Abstract
The marketing and sale of oxycodone (OxyContin) by Purdue Pharma has commanded a great deal of legal and policy attention due to the drug’s central role in the ongoing overdose crisis. However, little is known about the basis for OxyContin’s approval by regulators, such as Health Canada in 1996. Taking advantage of a recently created online database containing information pertaining to the safety and effectiveness of drugs, we conducted a retrospective analysis of Purdue Pharma’s submission to Health Canada, including both published and unpublished clinical trials. None of the trials sponsored by Purdue Pharma sought to meaningfully assess the risks of misuse or addiction associated with OxyContin. The trials were short in duration (maximum length was 24 days) and only assessed safety and efficacy of a 12-h dosing interval. Also, the two trial reports that explicitly mentioned (but did not formally evaluate) the risk of misuse were not published, making it unclear how Health Canada concluded that there was no risk of misuse. In our view, these findings underscore the need for transparency of not only of clinical trial data, but also the regulator’s interpretation of such data, which is currently lacking in Canada. Furthermore, they call into question why Health Canada’s role in precipitating the overdose crisis has not received greater scrutiny, including in the context of recent litigation surrounding OxyContin.
Purdue Pharma, Health Canada and the overdose crisis
Physician overprescribing of controlled-release oxycodone (OxyContin) following the drug’s approval in the 1990s is one of the early drivers of the overdose crisis in North America. OxyContin’s manufacturer, Purdue Pharma, and other companies engaged in its distribution, have been the subject of thousands of lawsuits in the United States 1 and, to a lesser degree, Canada 2 owing to the marketing tactics they allegedly employed.3,4 Despite this scrutiny, there is limited public knowledge about how OxyContin garnered regulatory approval in the first place. While opioid-based pharmaceuticals were understood to carry significant risks for decades,5,6 clinical resistance to prescribing opioids began to soften in the mid-1980s.7,8 It is unclear whether this shift dovetailed with, or fed into, the evidence supplied by Purdue to regulators.
Questions have been raised about whether regulators, including Health Canada, contributed to the current overdose crisis 9 by understating the risk of misuse in the original product monograph at the time of approval in 1996 10 and failing to take stronger enforcement actions in view of aggressive marketing tactics that were deployed by Purdue Pharma post-approval.3,11,12 Health Canada also neglected to follow the United States’ Food and Drug Administration in changing the OxyContin label to add a statement about the potential for misuse in 20019,13 despite the evidence that emerged post-approval revealing the increased demand for OxyContin among people who use drugs. 14 Until now, however, no systematic investigation of the clinical data originally submitted by Purdue Pharma to Health Canada has been conducted because the data were not publicly available.
Taking advantage of recent changes in Canadian law, we aim to shed light on the evidence underlying Health Canada’s approval of OxyContin. Specifically, following the passage of ‘Vanessa’s Law’ in 2014 Health Canada has put into place a new online portal named the ‘Public Release of Clinical Information’. 15 The portal includes a wealth of data pertaining to safety and effectiveness related to drugs approved by Health Canada after March 2019, when the portal was launched, and also pertaining to older drugs, including OxyContin. Downloading a range of data available from the portal, we report findings from our retrospective analysis of the clinical evidence underpinning Health Canada’s 1996 approval of OxyContin.
The evidence behind OxyContin’s approval
OxyContin clinical data that were submitted by Purdue Pharma to Health Canada for regulatory evaluation were retrieved from the portal’s website 16 for document-based retrospective analysis. Due to the voluminous amount of data in the submission, we first sought to identify all of the research studies that were included or referenced in the submission. We then categorized all studies by study type, including bioavailability reports, comparative bioavailability and bioequivalence reports, healthy subject pharmacodynamic and pharmacokinetic study reports, and clinical study reports. Focusing on the clinical trials sponsored by Purdue (see complete list of trials in Appendix 1), we determined whether or not each safety/efficacy trial had been published in a peer-reviewed journal and deduced the key elements of each trial’s design (trial phase, sample size, randomization and blinding, intervention and study timeframe, and number of subjects that reported adverse effects). In addition, we looked at side effect profiles, dosing regimens, misuse potential, and other literature referenced in the body of the submission. The findings in the studies that Purdue relied upon to establish OxyContin’s safety and efficacy were further compared against historical literature regarding the safety, efficacy, and misuse potential of opioid analgesics. Finally, the original OxyContin product monograph was used to ascertain what Health Canada concluded based on the evidence contained in the submission.
Safe and effective absent an evaluation of the risks of misuse and addiction?
OxyContin’s product monograph concluded that the drug was safe and effective for the treatment of ‘moderate’ – as opposed to acute end-of-life, cancer-related – pain. Furthermore, the monograph recommended that dosage should be increased if pain relief did not persist for the full 12 h, stating: ‘If a breakthrough pain repeatedly occurs at the end of the dosing interval it is generally an indication for a dosage increase rather than more frequent administration’. Consistent with how the drug was subsequently marketed by Purdue, 17 no maximum dose was specified in the monograph. As well, the monograph stated that the risk of misuse ‘is not a problem in patients with pain in whom [OxyContin] is indicated’. The monograph was silent with respect to the risk of addiction.
A variety of clinical evidence underlies the conclusions contained in the product monograph. Specifically, a total of six clinical trials designed to assess the safety and efficacy of OxyContin (including an open-label study continuing two randomized trials) were identified in Purdue’s submission (see Table 1 for key details). The trials ranged from 57 to 182 participants, and durations between 24 h and 24 days (excluding the open-label study that spanned 12 weeks beyond the two randomized trials it continued). Two of the six trials were not published in peer-reviewed journal articles. Beyond the six Purdue-sponsored trials included in the submission, an additional 24 clinical studies, reports and reviews were also referenced in the submission package (see Appendix 2 for a complete list).
Description and content of safety and efficacy trials of OxyContin submitted by Purdue Pharma to Health Canada by Purdue.
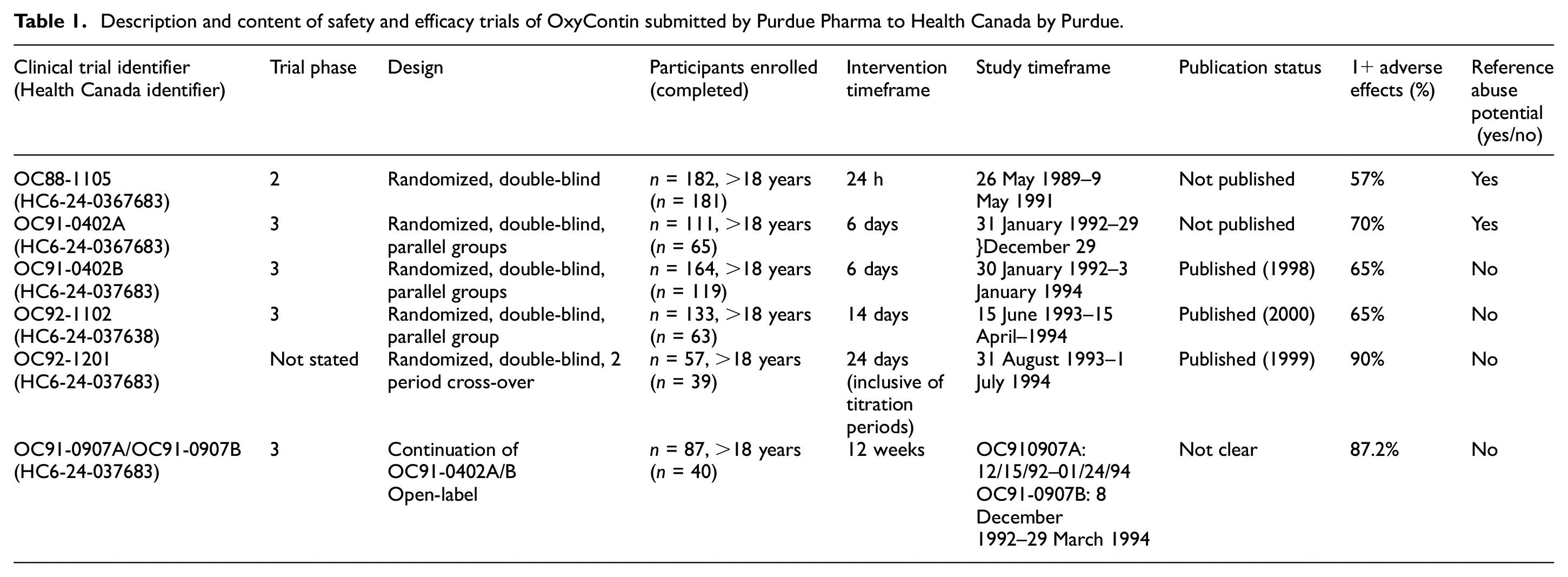
The conclusions reached in the product monograph were not supported by this body of clinical evidence. The trials performed by Purdue were all short in duration and did not assess OxyContin’s effectiveness for treating moderate, chronic pain.
Risk of misuse (described pejoratively as ‘abuse’) of OxyContin was mentioned in only two (OC88-1105 and OC91-402B) of the six safety and efficacy trials submitted to Health Canada. In the body of the report for trial OC88-1105, a statement is made that the controlled-release formulation of oxycodone may have less abuse potential than drugs such as Percodan [which also contains oxycodone] for several reasons. Most illicit drug abusers prefer a drug that is rapidly acting. The […] formulation will have longer acting effect without producing an immediate euphoria
Study OC88-1105 did not attempt to assess or show whether the abuse/misuse potential of OxyContin was in fact less than Percodan or other similar, but differently formulated, drugs. Similarly, OC91-402B listed withdrawal as an ‘unusual and rare adverse experience’. Yet, this statement was not formally measured nor supported by evidence generated in the trial.
Despite the absence of a statement about the risk of addiction in OxyContin’s product monograph, five (of the 24) studies that were referenced in Purdue’s submission (studies numbered 4, 6, 7, 8, and 22 in
Contrary to the findings of this referenced literature, a number of statements contained in Purdue’s submission were to the effect that the risk of addiction was ‘minimal’ due to the controlled-release formulation and the recommended time between doses. Purdue also did not account for – either in the trials it sponsored or in response to the literature it referenced in the body of its submission – the possibility of the tablets being consumed at intervals other than 12 h.
Finally, we found no reference to two publications – a letter to the editor by Porter and Jick that was published in the New England Journal of Medicine in 1980 21 and a 38-patient case series by Portenoy and Foley published in Pain in 1986 22 – that have been shown to have played an important role in liberalizing prescriber attitudes towards opioids during the 1980s, 1990s, and early 2000s.7,8,23
Gaps in transparency and a lack of accountability: continuing fallout from health Canada’s decision to approve OxyContin
Our retrospective analysis of the data tendered by Purdue in support of its submission to Health Canada reveals that the evidence base behind OxyContin’s approved indication for the treatment of non-cancer related chronic pain was weak. Although chronic pain has been long understood as persistent pain for over 3–6 months, 24 the longest Purdue-sponsored trial spanned a mere 24 days and dosing was only evaluated at 12-h intervals. Therefore, Purdue’s submission did not provide persuasive evidence in support of the indication for chronic pain, nor did it supply meaningful evidence pertaining to the risks of addiction and misuse associated with use of OxyContin.
Importantly, regulators, such as Health Canada had not – at the time of OxyContin’s original approval – fully developed their guidance to standardize how the risk of ‘abuse potential’ was to be assessed during the different phases of drug development. How best to assess that risk in the context of pre-clinical studies involving animal models was, for instance, the subject of significant scientific debate. 25 Regulators only formalized their guidance through the mid-2000s as concerns about addiction and misuse of OxyContin grew. 25 The fact that the preferred methods to evaluate these risks was not yet clear, however, does not mean that these risks can be ignored altogether. Indeed, at the time of OxyContin’s approval it was generally understood that substantial physiologic addiction occurs over periods of several days to weeks. 26 Yet, with the exception of one 24-day trial, none of the evidence generated by Purdue was long in duration. Thus, Purdue’s claim that the risk of misuse was low was not well supported.
Consistent with previous research, our analysis also shows that the evidence pertaining to OxyContin was only partially reported in the published medical literature. The two studies which included statements about – but did not actually evaluate – the risk of misuse related to OxyContin were not published.
It is also important to note the stigma towards people who use drugs that is, at times, explicit within, but also underlies the entirety of, Purdue’s submission. Purdue claimed that ‘the tablet formulation of the controlled release oxycodone will be more difficult to dissolve in a solution, hence not desired by the “street” addict who prefers an injectable solution’ (emphasis added). Given that stigma continues to plague the practice of addiction medicine, 27 this language is not surprising. Yet, the stated assumptions about what the ‘street addict’ or ‘abuser’ prefers and the inattention to the risks of misuse and addiction that is shot through Purdue’s entire submission, reveals a deeper disregard for people who use drugs.
Our study has two limitations. First, subsequent decisions involving the same sponsor and different formulations of OxyContin, in particular, the subsequent regulatory decision to replace controlled-release OxyContin with a tamper-resistant formulation in view of the overdose crisis which had by then developed, are not included in our analysis. Second, the study does not include information regarding Health Canada’s interpretation of the evidence provided by Purdue; Health Canada has long possessed the authority to make such interpretive information publicly available 28 but it chose not to include it within its portal.
The latter limitation, coupled with our findings, underscore both the potential of, and pitfalls associated with, the transparency that has been added to Canada’s regulatory system since the passage of Vanessa’s Law. In theory, the data now available from Health Canada have the potential to enhance evidence-based medicine by improving access to clinical trial data and other studies that were conducted but not incorporated into the published literature. 15 On the other hand, the evidence we examined raises more questions than it answers. How did Purdue persuade Health Canada to approve OxyContin given the lack of evidence about the potential risk of addiction and/or misuse? Studies that were subsequently used to promote OxyContin7,21,22 were not cited within the submission. What other evidence, if any, did Purdue provide in support of the claims that appeared in the product monograph? This is important because pharmaceutical companies are permitted by law to use regulator-approved information in their promotional practices. What impact, if any, did shifting clinical interpretations about opioid prescribing7,8 have upon the regulator’s evaluation? How much weight did Health Canada place upon the absence of treatment options for non-cancer chronic pain at the time? How did reviewers reconcile Purdue’s claims that the risk of misuse was non-existent for the proposed indication with the lack of evaluation of that very same risk in the course of clinical trials? None of these considerations is made transparent via the newly created portal. Transparency requires that both the underlying data and regulatory interpretations thereof; 29 without the latter, regulatory decision-making remains opaque as illustrated by the case of OxyContin.
Our study has policy and legal implications. First, it suggests that despite the creation of the portal significant gaps remain in Health Canada’s approach regulatory transparency. Building upon existing initiatives, there are steps that Health Canada can take to make its decision-making processes more transparent to enhance trust and public health.29,30 Second, the weak evidence underlying Health Canada’s decision to approve OxyContin for chronic pain raises questions about the regulator’s close relationship with industry, which may have played a role in precipitating the overdose crisis. 31 There has been a great deal of litigation against Purdue Pharma and other entities involved in the manufacture, sale, and distribution of OxyContin. 1 Including Health Canada as a defendant in those proceedings could provide important insight into why it approved OxyContin with minimal evidence about its potential risks of addiction and misuse.
Footnotes
Appendix 1
OC88-1105. Double-blind randomized, single-dose, parallel group study to assess the relative analgesic effectiveness and safety of graded doses of controlled-release oxycodone compared to immediate-release oxycodone, Percocet and placebo in patients with post-operative pain due to abdominal gynaecological surgery.
OC91-0402A. Controlled-release oxycodone tablets (30 mg Q12 h) versus immediate-release oxycodone tablets (15 mg, Q.I.D.): comparative efficacy, safety and acceptability in patients previously stabilized on fixed combination opioid analgesics for chronic cancer-related pain.
OC91-0402B. Controlled-release oxycodone tablets (Q12 h) versus immediate-release oxycodone tablets (Q.I.D.): comparative efficacy, safety and acceptability in patients previously stabilized on strong opioid analgesics for chronic cancer-related pain
OC92-1102. A randomized, double-blind, parallel group, analgesic efficacy, safety, acceptability and quality of life study of fixed doses of controlled-release oxycodone tablets versus placebo in chronic non-malignant pain due to osteoarthritis
OC92-1201. A randomized, double-blind, two-period crossover comparison of the pharmacokinetic and pharmacodynamic profiles of immediate-release oxycodone and controlled-release oxycodone in patients with chronic low back pain.
OC91-0907A/OC91-0907B . A 3-month open-label extension of protocol no. 0C91-0402AI0C91-0402B: controlled-release oxycodone tablets (q12 h) in patients with chronic cancer-related pain.
OC93-0202. Double-blind, randomized, two-period, crossover efficacy comparison of the pharmacokinetic and pharmacodynamic profiles of immediate-release oxycodone and controlled-release oxycodone in cancer patients with pain.
Appendix 2
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M. Herder is a member of the Patented Medicine Prices Review Board (PMPRB), Canada’s national drug pricing regulator, and reports receiving honoraria for his public service. The PMPRB had no role whatsoever in the design, conduct, or reporting of this research. No other conflicts of interest were reported.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Canadian Institutes of Health Research (grant no. CIHR PJT 156256)); and the Gladys Osman Estate Studentship Award provided by the Faculty of Medicine at Dalhousie University.
