Abstract
Background/Aims
To respect the rights and wellbeing of research participants, these should receive information at all stages of the trial, and procedures should be put in place to ensure a valid consent that promotes an informed, autonomous and voluntary decision-making. This review focuses on the extent and type of evidence available in relation to best practices in the information provision and consent processes for vaccine trials conducted in Sub-Saharan Africa.
Methods
Ancillary studies or evaluations assessing the information and/or consent processes used in vaccine trials implemented in Sub-Saharan Africa were eligible. The databases PubMed, CINAHL, Scopus, Web of Science, African Index Medicus Google Scholar and ProQuest dissertations and thesis citation index and Open Access Theses and Dissertations were searched, without time limits. Following a deductive approach, relevant data were extracted using an extraction tool and categorised into themes.
Results
The review included 46 sources reporting results from 37 studies implemented in 13 Sub-Saharan African countries. The studies covered: community engagement (n = 8); informants (n = 7); messages (n = 7); communication tools (n = 3); community groups (n = 4); consent process (n = 11); comprehension (n = 19) and dissemination of results (n = 4). They mostly represented the views of participants or parents of trial participants; researchers and trial site personnel; and community members and representatives, including those with formal informational roles. The studies showed gaps in information and consent processes leading to a lack of understanding and confusion or suspicion. The involvement of community members in information giving was essential. These were able to communicate in culturally–appropriate ways and also increase trust in the trial.
Conclusions
The studies highlight complexities involved in the information and consent processes for vaccine trials implemented in Sub-Saharan Africa. These processes would benefit from a stronger consideration to the context where research takes place, including culture, language, non-biomedical conceptions and power imbalances. The views from ethics review boards were mostly absent.
Keywords
Introduction
Providing information to participants and obtaining valid consent are key elements of good clinical trials and essential to respond to the ethical principle of respect for persons. 1 This requires using adequate language, considering participants’ needs and expectations and the context where research takes place,2–4 emphasising the importance of community engagement and stakeholders’ involvement2,4,5 to develop locally acceptable and effective information and consent procedures and materials.6–8 Nevertheless, implementing adapted information and consent processes remains a great concern in clinical trials in Sub-Saharan Africa. 9
Sub-Saharan Africa comprises a diverse and multicultural geographical region, with a vast number of spoken languages. It is also the world’s region with the highest illiteracy and poverty rates according to 2022 data.10,11 This is combined with widespread lack of adequate access to healthcare in a majority of countries 12 and an overall low investment in research, including against the prevalent infectious diseases. 13 The high burden of potentially vaccine-preventable diseases in Sub-Saharan Africa highlights the importance of conducting vaccine trials in this region.
Information and consent processes include here the multiple time points in which information is provided to participants and communities and understanding and voluntary decision-making promoted. Recent literature reviews focusing on comprehension 9 and community engagement practices14,15 in clinical trials conducted in Sub-Saharan Africa, have highlighted issues around the identification of appropriate stakeholders, adaptation of strategies to existing cultural practices and dealing with potential mistrust against research or the research institution, 14 as well as the need to address cultural and structural factors impacting on comprehension and ways to enhance and assess understanding. 9 Preventive vaccine trials aim to recruit healthy volunteers (i.e. unaffected by the condition under study) that do not receive any direct health benefit from participating, adding particularities to the way stakeholders are involved and communities informed about the trial, including information about risks and benefits. Moreover, in this region, the majority of vaccine trials are sponsored by international organisations. 16 This puts together two distinct spheres, the researchers linked to a different culture and socio-economic status, and the communities invited to participate, likely unfamiliar with research, who have low access to quality healthcare and who do not receive direct health benefits from trial’s participation.
This review aimed to describe information and consent practices used in vaccine trials that have been evaluated, highlighting research gaps and identifying how these could inform best practices and recommendations that strengthen valid consent in vaccine trials conducted in Sub-Saharan Africa.
Methods
Guidelines provided by the Joanna Briggs Institute 17 and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for scoping reviews (PRISMA-ScR) were followed. 18 The protocol was registered at OSF: https://doi.org/10.17605/OSF.IO/P2ZHR.
Eligibility criteria
Eligible studies evaluated aspects of information and/or consent processes from any stakeholder’s perspectives, in vaccine trials of any clinical phase and vaccine, including mock/hypothetical trials, conducted in Sub-Saharan Africa. Studies focusing on motivation, willingness to participate, or factors influencing participation were excluded. Eligibility was assessed by the main author using a predefined inclusion criteria discussed with the review team. Doubts about inclusion were resolved by consensus.
Types of studies
Qualitative, quantitative, mixed methods and systematic reviews were considered. Opinion papers or comments were excluded.
Search strategy
We aimed to locate published and unpublished studies, without language and time limit. A three-step search strategy was used:
Initial search of PubMed using: (Informed consent OR participation OR comprehension OR community sensitization OR engagement OR information dissemination) AND vaccine trial AND Sub-Saharan Africa. Full search strategy developed using keywords in titles and abstracts and index of relevant articles.
Search strategy adapted for each database: CINAHL, Scopus, Web of Science, African Index Medicus and Google Scholar. In addition, ProQuest dissertations and thesis citation index and Open Access Theses and Dissertations. The search included keywords, limited to the Title and Abstract field when possible, and related subject headings (Supplementary file, section 1).
Reference lists were hand searched and sources citing all included studies were screened using ResearchRabbit, a literature mapping tool, to identify additional studies.
Selection of studies and data extraction
References were imported to Covidence (Veritas Health Innovation, Melbourne, Australia). Titles and abstracts were screened according to predefined inclusion criteria by one reviewer (A.J.G.). Doubts about inclusion were resolved by discussion. All selected sources were assessed by a second reviewer (C.E.F. or E.C.A.).
Data extraction was done by one reviewer (A.J.G.) using a predefined tool developed by all authors and piloted and modified following extraction from the first 10 sources (Supplementary file, section 2). Data related to categorization of themes were extracted by a second reviewer (C.E.F. or E.C.A.). Discrepancies between reviewers were discussed and solved by consensus. Extracted data were also reviewed through regular discussions.
Data synthesis
A basic numerical analysis presented the extent, nature and distribution of included studies. Using qualitative content analysis, 19 and considering the objectives of the included studies, the studies were categorised deductively into themes and sub-themes, corresponding to a conceptual framework, grounded in recognised/key aspects of information and consent processes that we developed (Figure 1). For each theme, we described the processes used in the trials and included key findings, referring to time when relevant.
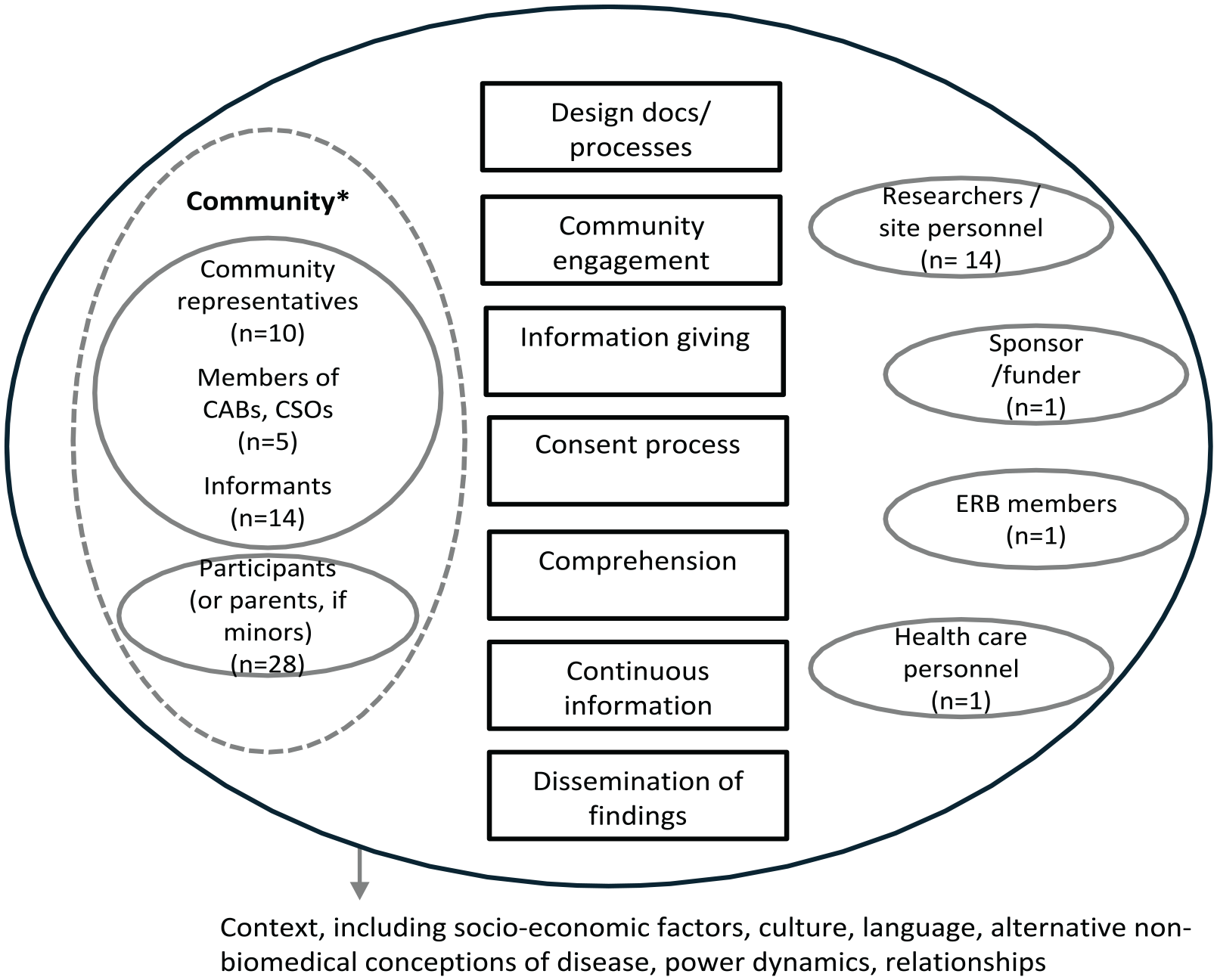
Conceptual framework of the information and consent processes and stakeholders involved in the studies included in the review.
Results
Study selection
The search was conducted between January and May 2024. In total, 843 records were retrieved and 506 remained after duplicates were removed. Of these, 149 were read in full and 46 included (Figure 2).
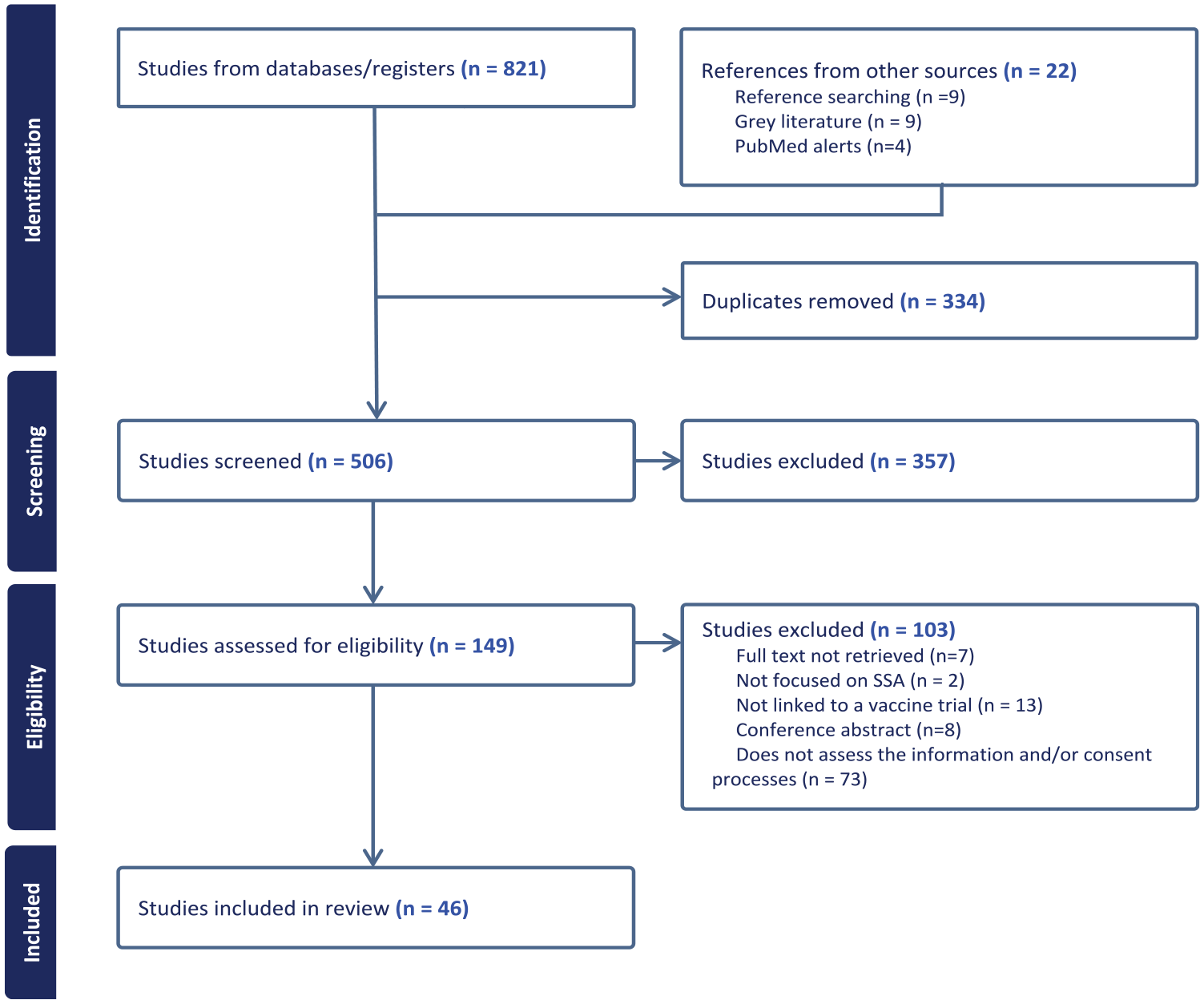
PRISMA-scoping reviews flow diagram.
Characteristics of included studies
We included 43 published articles and 3 thesis reports, one of these later published. These reported on 37 distinct studies, with 7 studies being reported by more than one source. Studies were published between 1997 and 2024 and conducted in 13 countries, with South Africa (n = 11), Kenya (n = 7) and The Gambia (n = 5) representing 56.1% (Figure 3).
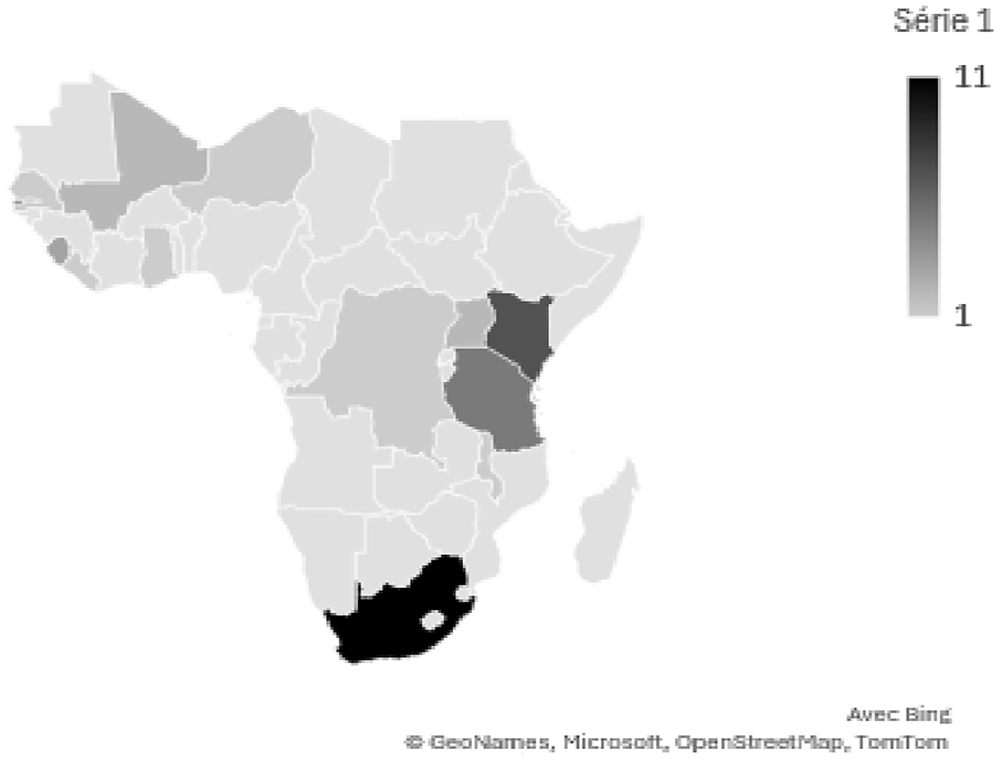
Geographical distribution of the studies included in the review.
Context
Studies were nested in an estimated 37 trials investigating vaccines against 14 different diseases including HIV (n = 10), malaria (n = 7) and Ebola (n = 5). Trials recruited infants or children <5 years of age (n = 18) or adults only (n = 19), with 2 enrolling women or pregnant women only. They covered all stages of clinical development. Two were mock trials. Sponsors included international organisations, with three trials nationally co-sponsored (Supplementary file, section 3):
Study methodologies. Studies were quantitative (n = 12),20–31 qualitative (n = 19)32–59 or mixed-methods (n = 5).33,60–64 One was an evaluation.
65
Themes. Studies covered eight identified themes (Figure 4 and Table 1). Studies published prior to 2001 focused on the consent process and the quantitative assessment of understanding. Assessment of community engagement activities started in 2012. Studies included a wide range of trial’s stakeholders (Figure 1).
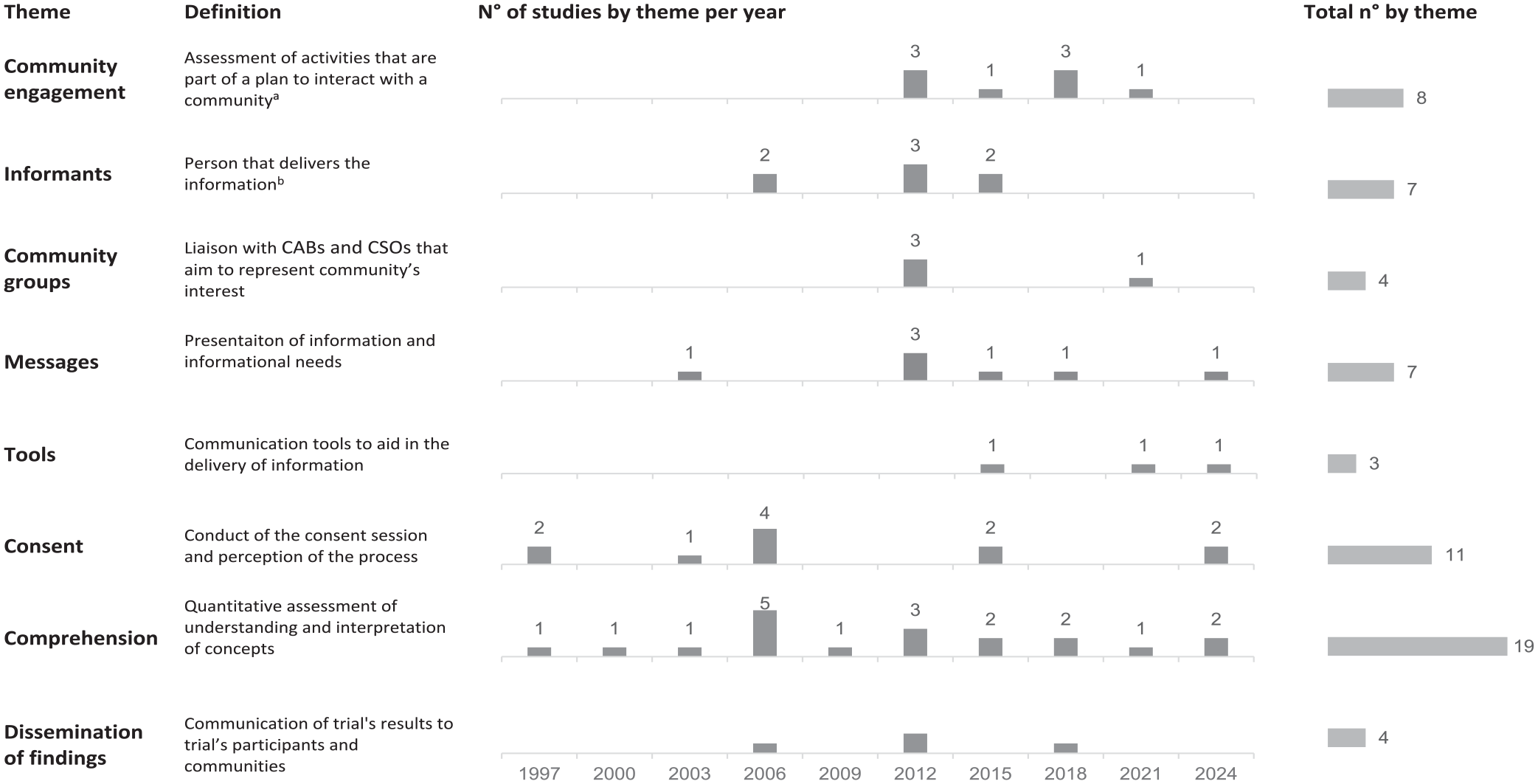
Themes identified in the included studies, per year and total.
Evidence table.
Community engagement
Community engagement activities were assessed in four paediatric vaccine trials in Kenya, Tanzania and Malawi,36,59,60,65 HIV trials in South Africa 38 and during the Ebola outbreak in Sierra Leone.39,66 These were published between 2012 and 2023. In these, community engagement consisted of a multi-step process. Engagement with community leaders, considered as gatekeepers, was generally a first step36,48,59,60,65 followed by community meetings36,37,39,60,65 at times assisted by leaders or other representatives.36,39,60 Engagement activities were also conducted to small groups at the community, 48 health facilities36,60 or during household visits.36,39,60,65 These were conducted by community members assigned with informational roles,36,39,48,60,65 and at times involved community advisory boards (CABs).36,38
Community engagement was directed to sensitise about the trial and promote understanding. This included handling rumours and concerns in five studies.39,59,60,65,66 Trust building also appeared a key goal of community engagement in the eight studies. Understanding and trust were enhanced when information was provided by community sources,36,39,48,65 who shared cultural and social norms. 66 This emphasised the importance of incorporating appropriate community stakeholders,36–39,48,59,60,65 identifying recognised leaders and considering power dynamics. 39 Nevertheless, this required caution as leaders could exert pressure to enrol. 60 Interactions with communities also played a role.37,39,59,60 These included establishing an inclusive dialogue, hearing and addressing communities’ concerns, described as essential in an Ebola vaccine trial.37,39
Community engagement was a complex process, framed by social relations and contextual specificities, including disparities in access to knowledge and resources. 36 In this way, community engagement activities were not limited to formal plans, but involved numerous and diverse interactions that occurred between researchers and community members through the trial, 59 influenced by personalities, backgrounds and expectations,36,60 and modified in responses to emerging issues, including rumours and concerns.60,65
Informants
Seven studies published between 2006 and 2018 assessed attributes and role of employed41,45,53,62 or hourly paid 35 field personnel, community health workers 33 and educators and CAB members47,54,56 in providing information to communities about the trial. Most of these informants were from the community where the trial took place and all shared culture and language. Researchers, communities and participants considered this proximity as facilitating communication in five studies.33,35,41,45,62
Along with providing information, informants aimed to enhance credibility and trust in the research and researchers in five studies.33,36,45,47,54,56,60,62 Factors such as familiarity or shared identity,54,56 and perceived independence to the researchers were attributed to ‘trustworthy’ communicators in HIV trials. 56 Trust was also built by being honest, transparent, consistent and reliable 54 and by exhibiting concern and fairness. 33 Trust building efforts appeared to respond to high levels of suspicion with blood sampling for instance being a key issue.36,41,45,56,60,62 To reduce suspicion, informants at times, implicated credible sources in information giving, including community leaders and ex-participants.33,54
Informants tried to provide adequate information, but were at times ill-equipped to respond to questions and concerns.33,62 In five studies they were found to use their own tactics to deliver information and increase trial’s acceptability, including using familiar examples, 54 emphasising benefits and positive aspects,33,35,41,62 building strong links with influential figures 41 and lengthening interactions with volunteers that appeared reluctant to make a decision about participation. 45 Their close proximity to communities also brought up some challenges, including requests for assistance from community members. 35 Informants, moreover, felt accountable in case of undesirable outcomes and perceived their informer’s and recruiter’s roles as conflictive, highlighting limits of adequately educating volunteers who knew little about research in a given timeframe.36,53
Messages
Seven studies assessed aspects related to the messages.22,30,34,44,47,54–56,60 Two studies highlighted the importance of adapting messages to participant’s needs,44,54,55 for instance differentiating between experimental and approved vaccines highlighted in an Ebola vaccine trial. 44 Pregnant women in Uganda highlighted the importance of clear and non-technical language to inform about risks and benefits, purpose of the study, side effects and withdrawal options. 55
One study suggested that messages framed as a gain (e.g. ‘If I take part in this study, I will have access to a risk-reduction counsellor and regular HIV testing’), as opposed to a loss (e.g. ‘If I do not take part in this study, […]’), could lead to higher willingness to participate. 22
Certain messages related to experimental vaccines and use of blood were sources of mistrust or suspicion in HIV trials.47,56 In these, messages were more likely to be trusted if they were congruent with everyday observations and community’s understanding, and consistent within different communicators. 56 Similarly, referring to safe, effective vaccines close to people’s experiences appeared to instil trust in medical research.47,54
Translation challenges were highlighted in three studies.30,34,60 A study in Niger, published in 2003 reported how the lack of validated translation of the consent information generated adaptations by the consenter, aiming for a positive image. 34 Translated terms in approved information sheets, such as compensation and randomisation, were reported to lead to different interpretations and were associated with rumours and concerns in study from 2014. 60 To enhance understanding and avoid adaptations, a study in Ghana evaluated a new procedure, where consent forms were written in English, and a scientific committee provided recommendations on the oral translation. 30
Communication tools
Communication tools were the focus of two studies.25,26 An audio-visual tool (i.e. speaking book) given to families participating in a paediatric vaccine trial was highly used and shared with friends and family. 26 The use of mobile communication was assessed in an HIV trial. 25 In this, 80% of participants preferred being called than messaged and 97% welcomed receiving regular updates. 25
Participants of one study suggested using short videos or audio, posters and flyers and to illustrate information notices. 55 The use of radio, television and social media, was also encouraged. 55
Community groups
Four studies published between 2010 and 2023 assessed the roles of CABs,36,38,52 and civil society organisations (CSOs). 46
Community advisory boards
CABs’ roles were addressed in three studies nested in HIV trials38,52 and malaria and rotavirus paediatric trials. 36 These involved providing advice to researchers about community norms and expectations; disseminating information; assisting in creating a supportive community environment and feeding back on community’s concerns; and reviewing participant information leaflets and consent documents.38,52 Their role in recruitment was disputed in a study published in 2010, with researchers expecting CAB’s assistance in recruitment and advisors considering this outside their boundaries. 52
CAB members felt responsible for presenting a positive image of the research organisation, overcoming suspicion and promoting acceptance. 36 This risked being viewed as ‘sell-outs’ or ‘brain-washed’. 47 The dependence of CABs on the trial, including financial support and training, was perceived as compromising their independence and actions in two studies published in 2010 and 2012.36,52
Civil society organisations
In HIV trials, CSO members viewed themselves as having informational and oversight roles, ensuring, for instance, that research benefitted communities and the voluntariness of consent. 46 They considered themselves as independent from the trial team and able to enhance community representativeness. 46 CSO members called for a transparent and inclusive involvement from trial’s onset, avoiding requests solely in case of difficulties. 46
Consent
Eleven studies published between 1997 and 2024 evaluated the consent processes.23,34,41,43,45,50,54,55,58,62,63 In this, consent was taken by researchers23,45,55 or fieldworkers, 41 at the study site 45 or volunteer’s home. 34 Consent documents were written in English/French and the local language.41,50,62,63 At times an oral translation to the local language was used.30,34,58
Consent process
The consent session was observed in two studies.23,58 In a paediatric malaria vaccine trial in Mali, the consenter provided an oral explanation to groups of 2 to 4 parents, taking breaks to summarise and allow questions. 23 In an HIV trial in South Africa, the counsellor took extensive, uninterrupted turns reading the consent document or explaining concepts, with little input from the volunteer. 58 To increase rapport, the counsellor at times relaxed from the formal role, by, for instance, commenting on the length of the document, and this appeared to increase participants’ comfort and trust. 58 Sessions lasted between 40 min and 1 h.23,58
Consent counsellors from HIV vaccine trials requested participants to explain concepts in their own words and switched languages to accommodate participants’ preferences. 54 Counsellors, however, reported limits in promoting understanding due to the volume of information and limited time. 54 They also found difficulties promoting voluntariness once information about compensation was disclosed. 54 These counsellors viewed consent as an ongoing process, with multiple opportunities to reinforce information. 54
Participants’ perceptions
Six studies assessed participants’ perceptions on the consent session.34,43,50,55,62,63 The three older studies published between 1997 and 2003, presented signing the consent as potentially causing mistrust or confusion, with parents questioning the motives of the investigator and their responsibility if something happened to the child 63 or not understanding why a signature was required.34,50 In two studies from Kenya and Tanzania published in 2008 and 2024, filling and signing a form was perceived as binding, limiting voluntariness 43 and protecting researchers. 62 However, in a Ugandan study from 2024, pregnant women considered signing as common and a proof of understanding and willingness to participate. 55 Assessment of literacy and requirement of a witness were questioned in two studies, with participants considering these procedures inappropriate in a study from 2003, 34 highlighting that literacy did not reassure understanding in 2024. 55 A study from 2024 stated that the consenters’ counselling skills helped participants understand procedures and develop trust. 43
Consent decisions were perceived as having wide implications, providing, for instance, access to medical service. 41 Moreover decisions were embedded in power dynamics and relationships, influenced by perceptions of respect and politeness towards heads of household and informants. 45
Trial participants provided suggestions to improve the consent process in two studies published in 1999 and 2024. The most recent suggested that consent could be improved by providing clear information, free from technical language. 55 The older study highlighted a polite and respectful process, without force. 63 Allowing time to share information and involving others in the decision-making, particularly husbands, was highly valued.55,63
Comprehension
A total of 19 studies published between 1999 and 2024 assessed comprehension of research-related concepts.
Measuring understanding
Fourteen studies assessed understanding using different questionnaires and types of questions, including true/false, multiple choice and open-ended. These were administered to trial participants,20,21,24,27,29,63 their parents,23,26,28,30,60,61,64 or less frequently to volunteers or refusals.24,31,63 Understanding was most often assessed on the consent day,21,23,26–28,31,64,30 or 3 months to 2 years after initial consent.27,29,31,60,61 Three studies assessed understanding at two points.26,27,31
Overall correct responses varied from 67% to 99%.21,24,26,28,30 Understanding of specific questions varied, with for instance placebo correctly understood by 10% to 49% of participants27,29,63 and trial’s aim by 35%. 64 In two studies, understanding decreased over time.26,63 Increased exposure to information, including repeated explanations and giving information sheets to take home, was suggested to increase understanding in seven studies.26–28,30,31,49,63
A study found that true/false questions overestimated comprehension, with open-ended or multiple choice providing a more accurate account. 24 Another study stated that measurement of understanding should be complemented with qualitative assessments. 64 One study found that a digitised, audio-recorded questionnaire including questions in different formats could increase objectivity and overcome challenges related to lack of standardised written languages. 20
Interpretations
The eight studies assessing understanding using qualitative methods described challenges in the representation of key information.32,43,44,49,51,60–62,64 For instance, a malaria trial was generally described as a form of healthcare assistance. 62 Differences between experimental and approved vaccines were presented as a source of misunderstanding in Ebola and HIV vaccines trials.43,44 43 Trial procedures were also associated with misunderstandings, with confidentiality, for instance, being hardly understood in a context where participation was known by community members. 60
Four studies highlighted how information was influenced by local knowledge and practices.32,51,60,61 In this way, health benefits attributed to the placebo in an Ebola vaccine trial were influenced by local understanding of injections and immunisation. 32 Understanding of trial’s procedures was also affected by people’s particular circumstances, including socio-economic, leading for instance to blood-stealing rumours in The Gambia. 61
Dissemination of findings
Four studies assessed dissemination activities.40,42,46,57,62 Results were disseminated by community meetings,42,46 at times supplemented by leaflets. 42 Dissemination of findings was understood as an ongoing process building up on initial community engagement activities.40,42,46 Two studies addressed the communication of negative findings.40,42,46 These were received with disappointment by community and CSO members, highlighting the need to enhance understanding during trial’s implementation.40,46
Three studies described trial’s end as a concern for participating communities as this was associated with interrupted access to improved healthcare42,57,62 and other benefits, such employment. 42 Dissemination activities involved negotiation of post-trial benefits and expectations.57,62 Participants perceived dissemination activities as a sign of respect and appreciation for their involvement.42,57,62 Ethics review board (ERB) members emphasised the importance of post-trial access. 57
Discussion
This review included studies assessing information-giving and consent processes used in vaccine trials implemented in Sub-Saharan Africa. The studies covered a variety of processes, ranging from community engagement to dissemination of results, including perspectives from participants, community stakeholders and researchers. The review highlights that informed consent for vaccine trials in Sub-Saharan Africa is not limited to a minimalist, dyadic model involving a researcher and a volunteer but involves various stakeholders. 67 Community leaders provided access to the community but also participated in sensitization activities and acted as credible sources. Informants from the community were able to facilitate communication and created positive relations. The involvement of community stakeholders appeared to increase trust. Nevertheless, the review highlights critical gaps in comprehension of key trial information after consent, questioning the adequacy of processes and extent of informed decision-making.
Considering the elements of our conceptual framework, comprehension received most attention, followed by consent and community engagement activities. Except for a formative assessment guiding communication for an Ebola vaccine trial, 44 included studies retrospectively assessed processes, lacking information on the design and adaptation of processes, including development and pilot of consent documents. Most studies on consent reported perceptions and only two studies observed the process, providing little insight on related challenges. In relation to stakeholders, most studies included trial participants. ERB members were only involved in one study, and didn’t address ERB’s approval and oversight roles.
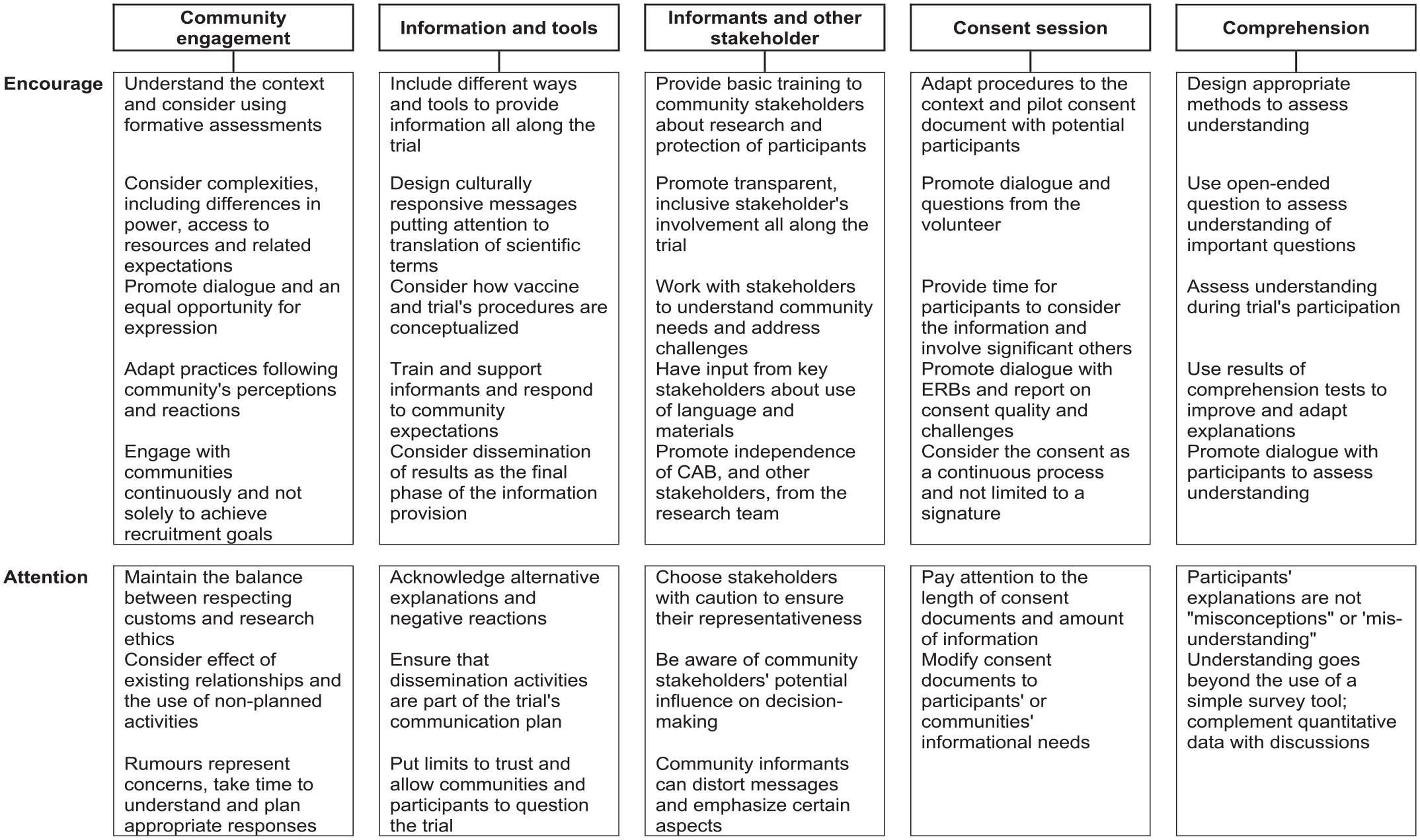
Despite these gaps, the studies highlight important recommendations to strengthen information provision and consent processes in vaccine trials in Sub-Saharan Africa (Table 2). These recommendations are aligned with international research guidelines1,3,4 and are applicable to clinical trials more broadly. Nevertheless, they also address unique challenges related to vaccine trials in Sub-Saharan Africa, particularly in relation to the recruitment of healthy volunteers from the communities, the implication of community stakeholders and potential effects of disparities between communities and researchers.
Main recommendations highlighted from the studies included in the review to strengthen the information provision and consent processes in vaccine trials in Sub-Saharan Africa.
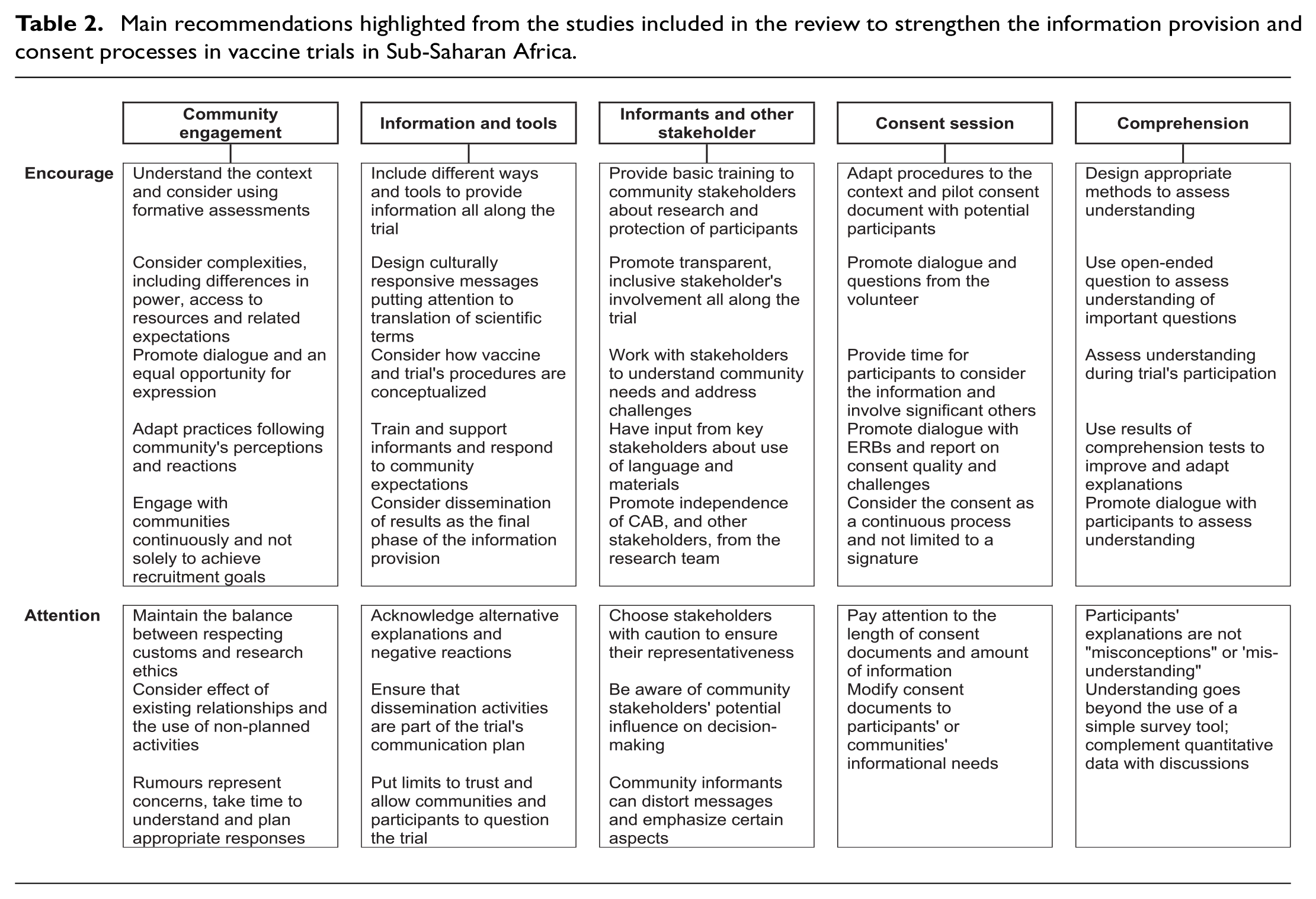
In these settings, the application of community and participant-centred principles that promote participants’ and communities’ views and needs would entail establishing an open dialogue between researchers, community representatives and potential volunteers in all stages of the vaccine trial, promoting an equal opportunity of expression,36,41,59 taking into account local framings of disease and health-related knowledge.32,48,51 The consideration of social relations and power, questioning who are considered as legitimate representatives’ and how these, in turn, act towards the interest of the community, should also be addressed. 15 Community stakeholders charged with information provision, including leaders and informants, should receive adequate training and support, along with continuous monitoring33,35,54,58,62,65 and adequate tools that help explain complex information in local languages. The dual role of informing and recruiting should be considered, addressing perceived conflicts and consequences. 53 Moreover, attention should be directed towards ensuring that local practices, such as involving community leaders, do not threaten ethical standards and that consent remains an individual’s informed voluntary decision.
Information and consent processes should be considered continuous,38,43,60 adapting messages in response to participants and communities’ evolving reactions and needs, assessing comprehension and desire to continue participation. Information should include post-trial access plans. Continuous, adapted communication should improve understanding and reduce feelings of unmet expectations at trial’s end that could negatively impact future research. However, this contradicts the rigidity of informed consent documents, difficult to modify once approved. 68 ERBs’ role in information and consent process should be expanded to include continuous oversight and monitoring and facilitation of participant-centred practices. Roles could be coordinated between in-country and sponsor’s country committees.
Over the years, different tools have been proposed to assist in tailoring requirements to the context. Rapid assessments have been demonstrated to be relatively quick and inexpensive. 69 Ethical frameworks, such as the principles and benchmarks proposed by Emanuel et al. 70 could also assist in adapting processes. None of the studies included in this review stated using specific tools.
Limitations
Categorization in themes was based on studies’ objectives and did not consider findings, leading to a potential underrepresentation of themes. Researchers conducting this review are experienced in vaccine trials in Sub-Saharan Africa, but are not from the region, representing an external view that might have affected representation.
Although practices changed over time, we did not set up a time limit and the first study identified dated from 1997. Older studies had a greater focus on the adequacy of individual informed consent and applicability of international standards.
Conclusion
The review raised ethical concerns about respect of participants’ rights and validity of informed consent in vaccine trials in Sub-Saharan Africa. The emphasis on promoting understanding and free decision-making is crucial for vaccine trials enrolling healthy individuals that do not obtain any healthcare benefit from participating. These highlight the need for greater emphasis to the context and participants and communities’ needs. To address these, funders, sponsors and researchers should allocate adequate resources to design processes that are adapted, working together with community members and representatives and adding flexibility to modify these as necessary. ERBs should also support these processes. The focus should be moved from meeting recruitment targets to also consider metrics related to how these were achieved and how responsive these processes were to the context.
Supplemental Material
sj-docx-1-ctj-10.1177_17407745251346134 – Supplemental material for Approaches to ensure quality of information provision and consent processes for vaccine clinical trial participation in Sub-Saharan Africa: A scoping review
Supplemental material, sj-docx-1-ctj-10.1177_17407745251346134 for Approaches to ensure quality of information provision and consent processes for vaccine clinical trial participation in Sub-Saharan Africa: A scoping review by Aitana Juan-Giner, Elena Carrillo-Alvarez and Cristina Enguita-Fernàndez in Clinical Trials
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Publishing costs were covered by a grant from The Víctor Grífols i Lucas Foundation. We received no other funding.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.