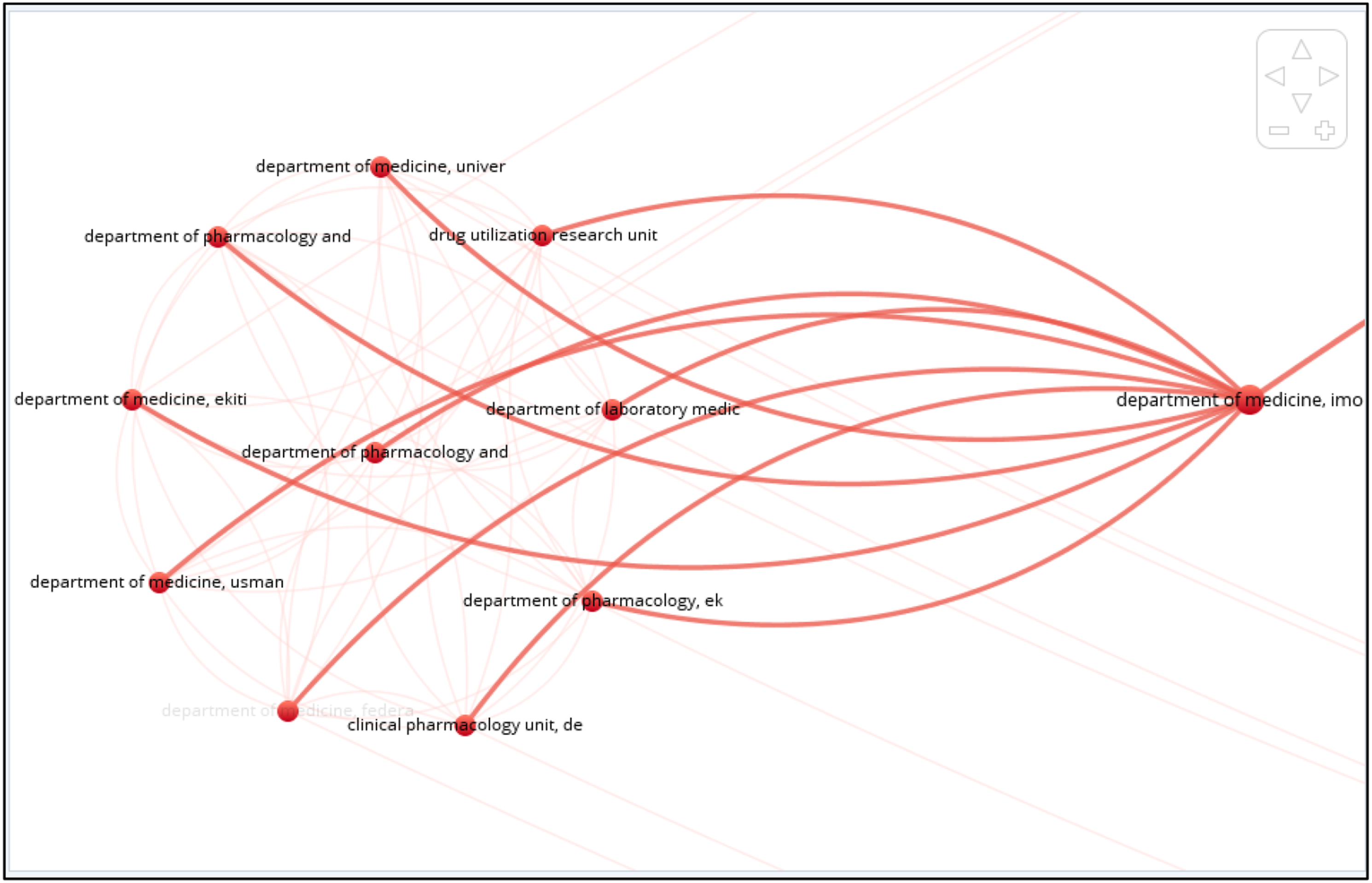

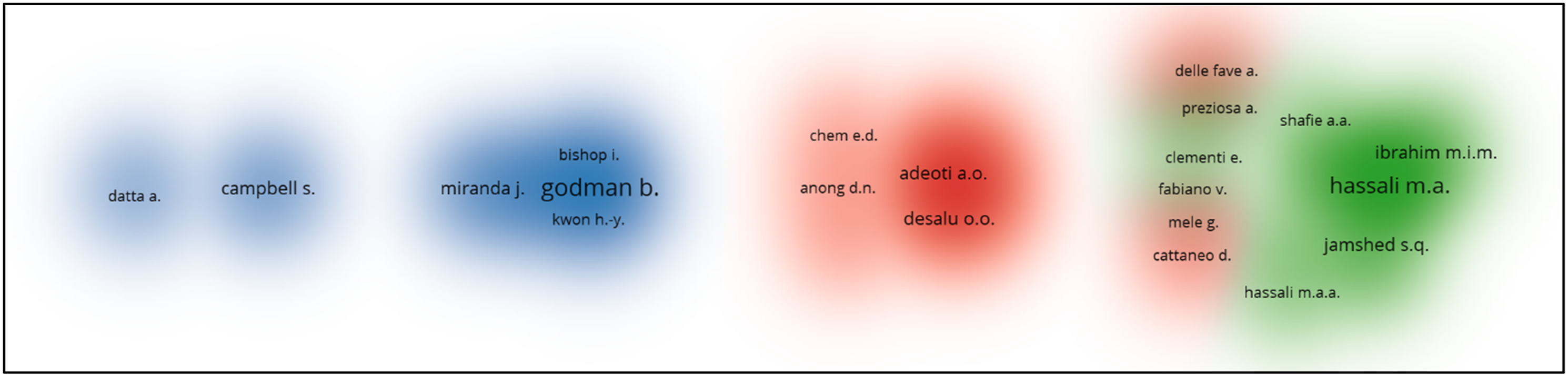
Bibliometric analysis of the co-authorship
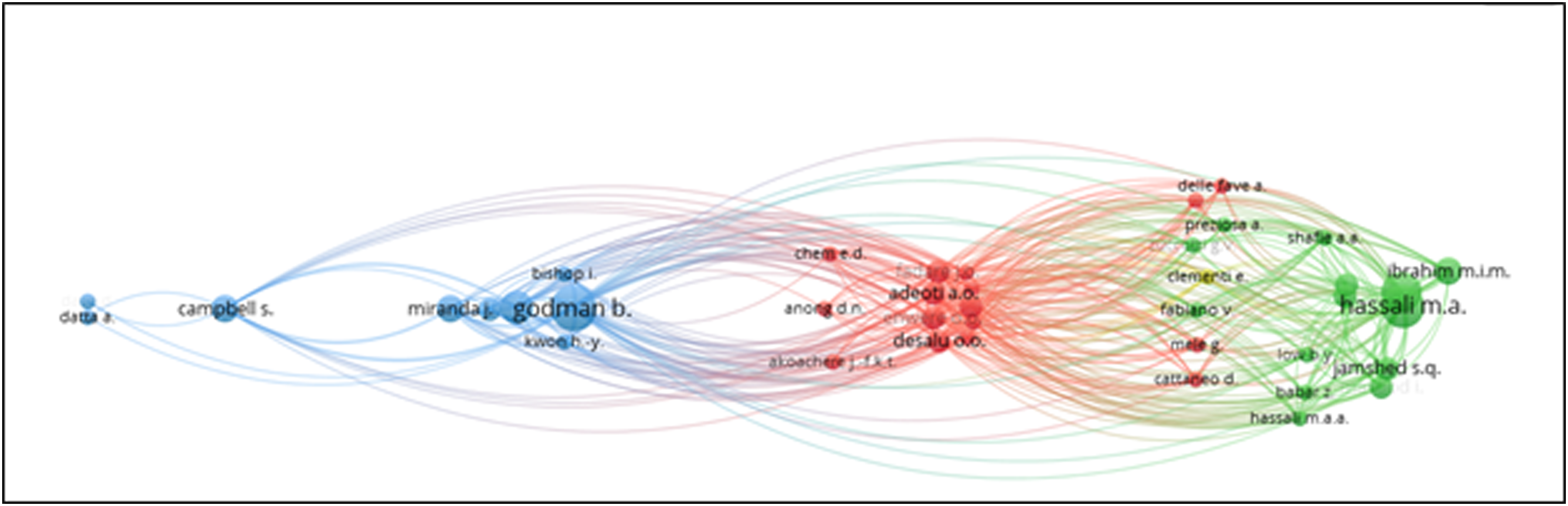
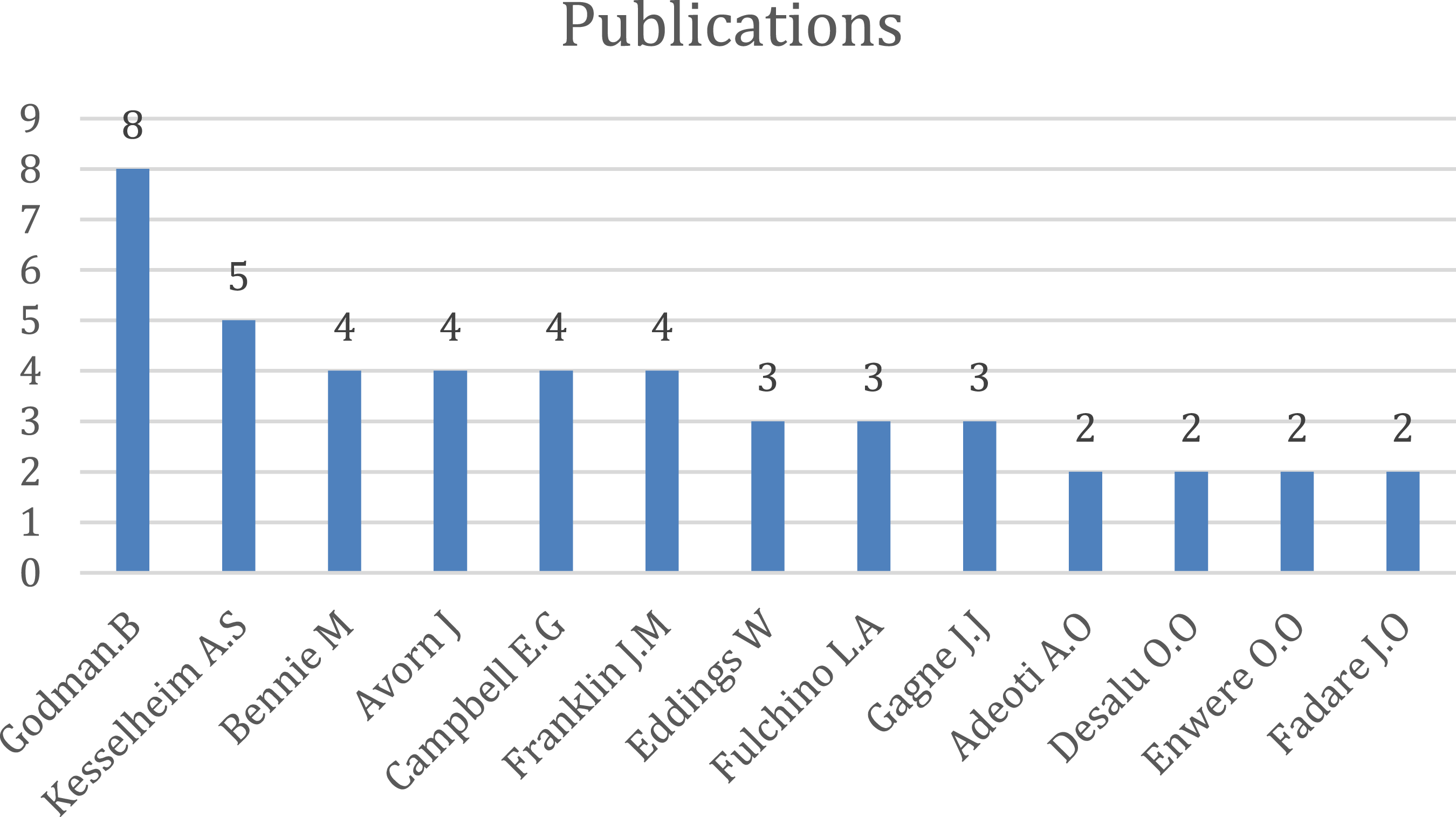
Among the authors, Godman B. from Division of Clinical Pharmacology, Department of Laboratory Medicine, Karolinska Institute, Karolinska University Hospital Huddinge, Stockholm has eight papers which mainly focus on the impact of generic drugs and the reforms necessary. The main collaborators with him are Fadare J.O., Adeoti A.O., and Desalu O.O.
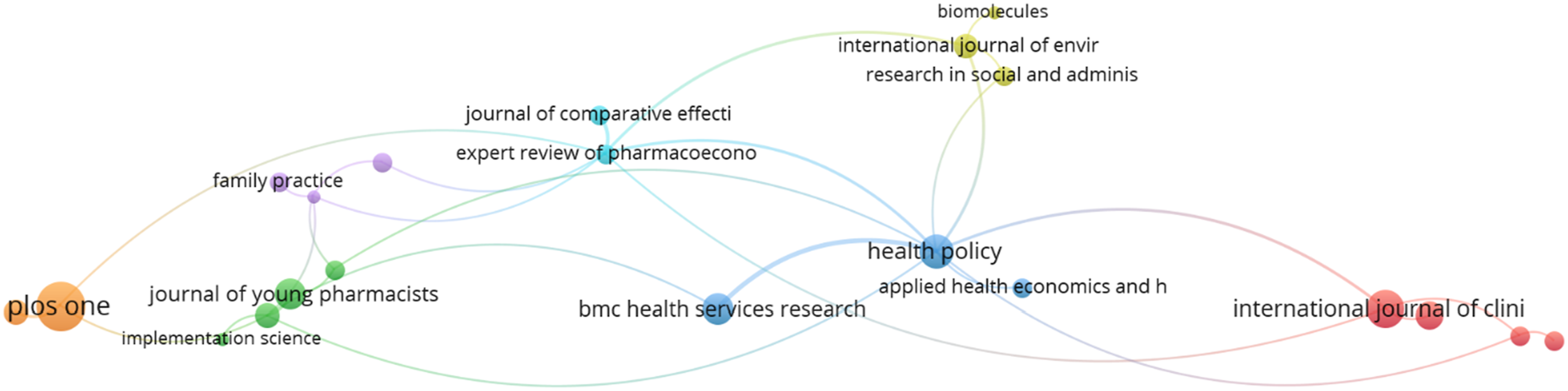
Bibliometric analysis of the bibliographic coupling and co-citation
The bibliographic coupling map and co-citation map of sources have been shown below. Figures 26–30 Three distinct clusters of the cited references have been obtained by bibliometric analysis. The top three clusters represent the research fields of clinical feature, health policy and Pharmacology. Bibliographic coupling Network map of sources. Bibliographic coupling Density map of sources. Co-citation network map of sources. Co-citation density map of sources. Bar graph of co-citation of sources.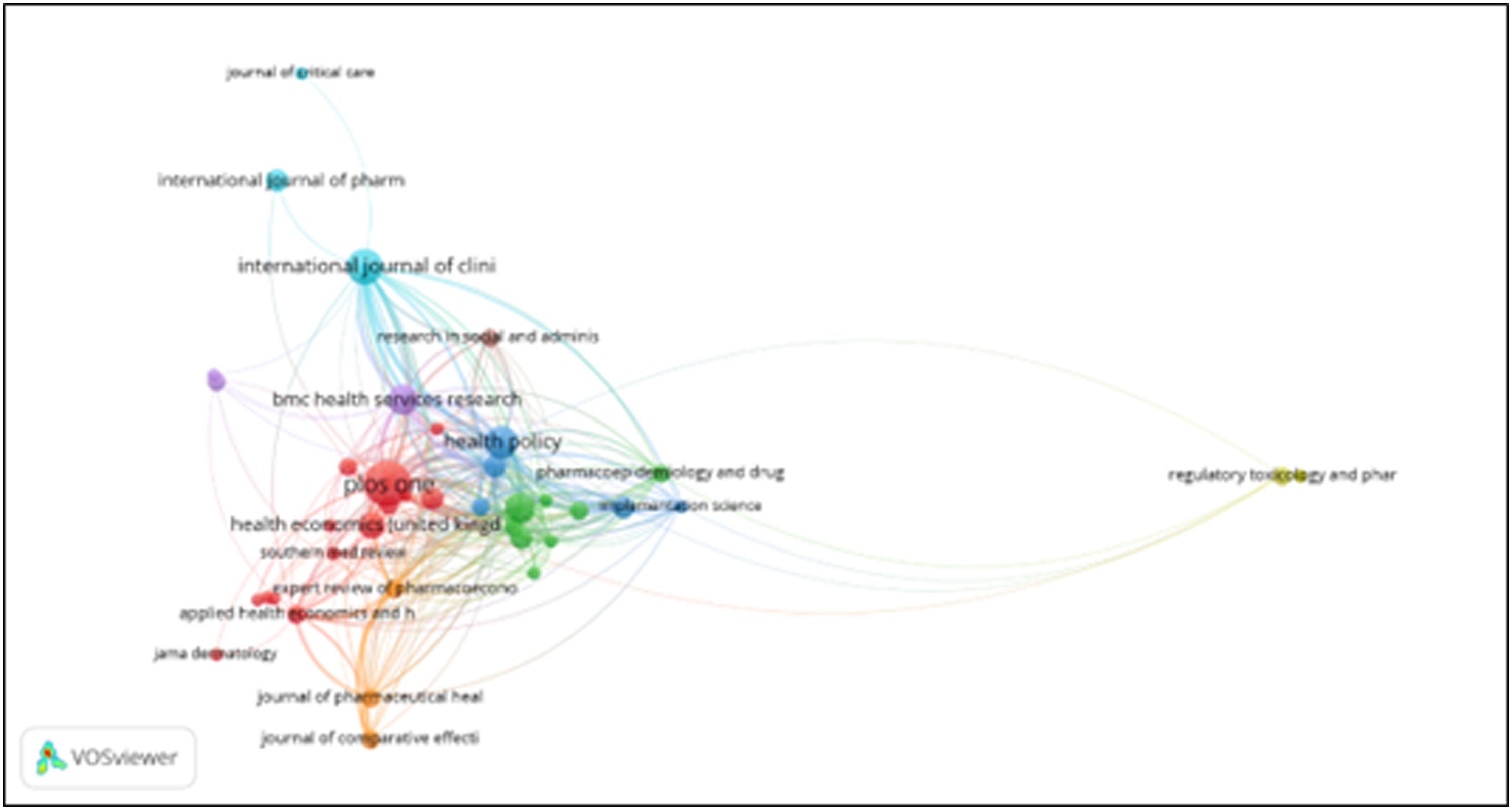
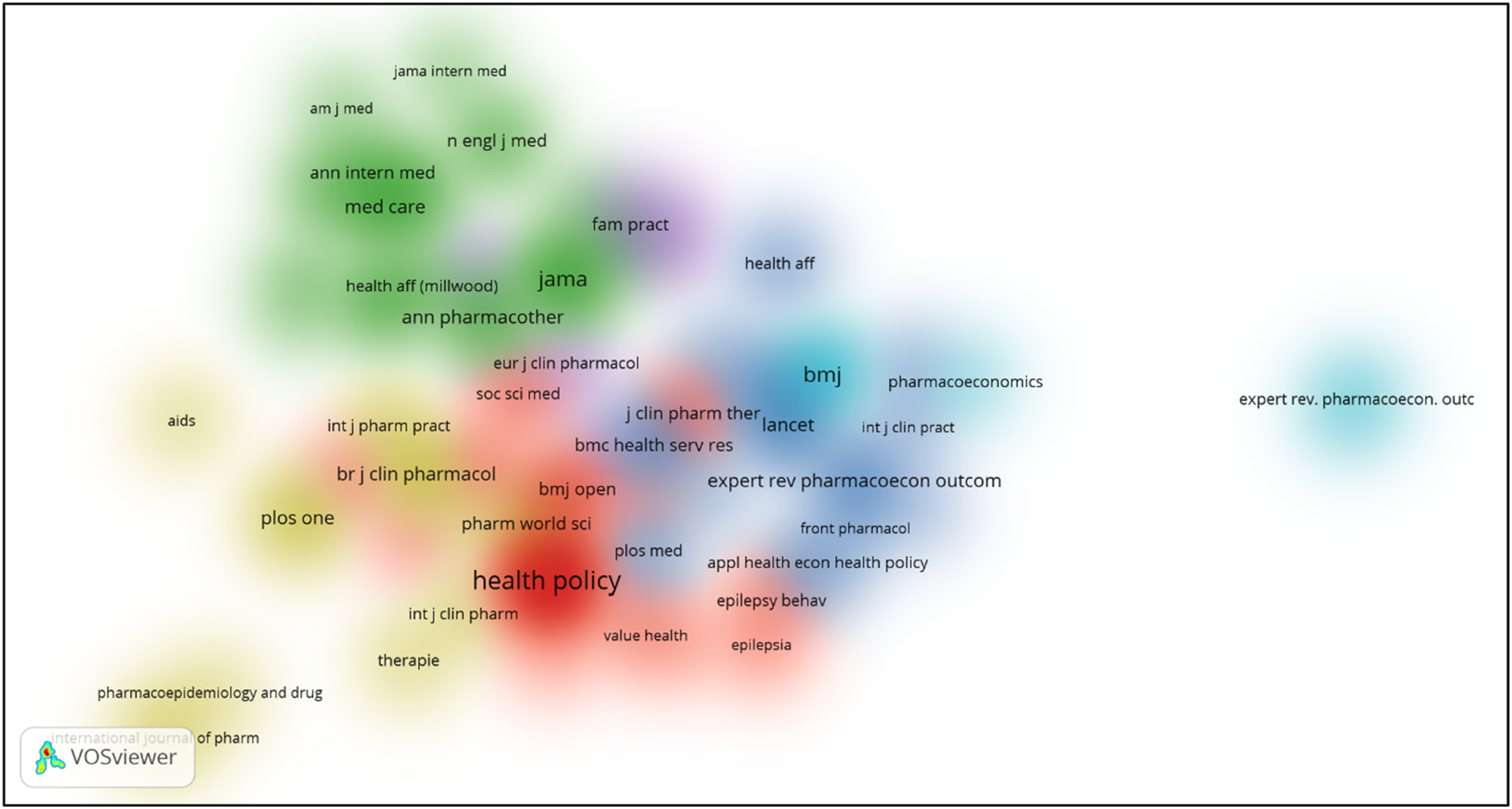
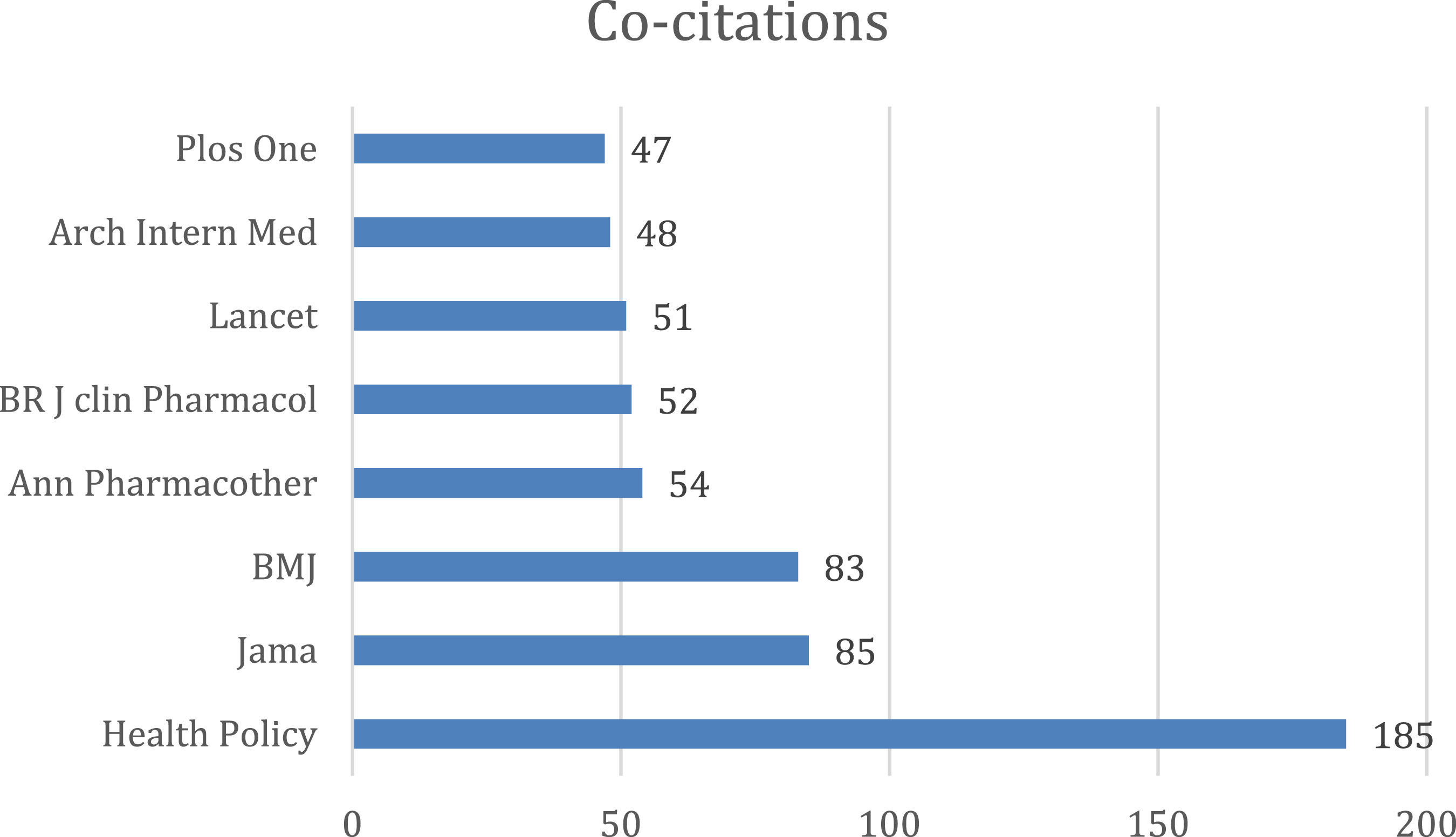
Discussion
This bibliometric study represents a pioneering endeavor in identifying the most highly cited articles and their key characteristics within the realm of generic drug prescription. Interesting trends have surfaced through the analysis of these articles, while at the same time future directions of research have been identified.
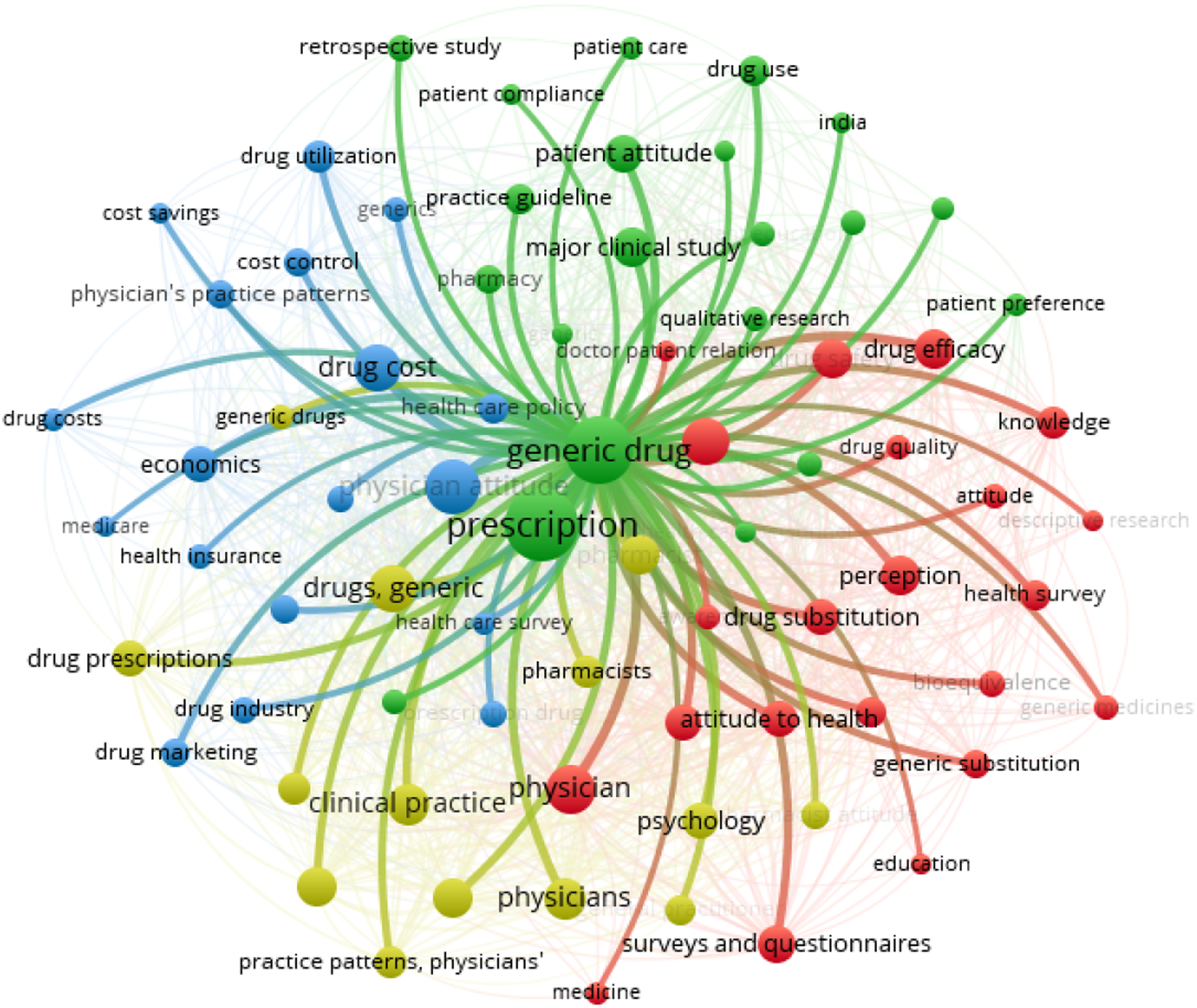
As indicated in Figure 31, four themes of studies were found. 1. Green cluster (Generic drug prescription) 2. Blue cluster (Drug cost) 3. Red cluster (Drug attitude factors) 4. Yellow cluster (Physician factors) Network map of the four identified clusters.
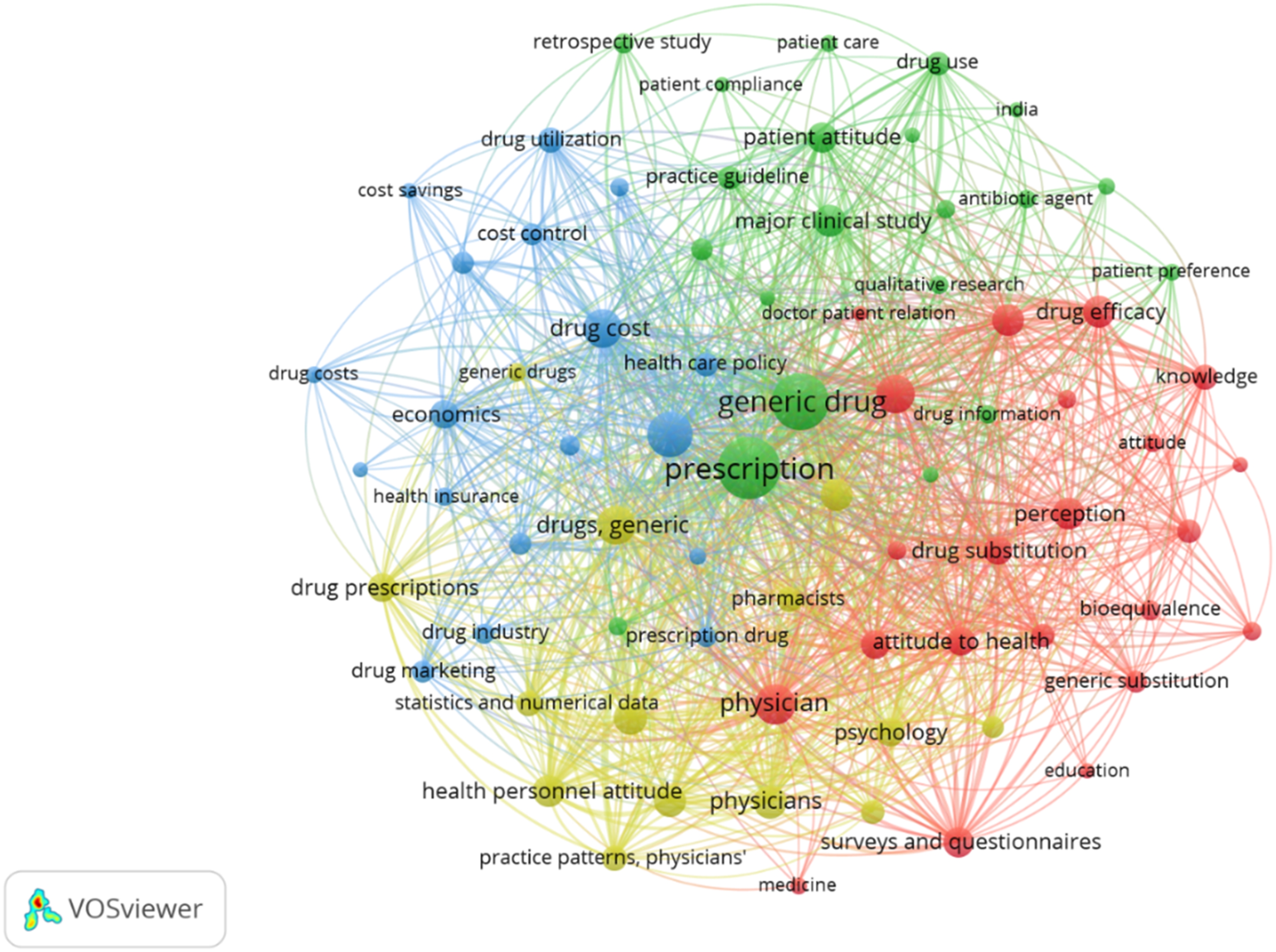
The blue cluster involves generic drug costs, potential savings, insurance, and related economic factors. The green cluster involves factors influencing generic drug prescription such as patient attitude, doctor-patient relation, patient preferences. The yellow cluster revolves around the physicians, and includes aspects like physician attitude, practice guidelines. The Red cluster involves the generic drug attitude factors such as perception, knowledge and awareness, drug efficacy, attitude. Most of the studies seem to be focused on theme of drug costs and perception factors.
Conclusion and future work
The assertion that the generic alternative is almost as effective as the parent substance (branded drug) on which the generic is based has led to its widespread adoption in an effort to decrease the skyrocketing cost of providing healthcare. Recognizing the same, the Indian government launched the “Jan Ausadhi" scheme in 2008, now called the Pradhan Mantri Bharatiya Janaushadi Pariyojana (PMBJP), to provide access to high quality, low-cost generic medicines. Physicians’ role, however, is crucial for successful implementation of a generic prescribing policy. Although physicians’ knowledge, attitudes and practice of generic drug substitution generally varies across countries, good knowledge of and positive attitude towards generic medicine could boost generic prescription practice anywhere in the world.
Physicians need to be well educated and informed about generic drug products during their academic careers in order to have a better appreciation of the same. Government health agencies should run awareness campaigns about generic medications to rationalize prescribing practices and enhance understanding among consumers and prescribers. In the interest of standardization, guidelines on generic drug replacement should be promulgated.
A limitation of this study is that we have only analyzed the doctor’s perception, knowledge, attitude, and practice of generic medicine and prescription. Future research could be taken up from other stakeholder view points like patients (chronic and acute), pharmacists, medical representatives and government regulatory bodies, to form a holistic understanding of the problems in the domain.
Supplemental Material
Supplemental Material - Decoding the perception of prescribers towards generic medicine: A bibliometric study
Supplemental Material for Decoding the perception of prescribers towards generic medicine: A bibliometric study by Aditya T Prasad, and Ashwani Panesar in Journal of Generic Medicines
Footnotes
Author contributions
Aditya Prasad has done the literature review, designed the study, collected the data and drafted the manuscript. Ashwani Panesar helped in reviewing the manuscript and provided assistance in interpretation of data. Both the authors approve this version of the manuscript and agree to be accountable for all aspects of the research work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Author biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
