Abstract
Background:
The results of rehabilitation clinical trials can be negatively affected by adherence to trial protocols. Adherence is multi-factorial, but studies often look at adherence factors separately. Therefore, a systematic review to appraise and synthesise the evidence is warranted to determine the barriers, facilitators and predictors associated with adherence to inpatient rehabilitation trial protocols, whether and how factors interact with one another, and how adherence to rehabilitation protocols can be optimised.
Methods:
A mixed-methods systematic review was conducted and reported in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). Databases searched were PubMed (Ovid), EMBASE (Ovid), MEDLINE (Ovid), CINAHL (Ovid), PsycINFO (Ovid), Cochrane Library, Health Technology Assessment Database, Web of Science and grey literature up to April 2024. A cohesive, integrated methodology was employed, leveraging the Consolidated Framework for Implementation Research (CFIR) 2.0, to transform, synthesise and integrate data from various methodologies to address the review objectives.
Results:
Twenty-seven studies met the inclusion criteria (randomised controlled trials, qualitative studies related to randomised controlled trials or mixed-methods). Most of the studies were in stroke (n = 17), but other studies included neurological, respiratory, cardiovascular, post-surgical, osteoarthritis and elderly medical. Multiple factors affecting adherence protocols were identified. Adherence was measured in various ways, and setting pre-specified adherence levels was uncommon.
Conclusion:
Adherence to inpatient rehabilitation trial protocols is multi-dimensional and multi-factorial. Consensus of adherence measurement and interpretation of adherence levels is needed to make meaningful comparisons between trials. A standardised approach, including adopting a traffic light system, would enable trialists to implement changes mid-trial or stop the trial to avoid research waste. Adopting approaches from behavioural science in the design and conduct of inpatient rehabilitation trials may overcome some of the behavioural barriers identified and optimise adherence for those delivering and receiving the intervention.
Review registration:
Prospective Register of Systematic Reviews, registration number CRD42021270121.
Background
People with stroke often experience a range of impairments affecting function and quality of life, and rehabilitation aims to promote recovery. Millions of pounds are spent on research evaluating rehabilitation interventions for people after stroke, but the results can be negatively affected by adherence to the trial protocols. Adherence is a key variable influencing the outcome of clinical trials and is the bottom-line measurement of implementation fidelity. 1 Thus, researchers need to identify ways to optimise adherence in the planning, design and delivery of stroke rehabilitation trials to ensure trial funds are well used and that trials accurately determine treatment effect and patient safety. Adherence to intervention protocols refers to the degree to which the behaviour of trial participants corresponds to the intervention assigned to them. 2 This implies the onus is on the participants; however, it is imperative to identify and address all factors affecting adherence in stroke rehabilitation trials, including the behaviour of those delivering and receiving the intervention and the context in which the trial is delivered.
Protocol adherence to a recently conducted multi-centre feasibility randomised controlled trial (fRCT, Standing Practice In Rehabilitation Early after Stroke (SPIRES)) for people with severe stroke during their inpatient stroke rehabilitation was low. Patient-, therapist- and organisation-factors all were identified as affecting adherence to the functional standing frame programme. 3 A recommendation from the fRCT was to determine what factors affect adherence to inpatient stroke rehabilitation trial protocols before progressing to a definitive trial. Adherence is multi-factorial, but studies often look at adherence factors separately, focusing on one component only. Therefore, a systematic review was warranted to determine the barriers and facilitators to adherence in inpatient stroke rehabilitation trial protocols, and whether, and how, factors interact with one another. It was unclear whether factors affecting adherence to SPIRES 3 were specific to stroke survivors; therefore, this review included all inpatient physical rehabilitation trials to determine any similarities and lessons that can be applied to future design of stroke rehabilitation inpatient trials.
The Consolidated Framework for Implementation Research (CFIR) 2.04,5 was used as a basis to synthesise and integrate the data. The CFIR is a meta-theoretical framework used by implementation scientists to systematically identify factors that may emerge in various, multi-level contexts that influence implementation.3–6 The updated CFIR 2.0 includes constructs from Michie et al.’s Behaviour Change Wheel (BCW). 7 The BCW is an evidence-based model consisting of three layers that recognise behaviour as part of an interacting system involving an individual’s capability to perform a behaviour (e.g. knowledge/skills), the opportunity (e.g. autonomy) and motivation (e.g. commitment) to carry out that behaviour. It is pertinent to both recipients and providers of inpatient rehabilitation trial protocols. Thus, the CFIR 2.0 provided a systematic way to classify factors affecting adherence and organise both qualitative and quantitative data to identify ways of optimising adherence to inpatient rehabilitation trial protocols.
The aim of this review was to determine the barriers, facilitators and predictors associated with adherence to inpatient rehabilitation trial protocols, whether and how factors interact with one another, and how adherence to rehabilitation protocols can be optimised.
Methods
The systematic review protocol was registered with the International Prospective Register of Systematic Reviews (CRD42021270121). The review was reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 8
Review question
Among adults participating in inpatient physical rehabilitation trials, what barriers, facilitators, and predictors influence adherence to trial protocols, how do these factors interact within inpatient settings, and what strategies can optimise adherence?
Eligibility
The review question and eligibility criteria were constructed using the PICCO method as follows:
Participants/population: aged ≥ 18 years participating in a physical rehabilitation trial.
Intervention(s), exposure(s): any inpatient physical rehabilitation trial which measures protocol adherence.
Comparator: Not applicable.
Context: Trials conducted in hospital-based healthcare/rehabilitation settings (e.g. rehabilitation unit/ward, hospital ward) in any country.
Outcomes: (1) Determine the barriers, facilitators and predictors associated with adherence to inpatient rehabilitation protocols; (2) determine how adherence to protocols can be optimised in inpatient rehabilitation trials, that is, how the effectiveness of an intervention can be improved, and its acceptability and feasibility be increased in practice 9 and (3) determine whether adherence to inpatient stroke rehabilitation trial protocols differs from those in other health conditions.
Exclusions include adolescents (≤17 years of age); people whose primary reason for admission to the rehabilitation ward/unit was for mental health/psychological issues; and people not participating in an inpatient rehabilitation trial.
Searches
Searches of databases were performed by two researchers (A.L. and E.C.) in November 2021. Focused searches were conducted in PubMed, EMBASE (Ovid), MEDLINE (Ovid), CINAHL (EBSCOhost) and PsycINFO (Ovid) using database-controlled thesauruses. Each database was searched separately. The Cochrane Library, Health Technology Assessment Database, Web of Science and grey literature (Open Grey, Google Scholar, ETHOS (British Library repository)) were also searched. A search of reference lists from all studies that met the inclusion criteria was conducted. Search terms were related to the concepts of adherence, rehabilitation, clinical trial protocol, intervention and fidelity and were peer reviewed by an independent Information Specialist. 10 Searches were restricted to randomised controlled trials (RCTs) and qualitative studies of those participating in RCTs to address the review objectives and English language, but not by date. The searches were repeated using the same methods in April 2024. In accordance with the PRISMA-S checklist, 11 see Supplementary Material for the search strategy.
Study selection
The selection process was conducted through Rayyan. 12 After removal of duplicates, four reviewers (A.L., J.M., J.F. and E.C.) independently screened the titles and abstracts of all retrieved articles to determine eligibility. Discrepancies were resolved through discussion to obtain consensus, with the option of a third reviewer if required. Screening of the full texts of preselected articles was carried out by the same researchers, but one reviewer (A.L.) screened all full texts, and all other reviewers (J.F., J.M., E.C.) independently reviewed their allocated studies.
Data extraction
The team of reviewers independently extracted data from each study using a pre-standardised data extraction template and the Template for Intervention Description and Replication (TIDieR) checklist and guide. 13 Additional extracted data included study characteristics, participant age, gender and health condition, adherence (how this was measured and outcomes), study outcomes, themes from qualitative data and key conclusions from the study authors. One reviewer (A.L.) extracted the data from all eligible studies, and three other reviewers (J.F., J.M., E.C.) independently extracted data from their allocated studies. One author (A.L.) collated and cross-checked all extracted data. Discrepancies in the data extraction were resolved through discussion with the relevant reviewers, with the option to involve a third reviewer if required.
Synthesis method
A convergent integrated approach was used to transform, synthesise and integrate data from different data and methods of analysis to address the review questions, following the Joanna Briggs Institute (JBI) methodological guidance for the conduct of mixed-methods systematic reviews. 14 The CFIR 2.04,5 was used as a basis to synthesise, integrate and visually display the data. Quantitative data were transformed into textual descriptions to qualitise the data. The author of one study was contacted to request additional data.
Quality assessment
The Mixed-Methods Appraisal Tool (MMAT) 15 was used to assess the methodological quality (risk of bias) of all included studies before data extraction. Each study was independently assessed by two authors. The MMAT was used due to its ability to evaluate study selection bias, study design, data collection methods, sample size, intervention integrity and analysis across multiple study designs. Previous versions of the MMAT have included allocating a methodological rating score, but a more detailed presentation of the ratings in each criterion is preferred 15 to better inform the quality of the studies.
Results
Of the 5188 records in the initial searches and 483 in the repeated searches (Figure 1), 27 studies were included in the review: 24 from the initial searches, two from reference lists and one from the repeated searches.
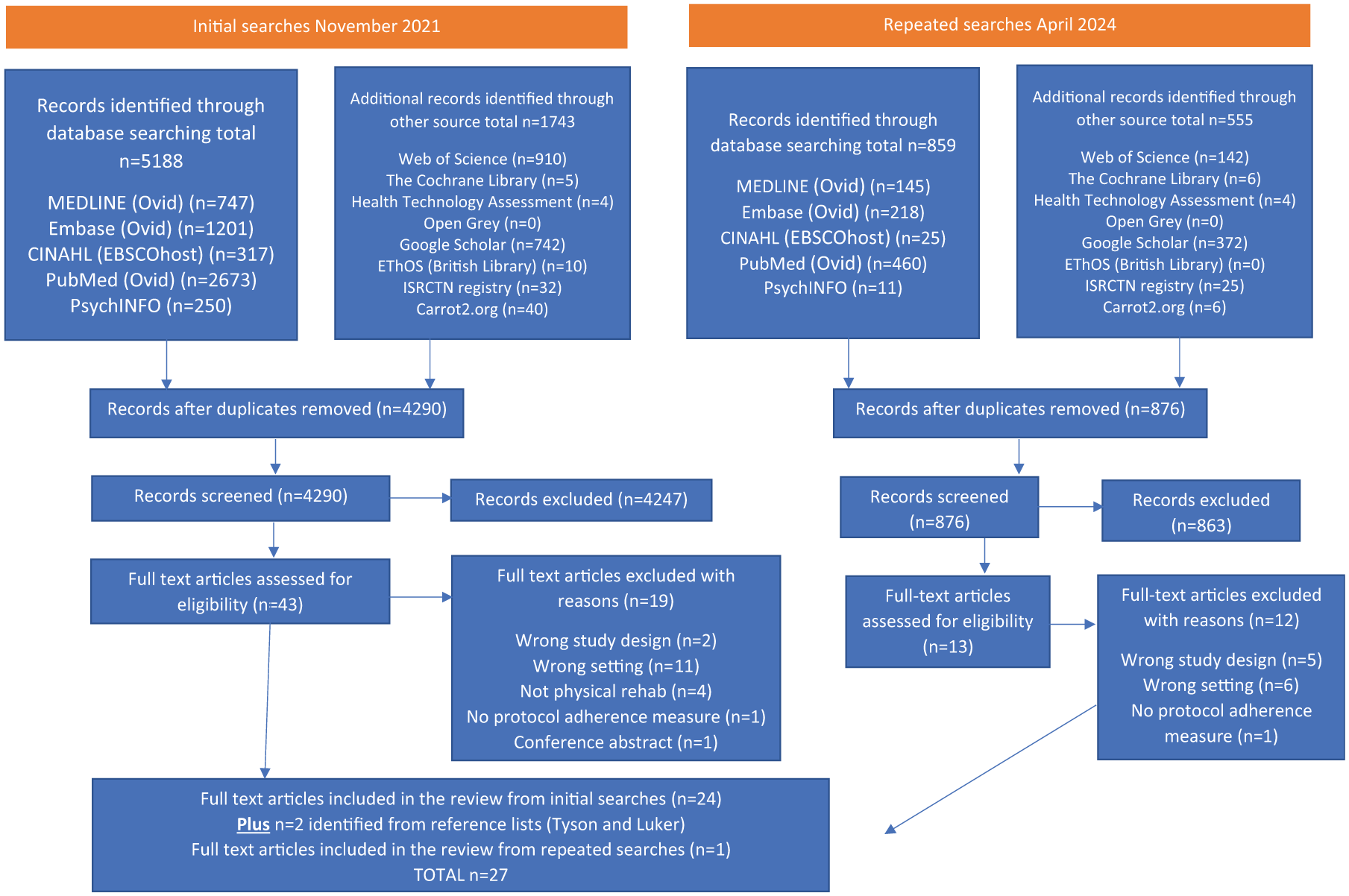
PRISMA diagram.
Characteristics of the included studies are shown in Table 1.
Characteristics of included studies.
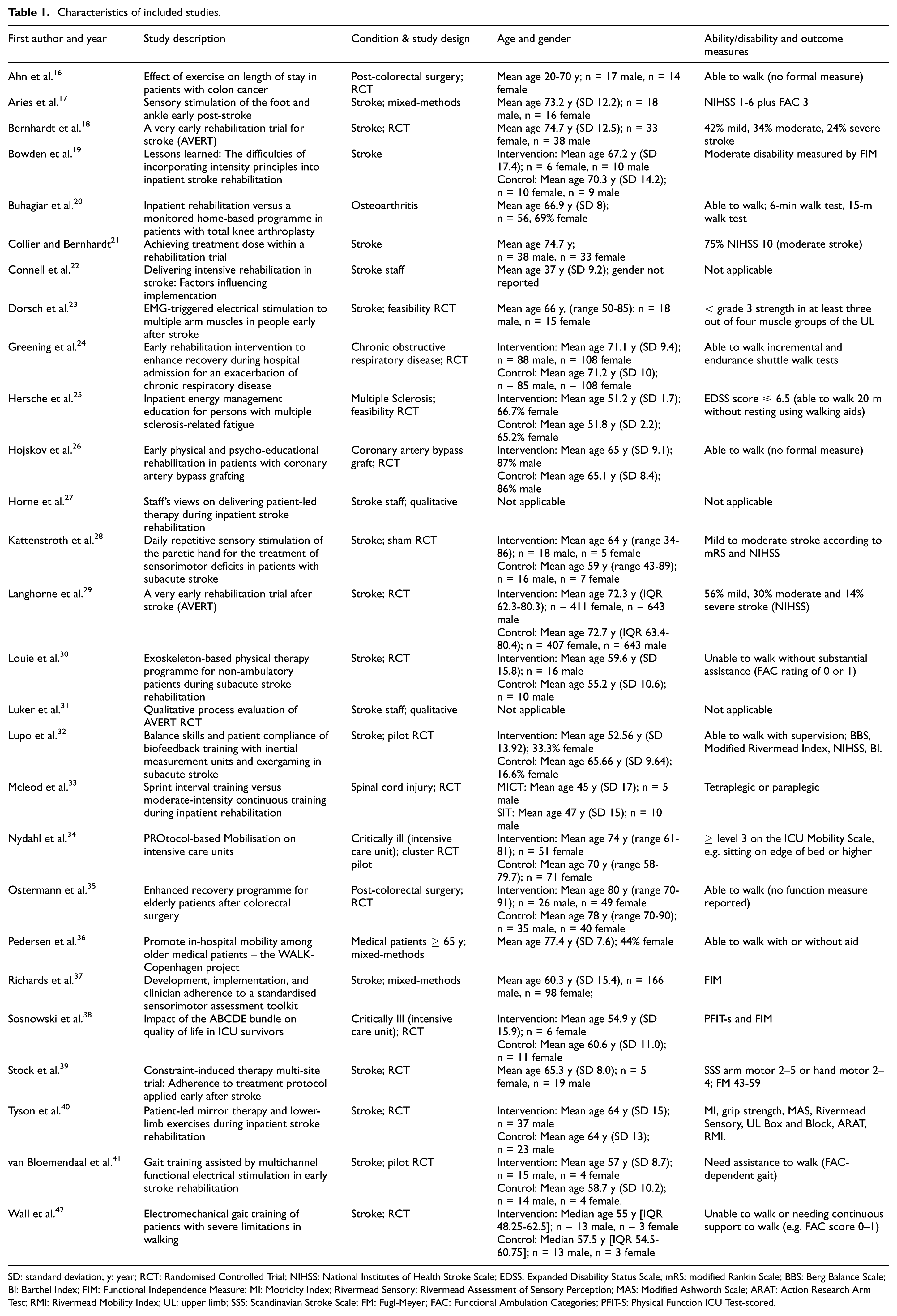
SD: standard deviation; y: year; RCT: Randomised Controlled Trial; NIHSS: National Institutes of Health Stroke Scale; EDSS: Expanded Disability Status Scale; mRS: modified Rankin Scale; BBS: Berg Balance Scale; BI: Barthel Index; FIM: Functional Independence Measure; MI: Motricity Index; Rivermead Sensory: Rivermead Assessment of Sensory Perception; MAS: Modified Ashworth Scale; ARAT: Action Research Arm Test; RMI: Rivermead Mobility Index; UL: upper limb; SSS: Scandinavian Stroke Scale; FM: Fugl-Meyer; FAC: Functional Ambulation Categories; PFIT-S: Physical Function ICU Test-scored.
Quality appraisal
All included studies were appraised for quality using the MMAT (Supplementary Table S1). The studies generally met the MMAT quality criteria, and in the case of the qualitative studies, all the criteria were met. Of the three mixed-methods studies, only one provided data to respond to all five of the criteria. Most of the RCTs met the criteria, apart from the outcome assessors being blinded. Overall, 85% (n = 23) of the studies met 60%–100% of the MMAT checklist, suggesting moderate to high quality.
Measuring adherence
Measurement of adherence varied greatly across trials in all health conditions and included adherence of both patient participants as well as staff (Supplementary Table S2). The most common adherence measure was the number of sessions (n = 17 studies; 10 out of the 14 stroke studies, two neurological condition studies, two post-surgical studies, one osteoarthritis, one chronic respiratory disease and one acute medical elderly study). Five of the six studies measuring duration of session (min) were in stroke. Only three studies used patient-reported measures (one stroke, one osteoarthritis, one post-surgical). Only seven of the included studies reported pre-specified adherence level(s). Pre-specified adherence levels included completing/attending a minimum number of sessions, delivering the protocol on a minimum number of days, the minimum/maximum duration of intervention sessions, the minimum patient satisfaction score and delivering a minimum percentage of the protocol activities. A rationale for the pre-specified adherence level was provided by one study. 34
Barriers and facilitators to intervention protocol adherence
Barriers and facilitators are presented in Tables 2–4 using the CFIR 2.0 components.4,5 Most of the data in Tables 2–4 are from stroke studies. Non-stroke data references are marked with †.
Barriers and facilitators to intervention protocol adherence (Innovation, Outer Setting and Inner Setting).
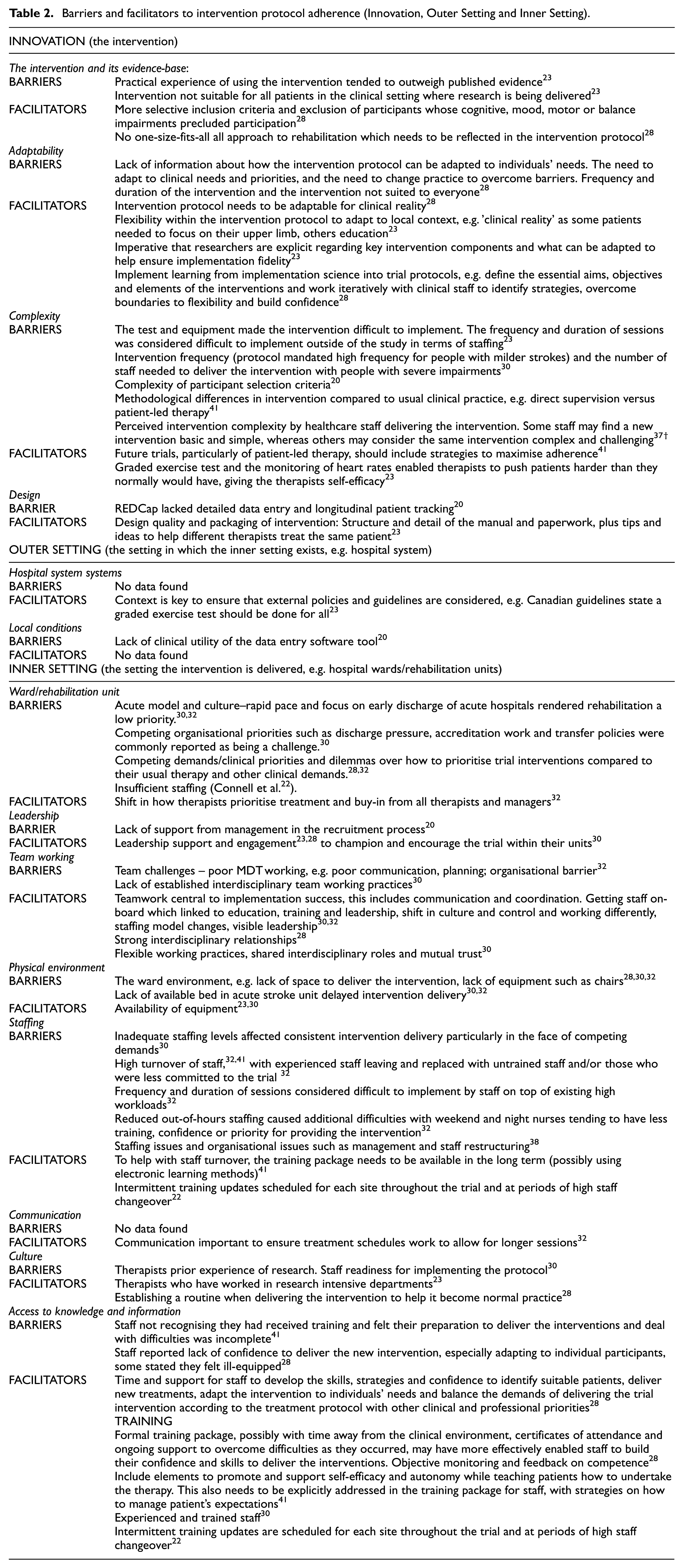
Barriers and facilitators to intervention protocol adherence (Individuals and Innovation).
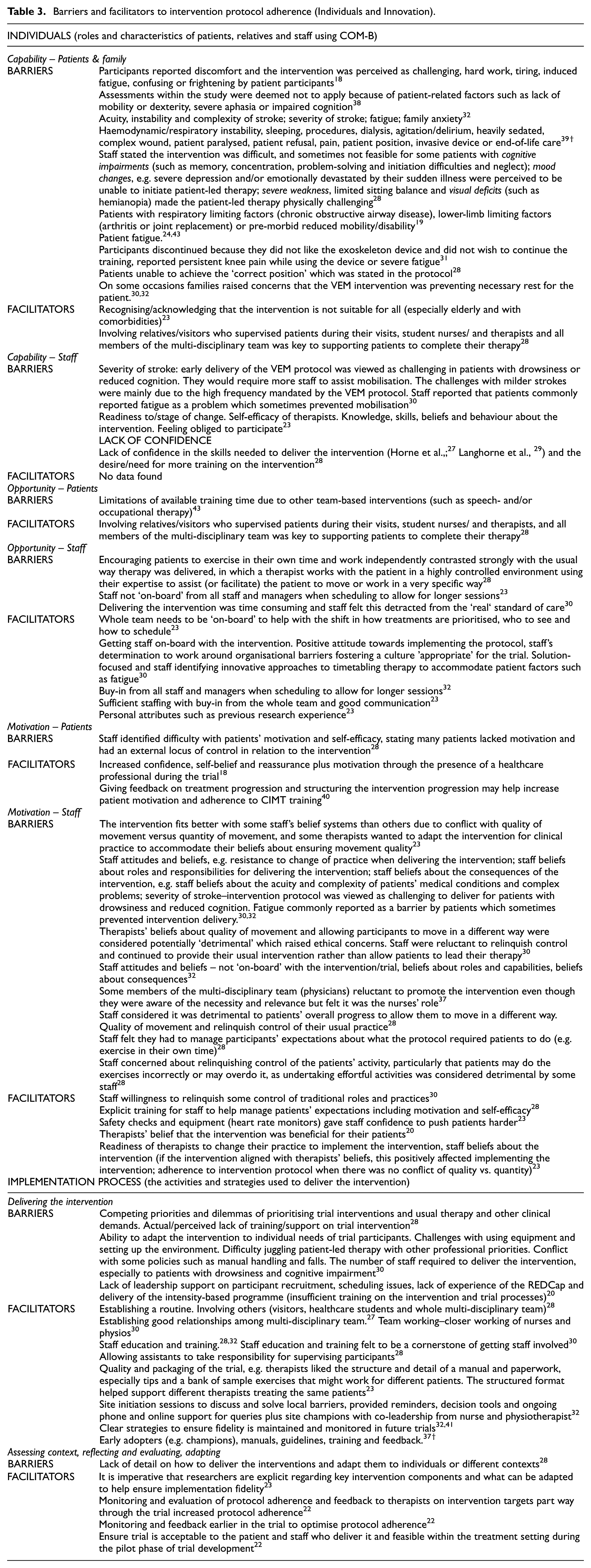
Staff and patient experiences of inpatient rehabilitation trials.

Protocol adherence in inpatient rehabilitation trials of stroke versus other health conditions
Protocol adherence in inpatient rehabilitation trials varied across all conditions, as well as across all stroke trials (see Supplementary Table S3).
Staff and patient experiences
Six studies captured staff and patient experiences of participating in inpatient rehabilitation trials.17,19,22,27,29,31 Only one study 17 captured patient experiences, via daily diaries and focus groups. The other five studies captured staff experiences via semi-structured interviews, focus groups or a quantitative survey (Table 4). No studies captured both patient and staff experiences.
Predictors of adherence
Only one study highlighted predictors of protocol adherence. 40 This stroke study identified demographic and clinical characteristics as predictors of protocol adherence (age, side of weakness, time since stroke, treatment group, neglect and sensation). Two other stroke studies identified patient demographic and clinical characteristics that should be considered in future inpatient rehabilitation trial protocols, although they were not specifically reported as predictors. Connell et al. 22 identified concerns about the suitability of the intervention in patients who were elderly or with comorbidities, and Stock et al. 39 suggested consideration of modifying protocols according to patients’ age, sex and stage after stroke.
Discussion
Adherence is a key variable influencing the outcome of clinical trials, and appropriate consideration of adherence is widely recommended.13,43,44 To accurately determine treatment effect and inform clinical practice, it is imperative that rehabilitation trialists identify ways to optimise adherence in the planning, design and delivery of physical rehabilitation trials, as well as the reporting of results. This systematic review highlights that adherence to inpatient rehabilitation trial protocols is complex and a multitude of factors influence adherence across all constructs of the CFIR: innovation, outer setting (hospital system), inner setting (hospital ward/rehabilitation unit), individual (those receiving and those delivering the intervention) and implementation process. These factors have been identified in several systematic reviews of physical exercise;45–47 thus, they are not isolated to inpatient rehabilitation trial protocols. Rehabilitation trials depend on behaviours of people such as those delivering or receiving a trial intervention, as well as team and organisational behaviour. 48
Adherence to intervention protocols refers to how closely the behaviour of trial participants aligns with the intervention assigned to them. 2 Conversely, intervention fidelity refers to how closely an intervention protocol is delivered by staff. 49 These definitions are somewhat simplistic and do not capture the complexity of the behaviours of those receiving and delivering the rehabilitation trial protocols and the context in which the trial is delivered. As such, behavioural science theories and frameworks have been used to understand and improve adherence to health interventions, including the Health Belief Model, the Theory of Reasoned Action and the Theory of Planned Behaviour, as summarised by Brawley and Culos-Reed. 50 A more recent framework, the BCW 51 provides a practice framework to design interventions that may optimise adherence to inpatient rehabilitation trial protocols. The BCW is incorporated into CFIR 2.0, 4 and we have mapped the barriers and facilitators to adherence to capacity, motivation and opportunity in this systematic review. This may help rehabilitation trialists understand and modify adherence behaviours, thereby informing protocol development and trial training to optimise adherence.
Staff beliefs were a common barrier to rehabilitation trial protocol adherence. Beliefs have been described as ‘a cognitive process resulting in a concrete cognition of how we think things are’ 52 and determines a person’s behaviour, in this case, clinical decision-making concerning adherence to rehabilitation trial protocols. Staff beliefs have been cited as a barrier to implementing evidence-based stroke rehabilitation as recommended in national clinical guidelines.53,54 This may be due to unconscious (implicit) biases that are intuitively held preferences or stereotypes of which we are unaware. 55 The presence of unconscious bias among healthcare professionals has been identified as a concern because it affects clinical decision-making and treatment selection. 56 When staff are delivering rehabilitation trial interventions, the research may challenge their biases if the intervention or potential outcome conflicts with their beliefs. Some of these issues could be related to the staff’s knowledge and understanding of research and their role in clinical trials. If staff lack knowledge of the core purpose of research and their role in clinical trials, they may not fully appreciate the importance of preventing their beliefs from impacting upon their behaviour, which can affect trial adherence. 56 Staff behaviour in rehabilitation trials is therefore acknowledged as being particularly challenging, but training to increase knowledge may overcome some of the barriers identified in our review.
There is no agreement on what comprises effective training, which is attributed to the heterogeneity of rehabilitation trial interventions. However, common barriers and facilitators were identified that could be used to develop a core set of standards for training in stroke and other rehabilitation trials. For example, beliefs and behaviours of those delivering and receiving the intervention and how they influence trial adherence, staff’s confidence in delivering the intervention, and their knowledge on the core purpose of research, roles and responsibilities in conducting rehabilitation clinical trials. These core standards have the potential to optimise adherence and facilitate recruitment, retention and intervention fidelity. However, consideration needs to be given to when and how the training is delivered. Data from these included studies suggest multi-modal training and support may facilitate adherence to trial protocols.
Multi-modal training and support could incorporate online resources, 57 vignettes, 58 mentoring 59 peer support, 60 a secure forum to share ideas or ask questions of other staff involved in the trial, and more in-depth training before commencing recruitment, could benefit adherence to inpatient trial protocols. Web-based training for staff delivering the intervention has been effectively used in rehabilitation trials, which have delivered information in multi-modal learning formats. 61 An advantage of web-based training is that it minimises training costs without compromising quality and effectiveness, allowing it to be delivered at the start and at regular intervals throughout the trial to maintain momentum and accommodate new staff.
In the United Kingdom, all staff involved in any clinical trial activities must complete Good Clinical Practice (GCP) training. 62 Currently, GCP training does not incorporate aspects that may optimise protocol adherence, such as clinical and personal equipoise, 63 gatekeeping 64 and unconscious bias. 55 Integrating these topics within a standardised multi-modal training package for rehabilitation trials offers a way of optimising adherence. There is a paucity of evidence on patient perspectives of adhering to inpatient rehabilitation trial protocols. It is possible that patient perspectives were sought from patient and public involvement and engagement partners 65 during trial design and development of studies included in our systematic review. Issues associated with the patients’ capability, opportunity and motivation were reported by staff, not patient participants themselves. Staff reported that intervention delivery was difficult and sometimes not feasible due to patient impairments, such as cognitive or communication impairments, mood changes, severe weakness, visual deficits, patient fatigue, and the general acuity of the patient’s condition. The lack of data from patient participants in the included studies makes it difficult to know whether patients and staff held different perceptions about barriers to adhering to the trial protocols.
Health conditions
This review included inpatient rehabilitation trials in stroke and other non-stroke conditions to determine whether adherence to the stroke trial protocols differs from those in other health conditions. It is important to acknowledge the individual different disease courses, for example, sudden (e.g. stroke, spinal cord injury, cancer or surgery), versus chronic conditions (e.g. respiratory disease or neuro-degenerative such as Multiple Sclerosis) and factors related to the condition and the individual, for example, physical, cognitive, psychological, demographic and socioeconomic. For example, in stroke and neurological conditions, cognitive and communication impairments, mood changes, fatigue and severe weakness/disability were reported as barriers to protocol adherence. However, trials in non-stroke health conditions identified similar barriers and facilitators in terms of the acuity of the patients’ condition and comorbidities. This suggests that the trajectories and disease characteristics of individual health conditions are of lesser importance when determining adherence to inpatient rehabilitation research protocols. However, rehabilitation trial protocols need to be adaptable to accommodate the varying severity of physical, cognitive and psychological impairments associated with neurological conditions. This in turn should be reflected in the rehabilitation protocol design and training provided to staff.
Adherence measures and levels of adherence
The variability of adherence measures makes comparison between studies or inclusion within a meta-analysis difficult. High variability of adherence measures and the absence of published standardised guidelines for measuring adherence in RCTs have been identified in a systematic review of current practice in the measurement and interpretation of adherence in RCTs. 66
There are no guidelines for categorising levels of adherence in rehabilitation or other non-pharmacological clinical trials. Only seven studies set and reported a pre-specified adherence level, with only one study providing a rationale for the specified level. It is unclear whether rehabilitation trialists are not considering protocol adherence and the impact it may have on trial results and implementation, or whether the lack of consensus of adherence levels means currently there is no benefit to categorising patients into arbitrary levels without evidence-based rationale. Acceptable adherence levels have been defined as 80%, but this does not appear to be based on a sound theoretical framework. 65 The development of evidence-based adherence levels is needed to apply to future inpatient rehabilitation trials.
Predictors of adherence
There is a lack of evidence evaluating predictors of inpatient trial protocol adherence in the included studies. The World Health Organization 2 identified multiple predictors of adherence and suggested adherence is multi-dimensional with a series of factors, rather than one single factor, determining adherence. They highlight the interplay of five dimensions, social and economic, health-care team and system-related, condition-related, therapy-related and patient-related, which align with the CFIR domains. Identifying factors that predict adherence to inpatient rehabilitation trial protocols and how these factors interact would be helpful in designing and implementing inpatient rehabilitation trials to optimise protocol adherence.
Implications for future research
This systematic review has highlighted the following implications for future research:
Using behavioural science methodologies and frameworks like the BCW and CFIR 2.0 during the design and formulation of inpatient rehabilitation trial protocols could help address identified behavioural challenges. This approach may enhance adherence among both providers and recipients of the intervention, potentially boosting its effectiveness and improving participants’ quality of life.
Consensus of adherence level categorisation would aid in interpretation of results. For example, a traffic light system, similar to that used for progression criteria 67 could be useful to standardise adherence levels. This would minimise research waste by implementing changes to improve adherence or stop the trial.
Designing and implementing inpatient rehabilitation trial protocols with consideration for the various dimensions of adherence and how they intersect may enhance adherence.
Standardised training in inpatient rehabilitation trials would provide consistency and may overcome multiple barriers and improve adherence; as yet this does not exist.
There is an absence of data related to outer settings such as hospital systems. Clinical trials operate within the broader context of a system, which faces its own stresses and demands for delivering healthcare (as distinct from research). For instance, UK hospitals are increasingly reaching Operations Pressure Escalation Level 4, 68 indicating a state where they are deemed ‘unable to deliver comprehensive care and patient safety is at risk’. Consequently, hospital staff are compelled to prioritise discharge, compromising research delivery unless additional funding is allocated to address staffing requirements. Trialists must assess how this situation impacts on adherence to overall trial delivery and consider this when designing research protocols.
Strengths and limitations
This is a comprehensive systematic review of adherence to inpatient rehabilitation trial protocols which identified published studies over the last 16 years. Adhering to the JBI Methodological guidance for the conduct of mixed-methods systematic reviews, utilising the MMAT and PRISMA reporting checklist, this review provides a robust synthesis of protocol adherence in inpatient rehabilitation trial literature. The review includes authors being contacted for raw data when this was not provided in the published article. Identifying relevant text within studies was a lengthy process because the language and definitions used were not standardised. This required subjective judgement and is open to interpretation. Other researchers could have reached different conclusions and mapped data to different CFIR constructs. The findings that some constructs had no barriers or facilitators mapped to them could be because of the mapping process itself. Only including studies published in English may be a limitation.
Conclusion
Adherence to inpatient rehabilitation trial protocols is multi-dimensional, with many factors interacting with one another. There are multiple barriers and facilitators to adherence to inpatient rehabilitation trial protocols, most related to the individual in terms of capability, opportunity and motivation. The beliefs of staff delivering the intervention appear to be a common barrier to protocol adherence. Understanding factors affecting adherence and how these interact with one another is key in the design, implementation and reporting of future inpatient rehabilitation trial protocols. Adherence in stroke and other health conditions appears to be similar. An absence of consensus on adherence measures and levels makes meaningful comparisons between trials difficult. A consensus about the categorisation of adherence levels, with a traffic light system would facilitate trialists to minimise research waste by systematically considering the need to either implement changes during the trial to improve adherence or to stop the trial.
Supplemental Material
sj-docx-1-ctj-10.1177_17407745251358262 – Supplemental material for Optimising adherence to inpatient rehabilitation trial protocols: A mixed-methods systematic review
Supplemental material, sj-docx-1-ctj-10.1177_17407745251358262 for Optimising adherence to inpatient rehabilitation trial protocols: A mixed-methods systematic review by Angela Logan, Jonathan Marsden, Jennifer Freeman, Emma Cork and Bridie Kent in Clinical Trials
Supplemental Material
sj-docx-2-ctj-10.1177_17407745251358262 – Supplemental material for Optimising adherence to inpatient rehabilitation trial protocols: A mixed-methods systematic review
Supplemental material, sj-docx-2-ctj-10.1177_17407745251358262 for Optimising adherence to inpatient rehabilitation trial protocols: A mixed-methods systematic review by Angela Logan, Jonathan Marsden, Jennifer Freeman, Emma Cork and Bridie Kent in Clinical Trials
Supplemental Material
sj-docx-3-ctj-10.1177_17407745251358262 – Supplemental material for Optimising adherence to inpatient rehabilitation trial protocols: A mixed-methods systematic review
Supplemental material, sj-docx-3-ctj-10.1177_17407745251358262 for Optimising adherence to inpatient rehabilitation trial protocols: A mixed-methods systematic review by Angela Logan, Jonathan Marsden, Jennifer Freeman, Emma Cork and Bridie Kent in Clinical Trials
Supplemental Material
sj-docx-4-ctj-10.1177_17407745251358262 – Supplemental material for Optimising adherence to inpatient rehabilitation trial protocols: A mixed-methods systematic review
Supplemental material, sj-docx-4-ctj-10.1177_17407745251358262 for Optimising adherence to inpatient rehabilitation trial protocols: A mixed-methods systematic review by Angela Logan, Jonathan Marsden, Jennifer Freeman, Emma Cork and Bridie Kent in Clinical Trials
Supplemental Material
sj-docx-5-ctj-10.1177_17407745251358262 – Supplemental material for Optimising adherence to inpatient rehabilitation trial protocols: A mixed-methods systematic review
Supplemental material, sj-docx-5-ctj-10.1177_17407745251358262 for Optimising adherence to inpatient rehabilitation trial protocols: A mixed-methods systematic review by Angela Logan, Jonathan Marsden, Jennifer Freeman, Emma Cork and Bridie Kent in Clinical Trials
Footnotes
Acknowledgements
The authors acknowledge the contribution of patient and public involvement and engagement partners in the conceptualisation and review of the manuscript. Thanks to Dr Wendy Clyne for her contribution to developing the search terms and PROSPERO protocol.
Authors’ note
The views expressed in this publication are those of the authors and not necessarily those of Royal Devon University Healthcare NHS Foundation Trust or the NHS.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The lead author received partial funding from the Royal Devon University Healthcare NHS Foundation Trust to prepare the manuscript.
Data availability
All data analysed in this systematic review are included in this published article and its supplementary materials. No primary data were collected. Additional information related to the search strategy, data extraction and analysis is available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
