Abstract
Background
Cost-effectiveness analyses embedded within randomized trials allow for evaluation of value alongside conventional efficacy outcomes; however, collection of resource utilization data can require considerable trial resources.
Methods
We re-analyzed the results from four phase III Canadian Cancer Trials Group trials that embedded cost-effectiveness analyses to determine the impact of minimizing potential cost categories on the incremental cost-effectiveness ratios. For each trial, we disaggregated total costs into component incremental cost categories and recalculated incremental cost-effectiveness ratios using (1) only the top 3 cost categories, (2) the top 5 cost categories, and (3) all cost components. Using individual trial-level data, confidence intervals for each incremental cost-effectiveness ratio simulation were generated by bootstrapping and descriptively presented with the original confidence intervals (and incremental cost-effectiveness ratios) from the publications.
Results
Drug acquisition costs represented the highest incremental cost category in three trials, while hospitalization costs represented the other consistent cost driver and the top incremental cost category in the fourth trial. Recalculated incremental cost-effectiveness ratios based on fewer cost components (top 3 and top 5) did not differ meaningfully from the original published results. Based on conventional willingness-to-pay thresholds (US$50,000–US$100,000 per quality-adjusted life-year), none of the re-analyses would have changed the original perception of whether the experimental therapies were considered cost-effective.
Conclusions
These results suggest that the collection of resource utilization data within cancer trials could be narrowed. Omission of certain cost categories that have minimal impact on incremental cost-effectiveness ratio, such as routine laboratory investigations, could reduce the costs and undue burden associated with the collection of data required for cancer trial cost-effectiveness analyses.
Keywords
Background
As the costs of cancer care continue to rise, clinical trials will increasingly need to demonstrate that new therapeutic options can offer both clinical benefit and value for money. Cost-effectiveness analyses embedded within randomized trials allow for formal evaluation of value alongside conventional efficacy determinations. Ideally, economic evaluations should capture all relevant costs in a large sample of patients. Ultimately, the perspective of the cost-effectiveness analysis (e.g. health system or societal) contributes to the determination of which resource utilization data should be collected alongside the usual clinical outcome elements.
However, at the level of the clinical trial execution, there is tension with respect to ideal study design to ensure comprehensive data collection without excessive use of trial resources (e.g. study coordinator time) or undue patient burden. Resource utilization data collected in the trials typically include parameters such as drug utilization, hospitalizations, emergency department and outpatient visits, diagnostics, laboratory, and personnel; subsequent cost calculations require that resource utilization parameters be multiplied by the unit cost of that parameter. Resources are collected within specifically designed case report forms and impart additional time and effort for participating patients and study personnel. Such burden may impact compliance with completion of required data elements and ultimately data validity. Following trial completion, central review processes (e.g. data validation, querying) are further required for the data incorporation into the trial database. The added costs and resources required for the completion of a prospective economic evaluation can themselves represent barriers to the conduct of such analyses.
Since trials are limited by the costs associated with patient accrual and with the practical collection of the resource utilization data, it would be important to determine the optimal data requirements to ensure judicious trial performance. The Canadian Cancer Trial Group is one of the only international cancer co-operative groups to maintain a Committee on Economic Analysis; its main aim is to build economic evaluations into phase III cancer trials. Re-analysis of previously completed cost-effectiveness analyses embedded into Canadian Cancer Trials Group trials could potentially clarify the optimal design elements and data requirements for future economic analyses. In particular, review of the costs and resources collected within these trials can determine which high-cost items have the largest impact on the economic sub-study results, including determination of an incremental cost-effectiveness ratio (ICER). Moreover, a resource’s influence on the overall costs might help determine which parameters should be collected within a trial while at the same time identifying parameters that have minimal impact and do not need to be collected. We hypothesize that the total cost of an intervention in a trial is often driven only by a few resource parameters, often drug acquisition or hospitalization costs, and that other resource parameters such as laboratory costs or diagnostic costs play a small role in the overall cost and therefore may be omitted from data collection. These data would inform the extent of information (and data collection) required to report a robust cost-effectiveness analysis as part of a prospective randomized cancer trial.
Methods
We sought to re-analyze the results from phase III trials that embedded economic evaluations to determine the impact of minimizing potential cost categories collected on the ICERs reported.
The Committee on Economic Analysis at the Canadian Cancer Trials Group has embedded a series of high-quality economic evaluations into a series of Canadian-led randomized controlled trials for new cancer therapies (n = 4). The LY.12 study was a non-inferiority trial that compared an outpatient salvage therapy (gemcitabine, dexamethasone, and cisplatin (GDP)) to an inpatient regimen (dexamethasone, cytarabine, and cisplatin (DHAP)) in patients with relapsed aggressive histology lymphoma (n = 619 overall trial population; n = 519 patients within the economic sub-study).1,2 The CO.17 trial evaluated the cost-effectiveness of cetuximab relative to best supportive care in patients with metastatic colorectal cancer (n = 572 overall trial population; n = 557 economic evaluation).3,4 BR.21 studied the cost-effectiveness of erlotinib versus best supportive care in patients with advanced non-small cell lung cancer who had failed cisplatin-based therapy (n = 731 patients in both the overall study population and economic evaluation).5,6 BR.10 studied the role of chemotherapy versus observation in patients with early-stage non-small cell lung cancer (n = 482 overall trial population; n = 172 economic evaluation).7,8
For each of the four trials, we disaggregated the total costs into component incremental cost categories (i.e. drug acquisition and administration costs, hospitalizations, outpatient visits, etc.), presented in tabular form (descriptive analysis). The total direct costs associated with the treatment strategies within the economic evaluations were recalculated in three ways: using only the top 3 cost categories, using only the top 5 cost categories, and then using all collected cost components. Using individual trial-level data, ICERs were subsequently recalculated in a stepwise manner. Confidence intervals for each of the ICER simulations were generated by bootstrapping and descriptively presented along with the original confidence intervals (and ICERs) from the publications. In the CO.17 trial, incremental cost-effectiveness (with life-years gained) and incremental cost-utility (with quality-adjusted life-years gained) analyses were completed in the overall trial population and in individuals with wild-type KRAS tumors. All ratios and confidence intervals were recalculated from this trial. In the LY.12 study, the experimental GDP arm was dominant, and no ICER was presented due to the non-inferiority trial design; as such, only the total direct cost recalculations were presented for this trial.
Results
Disaggregated drug cost components from the four clinical trials are presented in Table 1. Drug acquisition and administration costs represented the highest incremental cost category in three trials (BR.10, BR.14, and CO.17) and the second highest category in the LY.12 study. Hospitalization costs represented the other consistent cost driver, representing the top incremental cost category in one trial (LY.12) and the second highest category in two studies (BR.10 and CO.17).
Disaggregated incremental costs (top 5) associated with Canadian Cancer Trials Group phase III trials with embedded economic evaluations.
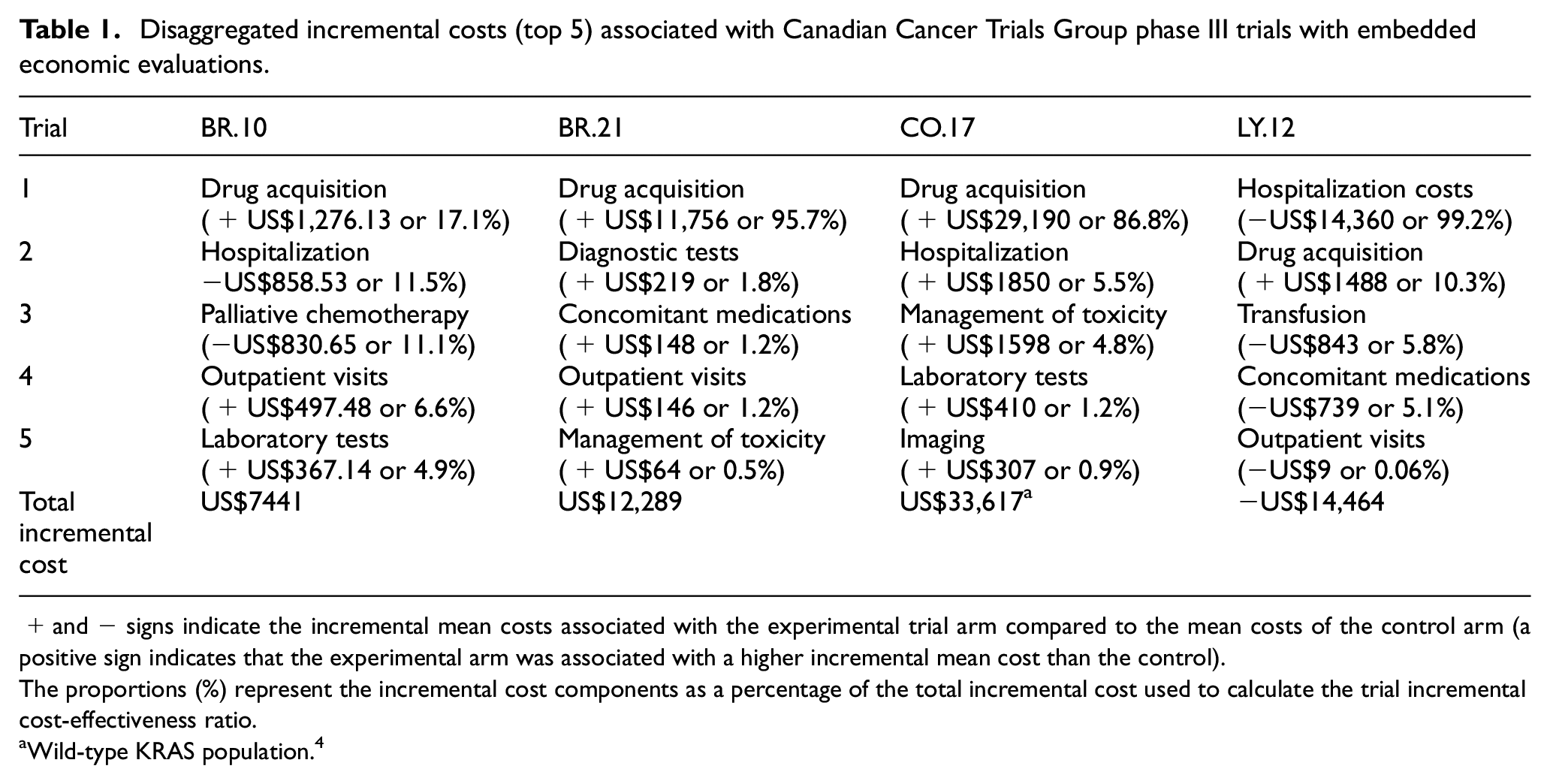
+ and − signs indicate the incremental mean costs associated with the experimental trial arm compared to the mean costs of the control arm (a positive sign indicates that the experimental arm was associated with a higher incremental mean cost than the control).
The proportions (%) represent the incremental cost components as a percentage of the total incremental cost used to calculate the trial incremental cost-effectiveness ratio.
Wild-type KRAS population. 4
Several cost components consistently contributed less than 5% of the total incremental cost used to calculate study ICERs (Table 1). For example, laboratory testing incremental costs for the CO.17 and BR.10 trials contributed only 1.2% and 4.9%, respectively, to the total incremental cost. Management of study drug toxicities (without requiring hospitalization) in BR.21 and CO.17 contributed only 0.5% and 4.8%, respectively, to the total incremental costs. Imaging studies in the CO.17 trial only contributed 0.9% to the total incremental cost.
ICERs were recalculated using top 3, top 5, and all cost components (original results) from the clinical trials, when available (Table 2). Only the total cost differences were presented for LY.12. The re-analyses based on fewer cost components (top 3 and top 5 components) did not appear to differ meaningfully from the original published results in that the re-analyzed ICERs would not have changed the original trial conclusions of cost-effectiveness. Specifically, none of the re-analyses would have changed the original perception of whether the experimental treatment arms were considered cost-effective or not, based on conventional willingness-to-pay thresholds in the range of US$50,000 to US$100,000 per quality-adjusted life-year gained.
Recalculated ICERs using top cost categories and all cost categories (published results).
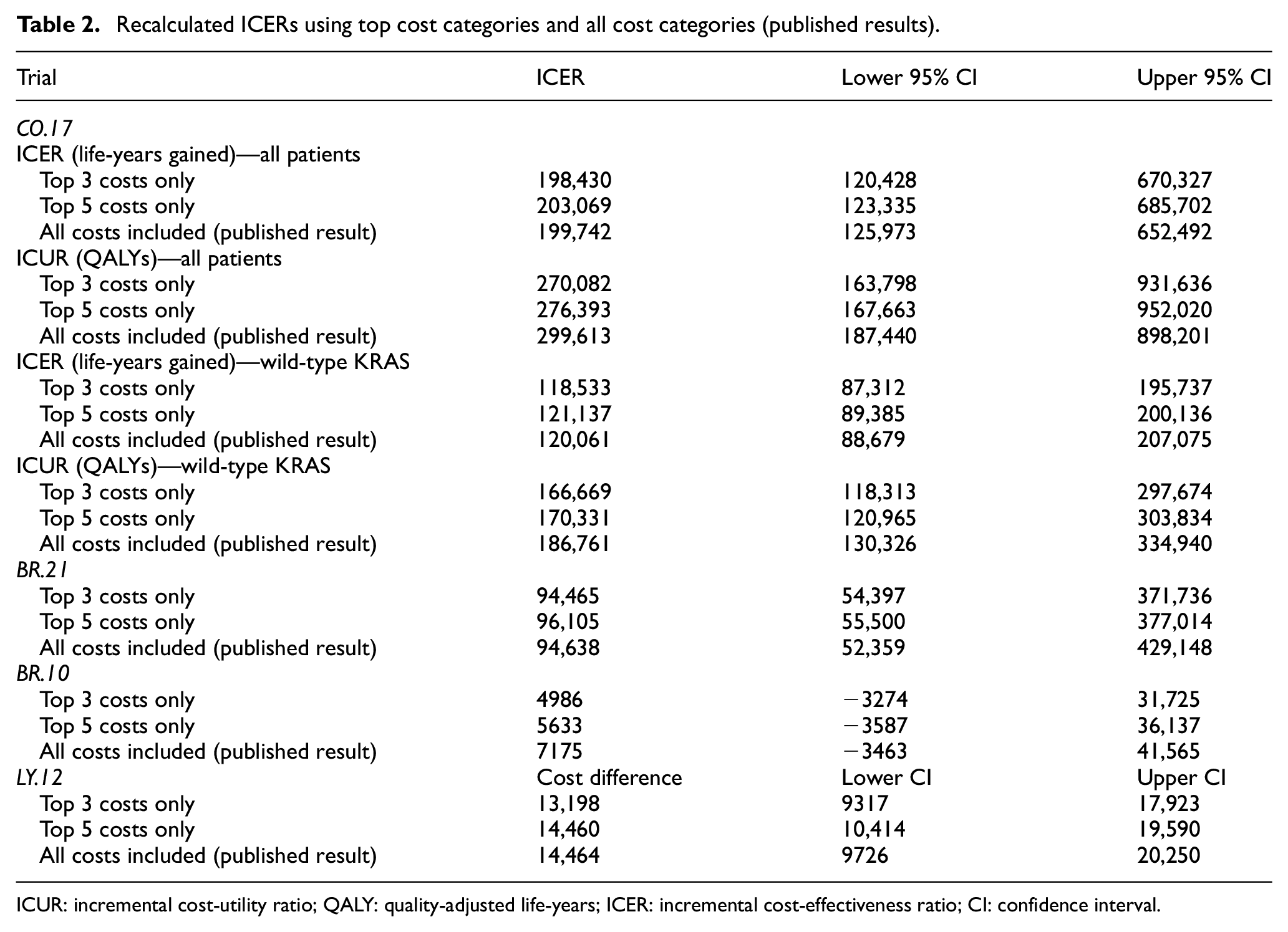
ICUR: incremental cost-utility ratio; QALY: quality-adjusted life-years; ICER: incremental cost-effectiveness ratio; CI: confidence interval.
Discussion
In individual-patient cost-effectiveness analyses conducted prospectively alongside cancer clinical trials, we ascertained that key cost drivers inform the calculation of ICERs, while other cost categories have minimal impact on the results. Within the sample of four Canadian Cancer Trials Group trials evaluated, drug costs and hospitalizations represented the top cost drivers of relevance. When re-calculating ICERs from the trials using only the top 3 and top 5 categories with the highest incremental costs, the resultant values did not meaningfully differ from the original published ICER result. In all of the re-calculations, there were no instances in our descriptive analyses in which the original conclusions regarding cost-effectiveness would have changed, based on currently accepted willingness-to-pay thresholds.
These results suggest that the collection of resource utilization data within cancer trials could be narrowed. Fewer cost categories or more targeted collection of data within cost categories might be considered in future clinical trials. We appreciate that this implies an a priori understanding of potential cost components or importance, which may not always be realized at the time of study design. Although our results suggest that drug acquisition costs and hospitalizations are consistently high-cost drivers, this may not invariably be the case. Moreover, certain costs might always warrant collection, if important to the trial outcomes or perspective of the analysis; for example, indirect costs including lost productivity and caregiver burden would likely be essential costs to collect in an economic evaluation that considers a societal perspective. Ultimately, cancer trialists and their health economic collaborators may be aware of cost categories that are likely to be of low impact based on their understanding of the clinical context to be studied. Collection of low-impact cost categories could be minimized on a trial-by-trial basis in future studies with embedded economic evaluations, ideally at the design phase of the cancer clinical trial.
The advantages associated with simplifying data collection are potentially significant. A recent analysis of two Canadian Cancer Trials Group studies (HN.6 with 320 patients and BR.26 with 385 patients) with embedded economic evaluations revealed that a considerable number of data queries were required for the resource utilization data (Penny Bradbury, Canadian Cancer Trials Group, personal communication); HN.6 required 1603 queries (690 of which were manual), while BR.26 required 3865 trials (2809 of which were manual). Efforts to minimize the burden of data collection with more pragmatic trial designs could have demonstrable impact on both human and financial resources, including site investigators and researches, data managers, central co-operative group analysts, and overall trial budgets. A formal analysis of the value of attaining perfect information, to quantify the additional cost to collect items that may not appear to impact outcomes, will be a future research endeavor. The use of routinely collected administrative data may also represent a novel, low-burden alternative to establishing resource utilization, with preliminary work demonstrating an ability to collect complete and highly concordant data. 9
Our analysis does have limitations. Our results were only applicable to studies that prospectively collected direct medical resources and costs from the health system perspective; our conclusions do not apply to the collection of indirect costs or caregiver burden, which may be substantial from a patient perspective. Our analysis only applied to a limited sample of previously completed trials that focused on drug interventions within Canada. This may represent a limitation in generalizing our results to all cancer clinical trials, and notably trials that might focus on radiation or surgical-based interventions; however, we feel that the concept that certain cost categories contribute little to the final results of a trial-based economic analysis can be broadly applied to cancer clinical trials in general.
This study serves to clarify the resource parameters required to calculate incremental costs borne when a new cancer therapy is considered. Key cost drivers including acquisition costs for novel cancer drugs and hospitalizations are identifiable and should be consistently collected. Other cost categories have a minimal impact on the final incremental cost calculations, and their omission does not change the value regarding cost-effectiveness. Limiting the collection of cost parameters that have minimal impact would ensure more efficient use of trial staff, improve allocation of trial resources, and ultimately reduce the burden on participating patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by a 2018 Seed Grant awarded by the Canadian Center for Applied Research in Cancer Control.
