Abstract
Background
Evidence-based methods for randomised controlled trial recruitment and retention are extremely valuable. Despite increased testing of these through studies within a trial, there remains limited high-certainty evidence for effective strategies. In addition, there has been little consideration as to whether recruitment interventions also have an impact on participant retention.
Methods
A systematic review was conducted. Studies were eligible if they were randomised controlled trials using a recruitment intervention and which also assessed the impact of this on retention at any time point. Searches were conducted through MEDLINE, EMBASE, Cochrane Library, and the Northern Ireland Hub for Trials Methodology Research SWAT Repository. Two independent reviewers screened the search results and extracted data for eligible studies using a piloted extraction form.
Results
A total of 7815 records were identified, resulting in 10 studies being included in the review. Most studies (n = 6, 60%) focussed on the information given to participants (n = 6, 60%), with two (20%) focussing on incentives, and two focussing on trial design and recruiter interventions. Due to intervention heterogeneity, none of the interventions could be meta-analysed. Only one study found any statistically significant effect of letters including a photograph (odds ratio: 5.40, 95% CI 1.12–26.15, p = 0.04).
Conclusion
Assessment of the impacts of recruitment strategies, evaluated in a SWAT, on retention of participants in the host trial remains limited. Assessment of the impact of recruitment interventions on retention is recommended to minimise future research costs and waste.
Introduction
Testing of interventions using randomised controlled trial (RCT) methodology is fundamental to health research, however, the validity of such research is dependent on the successful recruitment and retention of participants. 1 While there is much focus on the importance of participant recruitment to funders and trialists, this effort can be wasted if participants cannot be retained. 2 Previous evidence has suggested that around a quarter of trials experience primary outcome attrition of greater than 10%. 3 This has the potential to lead to research waste due to the need for additional costs and/or a study extension, reduced power and potential early termination of research activity.1,4 Where such instances occur, there is also the ethical impact to consider; those participating in the associated trial have invested time and effort to the study, however, a conclusion may not have been ascertained, thus leaving participants without a meaningful result from their participation.
As a result, evidence-based methods to increase the retention of RCTs are extremely valuable.5,6 One method is to ‘nest’ trials of trial process interventions in ongoing randomised trials, known as a study within a trial or SWAT. 5 Use of SWAT methodology for the testing of recruitment and retention interventions ensures causality of intervention effectiveness can be ascribed and allows identification of both effective and ineffective methods for trial retention. 7 Embedding this testing within an ongoing RCT avoids the potential for concerns regarding the feasibility of intervention effectiveness, and subsequent implementation, particularly where interventions have been assessed in a quasi-RCT or non-randomised setting.
Retention-specific strategies, tested using randomised SWAT methodology, were previously combined in a Cochrane Review 8 and this has recently been updated. 6 The recent update has identified a small number of interventions (n = 4) with moderate GRADE certainty evidence of their potential for increasing trial retention, however, no strategies have high GRADE certainty currently. 6 Three included SWATs used quasi-randomised designs, which may limit the applicability and effectiveness in a ‘real’ trial setting, however, sensitivity analyses conducted did not demonstrate any significant change in intervention effectiveness when these designs were removed. 6
The number of retention SWATs undertaken has seen a marked increase over recent years, from 38 included in the initial Cochrane Review of 2013, 8 rising to 71 included in the 2021 revision. 6 Despite this thus far, it appears that little consideration has been given to whether interventions implemented at the recruitment stage of an RCT can also have an impact on increasing participant retention. Identifying if any strategies have been assessed for dual effectiveness, and if so whether any are effective, will be beneficial in focussing future nested intervention designs and ultimately, where effective strategies can be identified, may help to minimise research costs and waste.
This review, therefore, sought to establish whether any nested, randomised, recruitment strategies have also been assessed in the context of long-term, retention, impacts. Where this was undertaken, the review aimed to assess which interventions had been shown to be effective and if cost-effectiveness had also been assessed for any effective strategies.
Methods
Protocol
A protocol for this systematic review was prospectively registered on the Open Science Framework on 28 July 2021 (Reference: 10.17605/OSF.IO/S78HY). No amendments were made to the protocol during the review.
Eligibility criteria
Trials were eligible for inclusion if they:
1) Enrolled trial participants (adults or children) into a randomised controlled trial (commonly referred to as the host trial) in the fields of health and social care. The restriction to health and social care trials was made on the basis that the vast proportion of SWATs have been undertaken in host trials within this field.
2) Used an intervention to improve recruitment and also assessed the impact of the recruitment intervention on retention at any time point.
Trials were not eligible if they were hypothetical.
Information sources and search strategy
Recruitment and retention trials are routinely collated within the Methodology Cochrane Database for Systematic Reviews by Treweek et al. 9 and Gillies et al. 6
Given the robust search strategy used for these reviews, the Cochrane Reviews were used to identify published randomised trials which fulfil the required inclusion/exclusion criteria. In addition, the search strategies for MEDLINE and EMBASE as used in each review were duplicated on 7 February 2022 to cover the period since the last Cochrane review date. A copy of the full search strategy is included in Supplementary File 1.
In addition, the Northern Ireland Hub for Trials Methodology Research SWAT Repository Store was searched from the date of inception to the date of the search on 7 February 2022. Article reference lists and bibliographic searches were also undertaken during the screening process. The PROMETHEUS programme 10 (hosted by York Trials Unit, University of York) was also contacted to obtain an update on the progress of any relevant SWATs. 10
Article inclusion was limited to articles that were published in English; no other limitations were used.
Selection process
Titles and abstracts retrieved from the searches were downloaded into Rayyan (https://www.rayyan.ai/) and de-duplicated. The remaining titles and abstracts were independently screened by two reviewers (C.A. and L.C.) against the pre-specified inclusion and exclusion criteria. Full-text copies were obtained for those articles deemed to be meeting inclusion criteria and these were again independently reviewed by two reviewers (C.A. and L.C.). Where necessary documentation relating to the host trial (for example registry entry, protocols, and published results) was obtained to aid eligibility assessment. In both instances, any disagreements were discussed and resolved.
Risk of bias assessment
The Cochrane Risk of Bias tool (version 2) was used to assess the risk of bias, 11 applying all domains of the tool. An assessment was made only of the SWATs and not of the host trials. Two reviewers independently assessed the risk of bias for each included trial and any disagreements in assessment were resolved by discussion.
Confidence in cumulative evidence
Assessment of strength of evidence was proposed to be completed using GRADE, 12 however as SWATs could not be combined, and a narrative synthesis was used, this was not completed.
Data collection and items
The authors developed a data extraction form. Each outcome (see section Data items) was extracted into its own cell within the spreadsheet, with multifaceted variables (e.g. sample size) separated into individual cells (e.g. planned sample size, actual sample size). The extraction form was piloted prior to full data extraction. An example data extraction form is provided in Supplementary File 2. Data extraction was completed independently by two reviewers (C.A. and L.C.) and compared for consistency.
Outcome data
The primary outcome of this review was the proportion of patients retained at the host trial primary outcome time point using each recruitment strategy. If the primary outcome time point was undefined, the first time point after intervention was used. Where the SWAT reported retention to another time point, this time point was used.
Secondary outcomes were cost-effectiveness (cost per patient retained) if reported, and retention of participants at subsequent, secondary analysis, time points. In a post hoc analysis, initial intervention effects on recruitment rate were also considered.
Data items
Data was collected regarding the characteristics of both the host trial and the SWAT. The host trial collected details on disease area, setting, trial design, intervention and comparator, sample size, primary outcome measure, and recruitment and retention methods used. The SWAT collected details on study design, participant characteristics (age, gender/sex, ethnicity), intervention details, number of participants in each SWAT arm, and retention outcome.
Synthesis
Data from trials or SWATs with multiple publications were extracted and reported as a single entity. Multiple recruitment or retention interventions tested within the same host trial were extracted and treated as separate entities. No sub-group or sensitivity analyses were planned.
A flowchart of the selection process is presented. Key characteristics are summarised in tables and with trials grouped by type of SWAT intervention.
A narrative synthesis is presented for each intervention. SWATs at high risk of bias are included in the results; however, all results are discussed within the context of the risk of bias assessment. Where available, data on the cost-effectiveness of an intervention is presented if an intervention has been shown to be effective.
Results
Searches identified 7815 records of which 90 were assessed for eligibility through full-text review. Data was subsequently extracted from 15 records,13–27 with 10 records ultimately being deemed eligible for inclusion.18–27 The remaining five records were excluded for the following reasons: Not a SWAT (n = 2),13,14 host trial not randomised (n = 2),16,17 and SWAT was hypothetical (n = 1). 15 A PRISMA flowchart is presented in Figure 1. Five additional SWATs were identified that were ongoing.28–32
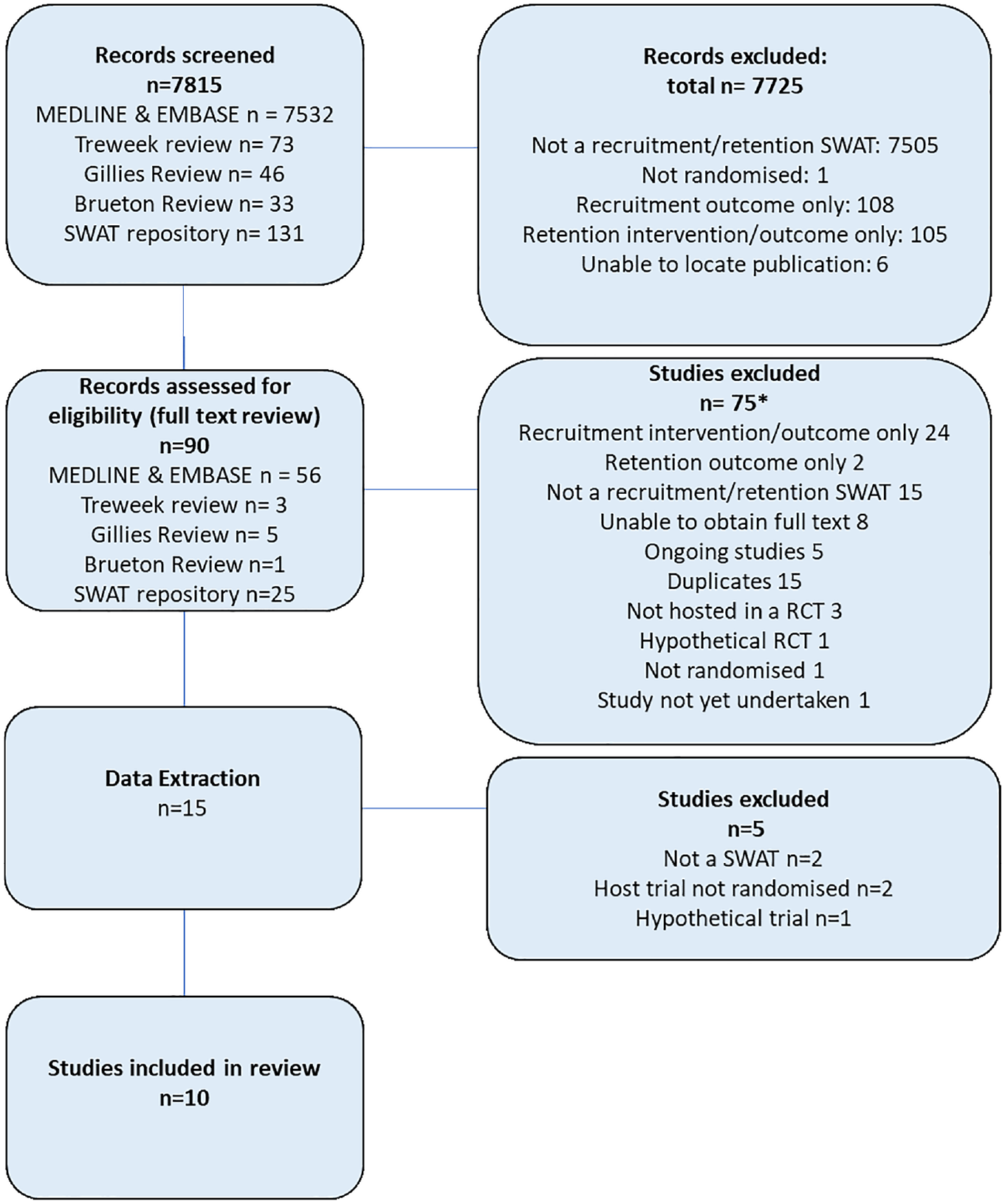
PRISMA flow diagram of screening.
SWAT and host trial characteristics
As summarised in Table 1, of the 10 SWATs included, there was a relatively even split in trial setting with six (60%) embedded in host trials in primary care18,19,23,25–27 and four (40%) in trials in secondary care.20–22,24 A broad range of disease areas were represented, with the most frequently represented being respiratory (n = 3, 30%),18,22,27 followed by podiatry23,25 and occupational therapy19,26 each with two trials. Most trials used patient-reported outcome measures (n = 6, 60%).22–27 Most trials (n = 6, 60%) used postal follow-up (patient self-completed diaries/calendars or questionnaires), with the remaining studies using follow-up in a clinic (n = 3, 30%) or telephone follow-up (n = 1, 10%). Four SWATs (40%) used unequal randomisation,19,21,23,27 with the majority (n = 3; 75%) favouring the SWAT control arm.19,21,23
Study characteristics (ongoing studies shaded in grey).
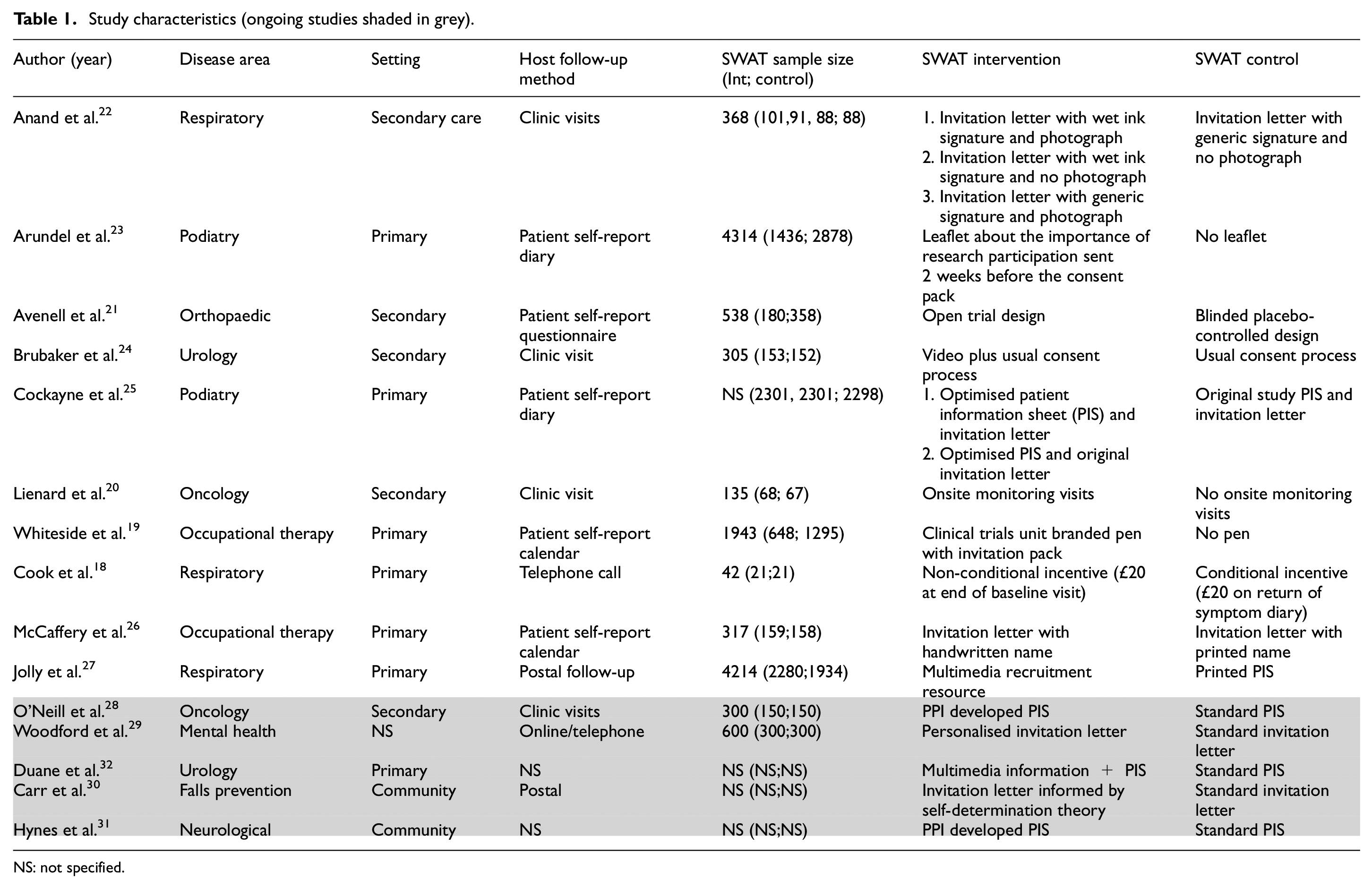
NS: not specified.
In line with the Online Resource for Recruitment research in Clinical Trials categorisation, and as detailed in Table 1, the majority of SWATs focussed on the information given to participants (n = 6, 60%),22–27 including modifications to patient information sheets (n = 2), modifications to invitation letters (n = 2), modifications to both (n = 1), and provision of a research information leaflet (n = 1). The remaining SWATs focussed on incentives (n = 2, 20%),18,19 and trial design (n = 1, 10%) 21 and recruiter interventions (n = 1, 10%). 20
Risk of bias
The 10 SWATs included in this review were assessed using either the Cochrane Risk of Bias 2 tool or the Cochrane Risk of Bias 2 Cluster Tool depending on design. 11 The results of the associated assessments are summarised in Figures 2 and 3.
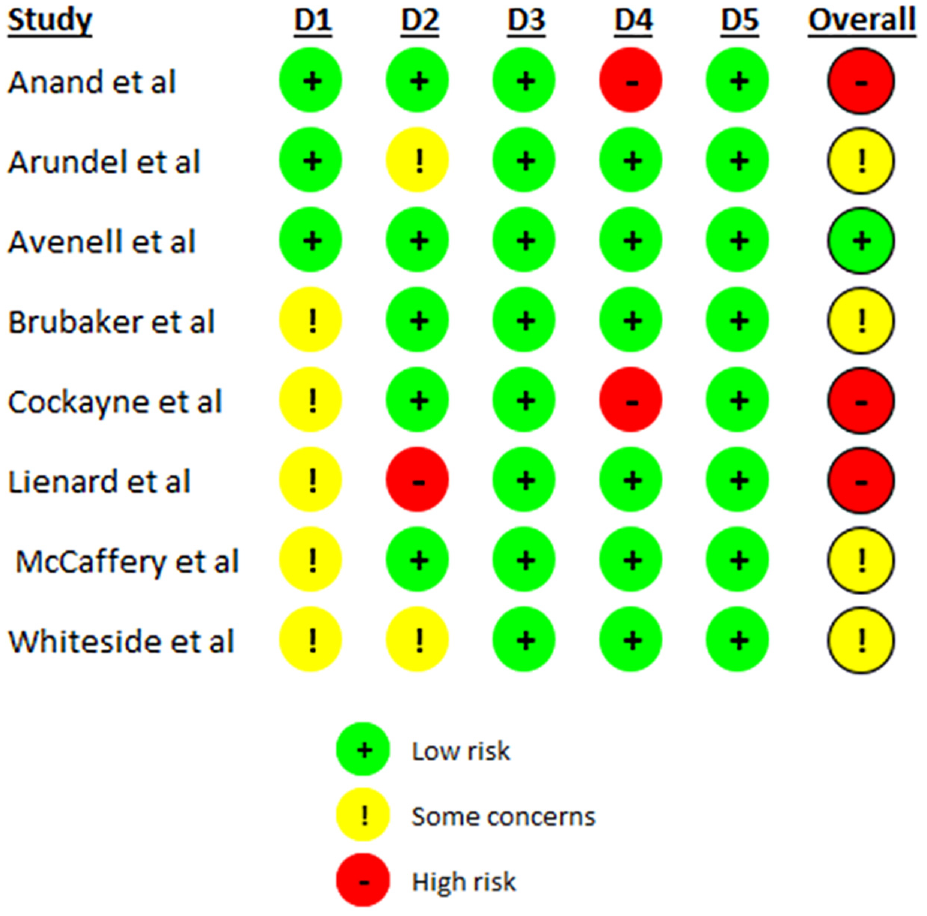
Risk of bias assessment of included individual RCTs.
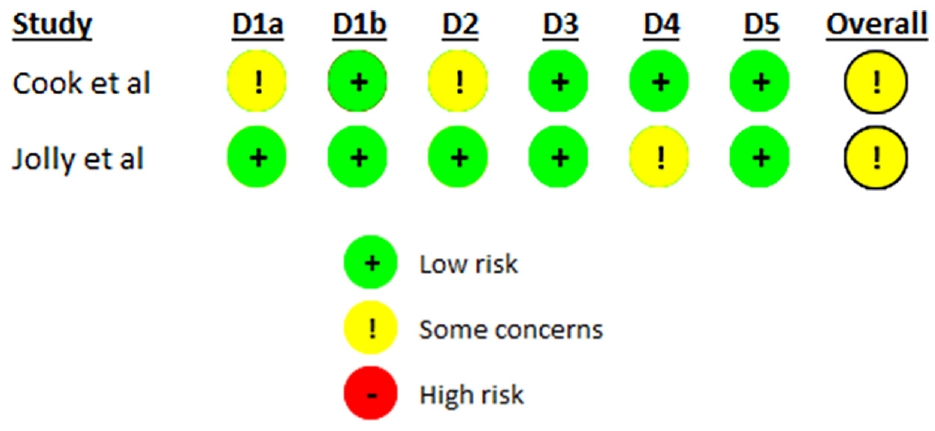
Risk of bias assessment of included cluster RCTs.
Overall, the majority of SWATs (n = 9) were assessed as having some concerns (n = 6) or a high risk of bias (n = 3).18–20,22–27 For all SWATs missing outcome data and selection of reported results were well reported. The reasons for some concerns were largely due to limited reporting of the randomisation process or outcome assessment or deviations from the intended intervention.
Analysis
Due to the lack of consistency in the interventions evaluated in the included SWATs, it was inappropriate to undertake a meta-analysis and hence a narrative synthesis was conducted.
Participant characteristics
Across the 10 included SWATs, patient demographic was not appropriate to report for one study given randomisation was at a centre level in relation to a recruiter intervention. 20 Of the remaining nine SWATs (90%), patient ethnicity was only reported in two instances,23,24 age in four SWATs,21,23–25 and gender or sex reported in only three SWATs.21,23,25 Aside from these no other demographic criteria were reported for any of the SWATs. Where data was reported, most participants were white (92%), females (92%) with an average age of 73 years.
Primary outcome
As shown in Table 2, of the six SWATs which focussed on the information given to participants,22–27 only five (83%) reported data on participant retention.22,24,25,27 Arundel et al. had proposed to conduct this but the results have not been reported with the reporting focussing on recruitment outcomes only. 23 Of these SWATs, the majority reported higher retention rates in the control group compared to the intervention group. The exceptions were the SWAT by Brubaker et al. who reported retention rates of 81% in the intervention group versus 78% in the control group, 24 and Anand et al. who reported 38% retention in the generic intervention letter with photograph compared to the control group (31% retention). 22 Only Anand et al. found any statistically significant difference between the groups, with an odds ratio of 5.40 (95% CI 1.12–26.15, p = 0.04) observed for the difference between letters including and excluding a photograph. 22
Summary of study results.
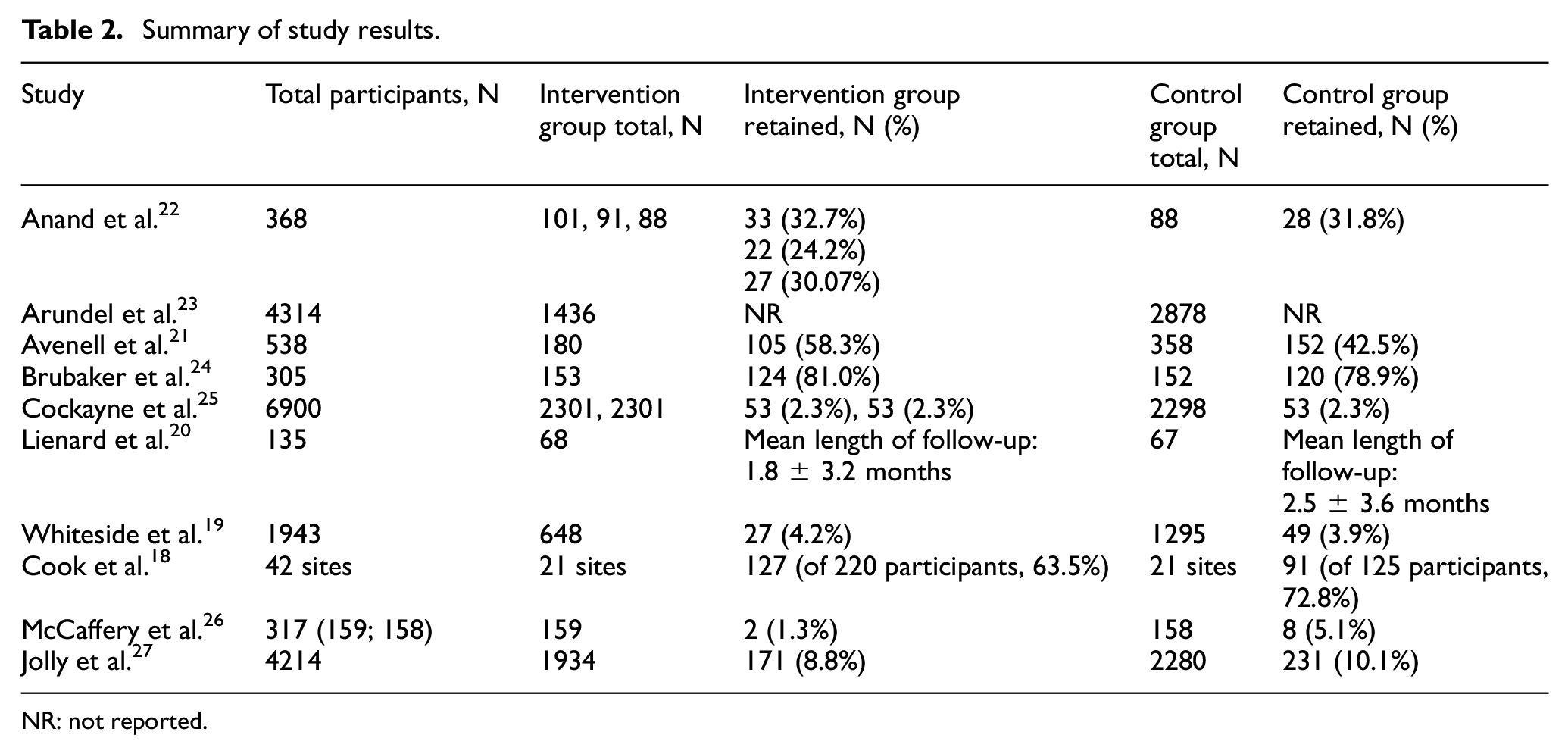
NR: not reported.
For the two SWATs which assessed incentives for participants, Whiteside et al. found that retention was higher in the intervention (pen) group compared to the control (no pen group); however, the result was not statistically significant (odds ratio: 2.63, p = 0.39). 19 Similarly, Cook et al. found higher retention in the conditional incentive (control) group; however, the finding was not statistically significant even when sensitivity analyses were conducted (p = 0.068). 18
The SWAT by Avenell et al., which assessed the impacts of presentation of trial design, found that more participants were retained in the intervention (open design) group compared to the control (blinded design) group (78.4% vs 65.2%); however, statistical significance testing of this finding was not reported. 21
Evaluation of the impacts of onsite monitoring versus no onsite monitoring by Lienard et al. did not assess retention in the strictest sense but assessed mean follow-up which was viewed within this review to be a proxy of retention. 20 The SWAT found that mean follow-up was longer in the control group compared to the intervention group (2.5 ± 3.6 months (control); 1.8 ± 3.2 months (intervention), however again the statistical significance of this result was not reported. 20
Secondary outcomes
Only two SWATs reported retention at other time points and neither found any statistically significant difference in retention; Brubaker at earlier time points (3 years: p = 0.13; 4 years: p = 0.13) 24 and Jolly at a later time point (12 months: odds ratio 0.79, 95% CI: 0.54–1.18). 27
The SWAT by Cook et al. was the only one to assess the cost-effectiveness of the intervention in the context of retention. 18 Including direct (voucher) and indirect (administrative time) costs the total cost was £5733 for the non-conditional incentive arm, equating to a cost of £45 per diary response received, compared with £2029 for the conditional incentive arm, equating to a per diary cost of £22. 18
Only the SWAT by Avenell reported evidence of intervention effectiveness on recruitment. 21 The use of an open design was found to increase recruitment by 9.4% (95% CI: 1.3–17.4) when compared to a blinded placebo-controlled trial. All other SWATs found no statistically significant effect of the intervention on study recruitment.
Discussion
This review identified only 10 eligible SWATs of recruitment interventions which also assessed the impacts of the intervention on trial retention. Given the heterogeneity between interventions, it was not possible to combine any of the included SWATs in a meta-analysis.
The majority of included SWATs focussed on the information given to participants which corresponds to the findings of Treweek et al., 9 where most recruitment SWATs (35/68, 51%) identified also focussed on modifications to the information given to participants. The majority of these reported higher retention rates in the control group compared to the intervention group and only one SWAT (letters including and excluding a photograph found any statistical significance between groups). 22 No statistically significant effects on retention were found for those SWATs assessing participant incentives,18,19 trial design presentation (open vs blinded), 21 or monitoring visits. 20
In this review, retention at other time points and cost-effectiveness of interventions were to be assessed; however, there was limited reporting of both secondary outcomes. No statistically significant differences were identified for retention at subsequent time points, and the only SWAT assessing cost-effectiveness, not unsurprisingly, found that conditional incentives were lower in cost than unconditional incentives. 18
Only one SWAT demonstrated intervention effectiveness in the context of recruitment, 21 with most of the remaining studies indicating a direction of intervention effect, albeit with no statistical significance.19,20,22,23,25–27 An important consideration here is that SWATs are often statistically underpowered, and hence require multiple replications and subsequent meta-analysis to identify evidence of effectiveness. Given that the individual SWATs included were single replications only, they are unlikely to have been able to ascertain effects individually. Subsequent replications may therefore indicate recruitment and retention effectiveness and thus contribute to the evidence base of interventions with dual effectiveness. Such strategies will undoubtedly be beneficial to the clinical trials community in terms of both trial efficiency and limiting future research waste. Similarly, there will also be benefits to participants if strategies can improve both recruitment and retention and result in participants seeing a timely, robust, and meaningful output from their participation.
A recent systematic review noted the lack of certainty with regard to cost-effective recruitment and retention interventions and recommended that economic evaluations be carried out alongside all SWATs to ensure cost-effectiveness can be appropriately evaluated. 33 This is particularly pertinent for recruitment SWATs assessing retention; if there are several effective recruitment strategies of similar cost, but one strategy is also better at improving retention, then it may be advantageous both from a deliver and a cost perspective to trials to utilise this method to minimise research waste. Given the paucity of evidence, it is therefore recommended that where trials undertake recruitment SWATs they also plan to and subsequently assess and report the impacts on retention to maximise trial, and SWAT, efficiency and thus limit research waste.
Limitations
Of the 7815 records screened for inclusion in this review, only 10 detailed assessments of the impacts of recruitment interventions on trial retention. This dearth of evidence therefore limited the ability to identify any potential interventions which may have an effect on both recruitment and retention in an RCT. Of the 10 included SWATs, only nine reported retention outcomes, with one detailing retention as a secondary outcome but noting the associated publication focussed on recruitment outcomes only. 23 A subsequent article on retention outcomes has not yet been developed. As a result, it is recommended that those undertaking recruitment SWATs plan to and subsequently undertake an assessment of intervention impacts on retention to allow the evidence base for effective strategies to build in this area.
Only SWATs embedded in RCTs in the fields of health and social care were included in this review, on the basis that the vast proportion of SWATs have been undertaken in host trials within this field. We acknowledge that this may have resulted in relevant SWATs outside of these fields being excluded; however, we do not anticipate any significant impact from this on the results presented.
Only publications in the English language were included in the review, thus introducing potential language bias. Given that no SWATs were identified in the search which were written in languages other than English, we view the impacts of this limitation to be limited.
While database search strategies exist for specific trial designs (e.g. RCT filters in MEDLINE), there is currently no search strategy for SWATs. As a result, we acknowledge the lack of use of a precise search strategy here; however, use of the search strategies used in Treweek’s Cochrane Review, 9 plus searching of a range of other resources aimed to ensure all relevant SWATs were captured. Irrespective, there is potential for some SWATs to have been missed; however, we anticipate minimal impact from this due to the range of databases and resources searched.
Difficulties in applying the domains in the Risk of Bias 2 tool 11 were identified given these do not necessarily fit easily with the SWAT design. Assessment of risk of bias independently by the two authors should have helped to mitigate any bias here.
Recommendations
There remains a need for a robust and rigorous assessment of the impacts of recruitment interventions on retention. It is therefore recommended that where trials undertake recruitment SWATs they also plan to and subsequently assess and report both the impacts on retention and the associated cost-effectiveness of the intervention. We acknowledge that this is not without its difficulties. One of the identified SWATs noted they would assess the impacts of the intervention on retention but did not subsequently undertake or report this. Given their timing, recruitment SWATs afford an opportunity for publication early in an RCT timeline, particularly where long-term retention is planned. This may also be driven by the need for timely reporting of SWATs to enable meta-analysis and so availability of definitive conclusions of intervention effectiveness. Where trials utilise long-term follow-up, trialists may wish to publish the initial recruitment SWAT results and follow with a separate publication on the impacts of retention.
Conclusion
This review has identified the limited assessment of the impacts of recruitment strategies, tested in a SWAT, on the retention of participants in the host trial. Assessment of the impact of recruitment interventions on retention is recommended to minimise future research costs and waste.
Supplemental Material
sj-docx-1-ctj-10.1177_17407745231206283 – Supplemental material for Do recruitment SWAT interventions have an impact on participant retention in randomised controlled trials? A systematic review
Supplemental material, sj-docx-1-ctj-10.1177_17407745231206283 for Do recruitment SWAT interventions have an impact on participant retention in randomised controlled trials? A systematic review by Catherine E Arundel and Laura Clark in Clinical Trials
Supplemental Material
sj-pdf-2-ctj-10.1177_17407745231206283 – Supplemental material for Do recruitment SWAT interventions have an impact on participant retention in randomised controlled trials? A systematic review
Supplemental material, sj-pdf-2-ctj-10.1177_17407745231206283 for Do recruitment SWAT interventions have an impact on participant retention in randomised controlled trials? A systematic review by Catherine E Arundel and Laura Clark in Clinical Trials
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Both C.E.A. and L.C. are employed at the University of York Trials Unit. Four SWATs included in this review had involvement by the York Trials Unit. C.E.A. is also the author of one of the included publications.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Availability of data
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
