Abstract
Background:
Intrapartum research (occurring during labour and birth) presents challenges to successful recruitment to clinical trials. These include limited time for discussion, decision-making for two (mother and baby), heightened emotional states (pain, anxiety and/or fatigue) and clinician hesitancy to discuss research in this setting. In the context of the Baby head ElevAtion Device Feasibility Study, where the event of interest (caesarean section at full dilatation) is both rare (fewer than 3% of all births) and unpredictable, we undertook a mixed-methods evaluation of the two-stage consent process: (1) abbreviated intrapartum consent and (2) full postpartum consent. The aim was to explore whether abbreviated intrapartum consent was acceptable to patients and clinicians.
Methods:
Eligible patients approached at full cervical dilatation (10 cm) to take part in the Baby head ElevAtion Device Feasibility Study were invited to complete a face-to-face survey of their experience of consent within 3 days after birth. We sampled those who consented and those who declined the study. Clinicians working at recruitment sites were invited to an individual semi-structured interview. Qualitative data were analysed using reflexive thematic analysis.
Results:
Over 12 months, 69% (128/186) of eligible patients consented to the Baby head ElevAtion Device Feasibility Study; 87% of consenters and 66% of decliners completed a follow-up survey. Most survey responders (78%) and clinicians found abbreviated intrapartum consent acceptable. Three themes shaped patient decision-making: perceived benefits, trust in healthcare, and feeling overwhelmed. Those who declined often wished they’d had more time or earlier information. Clinicians found the two-stage consent process feasible and appropriate for low-risk interventions, although time pressures and communication challenges affected consent quality. Many saw the model as respectful of autonomy and potentially useful for future intrapartum research.
Conclusion:
Our findings suggest that the two-stage consent process for this intrapartum study was acceptable to both patients and clinicians. We propose this as a useful consent model for peripartum studies where the clinical situation occurs infrequently, the intervention being studied is low risk, and where opt-out or deferred consent is not available.
Keywords
Background/Aims
Approximately half of all clinical trials fail to meet recruitment targets, and reasons for this are often multi-factorial.1,2 This has both ethical and financial implications and the potential to delay implementation of important clinical practices or treatments.3–5 Pregnant people invited to participate in research have the additional burden of considering the benefits and risks for both themselves and their baby. Research conducted in the intrapartum period (during labour and birth) adds challenges to recruitment including limited time for discussion and decision-making. Unexpected events in labour can add stress for families, and patients may be fatigued, distressed and in pain.4,6 Clinicians may feel hesitant to approach patients about research in these situations due to competing clinical demands, concerns about the patient’s vulnerability, and the potential risks or uncertainties associated with the proposed treatments. 7 Understandably concerns have arisen about whether informed consent is possible in this context. 8
Historically, ethical committees have been directed to consider pregnant people as ‘vulnerable’. It is possible that this view has led to a lack of advancement of the science around perinatal interventions and an underrepresentation of pregnant people in research.6,8,9 There is no evidence that pregnant people lose capacity to consent for research. Moreover, they are required to consent to a range of tests and procedures during the antepartum and intrapartum period. Previous research has identified that pregnant people value the opportunity to participate in research, and evidence-based medicine is important to providing high-quality intrapartum care.10,11
For these reasons, the clinical setting in which a trial is undertaken is crucial to the development of a suitable consent process. 12 While studies have looked at patient and/or clinician experience of consent in emergency settings, few have looked at maternity care specifically, and those that have present differing views on a recommended consent pathway.6,7,12–14
The Royal College of Obstetricians and Gynaecologists (RCOG) have produced guidance around obtaining valid consent for perinatal research where consent is time critical. This guidance considers oral consent in acute settings, followed by written consent when the situation allows, to be appropriate when the event is rare. 15 In the New Zealand setting, modifications to the standard consent process (e.g. opt-out, abbreviated or deferred consent) are considered on a case-by-case basis by ethics committees but are uncommonly used in maternity research. 16
The Baby head ElevAtion Device (BEAD) Feasibility Study was a double-blinded randomised controlled trial with patients becoming eligible for enrolment to the study in labour when their cervix was fully dilated, and there was a recommendation for the baby to be born by caesarean section. As a caesarean section at full dilatation (CSFD) is difficult to predict and occurs in fewer than 3% of all births, we obtained ethical approval to undertake a two-stage consent process using initial abbreviated intrapartum consent and full postpartum consent after the intervention. This Study Within a Trial (SWAT) aimed to better understand the patient and clinician experience of an approach to participate in a study using a two-stage consent process.
Methods
The research setting
New Zealand has a publicly funded, midwifery-led maternity care system. Most pregnant people receive continuity of care from a community-based lead maternity carer, typically a midwife. Maternity care is free for eligible residents and includes antenatal, labour and birth, and postnatal care. Those with complexities during pregnancy, labour and birth or postpartum receive additional care from hospital-based obstetric and midwifery teams.
The BEAD Feasibility Study was a randomised controlled trial assessing the feasibility of recruitment to a larger trial (BEAD Study) investigating the use of a head elevation device (the Fetal Pillow®) at CSFD. 17 The Fetal Pillow® is a small, inflatable silicone balloon inserted into the vagina under the baby’s head immediately prior to CSFD and inflated with 180 mL of saline. It is marketed to elevate the baby’s head out of the pelvis despite a lack of high-quality evidence for its effectiveness and is commonly used worldwide at surgeon discretion.18,19
Over 1300 births within retrospective studies and one small randomised controlled trial (RCT) have been reported in the literature with no documented complications. The absence of reported harm supports the classification of this study as low risk, with no added risks beyond standard clinical care. The study took place at two major tertiary maternity hospitals in Auckland, New Zealand (Auckland City and Middlemore Hospitals).
Consent process for BEAD Feasibility Study
The BEAD Feasibility Study used a two-stage consent process approved by the ethics committee, designed to accommodate the urgent nature of CSFD. Information was made available during pregnancy via a study website, patient brochures and posters in antenatal clinics and around the hospital. Patients were still eligible if they had not seen this prior to labour.
Stage 1: Abbreviated intrapartum consent
Eligible patients (aged ≥16 years, with a live, singleton, cephalic pregnancy at ≥37 weeks’ gestation) were invited to take part in the study. Intrapartum consent was sought at the time of surgical consent for CSFD. Depending on randomisation, the device was inflated with 180 mL of saline or sham-inflated, with both patient and surgeon blinded to allocation.
A study-specific standardised script (Supplementary File 1), designed to take approximately 1–2 min to read aloud, was used alongside verbal explanations and written materials to provide essential information about the intervention while minimising cognitive burden during labour. The time available for patients to decide about participation varied by clinical urgency; typically, patients had up to 60 min, while those requiring urgent Category 1 CS were often not approached. Both the patient and the clinician obtaining consent signed the abbreviated consent form to document agreement.
All clinicians involved received training and resources on the consent process, including an instructional video and written guidance, to ensure consistent and ethically sound communication. A midwife was present with patients during the intrapartum consent discussion, serving as an advocate and providing support.
Stage 2: Postpartum full consent
Following delivery, participants were re-approached by research staff to reaffirm their consent for the intervention, which was a one-off procedure, and to obtain additional consent for the collection of maternal, neonatal and future pregnancy data. This process used the standard Health and Disability Ethics Committees consent form and patient information sheet, allowing participants to ask questions and withdraw from the study at any time without affecting their clinical care.
Sample and recruitment
Patients
Recruitment for the BEAD Feasibility Study occurred at both hospital sites over 12 months. Patients who consented or declined during labour were approached, before discharge, to complete an opt-in consent experience survey co-developed with consumers, research staff, obstetricians and midwives (Supplementary File 2). Those who gave abbreviated intrapartum consent were approached shortly after giving full postpartum consent, while decliners were approached if research staff were available, leading to a lower approach rate.
Clinicians
Between February and July 2024, clinicians at both recruitment sites were invited via email to participate in semi-structured interviews exploring barriers and enablers to recruitment to the BEAD Study. Purposive sampling was used to include clinicians who actively recruited patients in labour and those who were more hesitant to approach patients. Those who agreed were sent a participant information sheet and consent form, and interviews were undertaken by an independent clinician.
Data collection
Patients
Patients were invited to complete the short, face-to-face consent experience survey with a member of the research team. Patients were often with family during the survey, and this was supported by the research team. Data collection took place between 14 August 2023 and 5 August 2024. The survey included a combination of open and closed questions (using Likert-type scales) about the experience of intrapartum consent (Table 1). De-identified survey responses were recorded directly into a REDCap database hosted by the University of Auckland. 20 Demographic details and the number of eligible patients in the time frame (to calculate the proportion of those eligible who were approached or recruited to the BEAD Feasibility Study) were obtained from the study screening log.
Topics explored in patient consent experience survey and clinician interviews.

Clinicians
Topic guides for the semi-structured interviews were developed, then piloted and conducted via Zoom using a clinician independent of the study team. Interviews were recorded, and study transcripts were checked for accuracy and anonymised.
Data analysis
Quantitative questions were analysed descriptively, and statistical comparisons were made using chi-square tests. Some responses were combined for statistical testing, and denominators varied excluding unanswered questions.
Reflexive thematic analysis was used for qualitative data and highlights the researcher’s active role in knowledge production. 21 We used a collaborative approach between three co-authors (J.W., R.C. and E.B.) to ensure a rich and ethnically appropriate interpretation of the data. 22 All data were read by J.W. and R.C. to allow familiarisation before using Microsoft Excel and NVivo version 12, to organise codes and develop themes using an inductive approach. EB (Ngāti Awa, Te Ātiawa ki Whakarongotai, Te Arawa and Ha’ano (Tonga)) provided a Māori (indigenous peoples of Aotearoa New Zealand) perspective and allowed us to explore responses from the sub-group of Māori patients, to support our consent approach being culturally appropriate. Coded data sets were subjected to further, in-depth analysis to identify additional themes and illustrative quotations. Quotes from survey participants (P) are coded (e.g. P12C, with C indicating consent for the BEAD Feasibility Study and D indicating decline) and clinician quotes DR for doctor and MW for midwife. Reflexivity was used throughout data analysis and collection as a methodological tool to enhance the credibility of the findings. 23 Ethical approval for the Feasibility Study and the qualitative research was given by Northern A Health and Disability Ethics Committee, New Zealand (2023 AM 15526).
Results
Over 12 months, 186 patients were approached to participate in the BEAD Feasibility Study, of whom 128/186 (69%) consented. A greater proportion of those who consented to the BEAD study (111/128, 87%) completed the survey, compared to those who declined to take part (38/58, 66%) (Figure 1). Two people who provided intrapartum consent did not reaffirm consent for the trial when seen postpartum, both declining for data privacy concerns.
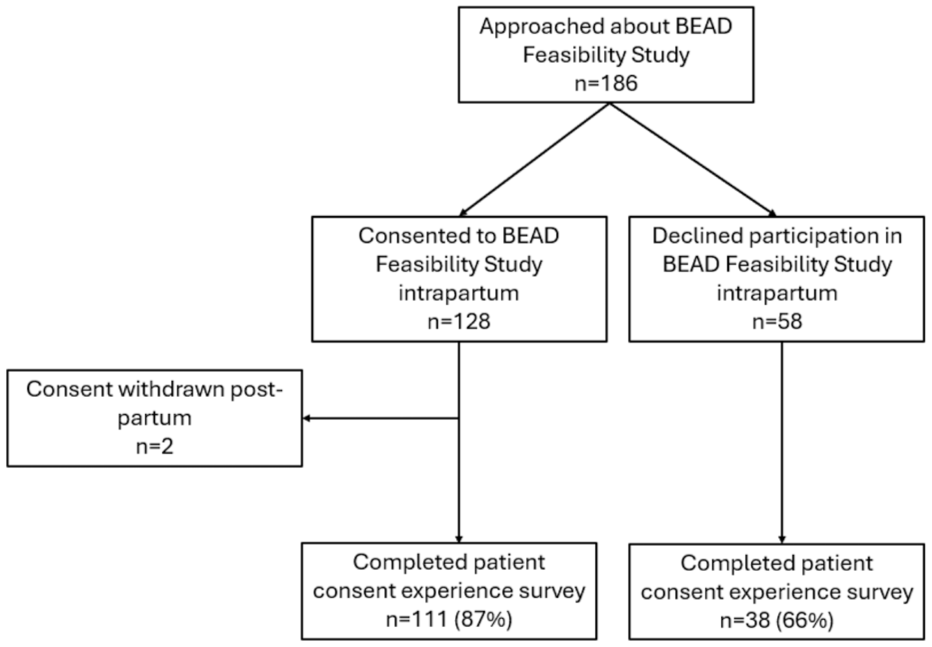
Flow chart relating to patient experience survey for those approached about the BEAD Feasibility Study in labour.
Survey responders were representative of the ethnic groups of those eligible for the BEAD Feasibility Study (Table 2). The quantitative elements of the patient experience survey are summarised in Table 3 along with illustrative comments. Most participants received verbal information and felt it was sufficient to make a decision; however, those who declined participation were significantly less likely to recall being approached and more likely to want information at an earlier time. Differences were also noted in perceptions of information adequacy between consenters and non-consenters (Table 3).
Patient survey responders compared to patients eligible for the BEAD Feasibility Study, by ethnicity.
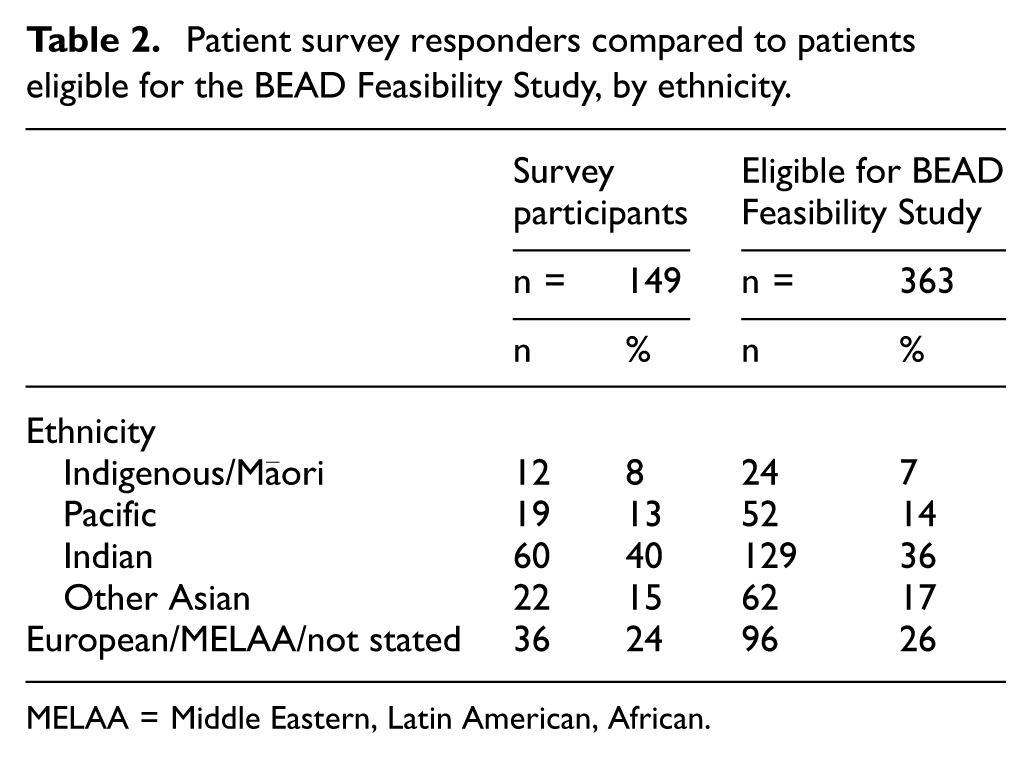
MELAA = Middle Eastern, Latin American, African.
Patient survey findings by consent status among responders.
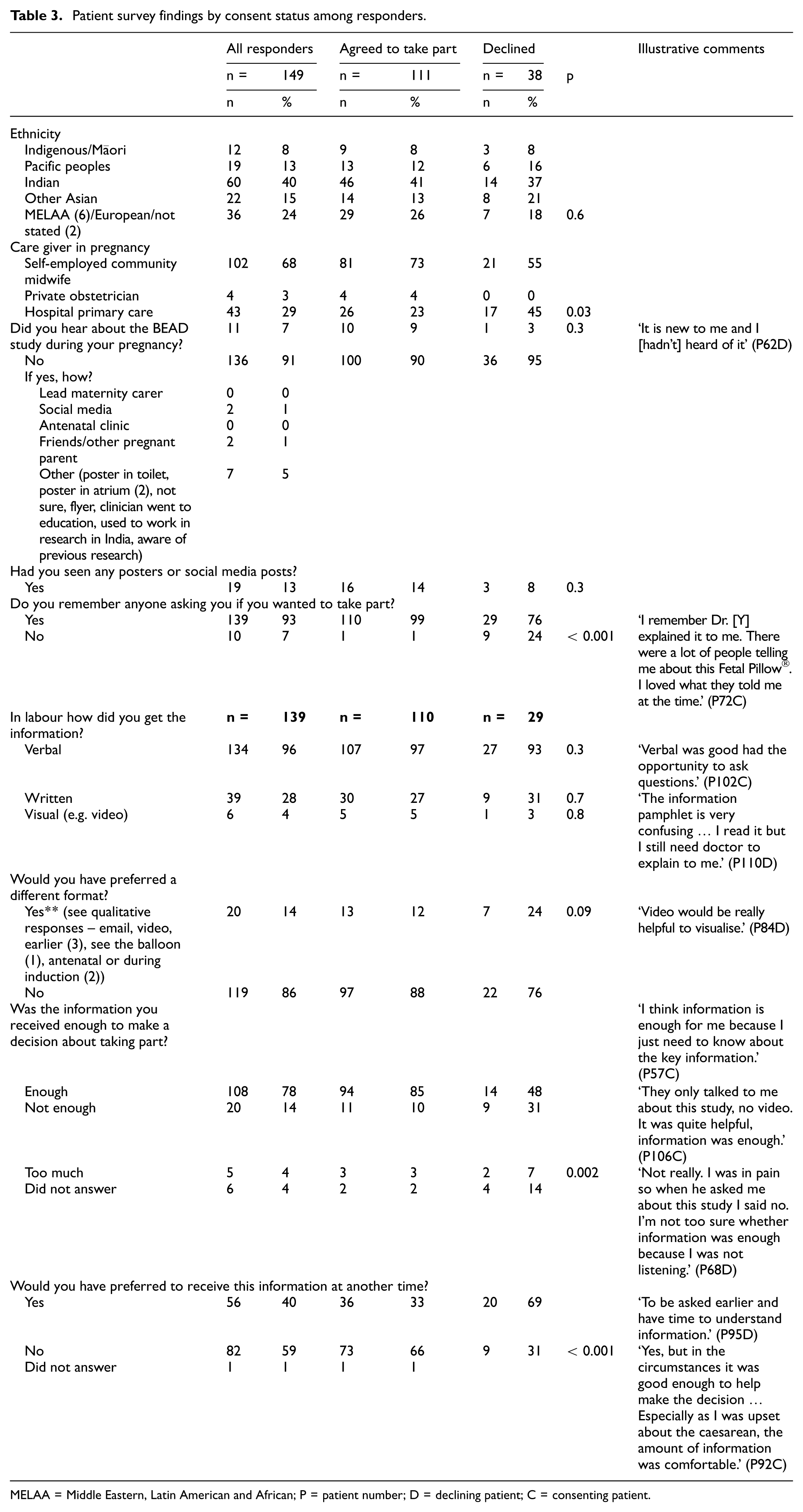
MELAA = Middle Eastern, Latin American and African; P = patient number; D = declining patient; C = consenting patient.
We asked patients who responded to the consent experience survey but had declined intrapartum participation in the BEAD Feasibility Study, if there was anything that could be changed that would make them more likely to participate if asked in a future pregnancy. Twenty-four of the 38 survey responders provided a comment, and many brought up the timing of information: Knowing ahead of time. Before the pain. Nothing else, just the timing or when still pregnant. Coming for my appointments would like my midwife to tell me about it and we could have done our own research on it. (P122D)
Another patient pointed out that: Would have been good to have had the information earlier and not during a vulnerable moment just before CS, as difficult to make a decision when you’re on drugs and being given so much information during a situation like that. (P143C)
We asked all survey responders how it felt to be asked about the study in labour and what were the reasons they chose to take part or not take part in the BEAD Feasibility Study. Three overarching themes (each with two sub-themes) were developed from the data that provided insight into their decision-making: (1) benefits of participation, (2) trust in healthcare and (3) overwhelmed in the moment (Table 4).
Patient barriers and enablers to participation in clinical trial when approached in labour.
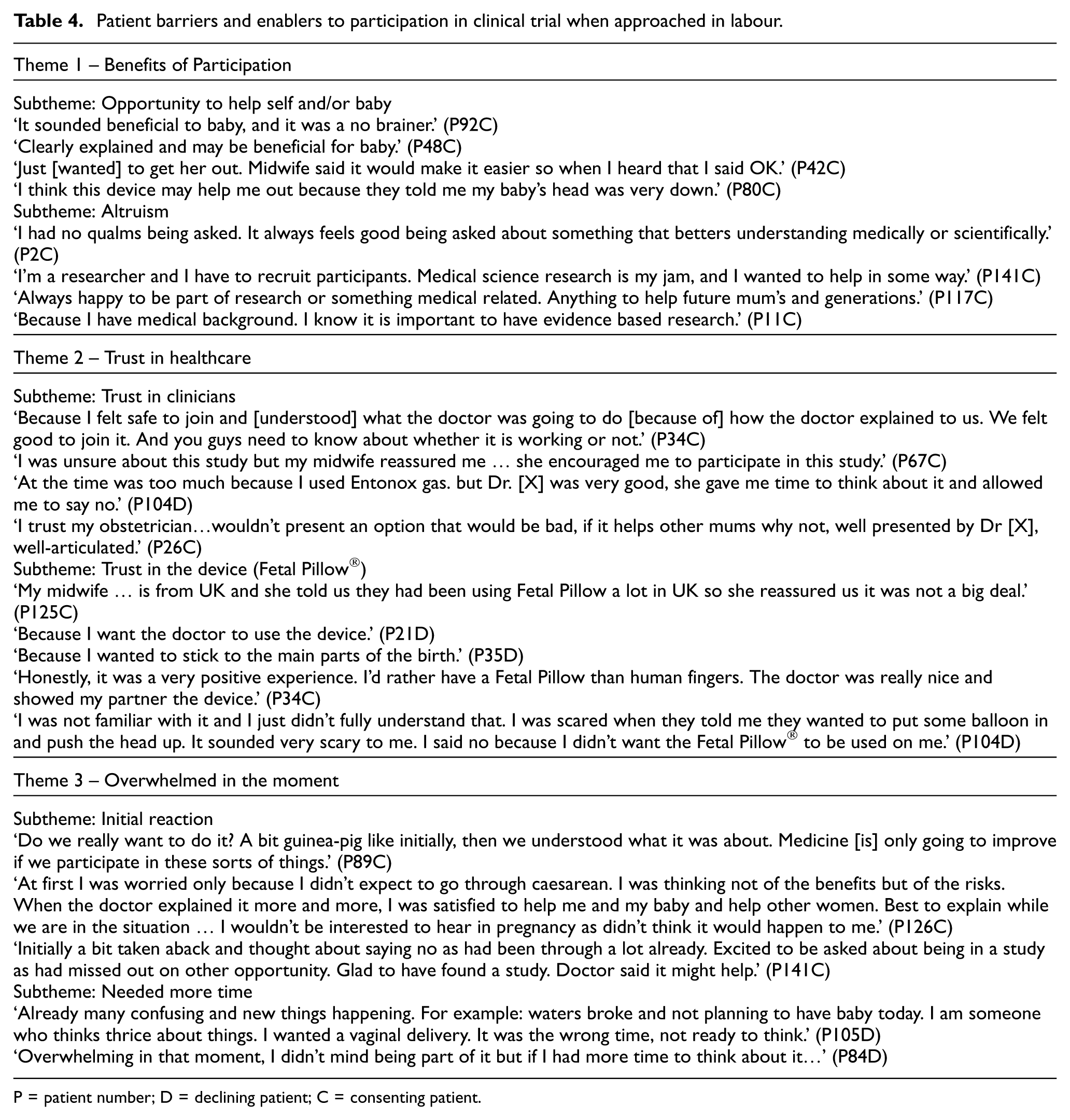
P = patient number; D = declining patient; C = consenting patient.
Benefits of participation
For self and/or baby
For those survey responders who chose to take part in the study, they commonly reported they were ‘happy to join’ and ‘give it a go’ and thought it would be ‘helpful’ or ‘beneficial’ either for their situation (needing a caesarean section in advanced labour) and/or for their baby. Clear explanations from clinicians about the relevance of the study were also important in their decision to take part.
Altruism
Other survey responders recognised an opportunity to make a difference to mums and babies in the future as well as appreciating the importance of research in women’s health. Despite the timing of discussion about this study (in advanced labour), for many, a combination of benefits for self and/or baby and of altruism was the motivation for their participation. ‘Made me feel like I was not only helping myself but potentially doing greater good. Happy to be asked’ (P41C).
Trust in healthcare
In clinicians
For those that consented to the BEAD Feasibility Study, trusting the clinician and the information they provided about the study during labour was an important factor in their decision to participate. This was reported less commonly for those who declined. Women looked to the healthcare professionals providing their care during labour for support and guidance.
In the device (Fetal Pillow®)
The Fetal Pillow® has been available for 10 years and used at surgeon discretion at one of our recruitment sites (as well as globally). There was mixed understanding about the device with some survey respondents misunderstanding the device to be ‘new’ and therefore a deviation from standard care, while others agreed or wanted the device to be used despite no evidence for benefit.
Overwhelmed in the moment
Initial reaction
Some survey responders described an initial reaction of feeling overwhelmed at having the study introduced to them in labour. However, when the study was explained clearly by clinicians, many felt comfortable to participate.
More time
For a small number of survey responders who declined participation in the BEAD Feasibility Study, the already stressful situation of an unexpected event (caesarean section) meant that it was not the right time for them to discuss research.
Sub-analysis of Māori responses (equity focus)
There were 11 survey responders who self-identified as Māori, representing 8% of all survey participants. The small sample size limited thematic analysis with an equity lens. However, their responses revealed similar themes to non-Māori participants but with less positivity. This was mainly related to the theme of being overwhelmed in the moment, with Māori participants more likely to report that being approached for intrapartum consent was a suboptimal experience, for example, ‘it felt a bit rushed …’ (P140D), and that the information presented was ‘very confusing’ (P110D). Yet it was reassuring that the majority of Māori patients (8/11) who were approached and completed the survey consented to participate in the BEAD Feasibility Study.
Clinician interviews
Interviews were conducted with 12 doctors (6 consultants and 6 registrars, half from each recruitment site) and 6 hospital midwives (all from Middlemore Hospital). Clinicians could appreciate that ‘trying to get consent in an emergency is difficult… not impossible… but a challenging thing to do’ (MW1), however feasible ‘as long as people can understand what’s involved’ (MW2) (Table 5).
Clinician responses to questions around the BEAD Feasibility Study consent process.
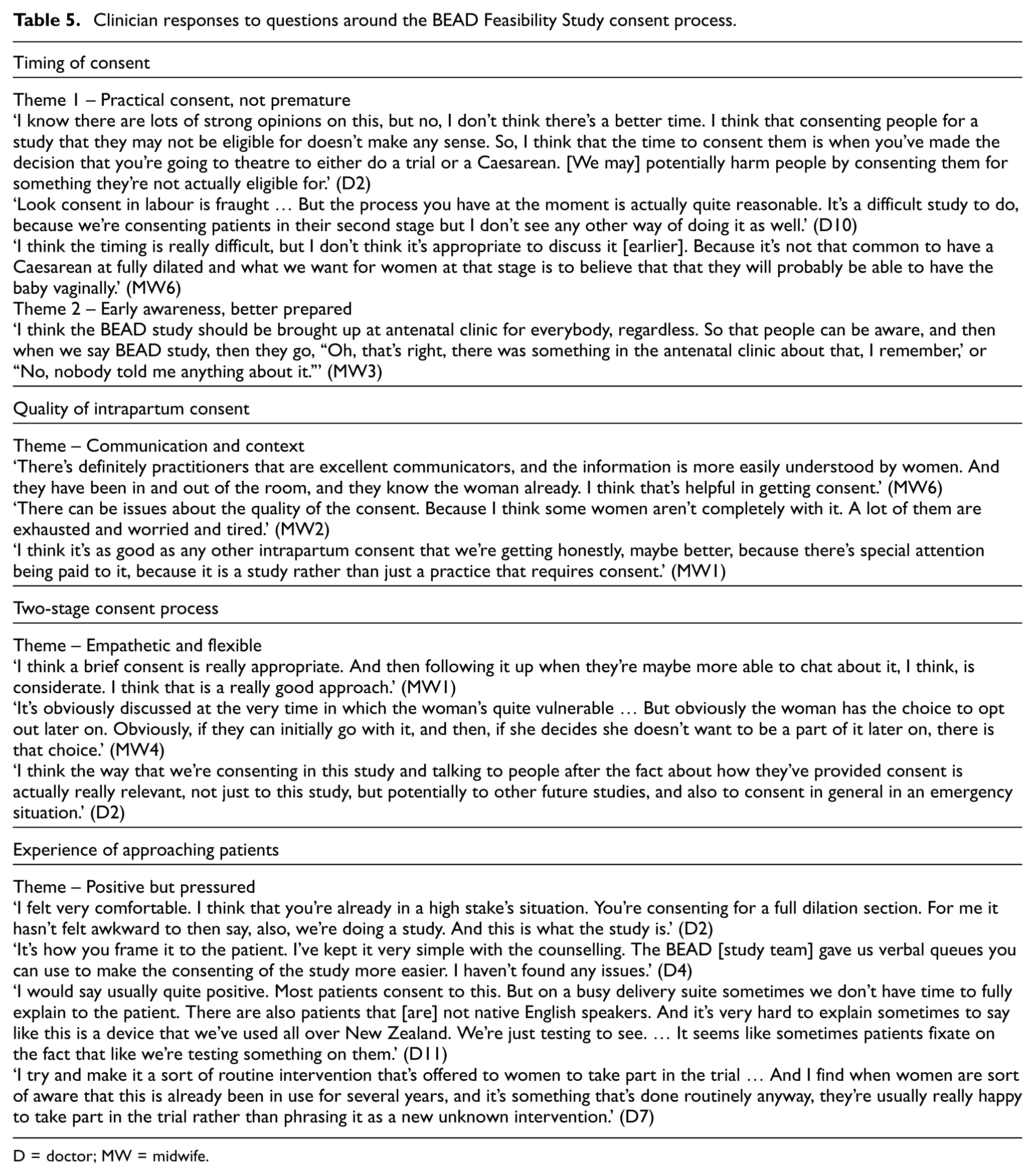
D = doctor; MW = midwife.
Timing of consent
Themes – (1) practical consent, not premature and (2) early awareness, better prepared
The optimal time to consent patients for the BEAD Study was felt to be ‘tricky’ or ‘difficult’ by most who were interviewed. Many felt discussing the study with those who become eligible (at decision for CSFD) was ‘practical’ and the most pragmatic way to conduct the study but that the addition of antenatal information for patients may be helpful.
Quality of intrapartum consent
Theme – communication and context
Clinicians observed that the quality of intrapartum consent varied due to factors such as time constraints, patient condition and how clearly clinicians were able to communicate study information, which sometimes limited patient understanding. Despite these challenges, many felt the process was appropriate and that the research context often improved consent quality through greater attention and repetition.
Two-stage consent process
Theme – empathetic and flexible
Clinicians viewed the two-stage consent process positively, noting it enabled informed decision-making despite the challenges of labour. They felt the abbreviated intrapartum consent followed by full postpartum consent was a ‘good process’ that respected women’s autonomy and could serve as a model for future studies.
Experience of approaching patients
Theme – positive but pressured
Doctors report mixed experiences when approaching patients in labour about the study. Some reported feeling ‘very comfortable’ (DR2) and had ‘no issues’ (DR4) and made trial participation a ‘routine intervention’ (DR7). Others felt on a busy delivery unit they didn’t always have time to ‘fully explain to the patient’ (DR11) or faced other challenges such as language barriers.
Discussion
This study explored the acceptability of a two-stage consent process – abbreviated intrapartum consent followed by full postpartum consent – for an intrapartum randomised controlled trial (the BEAD Feasibility Study) investigating use of the Fetal Pillow® during CSFD. The process was broadly acceptable to both patients and clinicians. Overall, 69% of those approached during labour consented to take part, and only two withdrew postpartum citing data privacy concerns. Recruitment exceeded feasibility targets, suggesting that this consent approach is practical in time-sensitive obstetric settings.
Clinicians predominantly used verbal explanations when introducing the study during labour, and most consenting patients felt this was sufficient. Patient experiences centred around three core themes: (1) benefits of participation, (2) trust in healthcare and (3) feeling overwhelmed in the moment. While the study context was generally well accepted, both clinicians and patients – especially patients who declined participation – highlighted the importance of more time to consider involvement. Clinicians also reflected that while intrapartum consent quality could be affected by clinical pressures, the two-stage model supported informed choice and may offer a respectful, flexible framework for future intrapartum research.
As recognised in other maternity studies and from our findings, improved antenatal awareness of a research study may help facilitate intrapartum consent discussions.12–14,24We need to balance the availability of information for all who wish to engage, with the potential to overburden and create excessive anxiety about an event (CSFD) which is unlikely. 15 Indeed, a recent study using verbal consent with written retrospective consent for a prospective cohort study around postpartum haemorrhage reported that patients were happy to be presented with pertinent information to decide about participation in research within an emergency setting, although only those who consented were included. 14
Fewer than 3% of birthing women were eligible for the BEAD Feasibility Study and we cannot predict prior to advanced labour who these will be. There is no evidence to suggest harm to mother or baby from use of the Fetal Pillow®, and therefore, we are continuing the two-stage consent process for the larger BEAD Study. Following our analysis of feedback from survey participants and clinicians, we will provide further education to community-based maternity providers about the study to share with pregnant people and are now evaluating providing study information at induction of labour. The abbreviated intrapartum consent has proven successful for recruitment in a time-limited (emergency) situation. The follow-up full postpartum consent provides a safety net, allows for more in-depth discussion and debrief, and an option for patients to withdraw their data from the study.
Strengths and limitations
A key strength of this study is the dual perspective of patients and clinicians. Importantly, we included patients who declined participation in the BEAD Feasibility Study when approached in labour. The rate (31%) of patients declining participation suggests that individuals felt able to make an autonomous decision, supporting the ethical integrity of the consent process. Their views and feedback are crucial for understanding and improving the patient experience and supporting recruitment. Survey participants were representative by ethnicity of those eligible for participation in the BEAD Study. The face-to-face approach provided a high response rate compared to other survey methods (e.g. email) and offered an opportunity for participants to discuss their experience of intrapartum consent and have their questions answered.
Limitations included fewer responses from those who declined participation in the BEAD Feasibility Study compared to those who consented. This is partly explained by an opportunistic approach to those who declined. The research team member who surveyed patients also requested postpartum consent which may have biased responses. In addition, it was not always possible or appropriate to complete the survey at the time the research staff visited due to patient factors which may have led to a less representative sample. This consent process has been designed and evaluated in a single study, and therefore, these findings may not be generalisable in all intrapartum settings.
Future research
Based on feedback from survey participants wanting earlier information, a further SWAT is planned.
Patients presenting for induction of labour at Auckland Hospital will receive a BEAD trial brochure. Regardless of mode of birth, they will be invited to complete a survey postpartum about how this information was received. This will help us assess the acceptability of providing trial information earlier, even to those ultimately ineligible for participation.
Conclusion
This mixed-methods study of patient and clinician experience of recruitment to an intrapartum study in an ‘emergency setting’ was largely supportive of abbreviated consent. Understanding the views of eligible patients on the consent process is crucial for optimising both patient experience and recruitment. We are continuing with our two-stage consent model including abbreviated intrapartum consent for the planned larger BEAD trial while trying to provide more information antepartum. This may be a useful consent model for other researchers to consider when undertaking peripartum trials involving recruitment in emergency situations and where opt-out or deferred consent is not available.
Supplemental Material
sj-docx-1-ctj-10.1177_17407745251412767 – Supplemental material for Exploring abbreviated intrapartum consent: A mixed-methods study of patient and clinician perspectives in the Baby head ElevAtion Device trial
Supplemental material, sj-docx-1-ctj-10.1177_17407745251412767 for Exploring abbreviated intrapartum consent: A mixed-methods study of patient and clinician perspectives in the Baby head ElevAtion Device trial by Jordon Wimsett, Charlotte Oyston, Robin Cronin, Erena Browne, Maria Boston, Lih Hwan Huang, Meghan G Hill and Lynn Sadler in Clinical Trials
Supplemental Material
sj-docx-2-ctj-10.1177_17407745251412767 – Supplemental material for Exploring abbreviated intrapartum consent: A mixed-methods study of patient and clinician perspectives in the Baby head ElevAtion Device trial
Supplemental material, sj-docx-2-ctj-10.1177_17407745251412767 for Exploring abbreviated intrapartum consent: A mixed-methods study of patient and clinician perspectives in the Baby head ElevAtion Device trial by Jordon Wimsett, Charlotte Oyston, Robin Cronin, Erena Browne, Maria Boston, Lih Hwan Huang, Meghan G Hill and Lynn Sadler in Clinical Trials
Footnotes
Acknowledgements
The authors would like to thank all the patients and clinicians who participated in the interviews.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Mercia Barnes Trust (MB 23-1 – Jordon Wimsett), HRC research delivery activation grant (HRC 23/735/A – Charlotte Oyston) and HRC Clinical Practitioner Research Fellowship (HRC 22/016 – Lynn Sadler).
Trial registration
ANZCTR. This mixed-methods research was undertaken as part of the BEAD Feasibility Study (trial registration number: ACTRN12623000758606; prospectively registered on 12 July 2023.
Supplemental material
Supplemental material for this article is available online.
