Abstract
Background/aims
Pantothenate kinase-associated neurodegeneration is a rare neurodegenerative disease with a variable clinical phenotype. Fosmetpantotenate is in clinical development as a replacement therapy that targets the underlying cause of pantothenate kinase-associated neurodegeneration. The FOsmetpantotenate Replacement Therapy pivotal trial—an ongoing phase 3, randomized, double-blind, placebo-controlled, multicenter trial—examines the efficacy and safety of fosmetpantotenate in patients with pantothenate kinase-associated neurodegeneration aged 6–65 years. The FOsmetpantotenate Replacement Therapy trial required the development and validation of a novel patient-reported outcome measure specifically relevant to pantothenate kinase-associated neurodegeneration. The Pantothenate Kinase-Associated Neurodegeneration–Activities of Daily Living scale was developed to assess activities of daily living related to motor functioning in patients with pantothenate kinase-associated neurodegeneration to evaluate clinically meaningful change as the primary efficacy endpoint in clinical trials. This article describes the design of the FOsmetpantotenate Replacement Therapy pivotal trial and the development of the Pantothenate Kinase-Associated Neurodegeneration–Activities of Daily Living scale.
Methods
A systematic, iterative process consistent with the US Food and Drug Administration guidance and advice from the Committee for Medicinal Products for Human Use at the European Medicines Agency was used to evaluate and adapt or remove scale items of an existing widely used instrument for movement disorders to be pantothenate kinase-associated neurodegeneration–specific, and to create new items. Modification of scale items was based on input from international experts, patient advocacy leaders, and primary caregivers. A clinimetric study of the Pantothenate Kinase-Associated Neurodegeneration–Activities of Daily Living scale conducted in patients with pantothenate kinase-associated neurodegeneration or their caregivers (N = 40 at first assessment; N = 39 at second assessment) demonstrated high content and construct validity and excellent test–retest reliability over an approximately 2-week period. The Pantothenate Kinase-Associated Neurodegeneration–Activities of Daily Living scale was developed to be broadly useful within clinical and research settings in the examination of patient response to pantothenate kinase-associated neurodegeneration therapies.
Results
Approximately 82 patients will be enrolled in the ongoing FOsmetpantotenate Replacement Therapy pivotal trial. Change from baseline in Pantothenate Kinase-Associated Neurodegeneration–Activities of Daily Living score over the 24-week double-blind period is the primary efficacy endpoint for the FOsmetpantotenate Replacement Therapy trial. Treatment effect will be evaluated using a mixed model for repeated measures analysis to assess data from all visits simultaneously.
Conclusion
The development and implementation of the Pantothenate Kinase-Associated Neurodegeneration–Activities of Daily Living scale in the FOsmetpantotenate Replacement Therapy trial illustrates the feasibility and potential patient benefit of putting into practice the current regulatory guidance on the use of patient-reported outcomes in clinical trials. These processes can be broadly applied to clinical trial methodology that requires newly created or revised patient-reported outcome measures to evaluate outcome change as a primary efficacy endpoint. The goal of such measures in patients with pantothenate kinase-associated neurodegeneration is to facilitate development of disease-modifying therapeutics in multiple drug development programs.
Keywords
Introduction
Pantothenate kinase-associated neurodegeneration (PKAN) is a rare neurodegenerative disease with a variable clinical phenotype for which disease-modifying treatment is in development. 1 It is the most frequent cause of neurodegeneration with brain iron accumulation, comprising 35%–50% of cases, and has an estimated prevalence of one to three per million persons worldwide.2,3 Clinical presentation includes dystonia with prominent craniofacial involvement, spasticity, rigidity, bradykinesia, postural instability, loss of ambulation and ability to communicate, feeding difficulties, psychiatric issues, and rarely, cognitive impairment. 1
PKAN is an autosomal recessive disorder caused by a mutation in the pantothenate kinase-2 gene. 4 Mutations in the pantothenate kinase-2 gene are thought to result in complete or partial deficiency of the pantothenate kinase-2 enzyme, with consequent reduction in concentrations of phosphopantothenate and coenzyme A, leading to downstream consequences, including defective membrane biosynthesis.5,6 Fosmetpantotenate, a phosphopantothenic acid prodrug currently in clinical development as a phosphopantothenic acid replacement therapy, aims to replenish phosphopantothenic acid and coenzyme A in patients with PKAN. 7 Preliminary findings from compassionate drug use of fosmetpantotenate in four adult patients with PKAN, in accordance with all local laws and regulations in the individual countries of these patient cases, have been reported.8–10 During open-label, uncontrolled treatment, these adult patients with PKAN showed improvement in assessed clinical parameters, including the Unified Parkinson’s Disease Rating Scale and the revised Movement Disorder Society-Unified Parkinson’s Disease Rating Scale, Barry-Albright Dystonia Scale, the EuroQol 5-dimensional 3-level scale, timed 25-foot walk test, and electroglottographic speech analysis.8,9 One patient showed continuous improvement in ability to walk through 2 months of fosmetpantotenate treatment, followed by disease stabilization through 12 months of treatment, along with improvements in dystonia, parkinsonism, rigidity, bradykinesia, and overall quality of life. 8 Two patients had improved gait, ability to walk unassisted for short distances, and overall functioning, including decreased impulsivity and dystonia and increased frustration tolerance and attentiveness in the first 6 months of fosmetpantotenate treatment, followed by stabilization of disease progression through 47 weeks. 9 Although these case reports were uncontrolled, their promising findings were the basis for fosmetpantotenate being granted fast track designation by the US Food and Drug Administration and orphan drug designation by the Food and Drug Administration and European Medicines Agency. The outcomes from the case studies also guided the generation of the hypotheses in the fosmetpantotenate pivotal clinical trial described below.
The FOsmetpantotenate Replacement Therapy (FORT) pivotal trial is an ongoing phase 3, randomized, double-blind, placebo-controlled clinical trial to assess the efficacy and safety of fosmetpantotenate in adult and pediatric patients with PKAN (NCT03041116). Patient-reported outcome assessments are being promoted by regulatory agencies to directly measure the effects of disease on patient health and functioning11,12 and to supplement other measures that rely on inferences about patient benefit. Due to the lack of PKAN-specific clinical outcome measures, the FORT trial required creation of a patient-reported outcome measure to assess functional abilities and activities of daily living from the perspective of patients with PKAN. To address this need for a patient-reported outcome assessment specifically relevant to PKAN for use in clinical trials, the PKAN-Activities of Daily Living scale was developed as a novel patient-reported outcome measure based on patient or primary caregiver report of PKAN-related patient functioning in everyday life. 13
We describe our methods for the design and validation of a patient-reported outcome measure, the PKAN-Activities of Daily Living scale, 13 and our implementation of change from baseline in PKAN-Activities of Daily Living score as the primary efficacy endpoint in the FORT pivotal trial. Our methods are consistent with the Food and Drug Administration’s guidance on development of patient-reported outcomes 11 and the process for patient-reported outcome assessment qualification as a drug development tool, 12 as well as advice received from the Committee for Medicinal Products for Human Use at the European Medicines Agency. The PKAN-Activities of Daily Living scale, applicable to patients with PKAN worldwide, allows for the measurement of real-world patient functioning in everyday life from the perspective of patients and primary caregivers. Our objectives in the article are to share our approaches to the design of the FORT pivotal trial and the development of the PKAN-Activities of Daily Living scale. These processes can be broadly applied to clinical trial methodology that requires newly created or revised patient-reported outcome measures used for the examination of change over the course of treatment as a primary efficacy endpoint.
Methods
FORT pivotal trial design
The FORT pivotal trial is a randomized, double-blind, placebo-controlled, multicenter, two-arm study that evaluates 24 weeks of treatment with fosmetpantotenate or placebo in patients with PKAN, followed by a 276-week open-label extension (Figure 1). Currently, there are no effective disease-modifying treatments available to consider as an active control arm in the study. Instead, physician- and patient-specific management is allowed for all enrollees during the study, with treatments and interventions presented at baseline, such as anticholinergics and deep brain stimulation, continued without any changes for the duration of the double-blind phase. The FORT study protocol was designed and is being conducted in compliance with the International Council for Harmonisation’s Harmonised Tripartite Guidelines for Good Clinical Practice, the International Council for Harmonisation’s Guidelines for Safety Data Management, the Code of Federal Regulations, and the European Union Clinical Trials Directive. Written approval has been obtained from the appropriate institutional review boards/independent ethics committee, applicable regulatory authorities, and host institutions, and written informed consent/assent was obtained prior to any screening procedures.
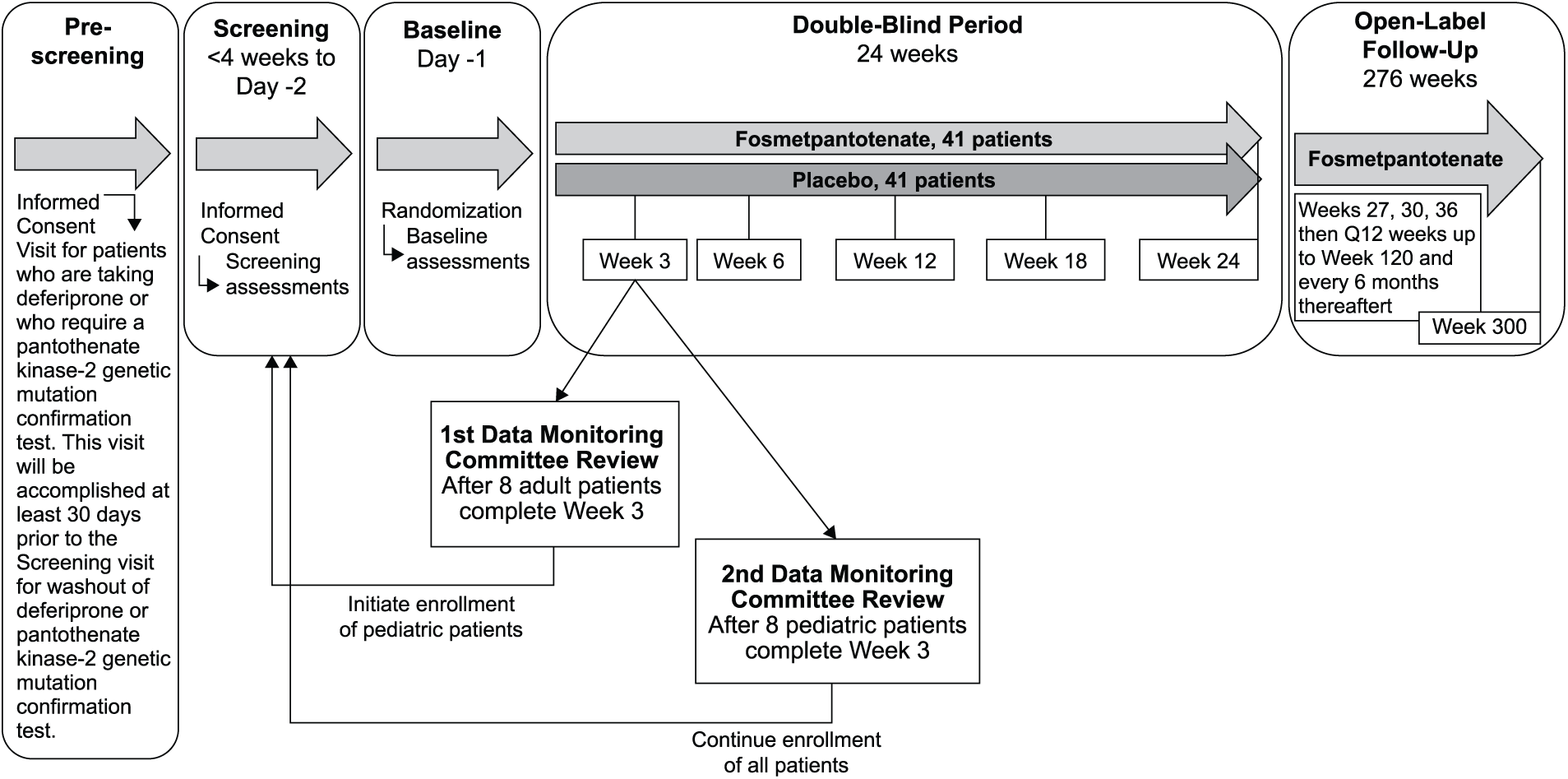
FORT study design.
Eligible patients include those who are aged 6–65 years (inclusive), with a genetically confirmed diagnosis of PKAN (i.e. confirmed homozygous or compound heterozygous pathogenic mutations in the pantothenate kinase-2 gene) and a score ≥6 on the PKAN-Activities of Daily Living scale. A score ≥6 on the PKAN-Activities of Daily Living scale indicates that the patient is experiencing at least mild impairment across multiple functional domains or moderate-to-severe impairment in at least one domain. Patients are not eligible to participate if they (1) have received deferiprone therapy within 30 days prior to screening; (2) have had a deep brain stimulation device implanted within 6 months prior to screening; (3) have required invasive ventilator support to maintain vital signs within 24 weeks of randomization; (4) have a serious, unstable medical or psychiatric condition unrelated to PKAN; or (5) are unwilling or unable to remain on their pre-study dose(s) of allowed concomitant PKAN maintenance medications and therapies during the double-blind period. Patients may be re-screened twice for eligibility. Enrolled patients are randomly assigned to a treatment group by an integrated web response system in a 1:1 ratio to receive either fosmetpantotenate or placebo during the 24-week double-blind period. Randomization is stratified by weight (≥40 kg, ≥20 kg but <40 kg, or <20 kg) and by age (pediatric vs adult) at screening. Study visits during the double-blind period occur at baseline (within 1 to 2 days prior to start of the double-blind treatment), Days 1–4, and Weeks 3, 6, 12, 18, and 24 (Table 1). Following the double-blind period, patients may enter the open-label extension period in which all patients receive fosmetpantotenate treatment.
FORT study visits in the double-blind period.
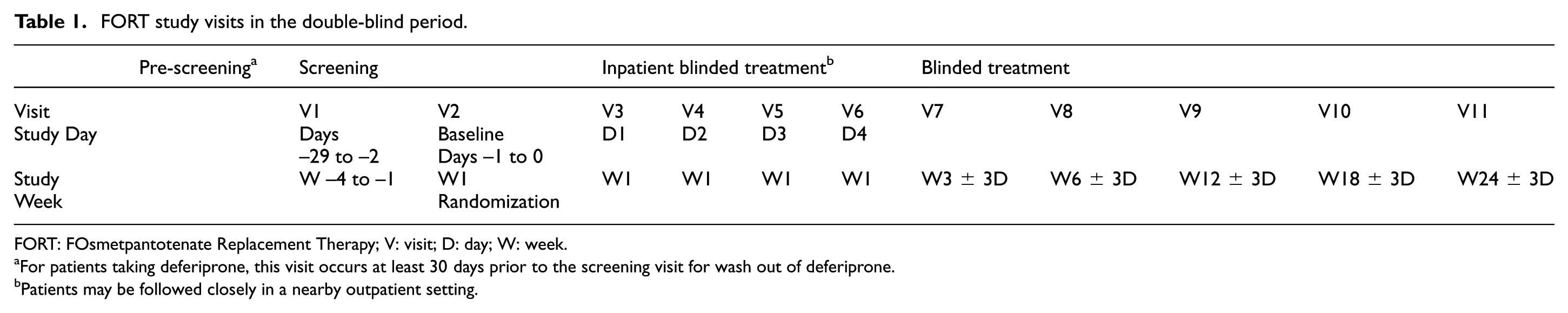
FORT: FOsmetpantotenate Replacement Therapy; V: visit; D: day; W: week.
For patients taking deferiprone, this visit occurs at least 30 days prior to the screening visit for wash out of deferiprone.
Patients may be followed closely in a nearby outpatient setting.
Development and validation of the PKAN-Activities of Daily Living scale
Due to the lack of a disease-specific, patient-centered, patient-reported outcome assessment for evaluating patient response to potential disease-modifying PKAN treatment, development and validation of a novel patient-reported outcome measure were necessary. A focus on functional consequences of motor symptoms, chosen following literature review and expert clinical input, led to the selection of the Unified Parkinson’s Disease Rating Scale Part II as the starting point due to its widespread use in clinical trials of movement disorders and by physicians treating PKAN patients with fosmetpantotenate in compassionate drug use case studies.8,9
A systematic, iterative process was used to evaluate the relevance and to adapt the Unified Parkinson’s Disease Rating Scale Part II items to be PKAN-specific. 13 In line with the Food and Drug Administration guidance, 11 revision of scale items was based on literature review, author input, and two rounds of interviews with PKAN experts, patient advocacy leaders, and primary caregivers to evaluate the content. Difficulty with speech is common in patients with PKAN, often making verbal interviews difficult or impossible. When required, interviews were conducted with the primary caregiver of a patient with PKAN rather than the patient due to the status of their PKAN symptoms or young age. The revised scale added clear examples for each item domain, one new item domain was added (getting into and out of a chair) and two item domains were removed (tremor and freezing when walking). This novel PKAN-Activities of Daily Living scale assesses 12 areas of motor functioning, each area with one item, and is validated for use in patients with PKAN aged 6–65 years (inclusive). 13
The PKAN-Activities of Daily Living scale may be completed by an individual with PKAN, alone or in collaboration with a primary caregiver, or by a primary caregiver. Primary caregivers are those who routinely assist the patient with PKAN with activities of daily living. When completed by the patient’s primary caregiver on behalf of the patient, the PKAN-Activities of Daily Living scale functions as an observer-reported outcome measure. The respondent is instructed to select the answer according to “your usual ability most of the time over the past week, including today.” This short recall period that includes the patient’s current state is consistent with the preference for short recall that is stated in the Food and Drug Administration guidance. 11
Next, a clinimetric study of the PKAN-Activities of Daily Living scale was completed in patients with a genetically confirmed diagnosis of PKAN (N = 40 at first assessment; N = 39 at second assessment). 13 The study showed high content, construct, convergent, divergent, and discriminant validity and excellent test–retest reliability within the context of the majority of patients reporting disease stability over the ~2 weeks between assessments. A semi-structured interview was conducted with the patients and primary caregivers who participated in the clinimetric study. They were asked about their overall impression of the PKAN-Activities of Daily Living scale, how well they understood the PKAN-Activities of Daily Living questions, and if they thought any content related to activities of daily living was missing. During cognitive debriefing, participants reported understanding the items, and most items were rated as highly relevant.
Results
Enrollment of approximately 82 patients aged 6–65 years (inclusive) is planned in the FORT pivotal trial to achieve the target of at least 74 patients completing the double-blind period with evaluable data. This will provide approximately 80% power to detect a 3-point difference between groups in the average change from baseline score in the PKAN-Activities of Daily Living scale total score, assuming a common standard deviation for the change from baseline scores of 4.5 points and using a two-sided Student’s t-test with alpha = 0.05. The schedule of key study outcome assessments is shown in Table 2. The novel primary efficacy endpoint for FORT is the change from baseline in PKAN-Activities of Daily Living score over the 24-week double-blind period. All efficacy analyses will include the full analysis set of patients, defined as all randomized patients with at least one post-baseline efficacy assessment, and study hypothesis testing uses a two-sided 5% significance level.
FORT study efficacy measures during screening and treatment in the double-blind period.

V: visit; D: day; W: week; FORT: FOsmetpantotenate Replacement Therapy; PKAN: pantothenate kinase-associated neurodegeneration; QoL: quality of life.
Administered to patients who can safely walk independently or with assistance. Screening evaluation determines whether the patient can be evaluated during the study.
Treatment effect will be evaluated using a mixed model for repeated measures analysis to assess data from all visits simultaneously. The model will include fixed effects for treatment, age stratum (pediatric or adult), visit and treatment-by-visit interaction, a random effect for patient within treatment group, and baseline PKAN-Activities of Daily Living score as a covariate. The primary treatment comparison is based on the test of contrast between treatment effects using the least squares mean estimate at Week 24 derived under the framework of the mixed model for repeated measures. Sensitivity analyses including results from analysis of covariance for the change from baseline to Week 24 in PKAN-Activities of Daily Living score using a model with fixed effects for treatment, age stratum, and baseline PKAN-Activities of Daily Living score as a covariate will be performed. Analysis of covariance will use both the observed case and last-observation-carried-forward procedures to account for drop-outs and ensure the treatment effect is robust.
The secondary efficacy endpoint within the FORT trial is the change from baseline to the end of the 24-week double-blind period in Unified Parkinson’s Disease Rating Scale Part III (motor examination). The same mixed model for repeated measures analysis approach will be used with the secondary efficacy endpoint as described for the primary efficacy endpoint. The serial gate-keeping approach is used to control the type 1 familywise rate at 5% in the test of the primary and secondary efficacy endpoints. The secondary efficacy endpoint will only be tested if the primary efficacy endpoint analysis is significant.
The FORT trial includes several exploratory efficacy endpoints evaluating change from baseline to the end of the 24-week double-blind period, including Clinician Global Impression of Improvement (ranging from 1 = very much improved to 7 = very much worse), Barry-Albright Dystonia Scale, Quality of Life in Neurological Disorders upper and lower extremity modules, EuroQol 5-dimensional 3-level scale (EuroQol 5-dimensional youth scale or proxy version with caregiver report for pediatric patients), Functional Independence Measure (Wee Functional Independence Measure for pediatric patients), ambulation 25-foot walk test (in appropriate patients), and speech diadochokinetic assessments. Notably important about the inclusion of these endpoints are the components with direct measurement of function, such as ambulation in the 25-foot walk test and speech in the diadochokinetic test. These exploratory endpoints are expected to provide complementary information to the primary and secondary endpoints.
Safety will be evaluated in the safety population, which consists of all patients who receive at least one dose of study treatment. Treatment-emergent adverse events, serious adverse events, vital signs, weight, physical examinations, clinical laboratory assessments, the Columbia Suicide Severity Rating Scale (in assessable patients), and 12-lead electrocardiograms will be evaluated. Patient enrollment in the FORT trial began with adult patients, and an independent data monitoring committee reviewed safety after 3 weeks of study treatment in the first eight patients to determine whether the study could continue (Figure 1). Enrollment of pediatric patients followed. After the first eight pediatric patients complete 3 weeks of study treatment, the data monitoring committee will review safety data from all patients to date. Enrollment of adult and up to eight additional pediatric patients may continue while this review is occurring. Following data monitoring committee review and recommendation that the study can continue as planned, enrollment of pediatric patients may be continued. Many of the patients with PKAN who will be eligible to participate in FORT will be children, and using this approach to safety review, balance will be achieved in the monitoring of safety and providing access to treatment.
Discussion
We report the design of the FORT pivotal trial and the methodology associated with development, validation, and implementation of a novel patient-reported outcome measure, the PKAN-Activities of Daily Living scale, specifically relevant to PKAN. The change from baseline in PKAN-Activities of Daily Living total score over the double-blind treatment period of the FORT trial is a novel primary efficacy endpoint for PKAN studies. The PKAN-Activities of Daily Living scale is a validated patient- or primary-caregiver-reported outcomes assessment of functional abilities in daily life related to motor function for patients with PKAN. The prior clinimetrics study established the reliability and validity of the PKAN-Activities of Daily Living scale in patients with PKAN. 13 Consistent with Food and Drug Administration guidance on the development of patient-reported outcome assessments that help to advance therapy development by being able to evaluate treatment outcome across therapeutics, 12 the PKAN-Activities of Daily Living scale is intended to be broadly useful within clinical and research settings in the examination of patient response to PKAN therapies. Inclusion in the FORT pivotal trial provides the first use of the PKAN-Activities of Daily Living scale in a clinical trial. The main goal of the FORT trial is to assess the efficacy and safety of fosmetpantotenate, a potential disease-modifying therapy, in a larger cohort of patients with PKAN. Use of the PKAN-Activities of Daily Living scale in the FORT trial provides the first validated, systematic, PKAN-specific patient-reported outcome assessing patient- or primary caregiver-reported functional abilities in daily activities related to patient motor function and degree of change in response to 24 weeks of treatment. The FORT trial will also allow evaluation of sensitivity to change properties of the PKAN-Activities of Daily Living.
There are several differences between the PKAN-Activities of Daily Living scale 13 and the recently available PKAN-Disease Rating Scale. 14 Whereas the PKAN-Activities of Daily Living scale is a patient-reported outcome assessment that focuses on the perspective of the patient across all 12 items, the 34-item, 6-subscale (Cognition (neuropsychiatric symptoms), Behavior, Disability, Parkinsonism, Dystonia, and Other Neurological Signs) PKAN-Disease Rating Scale involves multiple respondents, including patients and caregivers for questionnaire subscales and expert ratings of parkinsonism, dystonia, and other neurological signs using a videotaped motor examination protocol. 14 The PKAN-Activities of Daily Living scale and PKAN-Disease Rating Scale differ in their reporting timeframe, with the PKAN-Activities of Daily Living scale assessing the past week and the PKAN-Disease Rating Scale assessing the past month. The PKAN-Activities of Daily Living scale focuses on the functional consequences in daily activities of PKAN-specific motor function, without having respondents distinguish whether motor function is impaired due to symptoms of parkinsonism or dystonia, thus allowing assessment of clinically meaningful change in patient functions while minimizing respondent burden. The PKAN-Disease Rating Scale uses videotaped motor examination to distinguish parkinsonism and dystonia features, with the acknowledgment that this distinction is difficult to assess due to overlapping features in PKAN patients. 14 The PKAN-Activities of Daily Living scale and PKAN-Disease Rating Scale share the similarity of allowing primary caregivers to report patient responses when necessary due to PKAN-related oromandibular dystonia and dysarthria, which make patient speech very difficult to understand.
The need for patient-reported outcome-focused trials with high-quality reporting of outcomes to improve healthcare for patients has been acknowledged,15–17 and clinimetrically sound measures of patient-reported outcomes are urgently needed to facilitate therapeutics development in patients with PKAN. The focus on patient-reported outcome measures in clinical trials has led to the development of reporting guidelines for clinical trials with patient-reported outcome efficacy endpoints in an effort to improve the reporting quality of clinical trial patient-reported outcomes.18–20 Methods used in the development of the patient-reported outcome PKAN-Activities of Daily Living scale and its implementation within the ongoing FORT pivotal trial are consistent with these recommendations and with the recent Food and Drug Administration guidance.11,12 Key components related to patient-reported outcome development include patient and primary caregiver input during instrument development, documentation of the performance of the new instrument in the intended population, definition of the role of the patient-reported outcome as a primary endpoint intended to support an indication for treatment of symptoms of the condition being studied, and demonstration of the supporting conceptual and clinimetric evaluation prior to use in clinical trials.11,12 The inclusion of the PKAN-Activities of Daily Living scale in the FORT trial and use of change in the PKAN-Activities of Daily Living total score from baseline to the end of the double-blind treatment period as the primary efficacy endpoint in the FORT trial address the Food and Drug Administration guidance recommendations. 12 Specifically, use of the PKAN-Activities of Daily Living scale in the FORT trial provides adequate and well-controlled investigation of the newly developed, well-defined and reliable, novel patient-reported outcome measure with the goal to help advance PKAN therapy development and contribute to drug innovation, while supporting public health through the availability of the PKAN-Activities of Daily Living scale for use in multiple drug development programs. 12
Conclusion
The primary objective of the FORT pivotal trial is to examine efficacy and safety of fosmetpantotenate in patients with PKAN. To demonstrate efficacy, a novel patient-reported outcome tool specific to PKAN had to be developed and validated for use with patients with PKAN. It is a patient- or primary-caregiver-reported change in this novel outcome measure over the course of treatment that will establish treatment efficacy in the FORT trial. The methodology used in developing the PKAN-Activities of Daily Living scale and implementing it in the FORT trial illustrates the feasibility and potential patient benefit of putting into practice the current regulatory guidance on the use of patient-reported outcome assessments in clinical trials. As a quantitative measure of activities of daily living related to motor functioning in patients with PKAN, improvement in the PKAN-Activities of Daily Living scale score indicates clinically meaningful change for patients and can be used in clinical trials to evaluate treatment efficacy. Patient-reported outcome measures used to examine primary efficacy outcomes, such as the PKAN-Activities of Daily Living scale, may provide urgently needed patient-reported outcome data to both stimulate and support the evaluation of existing treatment strategies and facilitate development of new therapeutics.
Footnotes
Acknowledgements
The authors express their gratitude to patient advocacy leaders Patricia Wood, Angelika Klucken, and Antonio Lopez for their contributions to the design of the FOsmetpantotenate Replacement Therapy (FORT) trial. Their courage as caregivers and leadership as patient advocates are examples to us all. Writing support was provided by Lynanne McGuire, PhD, CMPP, of MedVal Scientific Information Services, LLC (Princeton, NJ, USA). This manuscript was prepared according to the International Society for Medical Publication Professionals’“Good Publication Practice for Communicating Company-Sponsored Medical Research: The GPP3 Guidelines.”
Declaration of conflicting interests
T.K. is a coordinating investigator of the FORT trial; receives research funding from Retrophin, Inc.; is a coordinating investigator of the deferiprone in PKAN randomized and extension trial; received research funding from ApoPharma Inc.; is supported by the European Commission 7th Framework Programme (FP7/2007-2013, HEALTH-F2-2011, grant agreement no. 277984, TIRCON); is supported by the European Reference Network for Rare Neurological Diseases (ERN-RND), co-funded by the European Commission (ERN-RND: 3HP 767231); provides consulting services to CoA Therapeutics, TM3 Therapeutics, and Retrophin, Inc; and received travel support from ApoPharma Inc. M.L.E. provides consulting services to Retrophin, Inc. R.D.M. and S.T. are former full-time employees of Retrophin, Inc. and may have an equity or other financial interest in Retrophin, Inc. B.P.-D. received grant support from Retrophin, Inc. to Hospital Universitari Vall d’Hebron, Barcelona, Spain. A.V. provides consulting services to Retrophin, Inc.; received active grant support from the National Institutes of Health; provides consulting services to Acadia, Jazz, and Roche; and served on DSMB for Wilsons’ Therapeutics and Acorda. F.G. is a full-time employee of Retrophin, Inc. and may have an equity or other financial interest in Retrophin, Inc.
Funding
The FORT pivotal trial and the PKAN-Activities of Daily Living study were supported by Retrophin, Inc. Writing support was funded by Retrophin, Inc.
