Abstract
‘Sample size requirements to detect a two- or three-way interaction in longitudinal cluster randomized clinical trials with second-level randomization’ by Moonseong Heo, Xiaonan Xue and Mimi Y Kim published in Clinical Trials 2014; 11: 503–507. DOI: 10.1177/1740774514532724.
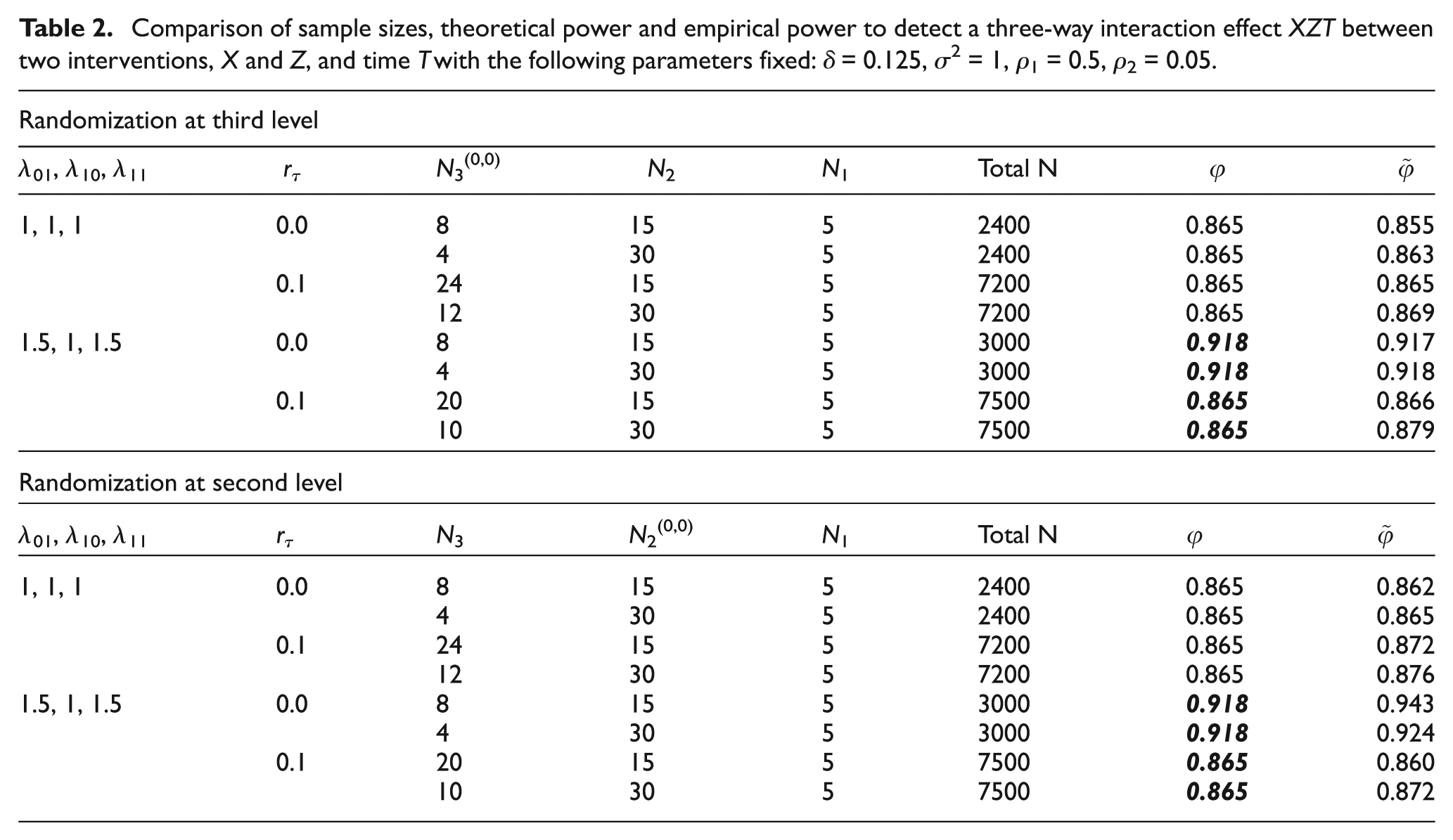
The contents of Table 2 should be corrected as indicated by the italicized bold-faced numbers.
Comparison of sample sizes, theoretical power and empirical power to detect a
three-way interaction effect XZT between two interventions,
X and Z, and time T with the
following parameters fixed: δ = 0.125,
σ2 = 1, ρ1 = 0.5,
ρ2 = 0.05.
Randomization at third level
λ01,
λ10, λ11
rτ
N
3
(0,0)
N
2
N
1
Total N
φ
1, 1, 1
0.0
8
15
5
2400
0.865
0.855
4
30
5
2400
0.865
0.863
0.1
24
15
5
7200
0.865
0.865
12
30
5
7200
0.865
0.869
1.5, 1, 1.5
0.0
8
15
5
3000
0.917
4
30
5
3000
0.918
0.1
20
15
5
7500
0.866
10
30
5
7500
0.879
Randomization at second level
λ01,
λ10, λ11
rτ
N
3
N
2
(0,0)
N
1
Total N
φ
1, 1, 1
0.0
8
15
5
2400
0.865
0.862
4
30
5
2400
0.865
0.865
0.1
24
15
5
7200
0.865
0.872
12
30
5
7200
0.865
0.876
1.5, 1, 1.5
0.0
8
15
5
3000
0.943
4
30
5
3000
0.924
0.1
20
15
5
7500
0.860
10
30
5
7500
0.872
Get full access to this article
View all access options for this article.
