Abstract
COVID-19 pandemic has shattered the global health economy. Advertisers take advantage of fear and emotions in society due to the pandemic outbreak. Rumors spread rapidly through digital media are another form of misinformation. False claims advertising is a combination of misinformation and disinformation that is fatal and moral deterioration to global society. True information is of central importance in society to avoid false information during the pandemic to control unexpected damage. Without scientific and evidence-based information about all kinds of products and services through advertisements on social media people often use to shift social attitudes. Society always relies on national and harmonious international regulatory bodies during any kind of epidemic and disaster credibility for true information. In most of the Countries, false advertisements are illegal. Countries need to continuously introduce a strict regulatory framework with the Suo Motu surveillance system to breach false advertisements on all media platforms about health care products and services.
Keywords
Introduction
The COVID-19 pandemic has drastically affected nearly every aspect of human life in most of the countries of the world. Misinformation rapidly spreads from the early days of the COVID-19 outbreak in various senses like fear, misrepresentations and contraindication assumption, etc. Millions of people are affected due to false and misleading advertisements.
1
According to the World Health Organization (WHO), most of the countries are not only fighting with COVID-19 but also with infodemic on TV, print and web-network platforms which are harder to control.
2
“There is currently no vaccine available.” ‘Quarantine’ is one of the most effective and oldest tools for controlling communicable disease outbreaks.
3
Companies seeking to capitalize on the market on a large scale are raising false advertisements and consumer effort due to the mentioned viruses.
4
COVID-19 disinformation campaigning is already in full swing. Some people are exploiting the present circumstances by false advertising products and services that claim to prevent and cure COVID-19. False advertisements generally claim to prevent and cure COVID-19 by unregistered products and services.5,6 Complementary and alternative traditional medicines that prevent and cure COVID-19 infection like Giloy (Tinospora Cordifolia
Advertisers must be careful about unwary information to avoid being caught in consumer and competitive lawsuits in hassle time. Recently there have been numerous advertisements for products and treatments on the web, in print and on TV, which have been falsely claiming how effective their products and services are. Giant digital and social media platform facing expanding regulatory examination over various types of content. In particular reflect fake news, false advertisements and ideologies. 11
Direct to consumer advertisements of pharmaceutical or healthcare products are illegal except in two countries, New Zealand and the USA. 12 The Objective of the present study is to highlight the effect of the present regulations to check the misleading claims by the companies, about their products. The outcome of the study emphasizes a need for more strict and effective regulation to address this issue, throughout the entire world. 16
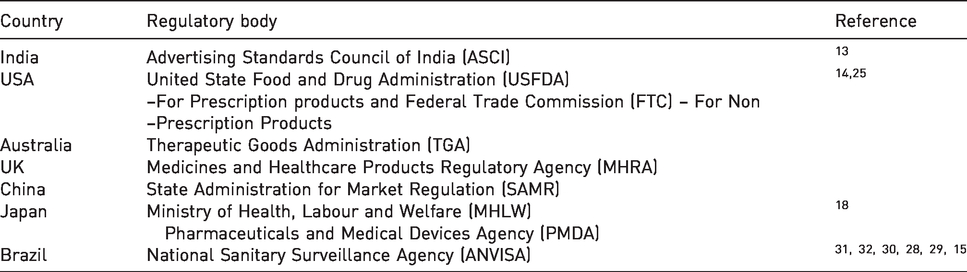
Advertisements of drugs and pharmaceuticals in some of the countries are regulated to some extent by the following regulatory bodies enlists in Table 1.
Regulatory authorities of pharmaceutical advertisements in different countries.
False testing advertisements claims
Negligent misrepresentations claim and common law flaws are a combination of false advertising which puzzles the healthcare experts about any mysterious pathogen, sometimes even about the known Infections.20-21
In Australia, on social media Pete Evans (Famous chef) was trying to sell $15,000 fancy light machines as Bio Charger to vulnerable and frightened people with a claim to treat and protect against COVID-19 infection. TGA Australia fined $25,000 over coronavirus claims relating to Bio Charger.22-24
FDA and FTC issued more than seven warning letters jointly to traders of misbranded and unapproved products, claiming that they can prevent or treat COVID-19 by various remedies in the USA. Companies products include colloidal silver, teas and essential oils. 25 , 26
Advertising started in California by Santa Monica based company RootMD as ‘at-home COVID-19 exposure and immunity tests’ for consumers concerned about the COVID-19. The company ensured that consumers could send thus sample back to “certified MD immunologists” for testing antibodies to COVID-19 and collect the results within two days. At the time, the company had sold about 800 kits while the FDA did not approve in-house testing for COVID-19. But company advertising material for RootMD Kits seemed recommended by the FDA. On Web-based platforms such as Twitter and Instagram, the company's messages included the hashtag “#Approved #FDA”. Company-paid five thousand dollars fine to the FDA as a settlement and stop all deals with consumers of COVID-19 test kits unless if it gets approval from the FDA.33–35
False claims on social media
Social media organizations are ensuring to take action against misinformation and disinformation on their platforms. Amazon’s footsteps, Facebook and Google also banned all misleading advertisements for products claiming to cure Coronavirus.27,36 Social networks, by their very nature, are social. Common forms of misinformation on social media are exaggerated, Untrue and dubious health claims due to fear. Nowadays there is tremendous flow of numerous unregistered traditional and alternative remedies to cure and prevent COVID-19 and other serious infectious diseases in society by the digital and social media platform. A viral video claims that COVID-19 can be treated by using hair dryer for breath. 37 Videos Claiming the cure of COVID-19 through injected of vitamin-C into the blood circulation are viral on the social media platform. Hypocritical baseless medical claims viral on web network are uncountable which create panic and disturb the society.38,39
India advertising regulatory scenario in COVID-19 outbreak
When the world was fighting against the COVID-19 pandemic. WHO has issued a guideline to use alcohol, soap and water for clean hands at least for 20-30 seconds. In India Lifebuoy soap merchandised by Hindustan Unilever Limited (HUL) filled plea in the Bombay high court against Dettol handwash, merchandise by Reckitt Benckiser Healthcare (RBH). 40 Dettol advertised handwash is 10 times more effective against bacteria and viruses as compared to soap. An advertisement of Dettol handwash trying to convey the message that soap is useless and not beneficial is against COVID-19. The High Court said that advertisement against the WHO guideline and misrepresentation of facts. So, the company will have to stop advertisement. Dettol suspends handwash advertisement. But Dettol gains false commercial importance in the market and interesting to see how the Bombay high court perceives and deals with this matter in the upcoming time.41,42
India’s Ministry of AYUSH promotes various practices and traditional healing therapies for boosting the immune system. Ministry has been promoted many traditional remedies to specifically ward off COVID-19. However, no scientific-based information is still available on how effective they are in COVID-19. The Indian government has already rejected health claims such as drinking hot water or gargling with salt or vinegar solution etc from a self-check system (PIB fact check).43,44 Another false claim by local company Arihant Mattress that advertised ‘anti-coronavirus' mattress. Image of commercial advertisement viral quickly on social media platforms like Facebook and Twitter. The half-page advertisement entitled “Anti Corona Virus Mattress pe soyega India toh badhega India (India will progress if it sleeps on this anti-coronavirus mattress)”. A case has been registered against the owner of the manufacturer of the mattress.7,45
In the crucial time of the COVID-19 Pandemic, millions of Indian people mislead by pharmaceutical companies. ASU product legislations are feeble in India. On 23rd June 2020, the National Institute of Medical Sciences & Research (NIMRS) and Patanjali Research Institute (PRI) jointly in the press conference launched a new innovative Ayurvedic formulation for the treatment of COVID-19. NIMSR and PRI have claimed to make Ayurvedic medication kit ‘Anu Taila, Swasari and Coronil ' to treat and cure COVID-19 which was based on scientific data as per their claims. PRI said in the conference that 69 percent of patients recovered within 3 days it’s a historical achievement. PRI claimed that Coronil gives 100 percent favourable results in the controlled clinical trial on mild and moderate patients. Swasari and Anu Taila were already available in the market. Coronil is a new Ayurveda product that comprises of the Tulsi, Ashwagandha and Giloy extracts. In the press conference, PRI announced the home delivery of the Ayurveda medicine kit via the OrderMe app. The medicine Kit has not yet been approved by the AYUSH ministry. But on the same day ministry of AYUSH takes Suo moto action and issued an official notification against the claim by PRI and orders to stop publishing and advertising claims of medicine to cure COVID -19 infection. Ministry of AYUSH had no evidence and claims information about the Ayurveda kit. These kinds of advertisements are regulated under Drug and Magic Remedies Act. After the claim, the ministry asked to share details regarding the medicine such as consent information, sample size, CPP and clinical trial certificate, etc.46–51 On 10th June 2020, PRI applied for a license and on 12th June 2020, the Uttarakhand Ayurveda Department (UAD) had issued a license for fever, cough and immunity boost, not for the COVID-19 cure. As per the WHO guidelines, immunity boosters are never considered as treatment or medicine for the COVID-19 pandemic. Coronil is being inspected for supposed infringement of the Drug and Magic Act,1954 and Drug and Cosmetic Act,1940. On 24th June 2020, the Central AYUSH minister said on the Ayurveda medicine kit which is developed by PRI and NIMRS should not be promoted before approval by the relevant government. Finally, the AYUSH ministry considered Coronil as an Immunity booster only.52–54
ASCI is a self -regulating body in India with no enforcement power over non-members. At present, numerous legislations are the display of obscene and misleading healthcare advertisements in India and other countries. The government of India and other regulatory bodies are encouraging people and web-based media platforms worldwide through awareness campaigns to stop the upload of false health claims news and advertisements.55
Conclusion
Advertisements that are true today may be wrong tomorrow because relevant data often change as more cases are reported in the prospect of COVID-19 pandemic. Advertising practices that claiming connection COVID -19 should follow ethics and remain within the boundary of regulations for the better post-COVID-19 era. New challenges come from internet advertisements. Web-based platforms are not designed for information with scientific and legal bases in society. In reality, most of the advertisements are unnoticed by the statutory bodies and ignored by the consumers also. There is also an urgent need for amendments in the Information Technology Act, 2000 and enactment of Cyber Law for the prevention of unlawful DTCA (Direct to Consumer Advertisements) over the internet in India.
Advertising in the specific and general area pertains to multiple laws at an extensive rate which creates confusion in the mind of the consumer as well as manufacturers. On the global level with Harmonized scale requires carefully collected unbiased data to avoid unfounded inferences false and misleading claims due to fear in Society. Policymakers should focus on online advertisement regulation for consumer privacy and security. Wasteful advertisement expenditure must be controlled to reduce the cost of pharmaceutical products. Pharmaceutical industries have to consider various attributes like legal and ethical issues, Socio-economic framework, etc of the countries before framing the advertisements of their products. Advertising research must go under ethical and regulatory boundaries. Advertising industries need advancement to prohibit misinformation and disinformation in real-time through Artificial intelligence and strict regulations about coronavirus or any future capricious emergencies in society is a need of the hour.
Footnotes
Acknowledgment
The authors are thankful to the Department of Pharmaceutical Sciences and MDU Library which provided the relevant data for the review article.
Authors’ note
This is to inform that the view, thoughts, and opinion expressed in this article belong solely to the author’s but not associated with employer, organization, committee or other group or individual.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
