Abstract
Background
Hyperbaric oxygen therapy, which consists of breathing 100% oxygen under a higher atmospheric pressure than normal, is utilized worldwide in the treatment of several diseases. With the growing demand for evidence-based research, hyperbaric oxygen therapy has been criticized for delivering too little high-quality research, mainly in the form of randomized controlled trials. While not always indispensable, the addition of a sham-controlled group to such a trial can contribute to the quality of the research. However, the design of a sham (hyperbaric) treatment is associated with several considerations regarding adequate blinding and the use of pressure and oxygen. This narrative review discusses information on the sham profile and the blinding and safety of double-blind trials in hyperbaric medicine, irrespective of the indication for treatment.
Methods
MEDLINE, Embase and CENTRAL were searched for sham-controlled trials on hyperbaric oxygen therapy. The control treatment was considered sham if patients were blinded to their allocation and treatment took place in a hyperbaric chamber, with no restrictions regarding pressurization, oxygen levels or indication. Studies involving children or only one session of hyperbaric oxygen were excluded. Information on (the choice of) treatment profile, blinding measures, patient’s perception regarding allocation and safety issues was extracted from eligible studies.
Results
A total of 42 eligible trials were included. The main strategies for sham treatment were (1) use of a lower pressure than that of the hyperbaric oxygen group, while breathing 21% oxygen; (2) use of the same pressure as the hyperbaric oxygen group, while breathing an adjusted percentage of oxygen; and (3) use of the same pressure as the hyperbaric oxygen group, while breathing 21% oxygen. The advantages and disadvantages of each strategy are discussed using the information provided by the trials.
Conclusion
Based on this review, using a lower pressure than the hyperbaric oxygen group while breathing 21% oxygen best matches the inertness of the placebo. Although studies show that use of a lower pressure does allow adequate blinding, this is associated with more practical issues than with the other strategies. The choice of which sham profile to use requires careful consideration; moreover, to ensure proper performance, a clear and detailed protocol is also required.
Keywords
Introduction
Hyperbaric oxygen (HBO) therapy, which consists of breathing 100% oxygen under a higher atmospheric pressure than normal (i.e. above 1.0 atmosphere absolute (ATA)), is utilized worldwide in the treatment of several diseases. The Undersea and Hyperbaric Medical Society, a nonprofit organization that plays an important role in providing scientific and medical information on hyperbaric medicine, lists 14 indications for HBO therapy. These include late radiation tissue injuries, diabetic foot ulcers and carbon monoxide poisoning. 1 Treatment for chronic problems (e.g. wound healing) usually involves giving daily sessions for several weeks, at pressures between 2.0 and 2.5 ATA. These sessions can be given in either a monoplace chamber (in which only one patient is treated per session) or in a multiplace chamber (where several patients are treated simultaneously, with the possibility of an attendant joining them to supervise treatment). The therapy is generally considered safe with few complications, with barotrauma of the ears or sinuses and transient myopia being the most common. 2
HBO therapy has been controversial from the start. For example, in 1987, Gabb et al. 3 stated in Chest that HBO was “… a therapy in search of diseases.” The currently available scientific research is still criticized. Although the theoretical basis for the use of HBO therapy seems rational, and well-performed studies in animals and humans show positive effects, a large proportion of the evidence is anecdotal, retrospective, uncontrolled and underpowered. The increased application of evidence-based medicine has raised concerns about the overtreatment of patients, possibly causing unnecessary risks and higher costs in health care. Despite efforts by the Undersea and Hyperbaric Medical Society and (recently) the European Committee for Hyperbaric Medicine to establish recognized clinical indications, a need still exists for more and better research, particularly in the form of randomized controlled trials. 4 Because the outcome of such a trial can be biased by a placebo effect (especially in the case of subjective parameters), a sham treatment for the control group can be a valuable addition.
However, the design of such a trial is associated with several considerations regarding the sham treatment. The goal of a sham treatment is to ensure that patients and investigators are unable to distinguish sham from actual HBO therapy (thus filtering out a potential placebo effect), while the sham procedure must not have any effect on the disease being treated. Because patients have to auto-inflate their ears when pressure is increased in HBO therapy, sham therapy also has to use pressure to create/mimic this experience. However, every increase in pressure has an effect on the partial pressures of gases, potentially causing the sham treatment to become an active agent.
This dilemma, together with considerations regarding practicality, safety and blinding, has resulted in different strategies being used in various trials over the years, each with their own advantages and disadvantages. The aim of this narrative review is to provide a structured overview of all past randomized controlled trials that included sham (hyperbaric) treatment. The methodology is examined and, if available, the authors’ considerations for the choice and the results of this methodology (regarding treatment profiles, blinding and safety) are presented. The findings of this review may help researchers to make a more balanced decision regarding the design of their specific trial.
Being a narrative review on the methodology of trials investigating hyperbaric medicine, there is no discussion on the indications for which HBO was applied, or on the efficacy and/or outcome of each trial.
Methods
Our aim was to provide a summary of past studies involving HBO therapy using a sham treatment. A systematic search of the literature was conducted using the following protocol.
Eligibility criteria
Included were all studies comparing actual HBO therapy with sham treatment, irrespective of the indication for treatment. It was expected that most of these studies would be randomized, but if any non-randomized controlled trial were encountered, these were also considered for inclusion. The control treatment was considered sham if no additional therapeutic effect to standard care was intended, and patients were blinded to their allocation. The minimum criterion for the blinding of patients was that the patients were located in a hyperbaric chamber (either monoplace or multiplace) during sham treatment; no restrictions were made regarding pressure or oxygen levels. Studies that did not meet this criteria (e.g. if patients were allowed to choose their own allocation, or if they received a gas mixture in a normal room) were excluded. Studies on children were excluded, because their treatment regimens (generally using a lower pressure) and blinding measures can differ from those applied in adults. Studies with only one session of HBO were also excluded, because that treatment regimen is dissimilar to a typical hyperbaric treatment which (generally) consists of multiple sessions, often administered over several weeks.
Database search
MEDLINE via PubMed, Embase via Ovid and the Cochrane Central Register of Controlled Trials were searched (up to April 2017) for sham-controlled studies on hyperbaric medicine. Medical subject heading (MeSH and Emtree) terms were used, in combination with keywords. Searches were limited to randomized controlled trials and trials including human adults and published in English. Details of the search strategies are presented in Appendix 1.
Other sources
The reference lists of the included studies and identified (systematic) reviews were screened to identify additional eligible studies. Also, the Database of Randomized Controlled Trials in Diving and Hyperbaric Medicine was searched by hand for articles on HBO.
Data collection
Data collection was performed by the first author (N.C.A.L.). Abstracts were screened for eligibility following the criteria mentioned above. After screening, information on the study sample, and on the sham and HBO profile (pressure, time, oxygen levels), was extracted from the full-text article.
The full text was also searched for considerations regarding the choice for a certain sham treatment profile, for information on blinding procedures and for the use of questionnaires on patients’ perceived allocation. Information on the safety of the sham procedure was also collected, including details on any type of complications.
Results
The search of the databases and other sources yielded 477 articles. After removal of duplicates (including separate articles reporting on the same trial) and initial screening, 45 articles remained. After assessing the eligibility based on the full-text articles, 42 studies were finally included in the present review (Figure 1). All of these studies were randomized controlled trials.
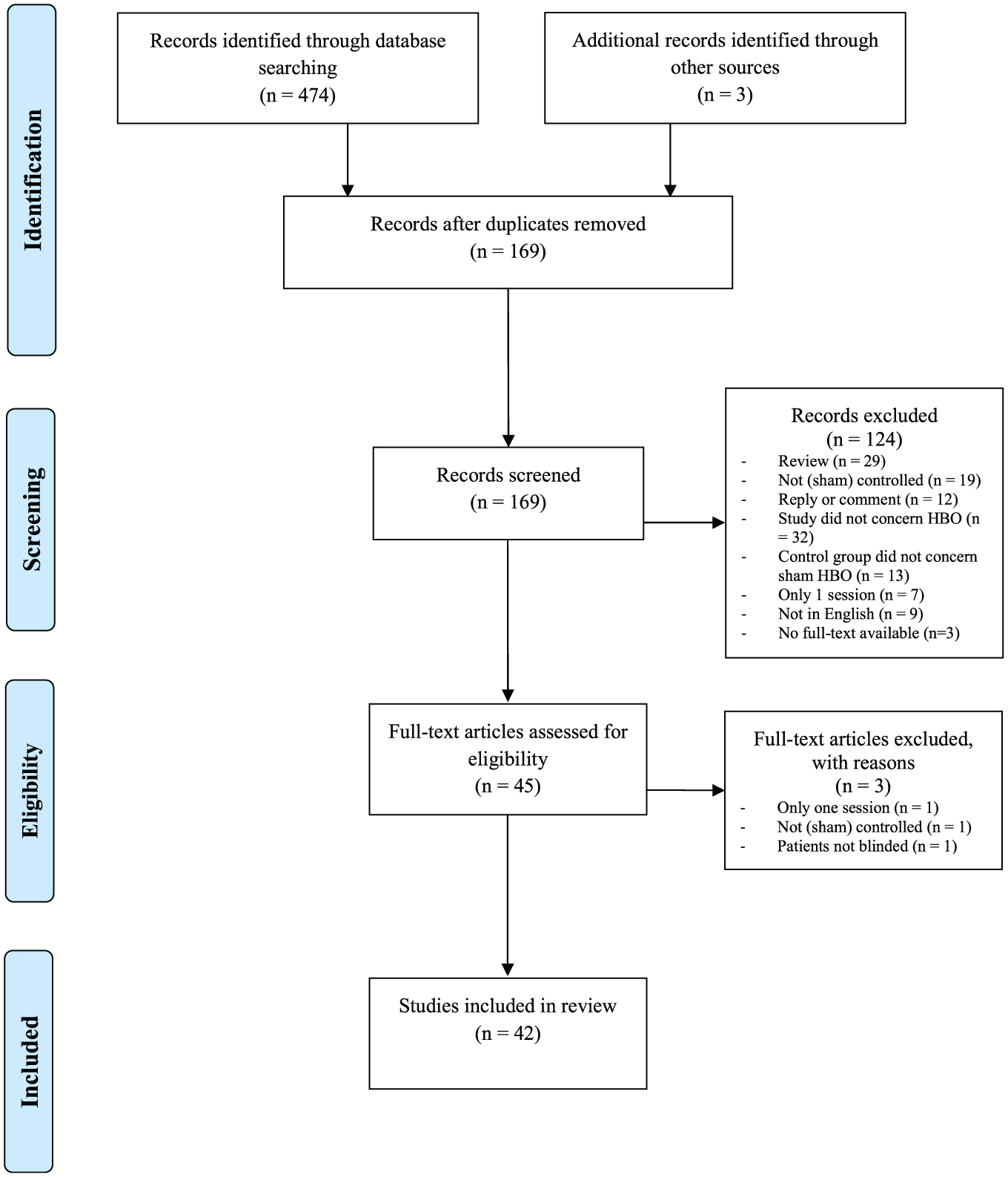
Flowchart showing identification of the eligible studies.
Of the 42 included studies, analysis of the sham profiles revealed the use of three main strategies. Presented in chronological order as described in the literature, these are (1) use of a lower pressure than the HBO group, while breathing 21% oxygen; (2) use of the same pressure as the HBO group, while breathing a mixture with an adjusted percentage of oxygen; and (3) use of the same pressure as the HBO group, while breathing 21% oxygen.
The first strategy was used in 23 studies, with pressures in the sham group ranging from 1.1 to 1.5 ATA. An overview of these studies including considerations for the choice of profile, blinding measures and complications in the sham group as provided in the articles is presented in Table 1. The second strategy, using an adjusted percentage of oxygen for sham therapy while maintaining the same pressure as the HBO group, was used in 11 studies. Adjusted oxygen levels ranged from 7% to 41%. An overview is provided in Table 2. The third and last strategy (use of the same pressure with 21% oxygen in the sham group) was used in eight studies, of which the information is presented in Table 3. The advantages and disadvantages of all three strategies, using the information provided in the articles, are discussed separately below.
Studies using lower pressure and 21% oxygen for sham treatment, and information on choice of profile, blinding and safety provided by these studies.
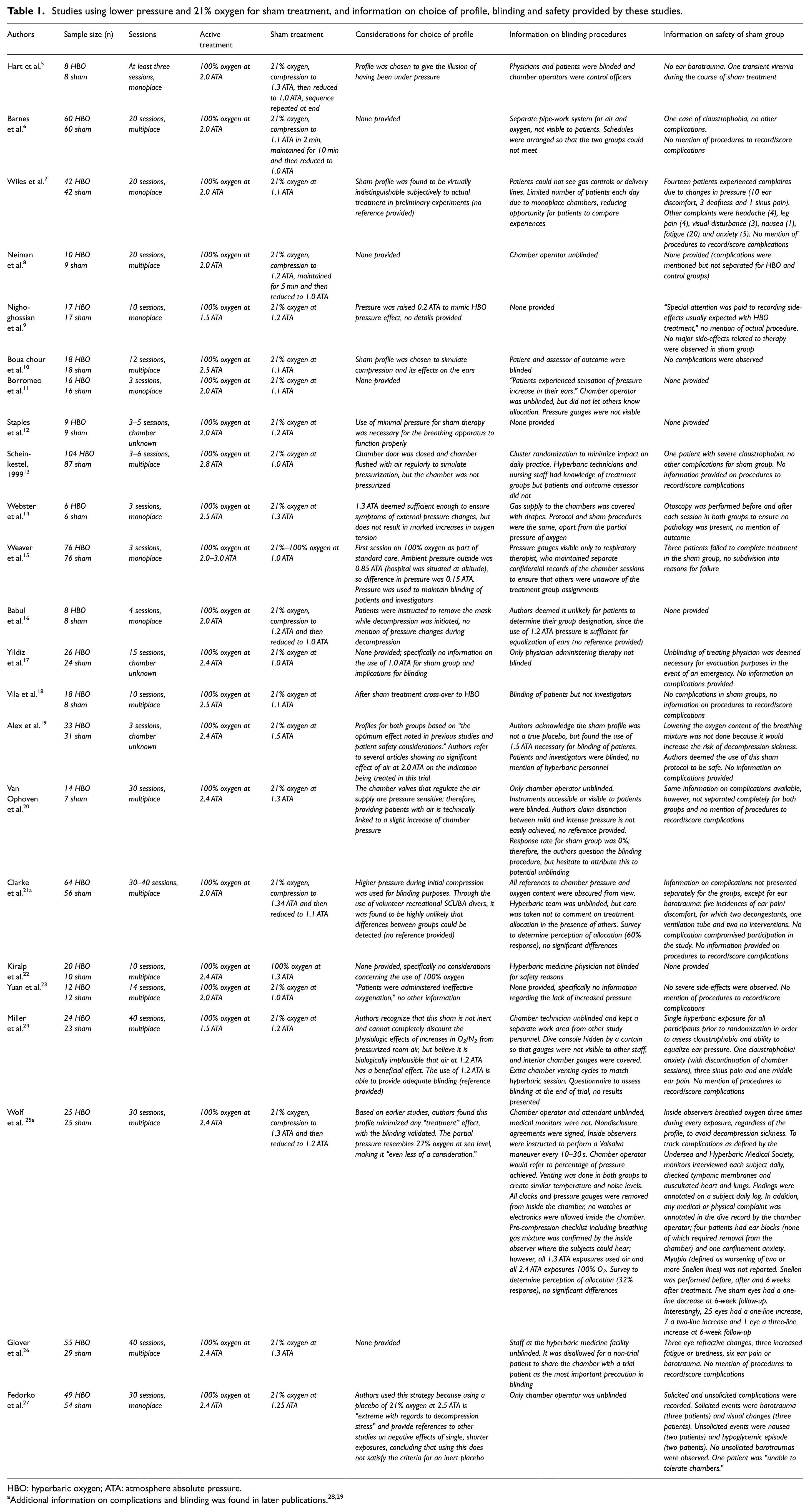
HBO: hyperbaric oxygen; ATA: atmosphere absolute pressure.
Studies using the same pressure and adjusted oxygen level for sham treatment, and information on choice of profile, blinding and safety provided by these studies.
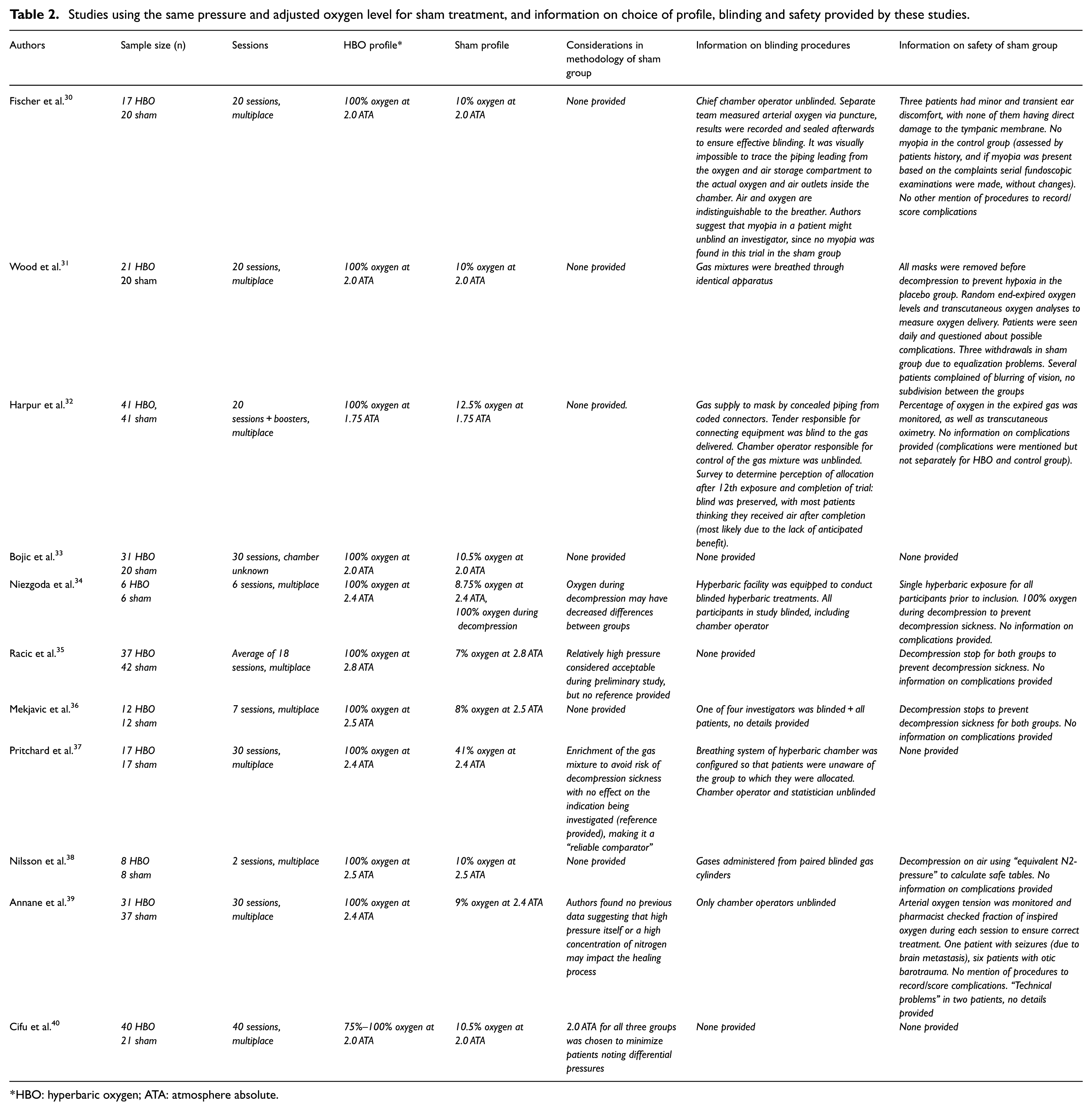
HBO: hyperbaric oxygen; ATA: atmosphere absolute.
Studies using the same pressure and 21% oxygen for sham treatment, and information on choice of profile, blinding and safety provided by these studies.
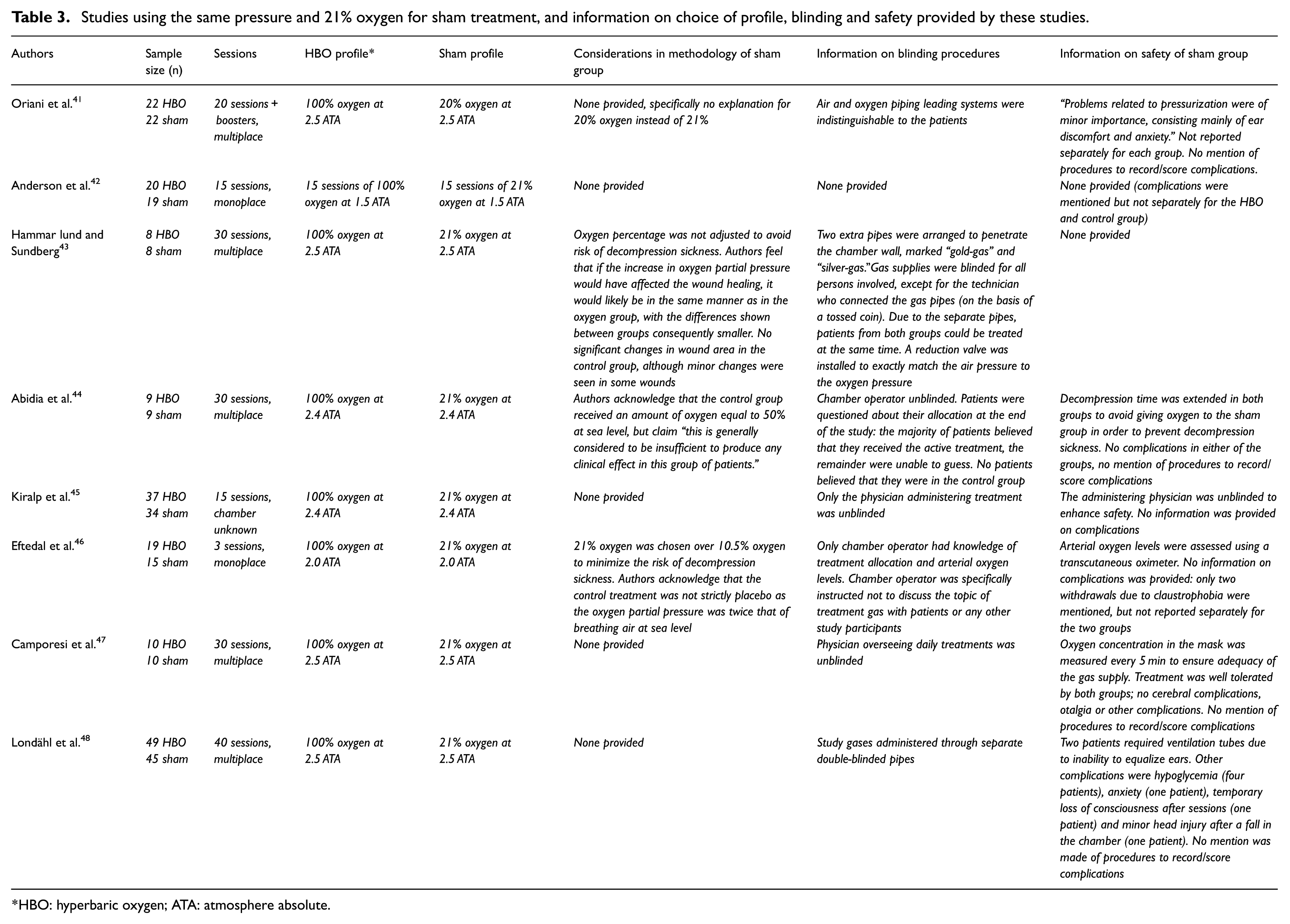
HBO: hyperbaric oxygen; ATA: atmosphere absolute.
Discussion
Sham treatment using lower pressure than the HBO group while breathing 21% oxygen
The first documented trial that involved patient blinding using a sham treatment was a study performed by Hart et al. 5 in 1974. In that study, patients were exposed to either real HBO therapy (at 2.0 ATA) or to a sham therapy consisting of breathing 21% oxygen (i.e. normal air) at a considerably lower pressure (1.1 ATA) than the active treatment. The choice for this profile was not explained by the authors.
The advantage of using only a slight increase in pressure is that the partial pressure of oxygen also increases only slightly. Normally, at atmospheric pressure (1.0 ATA), 21% of the air consists of oxygen; this is equal to a partial pressure of oxygen of 0.21 ATA. If the atmospheric pressure is doubled to 2.0 ATA, the partial pressure of oxygen also doubles to 0.42 ATA. This is the equivalent of 42% oxygen under normal circumstances (i.e. 1.0 ATA).
In the study by Hart et al., 5 21% oxygen was used under a pressure of 1.1 ATA. This is the equivalent of breathing 23% oxygen under atmospheric pressure and, therefore, minimizes the effect on the inertness of the placebo. However, some claim that even a slight increase in the partial pressure of oxygen, or the mere use of pressure alone (irrespective of the partial pressures of the gases), could inflict changes in the body and interfere with the realization of an inert placebo. Although a small number of animal and cell culture studies support this claim, it is debatable whether these results are substantial enough to be used as an argument for clinical practice.49,50
Following the trial by Hart et al., 5 concerns were raised about the effectiveness of blinding, given that no ear barotrauma occurred in the sham control group. If the use of 1.1–1.3 ATA had resulted in patients having to equalize their ears, one would also have expected more reports of complications.
Using a lower pressure for the sham treatment group has some practical implications related to blinding. Apart from masking the gas mixture that is being applied, care should also be taken to blind the interior/exterior of the chamber to prevent gauges or other devices being seen that might indicate the pressure. If the chamber operator or hyperbaric physician is unblinded, they should be instructed not to refer to the pressure used when in close vicinity to the patients and investigators and to use a percentage of pressure (rather than the actual pressure) when communicating with an (inside) chamber attendant. Using a different pressure also requires a separate daily session, preferably scheduled so that the two groups cannot meet and compare experiences. The use of cluster or block randomization can minimize the impact of these precautions on daily practice. Additional practical measures used to prevent unblinding in these trials are presented in Table 1.
Sham treatment using the same pressure as the HBO group while breathing a mixture with an adjusted percentage of oxygen
The second documented double-blind trial used a different strategy and was performed in 1983 by Fischer et al. 30 In that study, the same pressure was used for both the HBO and the control groups (2.0 ATA). This reduced concerns about patients’ perception of their allocation due to differences in pressure between the groups and made blinding easier because only the breathing mixture was different.
To correct for the increase in partial pressure of oxygen to 0.42 ATA for the control group, the breathing mixture for the control group was lowered in oxygen content by adding nitrogen. This resulted in a mixture of 10% oxygen and 90% nitrogen, allowing the control group to be exposed to about the same partial pressure of oxygen as normal (0.2 ATA). However, an increase in the nitrogen percentage in the breathing mixture also increases the risk for decompression sickness. In decompression sickness, nitrogen bubbles are formed inside the body once the patient is decompressed, resulting in symptoms ranging from joint pain and rashes to other neurological symptoms such as paresthesia and paresis. To prevent this, after the treatment, the period of time in which the pressure is lowered to normal has to be prolonged, by stopping once (or multiple times) during decompression; patients in the control group have to breathe 100% oxygen during decompression to get rid of any excess nitrogen.
In the present review, of the 11 trials applying this strategy, 3 used stops in the decompression to prevent decompression sickness,35,36,38 and 1 trial administered 100% oxygen during decompression. 34 Other trials did not mention the risk of decompression sickness or the way that this risk was minimalized. The authors of the study using 100% oxygen during decompression acknowledged that the use of this strategy may have negatively influenced the inertness of the placebo, possibly decreasing the differences in outcome between the groups. 34
When blinding measures are limited to the breathing gas alone, patients of the two groups can receive treatment during the same session (in case of a multichamber). Separate piping systems for air and oxygen can be installed relatively easily, as long as the systems are (visibly) indistinguishable from one another. To ensure that the appropriate breathing mixture is applied, oxygen concentration in the mask, or arterial oxygen tension, can be measured. For this reason and for other safety reasons (e.g. in case of an evacuation) in most studies, the chamber operator and/or the hyperbaric physician was unblinded. As mentioned, unblinded staff should be instructed not to discuss the topic of treatment gas with the patients or any other study participants.
Sham treatment using the same pressure as the HBO group while breathing 21% oxygen
In 1990, Oriani et al. 41 introduced a third strategy: 2.0 ATA for both groups, with the control group breathing 21% oxygen, instead of correcting the percentage of oxygen to keep the partial pressure of oxygen at 0.21 ATA. As mentioned, this resulted in the control group breathing the equivalent of 42% oxygen under atmospheric pressure. In the present review, seven other trials used this same strategy, with a maximal partial pressure of oxygen of 0.53 ATA. However, it is debatable whether this strategy can be considered to deliver a true placebo. For example, Greif et al. 51 showed that patients receiving 80% oxygen during and 2 h after colorectal resection had 50% fewer surgical wound infections than patients receiving 30% oxygen. The amount of oxygen that can be therapeutic is likely to depend on the disease being treated; however, in the absence of sufficient evidence regarding the different oxygen fractions in relation to (patho)physiology, the use of 53% oxygen for a placebo might be incorrect. Of the 42 studies in the present review, only two cited previous studies that showed no effect of an increased oxygen partial pressure on the condition being investigated, to justify their choice of a sham profile.19,37 In contrast, in 2016, Fedorko et al. 27 stated that using a placebo of 21% oxygen is “… extreme regarding decompression stress” and cited earlier studies reporting the negative effects (endothelial injury, proinflammatory changes and significant venous gas emboli) of single, shorter exposures; these authors concluded that using this particular profile does not satisfy the criteria for an inert placebo.
Of the eight trials that used 21% oxygen at the same pressure as the actual treatment, only three discussed the choice for this sham profile. Two groups preferred the use of 21% oxygen over an adjusted percentage to avoid the risk of decompression sickness,43,46 and the other claimed that the equivalent of 50% oxygen “… is generally considered to be insufficient to produce any clinical effect in this group of patients.” 44 Unfortunately, the authors presented no evidence for this claim.
Blinding perception as reported by patients
In the first double-blind trial (performed by Hart et al. 5 ), the lack of barotrauma raised questions about the adequacy of blinding measures, especially regarding the use of a lower pressure. However, sequential trials using the same strategy reported problems with equalization: even the use of ventilation tubes in the sham group was reported. 28 This would imply that blinding was adequate.
To further investigate blinding perception by patients, in some trials, the patients were asked about their allocation: at the end of treatment, patients were asked whether they thought they had received HBO therapy, sham treatment or if they did not know. In the present review, for each of the three strategies identified, at least one randomized controlled trial had provided information on this topic. All studies concluded that there was no relationship between patients’ perception and their actual allocation, thereby concluding that blinding had been adequate.25,28,32,44 In addition, in 2008, Clarke 28 stated that “… through the use of volunteer recreational Scuba divers it was found to be highly unlikely that differences between groups could be detected.” Unfortunately, no reference was provided to support their statement.
Apart from the information emerging from the trials in the present review, concerns were raised by others about the blinding perception of patients when using sham treatment. For example, in 2008, Rainolds and Long 52 used experienced scuba divers to assess whether they could differentiate between the use of 1.2 and 2.0 ATA; their study showed that even the most experienced divers (>500 dives) were unable to make this distinction. It is important to mention that this latter study used “… subtle pressure variations toward the attainment of the final target pressure” to ensure that patients in both groups had to constantly auto-inflate their ears during the first 10 min of compression. In the present review, none of the trials reported use of this latter strategy.
In 2009, Jansen et al. investigated the blinding of volunteers who had no prior experience in hyperbaric treatment or diving. They were asked to guess if they were pressurized to 1.2 or 2.5 ATA and how certain they were about this. Most volunteers reported to be quite certain they were exposed to 2.5 ATA, even though this opinion proved to be (statistically) invalid. 53
In 2012, Weaver et al. asked divers and experienced chamber attendants to estimate the pressure (1.2 or 1.5 ATA) and breathing gas (air or oxygen) that was being applied. Again, the conclusion was that no distinction could be made. 54 In 2015, this latter study was cited by Miller et al. 24 to justify the choice for 1.2 ATA in their sham group.
Based on the present research and the data from patient questionnaires in various randomized controlled trials, it can be concluded that adequate blinding of patients is possible, even when using a pressure lower than that of the actual treatment.
Complications in the sham group
A possible consequence of using pressure for the sham group to ensure adequate patient blinding is the occurrence of barotrauma. In our 42 studies, barotrauma was observed with the use of each of the three strategies. Even the use of minimal pressure resulted in complications: for example, in 1986, Wiles et al. 7 used 1.1 ATA for the sham group, with 14 patients experiencing complaints due to changes in pressure (10 ear discomfort, 3 deafness and 1 sinus pain). As mentioned, even the use of ventilation tubes was reported in sham treatment using lower pressures. 28 Two trials mentioned giving a single hyperbaric exposure to each participant prior to inclusion in the trial to prevent barotrauma during the study.24,34 Moreover, the manner in which barotrauma is scored and reported is often unclear: most studies seem to rely on patients’ complaints, with only two studies mentioning that patients were explicitly questioned about the occurrence of the most common complications.27,31 Only three studies mentioned the use of otoscopy to objectively assess barotrauma of the ears.14,29,30
With regard to another common complication, myopia, in 1983, Fischer et al. 30 suggested that the fact that no myopia was seen in their sham group might potentially unblind an investigator. However, later research showed that myopia appeared in the sham group with the use of each of the three strategies. This could either be an effect of the (slight) increase in pressure and/or oxygen partial pressure or a placebo effect. Unfortunately, information on the method used to determine myopia is often missing and is most likely based on patients’ complaints only. One study made a routine eye examination using a Snellen chart before, immediately after and 6 weeks after treatment to determine visual changes. No myopia (defined as a decrease of ≥2 Snellen lines) was seen in either the HBO or sham group. Interestingly, an increase in vision was reported in some patients, with up to three Snellen lines in one patient in the control group. 29 However, since the pathophysiology of myopia in HBO therapy remains unclear, it is uncertain how these results can best be interpreted.
Apart from barotrauma and myopia, claustrophobia was also reported in sham therapy. However, this can be expected, since the environment for patients in the sham group is similar to that for the HBO therapy group, in which claustrophobia is a well-known complication. 2 There were no reports of serious complications, such as oxygen toxicity (not expected with the partial pressures of oxygen used for sham treatment), decompression sickness or lung barotrauma.
Conclusion
This review examined different strategies used in the past to create a sham (hyperbaric) treatment. This is important because with the increased application of evidence-based medicine, randomized controlled trials are a frequently requested type of research. In such a trial, the addition of a sham treatment implies a considerable burden for patients not receiving actual treatment, including the time involved and the risk of complications (e.g. barotrauma of the ears). This means that performing such a trial is associated with ethical considerations, especially if performed with a vulnerable patient population, such as the elderly or the critically ill. However, especially in research where outcomes are patient reported, the inclusion of sham treatment can be a valuable addition to a trial.
All trials included in this review followed one of the three sham profiles: use of a lower pressure than the HBO group, while breathing 21% oxygen; use of the same pressure as the HBO group, while breathing a mixture with an adjusted percentage of oxygen; and use of the same pressure as the HBO group, while breathing 21% oxygen. The advantages and disadvantages of these strategies concerning blinding procedures, practicality, safety and inertness of placebo have been discussed.
The use of minimal pressurization and 21% oxygen was found to provide adequate blinding and cause the least interference on partial pressure of gases and, therefore, in creating an inert placebo. Although this strategy is associated with additional considerations regarding practicality and blinding measures, it is the most frequently used and documented profile. Considerable care is required when deciding which sham procedure to use; moreover, researchers need to report in detail the measures that were taken to ensure adequate performance of the chosen strategy.
Footnotes
Appendix
Search strategies used to search electronic databases.
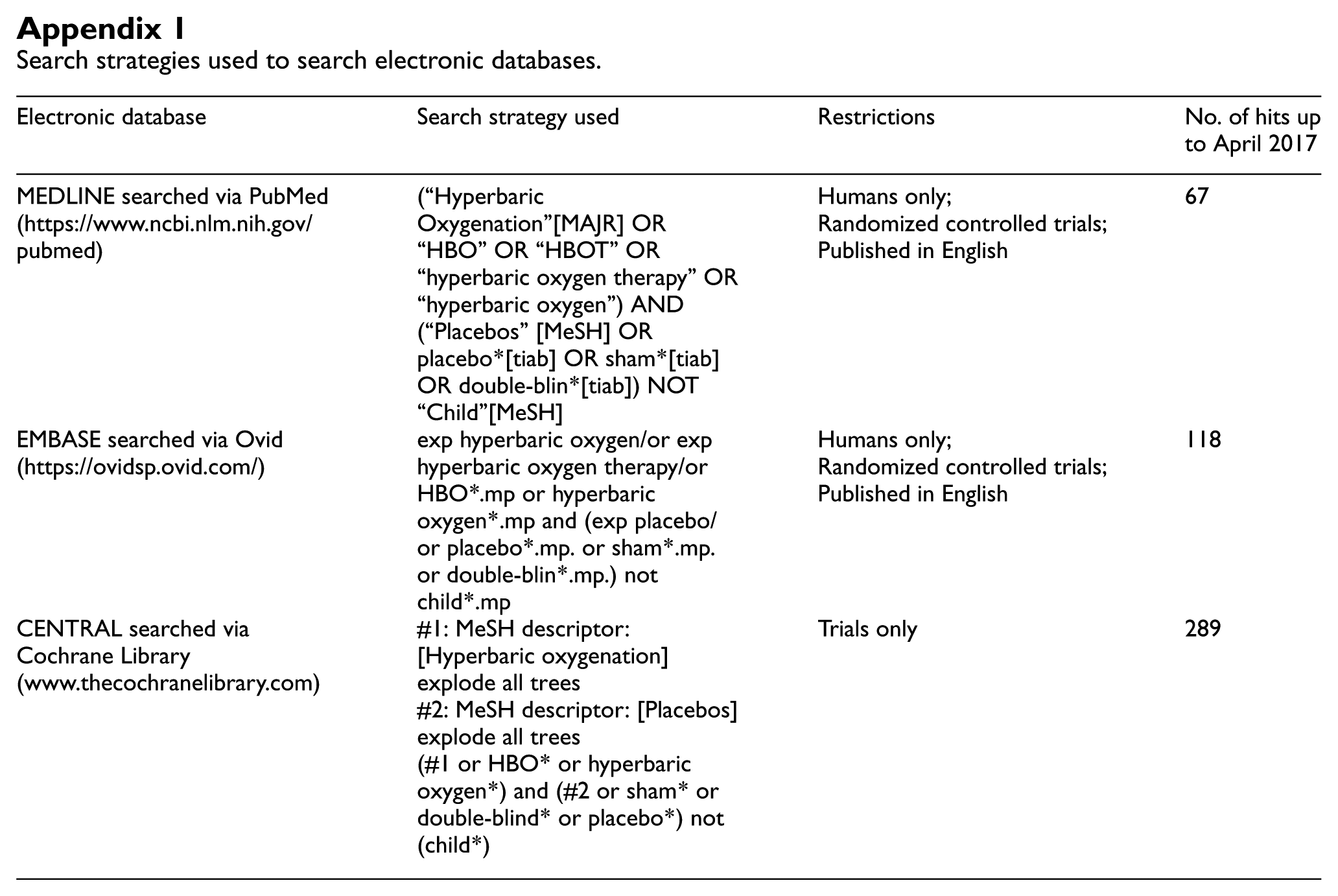
| Electronic database | Search strategy used | Restrictions | No. of hits up to April 2017 |
|---|---|---|---|
| MEDLINE searched via PubMed (https://www.ncbi.nlm.nih.gov/pubmed) | (“Hyperbaric Oxygenation”[MAJR] OR “HBO” OR “HBOT” OR “hyperbaric oxygen therapy” OR “hyperbaric oxygen”) AND (“Placebos” [MeSH] OR placebo*[tiab] OR sham*[tiab] OR double-blin*[tiab]) NOT “Child”[MeSH] | Humans only; Randomized controlled trials; Published in English |
67 |
| EMBASE searched via Ovid (https://ovidsp.ovid.com/) | exp hyperbaric oxygen/or exp hyperbaric oxygen therapy/or HBO*.mp or hyperbaric oxygen*.mp and (exp placebo/or placebo*.mp. or sham*.mp. or double-blin*.mp.) not child*.mp | Humans only; Randomized controlled trials; Published in English |
118 |
| CENTRAL searched via Cochrane Library (www.thecochranelibrary.com) | #1: MeSH descriptor: [Hyperbaric oxygenation] explode all trees #2: MeSH descriptor: [Placebos] explode all trees (#1 or HBO* or hyperbaric oxygen*) and (#2 or sham* or double-blind* or placebo*) not (child*) |
Trials only | 289 |
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
