Abstract
Background/Aims:
Global megatrends, including population growth, interconnectedness and artificial intelligence, are already shaping the clinical trials industry, and will do so for decades to come. Innovations from technology companies, greater collaboration with patients and caregivers, and increased diversity in clinical trials could transform clinical trials of the future. However, the industry has typically been slow to adopt new technologies, and certain barriers could stifle innovation and allow health inequalities to persist. Our aim was to explore the trends shaping clinical trials and identify initiatives that can be started now to move toward a future where patients are actively involved in clinical trial design and decision-making, and technologies like artificial intelligence are integrated responsibly and ethically into clinical trials.
Methods:
We used strategic foresight methodology to explore the trends that will shape the future of clinical trials and how the trajectory of these trends could lead to different future scenarios We then identified initiatives that could help us move toward our desired scenario.
Results:
We identified four possible scenarios for the future of clinical trials based on two key trends: data sharing and adaptation of regulatory frameworks. While the future will see collection of large amounts of data from different sources, such as databases and wearable devices, their usefulness could be limited if data remain in silos, limited by strict regulations and a lack of trust in data privacy. Alternatively, data may be integrated more efficiently into clinical trials if regulators adapt proactively to new technologies and patients are empowered to take control over their data, for example. We identified initiatives that can be started now to achieve our vision. Pharmaceutical companies, academic researchers, non-governmental organizations (including patient organizations), regulatory authorities, policymakers, technology companies and innovative service providers must join forces and work on initiatives to integrate technology into trials, enable data sharing, harmonize regulations across regions, co-design trials with patients and caregivers, strengthen decentralized trial capabilities and promote diversity in trials.
Conclusion:
Through a collaborative cross-industry effort that prioritizes proactive patient involvement and responsible use of technology, we can shape a clinical trial ecosystem that is inclusive, ethical and ready for the future. We urge all stakeholders to act now on these initiatives to positively influence the future of clinical trials.
Keywords
Background/aims
Clinical trials have become increasingly complex over time due to growing regulatory and consumer requirements,1,2 yet this has not necessarily improved efficiency or patient outcomes. The future of clinical trials will be influenced by global, societal, technological and economic changes known as global megatrends. Our goal was to understand these trends, map possible future scenarios and identify initiatives that we can start now to prepare for the future.
Megatrends are forces that will shape future outcomes for at least the next decade and are already observable. 3 For example, people are increasingly empowered and seek greater involvement in their healthcare decisions, 4 and there is greater demand to incorporate patient perspectives across clinical development. Yet, barriers to clinical trial access and participation persist, including limited geographic availability of trials, 5 social and financial barriers, and strict inclusion criteria, which contribute to underrepresentation of groups such as older adults, females and people with multiple health conditions.6–9 Regulatory authorities are introducing requirements to increase diversity in trial populations,10,11 which will benefit underserved communities and may lead to more robust and innovative science. 9 However, widening access is only part of the solution, and patients must also be supported to navigate complex and burdensome trial designs. 12
While advances in digitalization and artificial intelligence (AI) offer opportunities for clinical trials through data linkage, decentralization and automated data collection,13–15 the adoption of technology in clinical trials has typically been slow. This is due to stringent regulatory requirements, a risk-averse industry culture and challenges integrating innovations into existing systems.16,17 There is growing concern that AI may reinforce existing biases in healthcare related to socioeconomic status, race, gender and age, so inclusive data practices and vigilant oversight are needed.14,18 Technology companies and regulators must also reassure patients about data privacy and usage to facilitate trust in data sharing.15,18,19
Here, we present our systematic approach to mapping the future of clinical trials and identifying tangible actions to move toward our desired future outcome. We envision a future clinical trial landscape that prioritizes patient needs, leverages technologies responsibly and optimizes efficiency while respecting good clinical practice and the need for meaningful data. This work is a collaboration between patient advocates and healthcare professionals (collectively termed the Global Clinical Trial Experts Panel), representatives from the pharmaceutical industry (Boehringer Ingelheim), and the Copenhagen Institute for Futures Studies.
Methods
We used a four-step strategic foresight20,21 methodology (Figure 1) to explore potential future scenarios for clinical trials.
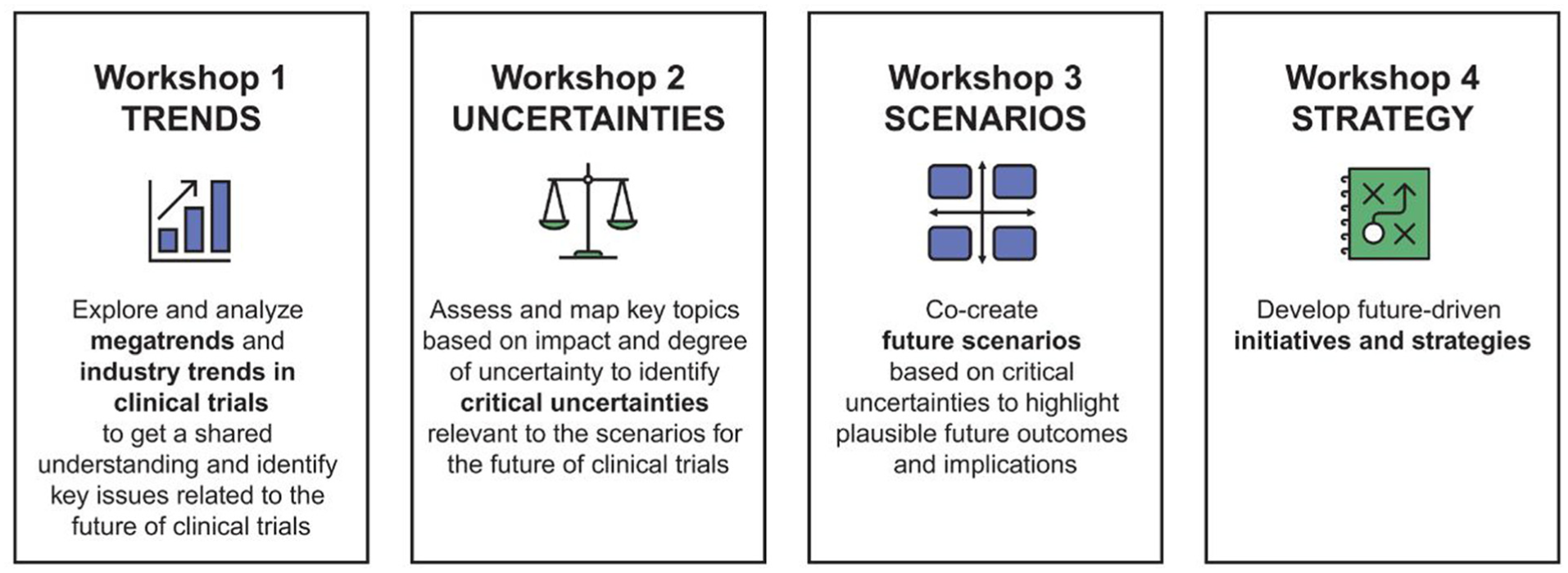
Workshop structure.
Between February and June 2023, we held four workshops with 48 participants from the Global Clinical Trial Experts Panel, Boehringer Ingelheim and the Copenhagen Institute for Futures Studies (see Supplementary material for participant list). Before the workshops, the Copenhagen Institute for Futures Studies identified 15 global megatrends 4 (see Supplementary material) and conducted desk research to understand their potential impact on clinical trials. Internal industry trends were gathered through interviews with patient advocates, healthcare professionals, pharmaceutical companies and regulators.
In Workshop 1, we assessed and prioritized the trends. Workshop 2 focused on refining their likely evolution and identifying critical uncertainties (trends with an uncertain trajectory that could lead to multiple plausible visions of the future). We also explored factors that could block or enable the achievement of our desired future outcome, as well as potential opportunities and challenges. Two critical uncertainties (that had the broadest “disagreement” on the future trajectory when discussed in Workshop 2) and their opposing outcomes formed a “scenario cross” in Workshop 3, outlining four plausible future scenarios for clinical trials.
In Workshop 4, we generated actionable initiatives aimed at steering clinical trials toward our preferred future. These initiatives were stress-tested across the four scenarios to ensure robustness and identify necessary adjustments.
Results
Workshop 1: trends that will shape the future of clinical trials
We analyzed how 15 global megatrends (Figure 2) may affect the future of clinical trials.
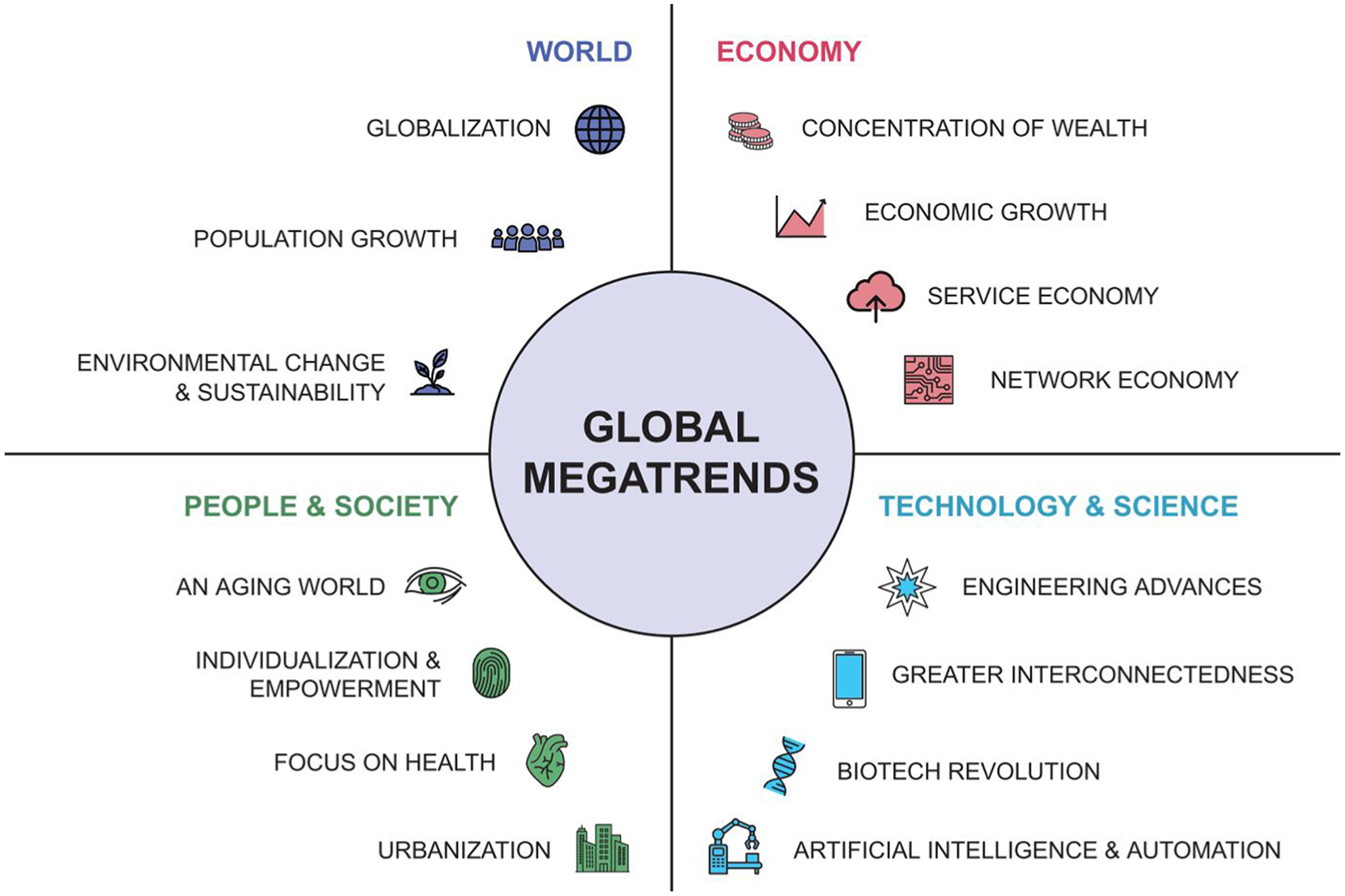
Global megatrends. 4
World trends: Globalization, population growth and sustainability. Globalization (the increasing connectedness of the world) will be shaped by political tensions, new alliances between countries and climate change. The flow of knowledge, science, technology, data and travel will accelerate, led by countries including China, India, Indonesia and Brazil. 4 Improved digital infrastructure will connect underserved regions, and AI-powered real-time translation could help overcome language barriers, enabling more inclusive collaboration.4,22 Globalization could facilitate access to hard-to-reach and diverse patient groups, but pharmaceutical companies will need to navigate different regulatory, cultural and social landscapes. Population growth will continue until the mid-2080s, 23 driven mainly by low- and middle-income countries.23,24 Africa’s population is expected to double by 2050, while Asia will remain the most populated region of the world. 25 The combination of unmet needs, large populations and a push for diversity may lead to more trials in these regions. Clinical trials must reflect the heterogeneity of characteristics observed in real-world populations. Real-world evidence could help address diversity gaps, and conducting these studies before regulatory approval may become mandatory. Initiatives such as Canada’s CanREValue collaboration are already working to incorporate real-world evidence into regulatory and reimbursement decisions. 26 Elsewhere, growing concerns about climate change and sustainability will increase scrutiny of the environmental impact of trials, including trial-related travel.
Economic trends: Economic growth, concentration of wealth and the service and network economies. Economic growth in emerging economies (especially Asia’s rapidly expanding middle class) 27 may boost healthcare infrastructure and increase trial diversity. However, growing economic inequality within countries 4 could perpetuate inequalities in clinical trials and limit trial access for people with low incomes due to financial, geographic and logistical barriers, including lack of caregiver support and inability to take time off work. 28 Also, sponsors may hesitate to run trials in countries where a limited population will be able to afford their drugs, highlighting a need for collaboration with regulators to facilitate patient participation, promote new research and support the use of new technologies.
Clinical trials may evolve into a service that is seamlessly integrated into participants’ everyday lives. Meanwhile, the network economy will foster stronger collaborations 4 between the pharmaceutical industry, academia 29 and non-governmental organizations (NGOs), including patient organizations. The growth of peer-to-peer platforms could enable experienced trial participants to connect and share experiences with those considering participation.
Trends in people and society: An aging world, empowered individuals, a focus on health and urbanization. Older adults have historically been excluded from clinical trials for many reasons, including age-related changes and risks, comorbidities and being prescribed multiple medications.30–33 While representation of older people in trials is increasing, progress is slow.34,35 Better understanding of drug–drug, drug–gene and drug–age interactions may help expand trials to older adults. Wider use of digital and decentralized trial approaches will require investments in health and digital literacy. These approaches rely on patient trust and, therefore, must be co-created with patients. Greater patient involvement in healthcare could encourage more patient-centered trial designs, improved data sharing, and more efficient and cost-effective clinical trials. 36
The future will see a shift from reactive to preventive healthcare. 4 Improved public understanding of clinical trials, supported by educational campaigns, could transform the perception of trials from a “last resort” to a legitimate healthcare option, especially for rare conditions. Increased urbanization risks widening inequalities between those living in cities (who have better access to healthcare) and rural areas. Decentralized trials may help address this issue, but ensuring people can access and use these technologies is crucial.
Trends in technology and science: Engineering advances, greater interconnectedness, biotech and AI. The number of Internet-connected devices will continue to increase 4 and could help reach more prospective trial participants. Connectivity is one cornerstone of decentralized trials, enabling digitalization of process, data capture and consultations. 37 Challenges exist in integrating multiple data sources, utilizing collected data and preserving data privacy. 37 Also, certain data can only be obtained through in-person physical examination. A lack of connectivity in remote areas, the financial cost of devices and a lack of digital literacy could continue to prevent universal access to the Internet. 38 Services and devices aimed at trial participants must be co-designed with the patient community from the outset to maximize acceptance, usage and adherence.
The use of “big data” will allow for greater personalization and stratification of clinical trial participants and create new treatments. AI may be used to optimize clinical trial processes, including recruitment, participant stratification and data collection. Developing regulations that enable innovation, such as platform trials (which can quickly study multiple treatments),39,40 while ensuring effectiveness, safety and cost efficiency, will be a major challenge. Some countries, like Canada, are already updating their clinical trial regulatory frameworks to keep pace with trends and emerging technologies. 41
Workshop 2: uncertainties
We explored 16 clinical trial industry trends (Figure 3 and Supplementary Figures S1 and S2). Nine trends were prioritized for further analysis and are summarized below. For each, we identified “critical uncertainties” (uncertain future trajectories) and mapped opposing outcomes by the year 2040.
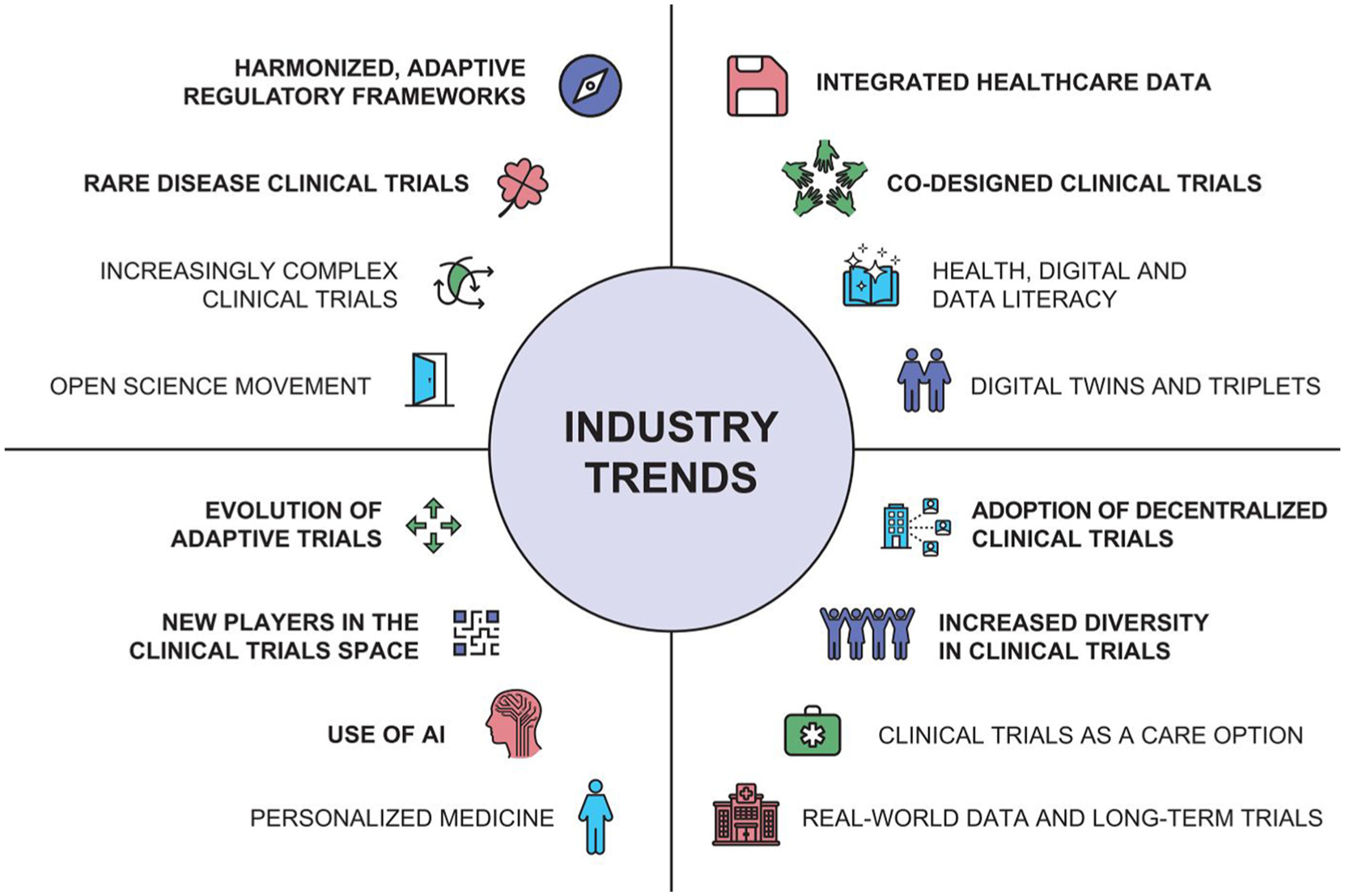
Clinical trial industry trends.
Harmonized, adaptive regulatory frameworks for clinical trials
Regulatory clinical trial frameworks could become more harmonized, leading to closer collaboration between authorities, simplified data sharing and adoption of innovative approaches. This could accelerate drug development and improve patient access. The openness of regulators to these new concepts is critical to whether this scenario is adopted. Hesitancy to work with industry, limited harmonization of regulations and limited data sharing across regions could result in siloed regulatory frameworks. In this scenario, many regions will still have their own regulatory authorities and laws, making collaboration difficult and causing delays in the approval of new treatments.
Integrated healthcare data
Collaboration between stakeholders committed to innovation may enable the collection, integration and use of meaningful, high-quality data. There may be widespread willingness among citizens to share their data when they see a personal benefit in return. Governments will support this through regulatory frameworks to limit data breaches.
Alternatively, the rapid growth of data availability could mean an uncoordinated approach to data collection, hesitancy around data sharing, and unclear regulations on data collection and ownership. Governments may respond by developing strict regulatory frameworks around systems and compliance. These fragmented data infrastructures could be a challenge for globally conducted trials.
Co-designed clinical trials
Increased patient involvement in trial design, and new tools for providing information and allowing participant feedback could improve recruitment and significantly increase participant engagement, retention and satisfaction. Yet, challenges could arise if stakeholders do not have the capability to discuss trial design. An alternative scenario could see pharmaceutical companies and regulatory authorities avoid co-designing trials with patients, fearing that this may delay trials. As a result, patients face barriers to trial participation, delaying recruitment and regulatory approval of new medicines.
Increased diversity in clinical trials
Research protocols that emphasize culturally sensitive methodologies will foster trust and participation among Indigenous communities. Clinical trials could become a seamless component of patient care, with patients and healthcare providers easily identifying relevant trials. Diversity and access may be driven by pharmaceutical companies, who work with governments, payers and patients to fully engage underrepresented populations. Increasing the value of drugs that truly have an impact on health outcomes across diverse and underserved patient populations will have been the guiding principle in this scenario.
Alternatively, access to clinical trials may remain limited due to traditional inclusion criteria and restrictive infrastructure and regulations. Diversity and access could instead be driven by governments, who would support patient organizations and drug developers to conduct clinical trials in traditionally underrepresented regions such as Africa, South America and South-East Asia, and across different socioeconomic groups.
Use of AI
Technology companies may drive the use of AI in future clinical trials, making processes more efficient and successfully recruiting participants through social media channels, targeted communication and automated review of large clinical databases. Enabling trial participants to use tools and devices that they use every day will help retention. Algorithms will be carefully designed and governed by a diverse group of stakeholders to reduce bias. AI offers opportunities for better-informed trials, analysis of big data to detect small signals (hints about emerging trends), value-based healthcare and prediction of risk for disease prevention approaches. Greater AI literacy among new generations could enable its adoption in trials.
In an alternative future, AI in clinical trials may be driven by the life sciences sector under strict regulations, potentially slowing its progress. AI could be used in a limited capacity to assist diagnosis, identify new drug targets and effective drug combinations, predict the efficacy of new drugs, identify patients who are most likely to benefit from a new drug, screen trial participants and support with administrative tasks.
Evolution of adaptive and platform trials
Adaptive trials (that combine all phases into a single, seamless trial) could become the norm in 2040. This could result in reduced waste, shorter recruitment time, fewer control-arm participants needed, more efficient regulatory approval and better outcomes. A Bayesian approach to analysis, where new information can be added to existing findings, has facilitated this faster approach. Alternatively, traditional Phase 1–4 trials could continue. The costs and increased complexity of adaptive trials, reluctance to compare directly against competitors and lack of awareness of the benefits could limit their adoption.
New players (such as technology companies) in the clinical trials space
Technology companies will create innovative trial solutions to reach diverse populations and personalize trial experiences, but their acceptance by industry and academia remains uncertain. If technology companies are embraced, multi-sponsor clinical trials could become the norm. While data ownership and financial incentives could remain a challenge, the mutual benefit of collaboration may outweigh the issues. If technology companies are not embraced, they will have to compete with the established system. Industry and academia may be reluctant to collaborate due to a lack of clarity around data ownership, lack of financial incentives and regulatory issues.
Adoption of decentralized trials
By 2040, most clinical trials may be decentralized or hybrid. The complexity of decentralized trials could be overcome through effective regulation, increased integration of app and wearable data into site systems, and investment in training and education from sponsors, trial sites and governments. Training and monitoring will be needed to ensure the continuity of high-quality care at local sites.
Widespread adoption of decentralized trials faces several challenges, including fragmentation of regulations across regions, concerns about data quality and data privacy, and a shortage of suppliers able to provide global support for these trials. A perceived loss of human contact could negatively impact retention, especially if a trusted clinician is replaced with a different provider at each home visit, and participants are unsure who to approach for support. Thus, retaining the human connection in decentralized trials will be crucial. Low health and digital literacy also pose barriers, requiring sponsors to consider alternative ways to include participants. Decentralized trials may be adopted primarily in regions with high societal readiness, adequate infrastructure and staffing, and high digital and health literacy. The investment required in educating participants, the burden trial sites face in adapting to these trials, and data privacy concerns could limit uptake.
Rare disease clinical trials
Engaged groups of NGOs, academic communities and specialist rare disease centers will coordinate clinical trials for rare diseases (and rare subgroups of common diseases). They will have successfully connected patients with the same rare disease, making running clinical trials easier. Combined with new regulations, this will allow for better collaboration and sharing of data, and facilitate drug development. This approach could provide opportunities for running decentralized trials, enabling patients to participate in clinical trials abroad, addressing resource and infrastructure challenges, and setting up umbrella sites to support coordination. However, challenges with patient recruitment and the high costs of running rare disease trials could limit coordination. Rare disease trials may continue primarily in high-income countries, excluding most of the world.
Workshop 3: dealing with the uncertainties: four scenarios for the future of clinical trials
The two critical uncertainties we focused on were as follows: (1) How regulators will adapt to change: regulators may act reactively to change, addressing problems as they occur, or proactively, attempting to foresee and plan ahead; and (2) How widely patient data will be shared: data sharing may be limited if data privacy concerns are not mitigated or patients are unaware of the potential value their data can provide. Alternatively, empowering patients to take control over the sharing and use of their data may enable wide data sharing.
The opposing outcomes of these two critical uncertainties formed a “scenario cross,” outlining four plausible future scenarios (see Figure 4 and Supplementary Table S1).
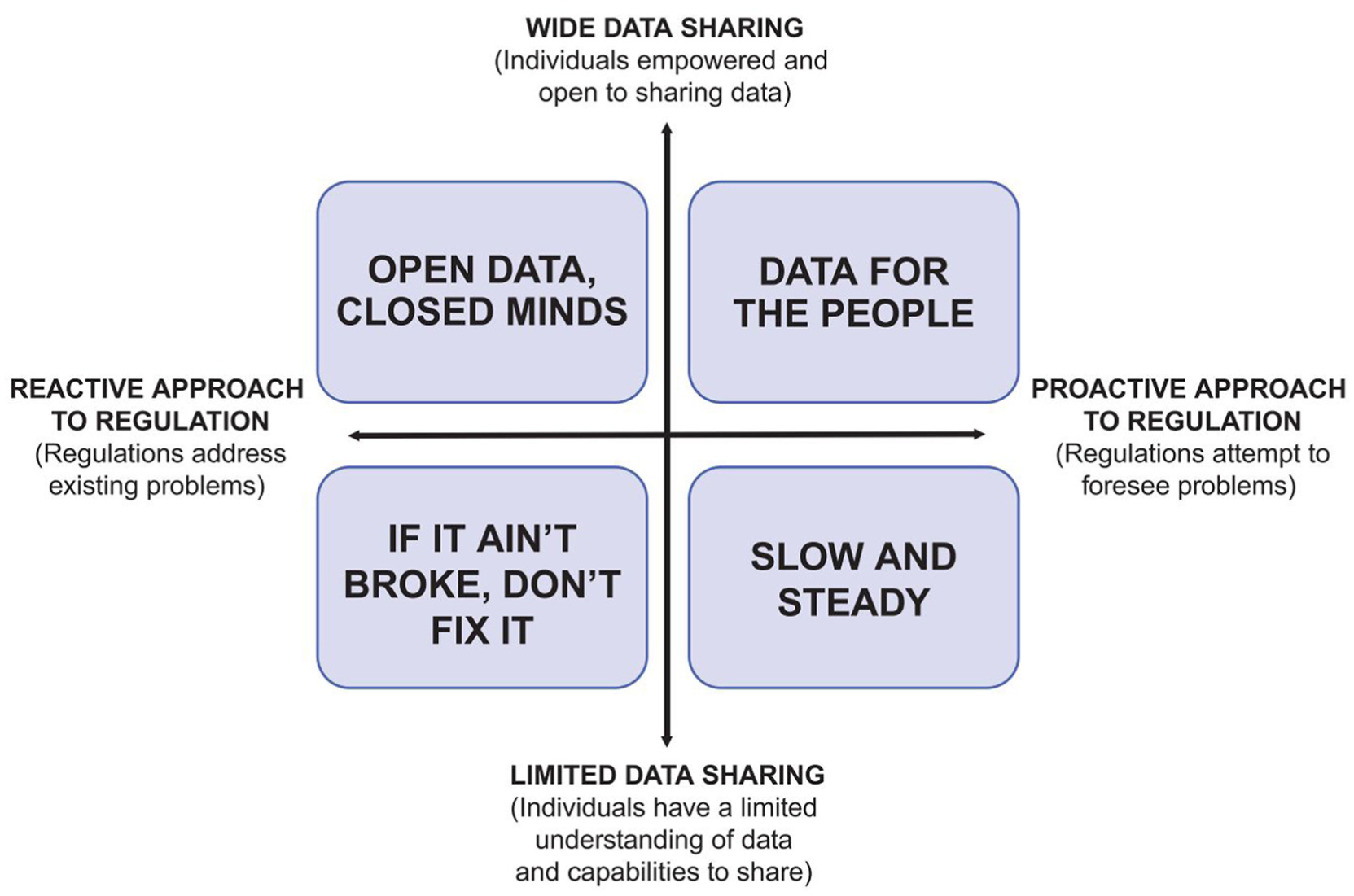
Four scenarios for the future of clinical trials.
Data for the people
Our preferred scenario envisions wide data sharing supported by a proactive approach to regulation, facilitating technology integration and stakeholder collaboration, giving patients guaranteed access to, and control over, their clinical trial data. Patients could even allow trusted patient organizations to manage their health data for them, akin to financial managers. Real-world data from wearable devices will shape the selection and design of trials, while technological advancements such as a “gene passport” could allow for personalized treatment selection.
Open data, closed minds
Wide data-sharing capabilities combined with a reactive approach from regulators could create a complex scenario in which technology thrives, but disparities persist in clinical trial access, and digital and health literacy. NGOs will play a major role in driving change and co-designing trials that benefit from a diverse range of perspectives. Participation of NGOs in data governance bodies and data altruism organizations will increase patient trust and their likelihood of consenting to data sharing for research. However, regulators may act slowly and cautiously, stifling the potential of data sharing and innovation, while companies are reluctant to share potentially sensitive data with competitors. Patients with low digital or health literacy may be reluctant to share their data, unaware of the value it can provide.
Slow and steady
This scenario imagines that infrastructural constraints and data privacy concerns lead to a cautious approach to technology integration and limited data sharing. To address these issues, a comprehensive technical and legal framework for data sharing is needed, combined with a secure data infrastructure that addresses data privacy concerns. A proactive approach to regulation will champion change, advocating for diversity representation, ethical data practices and integration of technologies providing real-world evidence. The clinical trial landscape will slowly progress toward an inclusive, transparent and personalized healthcare system, led by regulations that prioritize patients’ needs.
If it ain’t broke, don’t fix it
Technology companies will disrupt the clinical trial industry, but low digital literacy could hinder integration. Regulators will operate reactively to rein in technology companies, prioritizing thoroughness over speed. While data breaches and misuse may become rare, there is limited data sharing. Data may be locked in silos, with no access for trial participants or other researchers. The slow approach from regulators will hinder the evolution of clinical trials and growth of personalized medicine, and make diversifying trial participation more challenging.
Workshop 4: initiatives to shape the future of clinical trials
The following initiatives aim to address critical uncertainties and drive us toward our preferred scenario: “data for the people” (see Table 1 for further details).
Initiatives to shape the future of clinical trials:
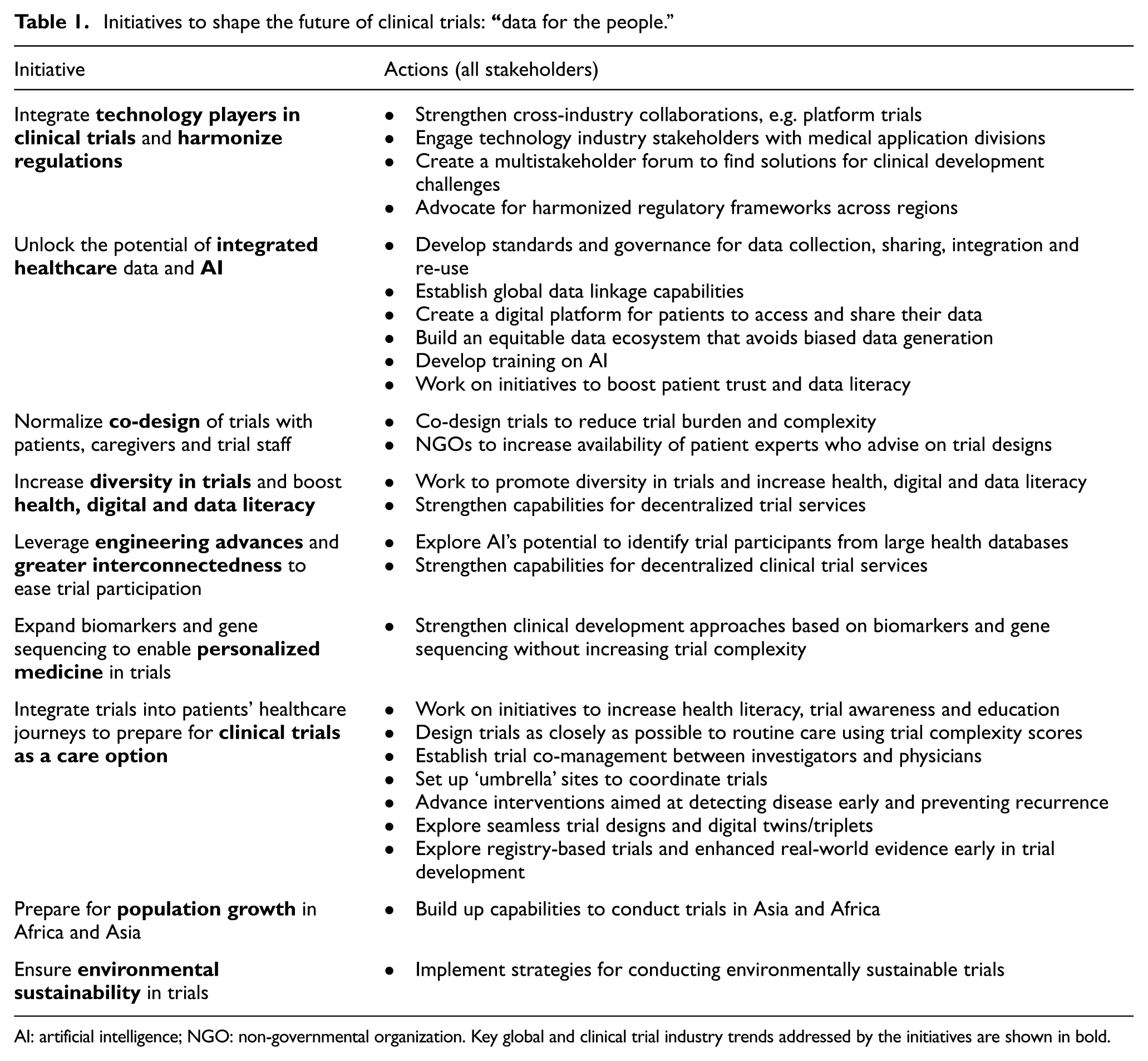
AI: artificial intelligence; NGO: non-governmental organization. Key global and clinical trial industry trends addressed by the initiatives are shown in bold.
To unlock the potential of technology in clinical trials, we must build global, multistakeholder partnerships to establish data collection standards, create governance frameworks that empower patients with control over their data and implement regulations that facilitate data integration. Advocating for harmonization of regulations across regions will be crucial to enable collaboration and innovation. To create a future where trials are less complex and more patient-centric, co-design of trials with patients, caregivers and trial site staff must become standard practice. Cross-industry partnerships should be strengthened (e.g. through platform trials), and we should make efforts to collaborate with technology companies entering the clinical trials space. Establishing a forum that brings together multiple stakeholders (e.g. industry, patient organizations, regulatory authorities and medical associations) will enable ideation of solutions to common clinical trial challenges. As global inequality rises, we can break down barriers to trial access by prioritizing diversity in trials, working on initiatives to improve health and digital and data literacy, and strengthening decentralized trial capabilities. We must also make efforts to seamlessly integrate trials into patients’ healthcare journeys by designing trials that closely mirror routine care. Working as a collective to implement these initiatives will shape a more inclusive, efficient and patient-centered future for clinical trials.
Conclusion
To truly transform the current landscape and maximize the benefits of technology responsibly, we must be willing to take bold steps, some of which may challenge existing policies but will likely evolve into regulatory expectations by the time a trial completes.
We call on pharmaceutical companies, academic researchers, NGOs (including patient organizations), regulatory authorities, policymakers, technology companies and innovative service providers to join forces in multistakeholder partnerships to drive our recommended initiatives forward. We must work to integrate technology into trials, enable data sharing, harmonize regulations across regions, co-design trials with patients and caregivers, strengthen decentralized trial capabilities and promote diversity in trials. Through a collaborative cross-industry effort that prioritizes proactive patient involvement and responsible use of technology, we can shape a clinical trial ecosystem that is inclusive, ethical and ready for the future. We urge all stakeholders to act now to help transform this vision into reality.
Supplemental Material
sj-docx-1-ctj-10.1177_17407745251414680 – Supplemental material for Shaping the future of clinical trials through strategic foresight
Supplemental material, sj-docx-1-ctj-10.1177_17407745251414680 for Shaping the future of clinical trials through strategic foresight by Sharon Lee, Jeff McIntyre, Tracey Robertson, Kimberley Kallsen, Martin Bauer, Rossana Berardi, Christian Boisvert-Huneault, Emmanuelle Clerisme-Beaty, Christopher P Denton, Eric Fakra, Jan Geissler, David Goldstein, Kazuo Hasegawa, Manya Lind, Patricia LoRusso, Keith Perry, Claas Röhl, Jose Francisco Kerr Saraiva, Nidhi Swarup, Aron Szpisjak and Allan Flyvbjerg in Clinical Trials
Footnotes
Acknowledgements
The authors thank the following people for their participation in the workshops on which this publication is based: Peter Aadal Nielsen (Copenhagen Institute for Futures Studies), Axel Aakerblom (Boehringer Ingelheim), Uli Broedl (Boehringer Ingelheim at the time of the research conduct), Steven Brunette (Boehringer Ingelheim), Bogi Eliasen (Copenhagen Institute for Futures Studies), Keith Ferdinand (Global Clinical Trial Experts Panel), Candice Fitzgerald (Boehringer Ingelheim), Katie Gillies (Global Clinical Trial Experts Panel), Tristan Gloede (Boehringer Ingelheim), Sabine Harning (Boehringer Ingelheim), Anne Hildebrandt (Boehringer Ingelheim), José Manuel Jerez-Pombo (Copenhagen Institute for Futures Studies), Sandra de Kruif (Boehringer Ingelheim), Vera Langer (Boehringer Ingelheim), Bronwyn Lewis (Boehringer Ingelheim), Baldwin Mak (Boehringer Ingelheim), Marina Medved (Boehringer Ingelheim), Katrin Ong (Boehringer Ingelheim), Anne Rau (Global Clinical Trial Experts Panel), Jessie Song (Boehringer Ingelheim), Katharina Tomala (Boehringer Ingelheim), Nobutaka Yagi (Boehringer Ingelheim), Erik Van der Eycken (Global Clinical Trial Experts Panel), Diego Villalón (Global Clinical Trial Experts Panel), Maarten Voorhaar (Boehringer Ingelheim), Joep Welling (Global Clinical Trial Experts Panel) and Alison Zheng (Global Clinical Trial Experts Panel).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.C-B., K.K. and T.R. are employees of Boehringer Ingelheim. J.M., E.F., J.G., D.G., P.L., K.P. and J.F.K.S. received remuneration and/or travel support from Boehringer Ingelheim in support of this project.
No conflicts were declared in relation to this work for all other authors.
Outside of the submitted work:
J.M. reports receiving support for attending meetings and/or travel from Boehringer Ingelheim. M.B. reports receiving consulting fees from Boehringer Ingelheim and Lundbeck; support for attending meetings and/or travel from Rovi; and advisory board participation for Boehringer Ingelheim. R.B. reports receiving grant funding at their institution from AstraZeneca, Pfizer and Roche; personal consulting fees from Astellas, AstraZeneca, Boehringer Ingelheim, Eisai, Gilead, Menarini and MSD; and payment or honoraria for lectures, presentations, manuscript writing or educational events from Bayer, Bristol Myers Squibb, Daiichi Sankyo, Incyte, Lilly, Pierre Fabre and Seagen. C.B-H. reports funding paid to their organization by Boehringer Ingelheim; personal grants or contracts from UCB and Janssen; payment or honoraria from Boehringer Ingelheim for lectures, presentations, manuscript writing or educational events; and payment for expert testimony from UCB and Janssen. C.B-H. is an unpaid co-chair of Psoriasis Canada. C.D. reports receiving consulting fees from Acceleron, Arxx Therapeutics, Boehringer Ingelheim, Certa Therapeutics, Corbus, CSL Behring, GlaxoSmithKline, Horizon, Lilly, Novartis, Roche and Zurabio; and payment or honoraria for lectures, presentations, manuscript writing or educational events from Boehringer Ingelheim, GlaxoSmithKline and Janssen. E.F. reports receiving payment or honoraria for lectures, presentations, manuscript writing or educational events from AbbVie, Boehringer Ingelheim, Eisai, Ethypharm, Janssen, Lundbeck, MSD, Neuraxpharm, Otsuka and Sanofi; payment for expert testimony from Boehringer Ingelheim, Ethypharm, Janssen, Lundbeck and Otsuka; support from Lundbeck for attending meetings; and/or travel and advisory board participation for Ethypharm, Lundbeck, Otsuka and Newron. J.G. reports grants or contracts paid to their organization by Astellas, AstraZeneca, Bayer, BeiGene, Boehringer Ingelheim, Bristol Myers Squibb, Daiichi Sankyo, Eli Lilly, Gilead, GlaxoSmithKline, Janssen, Jazz, Merck, Novartis, Novavax, Pfizer, Roche, Recordati, Servier, UCB, Takeda and Vifor. JG is trustee, vice president and managing director of Leukemia Patient Advocates Foundation; managing director of European Patient Advocacy Institute; owner and chief executive officer of Patvocates; unpaid chair of LeukaNET and Acute Leukemia Advocates Network; unpaid trustee of HARMONY foundation; steering committee member of CML Advocates Network; and committee member of the European Cancer Organisation, the European Hematology Association and the Ethics Committee of the Bavarian Chamber of Physicians. J.G. is also a board member of Patient Focused Medicines Development, ECHoS and National Decade Against Cancer. D.G. reports receiving grant funding at their institution from Amplicare, Bayer and Pfizer; consulting fees from Biontech, MSD and Takeda; and data safety monitoring board participation for Australian and New Zealand Urogenital and Prostate Cancer Trials Group, Omico and Panbela. D.G. is an unpaid scientific advisory board member of Australasian Gastro-Intestinal Trials Group. K.H. reports consulting fees paid to their organization from Boehringer Ingelheim. P.L. reports receiving grant funding and medical writing support paid to their institution from Boehringer Ingelheim, consulting fees from Boehringer Ingelheim and advisory board participation for Boehringer Ingelheim. K.P. reports receiving consulting fees and support for attending meetings and/or travel from Boehringer Ingelheim. K.P. is an employee of Stanford University. C.R. reports receiving payment or honoraria from AOP Austria for a panel discussion and support for attending meetings and/or travel from Boehringer Ingelheim and Novartis. C.R. is chair of NF Kinder and EUPATI Austria. C.R. is board member of Pro Rare Austria, ERN GENTURIS and Die Allianz onkologischer Patient:innenorganisationen. J.F.K.S. reports receiving grant funding from Boehringer Ingelheim; consulting fees from Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Hypera, Lilly, Medtronic, MSD, Novartis, Novo Nordisk and Servier; payment or honoraria for lectures, presentations, manuscript writing or educational events from AstraZeneca, Bayer, Hypera/Mantecorp, Lilly, Novartis and Novo Nordisk; and advisory board participation for Bayer, Lilly, Novartis and Novo Nordisk. N.S. reports consulting fees paid to their organization from Boehringer Ingelheim; honorarium from PxP Canada for a presentation; and support for attending meetings and/or travel from Asia Pacific Alliance of Rare Disease Organisations, Boehringer Ingelheim, European Federation of Crohn’s and Ulcerative Colitis Associations, International Alliance of Patients’ Organizations, International Consortium for Health Outcomes Measurement, International Society for Quality in Health Care, Patient Focused Medicines Development and Roche. NS is founding chair of the Alliance of Patients’ Organizations Singapore and a member of the Ministry of Health Singapore National Pharmacy Advisory Committee and the Ministry of Health Singapore Agency for Care Effectiveness Consumer Panel. N.S. is a member of the Strategic Advisory Committee of the Lee Kong Chian School of Medicine Office of Patient Engagement and is director and founder of Nidhi-Kintsugi PL. A.F. reports receiving consulting fees from Boehringer Ingelheim.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported and funded by Boehringer Ingelheim. Alice Kirby of MEDiSTRAVA provided writing, editorial and formatting support, which was contracted and funded by Boehringer Ingelheim.
Supplemental Material
Supplemental material for this article is available online.
