Abstract
Background
Using information and communication technologies to seek, discuss, and share health-related information influences people’s trust and knowledge of several health practices. However, we know little about the associations between individuals’ information and communication technology use and their perceptions of trust and knowledge of clinical trials. Examining these associations may lead to the identification of target audiences and channels for developing effective educational interventions and campaigns about clinical trials.
Methods
In this study, we analyzed Health Information National Trends Survey data to document perceptions of clinical trial-related knowledge and trust that were recently added as questions in this annual national survey of US adults. We also examined correlates of these clinical trial perceptions that included sociodemographic factors and individuals’ use of information and communication technologies to seek health information, discuss such information with their healthcare providers, and share the information in their network.
Results
More than 90% of participants had no or limited perceived knowledge about clinical trials. Knowledge was higher among those who seek or discuss health-related information online. Differences in perceived knowledge and trust emerged for some racial/ethnic subgroups and other demographic factors. Providers were considered the most trusted source of information (73.6%), followed by health organizations (19.4%) and social support (7.1%). Trust in health organizations compared to health providers was higher among those who used online resources to share health information online with others. Trust in social support was significantly higher among those who used information and communication technologies to communicate about health.
Conclusion
Based on these findings, we recommend developing online resources about clinical trials to be distributed through social media. These resources should facilitate a dialogue and be targeted to several groups considering their information and communication technologies’ use.
Keywords
Introduction
Researchers involved in developing new medical treatments and procedures rely on clinical trials to compare the safety and efficacy of potential innovations with existing standards of care. 1 Successful clinical trials require accruing an adequate number of participants to meet enrollment targets, which can be either patients or healthy individuals, depending on the scope of the trial. Despite their importance, recruiting participants for clinical trials is very challenging for several reasons, and enrollment rates remain low.2,3 For example, despite a proliferation of clinical trials for cancer treatment, less than 10% of all cancer patients enroll in trials. 4 Several barriers may influence individuals’ decision to participate in clinical trials, including trial design features, participation costs, or travel distance to the research facility.5,6 Poor communication about clinical trials with potential participants has emerged as an additional critical barrier to enrollment.7,8 The Health Interview National Trends Survey (HINTS) is a nationwide study that has been administered annually or biannually in the United States since 2003 and is designed to facilitate research into the relationships between communication practices and health-related knowledge and perceptions. 9 Several new survey items related to clinical trials were added to the 2020 HINTS, presenting a unique opportunity to explore the relationships between communication behaviors and clinical trial perceptions among a national US sample. 10
Challenges of communicating about clinical trials
Several communication challenges contribute to individuals’ negative reactions toward medical research.7,8 First, healthcare providers and clinical researchers rarely invite patients to join clinical trials. 11 They are reluctant to talk with patients about clinical trials for a variety of reasons, including the intrinsic uncertainty of the research inquiry, the fear of damaging relationships with their patients, and implicit bias. 12 Many healthcare providers also lament limited time during medical appointments and a lack of resources available to support challenging discussions13,14 as reasons for limited conversations about joining clinical trials. Overall, patients are more likely to join clinical trials when their physicians openly discuss clinical trials.7,15 Beyond the critical role of doctor–patient communication in clinical trial recruitment and patients’ understanding of clinical research, data indicate that many patients learn about clinical trials from other patients, and friends or family members.16,17
Second, many information resources for patients are lengthy and complex to understand. 18 For example, the key takeaways from informed consent documents are understood by only 75% of individuals who are offered an opportunity to join clinical trials. 19 Some specific concepts, such as placebo and randomization, are complicated to understand and require communicators to simplify them by considering cultural adaptations20,21 and more engaging strategies to reach patients.22–25
Third, there are discrepancies in how clinical research has been experienced and discussed by individuals depending on their sociodemographic characteristics.12,26 Individuals from historically underrepresented groups, such as African Americans, have suffered abuses in the past that still influence their perceptions and experiences with clinical research. 26 These groups also have specific individual and relational needs that should be considered with greater attention when engaging in clinical research–related discussions.27–29 Everyday discriminations and lack of representation are other barriers that affect such discussions. 26
Finally, patient-specific factors should be taken into consideration when communicating about scientific discoveries and medical research. These include strategies to seek information and process messages. For example, Occa and Morgan have observed that individuals react differently to specific message formats depending on their levels of ability and motivation to process clinical trial–related information. 24 Hillyer and colleagues 30 interviewed cancer patients and discovered that about a quarter of participants in their sample did not know what clinical trials are. Their study echoes the findings from other projects 31 in which patients showed limited scientific literacy and challenges in understanding clinical trial–related information. In addition to challenges in processing and understanding clinical research, several segments of the population also show a marked mistrust, the belief that someone would harm their health or, more generally, would not act in their best interest, toward participating in clinical trials.28,32 Mistrust can be directed not only toward healthcare institutions, in general, such as hospitals, not-for-profit organizations, and governmental institutions, but also toward specific individuals involved in the research process, such as researchers, physicians, and clinical personnel.33,34 Mistrust in clinical research needs to be addressed by improving individuals’ perceptions of scientific institutions and researchers, and increasing their knowledge and understanding of the nature of the clinical research process.33,35 To do so, it is necessary to explore both the information resources that individuals trust when considering information about clinical research and what people think they know about clinical research. The source and resulting perceptions are likely to impact communication behaviors, including how individuals seek information, whether they discuss such information with their healthcare providers, and whether and how they share the information in their network.36–38 In this regard, special attention needs to be paid to the channels and resources used by individuals to seek health information.
Influence of information and communication technologies
Information and communication technologies (ICTs), such as phones and computers, applications, online resources, and social media, are key vehicles for individuals to retrieve health-related information and form opinions about health issues. 39 While there are no commonly agreed definitions of ICTs, researchers use the term to identify technologies, platforms, and programs which can improve access, effectiveness, and quality of healthcare information and practices. 40 The availability of multiple social media and technological platforms has resulted in more proactive health information seeking from sources other than healthcare providers. 41 People are also increasingly using social networks, blogs, virtual worlds, online games, and a variety of text messaging platforms to communicate about their health and to make decisions about health in general42,43 and clinical trials specifically. 44 The increased use of ICTs for healthcare purposes has been facilitated by the low cost of the Internet, continuous technological advancements, and the potential to customize the information retrieved to one’s own level of knowledge and understanding.40,42 ICTs have allowed individuals to be exposed to a variety of information with more autonomy than ever before.42,45 Individuals rely on several cues to judge the credibility of online information, 36 and some use such information in discussions with their healthcare providers.38,46
Although these technologies and new ways of communicating have improved access to health information, healthcare services, and providers, research focused on concerns with social media information has emerged. For example, individuals can struggle to identify reliable information due to the high volumes and dramatical tone of health risk messages online. 47 Individuals’ understanding and opinions about health issues is also affected by the structure of social media platforms. Researchers have conceptualized the existence of “echo-chambers” of like-minded individuals, which limit the consumption of diverse perspectives and information. 48 While these echo-chambers may be considered as target groups to address with dedicated communication efforts, it is also clear that creating interventions for highly polarized groups presents significant challenges, and without adequate understanding of the target audience, these interventions may also backfire in the long term. 49
Rationale for the current study
Researchers and public health experts need to identify target audiences and channels for developing effective educational interventions and campaigns about clinical trials. To support these efforts, this study has two key objectives. First, it aims to measure differences in perceptions of trust and knowledge about clinical trials by sociodemographic factors. Second, it aims to examine how differences in trust and knowledge varied depending on whether individuals used information technologies or online resources to seek health information, discuss with their healthcare providers, and make health decisions.
Methods
Data source
For this study, we analyzed data from the Health Information National Trends Survey 5 (HINTS 5) Cycle 4 (2020). 9 HINTS is administered by the National Cancer Institute and is a cross-sectional, nationwide survey of health communication and information among civilian, non-institutionalized US adults aged at least 18 years. HINTS investigators use a probability design and recruit participants from across the United States to produce a sample of individuals with diversity in racial/ethnic and sociodemographic characteristics. 10 HINTS data include analysis weights to account for selection probabilities and adjust for non-responsive households. HINTS 5 cycle 4 was fielded between February and June 2020, and participants completed a self-administered mailed questionnaire in English or Spanish. The response rate for HINTS 5.4 was 37%, and a total of 3865 respondents returned completed surveys.
Study outcomes and correlates
Primary outcomes
This study includes two main outcomes. The first is perceived knowledge of clinical trials, which was measured with one item: “How would you describe your level of knowledge about clinical trials?” Response options included “I don’t know anything about clinical trials,”“I know a little bit about clinical trials,” and “I know a lot about clinical trials.”
The second outcome is health information source trust, assessed using one item: “Imagine you had a need to get information about clinical trials. Which of the following would you most trust as a source of information about clinical trials?” Response options included “My healthcare provider,”“My family and friends,”“Government health agencies,”“Health organizations or groups,”“Disease-specific patient support groups,” and “Drug companies.” For the purposes of the current analyses, we combined the responses of “Government health agencies” and “Health organizations or groups” into a single response which we labeled “Professional Health Organizations.” The responses “my family and friends” and “disease-specific patient support groups” were also combined into a single response labeled “social support.” We excluded “drug companies” from the analyses due to a small number of responses (n = 16, 0.4% of the sample).
Sociodemographic and information correlates
Correlates were selected considering standard sociodemographic variables and a review of the existing literature on barriers to clinical trials and ICTs to support health communication. 50 Sociodemographic factors included gender, race/ethnicity, age, education, income, geographical location, and cancer history. Health communication measures were designed to assess whether individuals used ICTs to seek clinical trial–related information, discuss such information with their healthcare providers, and share information in their networks. Although these items did not specifically ask about the use of ICTs for healthcare research, prior research has demonstrated a relationship between these general communication constructs and several health-relevant behaviors.51,52 Table 1 indicates the ICT-related items used from the HINTS dataset which all have dichotomous yes or no response options: (a) used the Internet to look for health-related information, (b) used emails to communicate with their healthcare providers, (c) used ICTs (tables or smartphones) to discuss or take decisions about their health, (d) visited a social networking site or used a social networking site to share health-related information, (e) watched health-related videos on YouTube, and (f) used the Internet for social support.
Information and communication technologies–related items from the Health Interview Trends Survey.
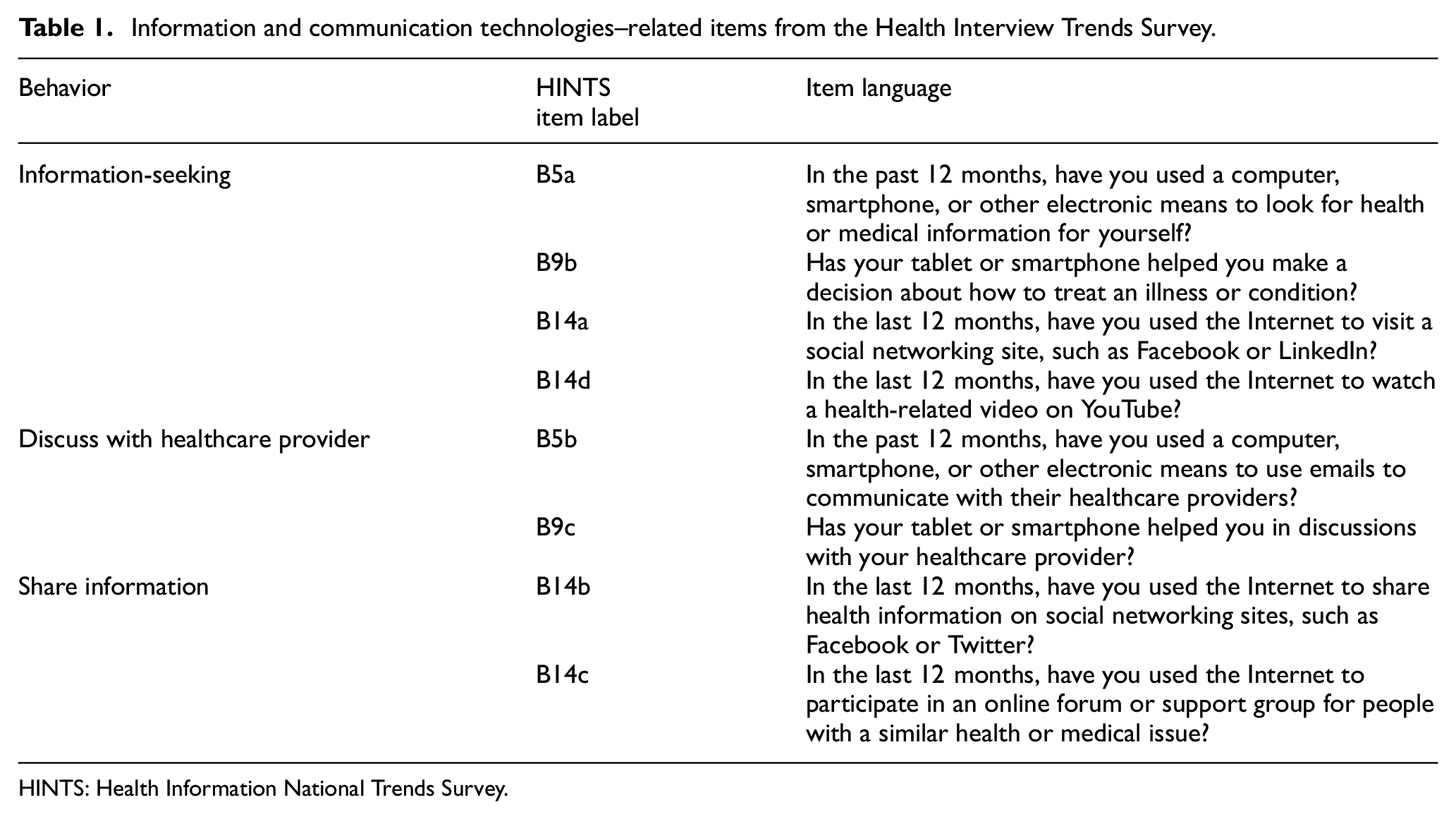
HINTS: Health Information National Trends Survey.
Data analysis
All analyses were estimated using Complex Samples in SPSS Version 26 53 to incorporate analysis weights that correct for nonresponse and noncoverage biases. Prevalence estimates are percentages and corresponding 95% confidence estimates generated using the SPSS Complex Samples Crosstabs procedure. Associations were first examined with unadjusted (bivariable) logistic regression models to identify whether there are independent relationships between each correlate and outcome. An additional adjusted (multivariable) model was conducted with all the correlates included as independent variables in the same model to account for potential collinearity in sociodemographic and information communication factors and to account for differing levels of missing data across individual variables. Nominal, two-sided 5% significance levels were applied for generating confidence intervals. Data analysis for this project was approved by the University of Kentucky Medical Internal Review Board (IRB# 67898).
Results
Participants
Participants’ non-weighted responses to correlates are presented in Table 2. Percentages of the largest race/ethnicity response categories are similar to 2020 US Census estimates: Non-Hispanic White: 61.6% Census/61.1% HINTS; Hispanic: 18.7% Census/17.1% HINTS; Non-Hispanic Black: 12.4% Census/13.8% HINTS; Non-Hispanic Asian: 6.0% Census/4.6% HINTS. The majority of participants were female (57.9%) and 50 years of age or older (combined 68%). For socioeconomic status–related variables, 43% of participants completed a Bachelor’s Degree or higher and 34% reported an annual income of US$75,000 or higher.
Frequencies of outcomes, sociodemographic, and information and communication technologies–related survey questions from the Health Interview Trends Survey.
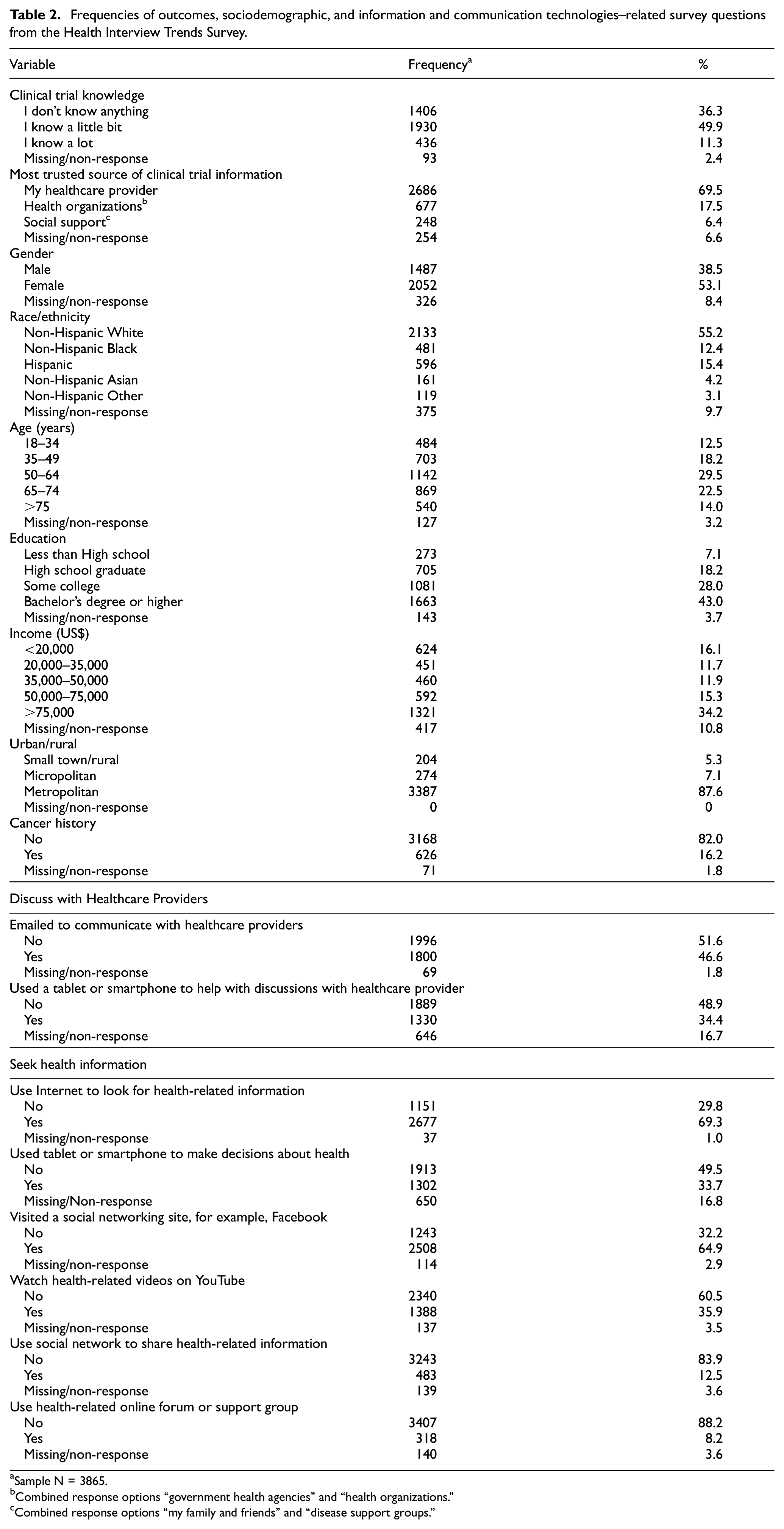
Sample N = 3865.
Combined response options “government health agencies” and “health organizations.”
Combined response options “my family and friends” and “disease support groups.”
Perceived knowledge of clinical trials: unadjusted models
For the entire sample, 41.3% of participants indicated that they don’t know anything about clinical trials, 49.3% perceived knowing a little, and 9.4% perceived knowing a lot about clinical trials (Table 3). In unadjusted models, non-significant differences were observed for knowledge based on gender, urban/rural, and cancer history. Differences in knowledge were observed for some racial/ethnic subgroup comparisons. Specifically, Hispanics were significantly less likely than non-Hispanic Whites to report knowing a little (odds ratio (OR): 0.54, 95% confidence interval (CI): 0.37–0.79) or knowing a lot (OR: 0.52, 95% CI: 0.27–1.00) compared to don’t know. Non-Hispanic Asians were significantly less likely than Whites to know a little compared to don’t know (OR: 0.52, 95% CI: 0.33–0.82). Participants above 75 years old were significantly less likely than adults between 18 and 34 years old to report knowing a little (OR: 0.62, 95% CI: 0.43–0.89) or knowing a lot (OR: 0.47, 95% CI: 0.25–0.89) compared to don’t know. Higher levels of education were associated with more knowledge compared to less than a high school degree in several comparisons. Significant differences were found in comparing “know a little” and “don’t know” with the two highest income levels (US$50,000–US$75,000 versus <US$20,000: OR: 1.69, 95% CI: 1.12–2.55; >US$75,000 versus <US$20,000: OR: 1.76, 95% CI: 1.25–2.47). Individuals in the highest income category were also significantly more likely to report “know a lot” compared to the reference category (> US$75,000 versus <US$20,000: OR: 1.95, 95% CI: 1.05–3.63).
Prevalence and correlates of perceptions of knowledge of clinical trials by demographics, health status, and health communication behaviors.
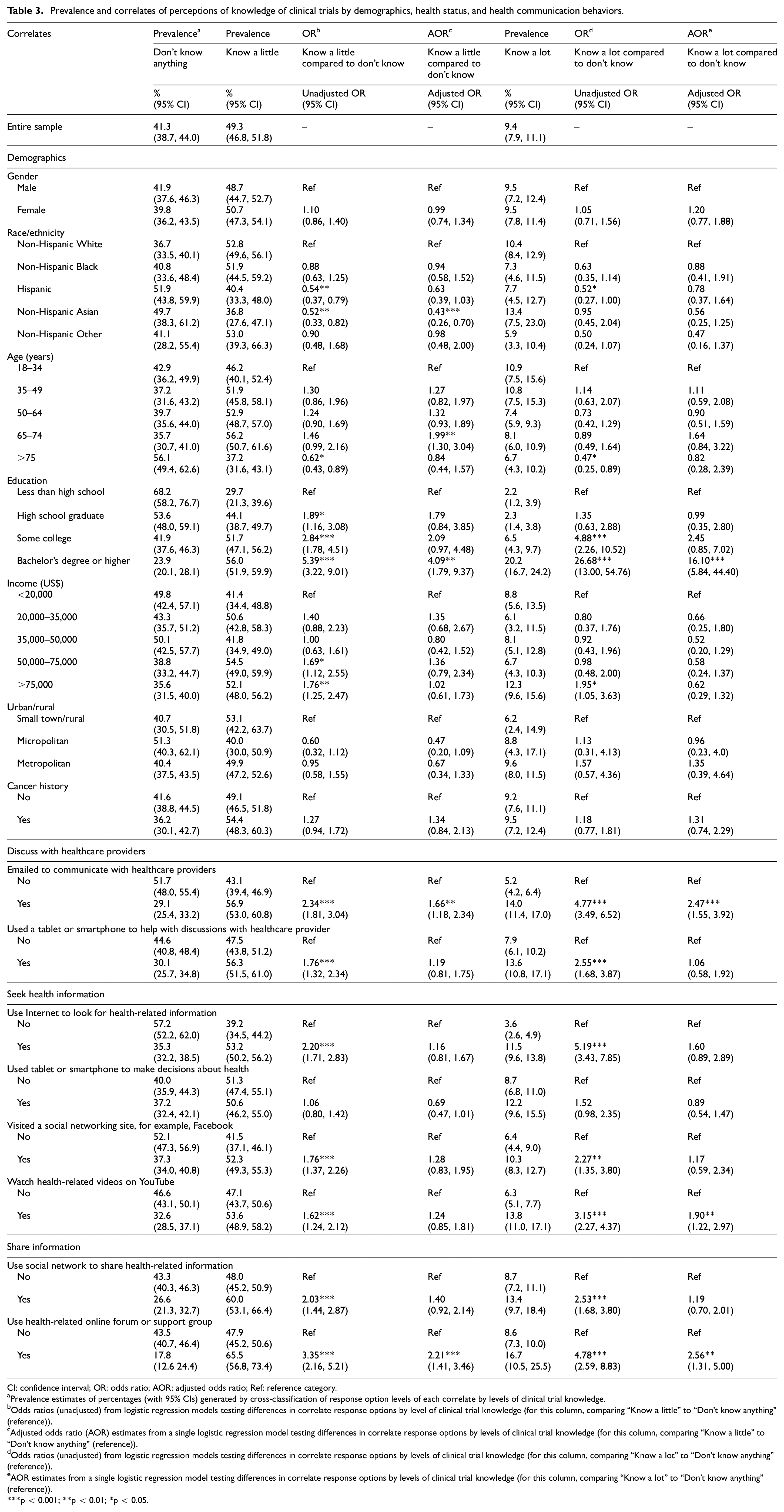
CI: confidence interval; OR: odds ratio; AOR: adjusted odds ratio; Ref: reference category.
Prevalence estimates of percentages (with 95% CIs) generated by cross-classification of response option levels of each correlate by levels of clinical trial knowledge.
Odds ratios (unadjusted) from logistic regression models testing differences in correlate response options by level of clinical trial knowledge (for this column, comparing “Know a little” to “Don’t know anything” (reference)).
Adjusted odds ratio (AOR) estimates from a single logistic regression model testing differences in correlate response options by levels of clinical trial knowledge (for this column, comparing “Know a little” to “Don’t know anything” (reference)).
Odds ratios (unadjusted) from logistic regression models testing differences in correlate response options by levels of clinical trial knowledge (for this column, comparing “Know a lot” to “Don’t know anything” (reference)).
AOR estimates from a single logistic regression model testing differences in correlate response options by levels of clinical trial knowledge (for this column, comparing “Know a lot” to “Don’t know anything” (reference)).
p < 0.001; **p < 0.01; *p < 0.05.
In unadjusted analyses of health communication behaviors, knowledge was higher among those who used email to communicate with healthcare providers, used technology to discuss their health, used the Internet to look for health-related information, visited social networking sites, watched health-related videos on YouTube, shared health information on social networks, and used the Internet for social support.
Perceived knowledge of clinical trials: Adjusted models
As in the unadjusted models, non-significant differences were observed for perceptions of knowledge based on gender, urban/rural, and cancer history in adjusted analyses. Differences in knowledge among Hispanics were non-significant but non-Hispanic Asians were significantly less likely than Whites to know a little (adjusted odds ratio (AOR): 0.43, 95% CI: 0.26–0.70) compared to don’t know. Participants between 65 and 74 years old were significantly more likely than between 18 and 34 years old to report knowing a little (AOR: 1.99, 95% CI: 1.30–3.04) compared to don’t know. Several effects of education levels were no longer significant in adjusted models except among when comparing the highest and lowest education groups in both higher levels of knowledge (AOR: 4.09, 95% CI: 1.79–9.37; AOR: 16.10, 95% CI: 5.84–44.40). Two ICT variables were significantly associated with both higher levels of knowledge: the use of email to communicate with providers (AOR: 1.66, 95% CI: 1.18–2.34; AOR: 2.47, 95% CI: 1.55–3.92) and use of online forums and health groups (AOR: 2.21 95% CI: 1.41–3.46; AOR: 2.56, 95% CI: 1.31–5.00). Participants who watched health-related videos on YouTube were more likely to report knowing a lot about trials (AOR: 1.90, 95% CI: 1.22–2.97).
Most trusted source of health information regarding clinical trials: unadjusted models
For the entire sample, 73.6% of participants indicated they most trusted providers, 19.4% indicated health organizations, and 7.1% indicated social support (Table 4). In unadjusted models, non-significant differences were observed for sources of trusted information based on gender, income, and cancer history. Hispanic participants were more likely to trust health organizations over providers (OR: 1.58, 95% CI: 1.07–2.35) compared to non-Hispanic White participants. Compared to participants between the ages of 18 and 34 years, participants aged 50–64 (OR: 0.58, 95% CI: 0.39–0.86), 65–74 (OR: 0.57, 95% CI: 0.39–0.85), and 75 years and older (OR: 0.51, 95% CI: 0.27–0.94) were less likely to trust health organizations versus providers. Participants with some college education (OR: 2.34, 95% CI: 1.08–5.07) or a Bachelor’s degree or higher (OR: 2.56, 95% CI: 1.20–5.49) were more likely to report trusting healthcare organizations versus providers compared to participants with less than a high school education. Some college education was also associated with higher trust in social support versus providers (OR: 2.71, 95% CI: 1.05–7.04). Participants in the highest income category were significantly more like to trust social support over providers (OR: 3.23, 95% CI: 1.70–6.11). Those with a history of cancer were less likely to trust social support over providers (OR: 0.51, 95% CI: 0.32–0.83). For the ICT variables, trust in health organizations compared to health providers was higher among those who used email to communicate with healthcare providers, used the Internet to look for health-related information, watched health-related videos, and used health support Internet group. Trust in social support was significantly lower than providers among those who used technology to help with provider discussions (OR: 0.49, 95% CI: 0.31–0.7). However, trust in social support was significantly higher than providers for those who used the Internet to seek health information, visited a social network site to share health information, and used the Internet for health videos.
Prevalence and correlates of most trusted source for clinical trials-related health information by demographics, health status, and health communication behaviors.
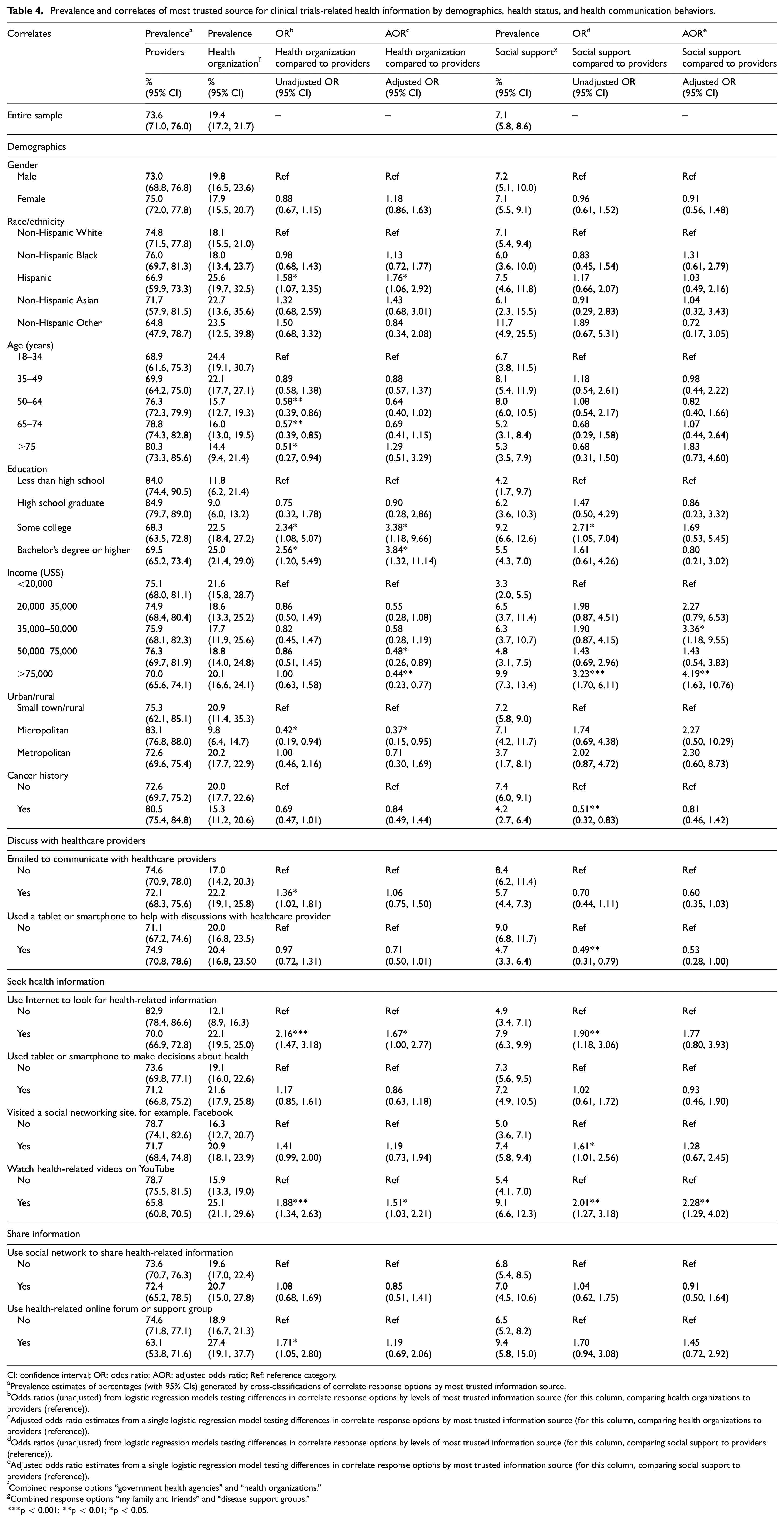
CI: confidence interval; OR: odds ratio; AOR: adjusted odds ratio; Ref: reference category.
Prevalence estimates of percentages (with 95% CIs) generated by cross-classifications of correlate response options by most trusted information source.
Odds ratios (unadjusted) from logistic regression models testing differences in correlate response options by levels of most trusted information source (for this column, comparing health organizations to providers (reference)).
Adjusted odds ratio estimates from a single logistic regression model testing differences in correlate response options by most trusted information source (for this column, comparing health organizations to providers (reference)).
Odds ratios (unadjusted) from logistic regression models testing differences in correlate response options by levels of most trusted information source (for this column, comparing social support to providers (reference)).
Adjusted odds ratio estimates from a single logistic regression model testing differences in correlate response options by most trusted information source (for this column, comparing social support to providers (reference)).
Combined response options “government health agencies” and “health organizations.”
Combined response options “my family and friends” and “disease support groups.”
p < 0.001; **p < 0.01; *p < 0.05.
Most trusted source of health information regarding clinical trials: adjusted models
Findings were similar in adjusted models for gender, race/ethnicity, education, and urban/rural status. However, age differences in trust of health organizations over providers became non-significant in adjusted models. Participants in the two highest income categories were significantly less likely to trust health organizations over providers (US$50,000–US$75,000 versus <US$20,000: OR: 0.48, 95% CI: 0.26–0.89; >US$75,000 versus <US$20,000: OR: 0.44, 95% CI: 0.23–0.77). Differences in trust based on cancer history were non-significant. For ICT variables, both who used the Internet for health-related information and watched videos were associated with significantly more trust in health organizations compared to providers. In adjusted models, watching for health videos remained significantly associated with trust in social support over providers.
Discussion
Statement of principal findings
In this study, we used HINTS data to explore differences in perceptions of trust and knowledge about clinical trials in a sample of US adults. We also explored correlates of these perceptions that included sociodemographic factors and whether individuals used ICTs to seek health information, engage in discussions with their healthcare providers, or make health decisions. Several interesting findings emerged.
First, our findings suggest most US adults perceive little to no knowledge about clinical trials, supporting the need to more systematically provide informational and educational resources for the general population about clinical trials and medical research. In addition, Hispanics and Asian respondents reported lower knowledge perceptions compared to non-Hispanic Whites. However, these observed differences for Hispanics were non-significant after adjusting for other sociodemographic and information behaviors. In line with previous research,54,55 our results support the need to further document the informational needs of specific groups and the potential value of developing culturally sensitive educational and informational resources.
A second important finding was that knowledge perceptions varied along with patients’ general use of social media, online support groups, and YouTube to retrieve health-related information. Although HINTS survey questions did not specifically focus on social media use for retrieving clinical trial information, this finding extends existing literature suggesting that patients may be exposed to clinical trial–related information when online and they may be actively looking for this information on social media and among peers, or among patients with similar experiences or conditions. 44 Therefore, it may be beneficial to identify and provide online resources and links that eligible patients can consult on their own before and after having been introduced to an opportunity to join a clinical trial or participate in a medical research study. Internet links and information may also need to be distributed through social media to facilitate patients in identifying available clinical trials through government websites and other official resources. These materials for online distribution should be designed to consider both the information needs of patients and the social dynamics happening online. In adjusted models, using email to communicate with providers and engaging in online support groups related to health emerged as significant factors driving knowledge. This suggests including patient activation strategies as part of clinical trials enrollment efforts could lead to improvements in potential participants’ clinical trials–related literacy. These strategies have been successful in improving psychological and behavioral outcomes of patients dealing with complex illnesses. 56
A third finding concerned the role of trust in clinical trials sources of information and how trust varied considering social media use. It is promising that most participants indicated their provider was their most trusted source of information (73.6%). The remaining participants trusted either professional health organizations (19.4%) or social support (7.1%) which demonstrates the value of providers integrating messages from health organizations or government sources in their clinical trial communications and including patients’ family or caregivers in clinical trial–related discussions. Using the Internet to gather health information and watching health-related videos online were associated with greater trust in professional health organizations. Contrasted with growing concerns about the proliferation of inaccurate health information and increasing mistrust in the medical field, it is encouraging that professional health organizations appear to be trusted sources among those seeking health information online. There was little evidence of systematic differences in trust for other sociodemographic or communication factors. Given that trust is a significant component of a perceived source credibility 58 and that being credible is essential when communicating health information, it is imperative to continue examining how to build trustful relationships and online exchanges with patients when discussing clinical trials.
Strengths and limitations
This study contributes to the literature in several meaningful ways. First, it is one of the few studies examining associations between ICTs use and trust and knowledge of clinical trials. Second, it takes advantage of the most recent HINTS, a publicly available dataset that captures health and communication–related perceptions from individuals across the United States. The distribution of race and ethnicity of HINTS participants in the current sample closely mirrors 2020 US Census results 57 which allowed us to also explore clinical trial perceptions in a diverse sample.
A few limitations should be taken into consideration when evaluating the implications from this study. First, we used cross-sectional data, which prevented us from examining causal inferences. Second, the existing HINTS measures capture an important but narrow range of ICTs. This range of ICTs behaviors is collected through simple categorical measures. This limits the opportunity to examine a detailed picture of ICTs use to communicate about clinical research and health. Third, self-reported data may be affected by reporting or recall bias. Fourth, some of the differences observed by groups may have been exacerbated by the COVID-19 crisis and should be considered with caution when the crisis would be resolved. Fifth, we examined associations between ICTs use and trust and knowledge of clinical trials. While this association represents useful information, it is important to specify that there were no specific questions in the HINTS survey that explicitly examined whether ICTs were used for healthcare research. Finally, missing data is present for each variable and outcome which means that although HINTS does provide a nationwide sample of adults, results of analysis with missing data are not truly representative of the US population and may not be directly comparable across analyses.
Implications for policy, practice, and research
These finding have implications for developing communication and education interventions to address some of the challenges and needs that emerged from this analysis of HINTS data. Such interventions should strive to provide informational and educational resources for the general population about clinical trials and medical research, especially targeting specific groups. Considering the important role of trust and online behaviors, healthcare professionals should focus on creating a dialogue rather than a top-down form of communication about clinical trials and participation in medical research. Links and information should be made available through social media to facilitate patients in identifying available clinical trials through governmental websites and other official channels. However, future studies should focus on identifying the best strategies to distribute such materials to reach patients and citizens in general more effectively. Our study suggests YouTube may be an important channel in efforts to recruit participants or communicate about medical research innovations. Future studies are needed to identify strategies to optimize communication on YouTube and to explore its use in complementing other resources or social media outlets.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by pilot funding provided by the National Cancer Institute (NCI) through the University of Kentucky Markey Cancer Center’s Cancer Center Support Grant (P30 CA177558). The study sponsor had no role in the conduct, review, or generation of this report. Content is solely the responsibility of the authors and does not necessarily represent the official views of the NCI.
