Abstract
Introduction
The contribution of heritable factors to the development of type 2 diabetes (T2D) has been estimated to be as high as 40% [1]. With continuing advances in ‘next-generation’ genetic sequencing technologies, increased ability to identify rare risk loci, and the identification of alternative heritable factors (such as copy number variation, epigenetic marks), we are now poised on the threshold of applying personalized genetic risk information to the clinical management of diabetes [2,3]. Given the increased costs, uncertain efficacy, and potential for adverse impact of such testing, randomized clinical trials are needed for rigorous evaluation of the impact of applied genetic testing on patient behavior and on clinical management [4–6].
One potential role for diabetes genetic testing may be to motivate patients to adopt healthy behaviors to prevent T2D [7]. Lifestyle modification (such as increased exercise and sustained dietary changes to achieve >7% weight loss) has been conclusively demonstrated in several studies to significantly reduce T2D incidence among patients at increased phenotypic risk [8–10]. However, translation of these results into clinical practice has been limited by difficulty in motivating patients to adopt and sustain the behavioral changes necessary to prevent diabetes [11,12].
Recent advances in genetic epidemiology have now identified over three dozen loci associated with increased T2D risk [13]. It is not known whether communicating personal diabetes genetic risk information can be used in clinical practice to increase and sustain patient motivation effectively. Conversely, genetic risk assessment of phenotypically at-risk patients raises the paradox that testing often identifies individuals at relatively low genetic risk [14]. Such ‘lower’ genetic risk information could inappropriately reassure individuals who would clearly benefit from lifestyle changes based on phenotype.
We designed a randomized clinical trial—The Genetic Counseling/Lifestyle Change (GC/LC) Study for Diabetes Prevention—to evaluate the impact of T2D genetic counseling on behavior change among patients who are aware that they are at increased phenotypic risk for diabetes. Funded by the National Institute for Diabetes and Digestive and Kidney Diseases (NIDDK), our study is specifically designed to test two separate hypotheses in comparison to individuals randomized to no genetic testing: (1) individuals with ‘higher’ diabetes genetic risk would be more motivated to sustain lifestyle changes; and (2) individuals with 'lower’ diabetes genetic risk would be less motivated to sustain lifestyle changes.
In this report, we describe important study design decisions related to: (1) implementing an individualized diabetes genetic risk score, (2) creating an allocation scheme based on the novel application of ‘Mendelian randomization’ to exclude the majority of individuals who fall into a relatively narrow ‘average’ diabetes genetic risk range, and (3) designing an ethical genetic counseling intervention to motivate participants regardless of their genetic risk score.
Methods
Patient eligibility and recruitment
We enrolled patients at high risk for T2D based on phenotypic criteria by identifying individuals in our clinical network with the metabolic syndrome (MetS; defined as meeting at least three of five of the following clinical criteria: fasting glucose ≥ 100 mg/dL, waist circumference > 35 in (women) or > 40 in (men), HDL cholesterol < 50 mg/dL (women) or < 40 mg/dL (men), triglycerides ≥150 mg/dL, and blood pressure ≥ 130/85 mmHg or prescribed antihypertensive medication) [15]. Patients in this source population were predominantly white (79%), with mean age 56.5 (±14) years, and evenly divided between men and women. National data indicate that patients with MetS have a 3- to almost 30-fold increased diabetes incidence compared to similar patients without MetS [16,17]. MetS has similar sensitivity (but lower specificity) than the more burdensome oral glucose tolerance test [18] for identifying individuals at risk for T2D and has the distinct advantages of using readily available clinical measures and of identifying a larger at-risk patient population (47 million in the US with MetS [19]), making this approach more readily applicable to the ‘real world’ setting.
Potentially eligible study participants were identified initially through automated medical record review using a previously validated algorithm that had 73% sensitivity and 91% specificity for identifying patients with MetS [20]. Eligibility of potential participants was then verified using a manual medical record review and, with permission from the patient’s primary care provider, confirmed via a brief telephone screen.
Study schema—Phase 1
Our protocol was implemented in two phases (Figure 1). In Phase 1, we enrolled and consented eligible participants to the GC/LC Study. At this first visit, all participants completed baseline surveys to assess baseline diabetes risk perception and current motivation to change diabetes-related health behaviors. After survey completion, participants were randomized in a 4:1 ratio to diabetes genetic risk testing vs no testing. The larger proportion of participants allocated to genetic testing was necessary in order to identify sufficient numbers of patients in the top and bottom risk quartiles (‘higher’ and ‘lower’ diabetes genetic risk) for the more resource-intensive Phase 2. Participants found to have ‘average’ diabetes genetic risk results after genotyping were excluded from Phase 2. These individuals received their diabetes genetic test results directly from the principal investigator and were advised to follow up with their primary care physicians to address modifiable risk factors.
Study schema: Phase 1 (2-stage randomization) and Phase 2 (12-week diabetes prevention curriculum)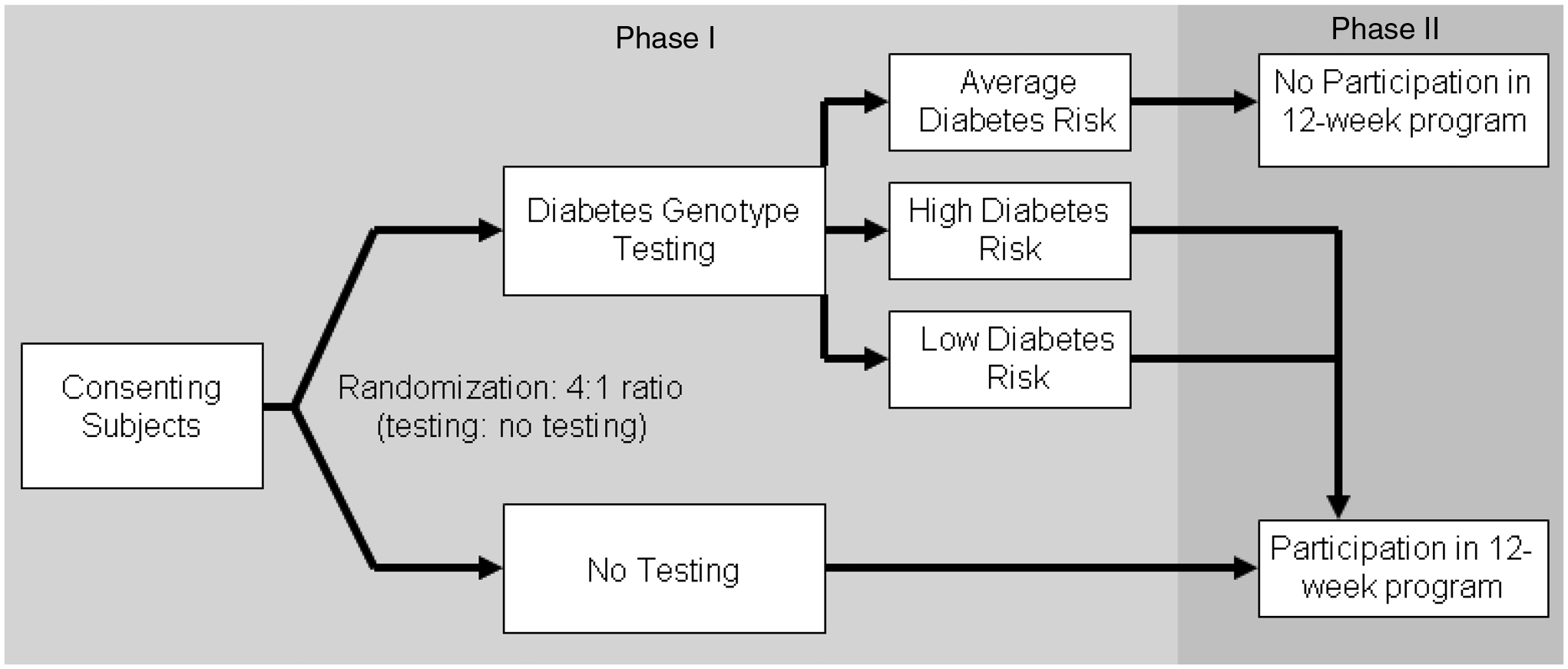
Study schema—Phase 2
Participants allocated to Phase 2 included the ‘higher’ and ‘lower’ diabetes genetic risk result recipients (intervention patients) and the untested controls. These patients returned for a second research visit once genotyping was completed (3–4 weeks after first research visit). At this visit, intervention patients received their diabetes genetic risk results and underwent a 15-min individual diabetes genetic risk counseling session. After the counseling session, intervention patients completed the repeat survey and were scheduled for the 12-week diabetes prevention course. Control patients attending the second research visit completed the repeat survey and were scheduled for the 12-week prevention course. In the weeks following completion of the curriculum, participants returned for their third and final research visit and completed the third survey. Thus, apart from the diabetes genetic risk counseling session, intervention and control patients in Phase 2 received identical exposure to diabetes risk education and to study staff over the approximately 4 months of their study participation.
Creating a diabetes genetic risk score
For the GC/LC Study we used 36 validated single nucleotide polymorphisms (SNPs) independently associated with T2D [13]. At each locus, an individual can have 0, 1, or 2 risk-associated alleles. We created a single diabetes genotype risk score by summing up the total number of risk alleles (range 0–72). Prior research has shown that for risk ranking simple summing provides substantially similar results as various approaches that weight individual alleles according to effect size [21].
Relative risk for type 2 diabetes by diabetes genotype risk score derived from the Framingham Offspring Study
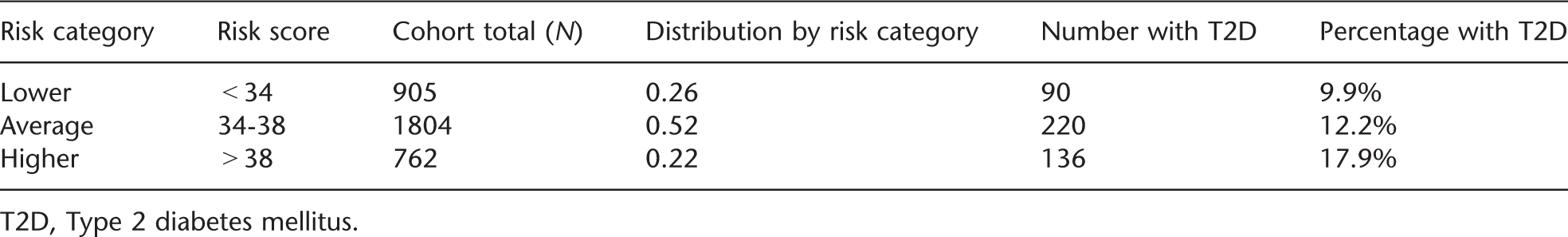
T2D, Type 2 diabetes mellitus.
For the GC/LC Study, we provided our study participants with an overall estimate of their risk for developing diabetes that combined their known phenotypic risk (based on MetS) with their measured genetic risk (derived from the Framingham Offspring Study data). Phenotypic risk for our study cohort was determined using data from our own hospital primary care practices [20]. Based on an analysis of 3-year diabetes incidence among patients with MetS, we estimated an 11% risk of developing diabetes over the next 3 years for our overall study population. Applying the relative risks determined using the Framingham Offspring Study data (46% relative increase for the ‘higher’ and 18% relative decrease for the ‘lower’ risk groups), we estimated that patients with ‘higher’ genetic risk results had a 3-year diabetes risk of 17% and patients with ‘lower’ genetic risk results had a 3-year diabetes risk of 9%.
Mendelian randomization and post-randomization allocation
Since roughly 50% of genotyped individuals fall within the relatively narrow numeric range of ‘average’ genetic risk, we chose a post-randomization allocation strategy that excluded ‘average’ genetic risk patients in order to substantially reduce the size and cost of Phase 2 (two further research visits and the 12-week diabetes prevention group sessions) without reducing the trial’s power to test our two study hypotheses. The rationale for this post-randomization allocation was based on the concept of ‘Mendelian randomization.’
In classic clinical trial design, post-randomization allocation to study arm is forbidden because the criteria used for allocation may introduce confounding and thereby undermine the benefit of randomization. Mendelian randomization is a term coined to describe the concept that a given individual’s genotype represents the random allocation of paternal and maternal alleles [23]. This concept has been used to support observational data analyses [24–26]. The GC/LC Study represents one of the first examples of this concept being used to increase the efficiency of a randomized trial. Since the genetic factors (in this case, alleles associated with diabetes) are not believed to be causally associated with the primary study outcomes (such as program attendance behavior), the diabetes genotype risk score acts as an instrumental variable that was assigned randomly at gametogenesis. In this context, post-randomization allocation to study arm does not undermine the random allocation of risk SNPs within study arms.
Design of genetic counseling intervention
The goals of the 15-min genetic counseling session were to explain the basic concept of genetic testing, present participants with their diabetes genotype score, and explain how their test results modified their phenotypically estimated risk. Participants received a one-page printed report that listed their results for each SNP tested and their total score. The score was also interpreted as conveying ‘higher’ or ‘lower’ diabetes genetic risk. This report was used as the basis for ‘personalized’ diabetes genetic risk counseling. To graphically represent risk to the intervention patients, we first presented a diagram to illustrate the combined influence of genetic (non-modifiable) and environmental (modifiable) factors. The added risk information generated from the genetic testing results was communicated to patients using the visual concept of a balance scale, in which the individual’s 3-year absolute diabetes risk was either increased or decreased based on the additional information provided by their diabetes genetic risk assessment (for example, the scale tipped from 11% absolute 3-year risk to 17% absolute risk among patients with ‘higher’ genetic risk results). The initial development of the counseling session and visual materials was piloted using cognitive interviews with individuals who would have met eligibility criteria for the main study [27].
A key ethical consideration in our intervention development was that the diabetes genetic risk counseling session should be designed to avoid discouraging phenotypically and genetically high-risk patients from participating in a diabetes prevention program. Thus, the genetic counseling intervention had to be structured so that both ‘higher’ and ‘lower’ diabetes genetic risk result recipients would be motivated to make behavior changes. Accordingly, ‘higher’ diabetes risk result disclosure focused on the need to adopt lifestyle changes to counterbalance the increased diabetes genetic risk, whereas ‘lower’ diabetes risk result disclosure emphasized the greater relative contribution of modifiable, non-genetic risk factors to the individual’s overall risk for diabetes. The goal was to avoid discouraging ‘higher’ diabetes risk recipients who might feel a sense of genetic fatalism while avoiding in ‘lower’ diabetes genetic risk recipients the sense of being protected from developing diabetes [28].
Choice of study outcomes
For rigorous assessment of the efficacy of the genetic testing and counseling, all participants in Phase 2 were enrolled into a 12-week Group Lifestyle Balance diabetes prevention curriculum modeled after the Diabetes Prevention Program. This format was developed by Seidel et al. and first implemented in patients with MetS [29]. The curriculum provided educational content (such as identifying healthy eating habits, planning an exercise program) and also used motivational tools (dietary logs, weekly weigh-ins) to initiate behavior changes associated with diabetes prevention. Each 12-week program was conducted by clinical staff who were unaware of participants’ genetic risk test results. Although we could not blind patients to their randomization status—genetic testing or no testing—we instructed the group session educators to request that participants did not disclose their results to the group. To assess the adequacy of masking, we asked the group leaders to predict allocation status for all group participants after completion of the 12-week curriculum.
Our primary study outcomes were the number of 12-week program sessions attended (behavior) and changes in self-reported readiness to adopt the behavioral changes recommended by the diabetes prevention program (motivation). Secondary outcomes included objective measures of weight change before and after the 12-week session and number of weekly exercise and dietary log books turned in during the 12-week session.
Surveys administered at the baseline and follow-up research visits included: Diabetes Risk Perception [30], Stage of Change for a Low Fat Diet [31], Stage of Change for Exercise [32], and Stage of Change for Weight Loss [33]. The three Stage of Change instruments are each validated measures designed to assess motivation by classifying respondents into one of five stages (precontemplation, contemplation, preparation, action, or maintenance) for each of the three behavioral domains (following a low-fat diet, exercising, and losing weight) related to diabetes prevention. For example, the Stage of Change for Exercise instrument asks: ‘Please describe your current exercise behavior,’ with five choices [Currently, I do not exercise and I do not intend to start exercising in the next 6 months; Currently, I do not exercise but I am thinking about starting to exercise in the next 6 months; Currently, I exercise some but not regularly; Currently, I exercise regularly but I have only begun doing so within the last 6 months; and Currently, I exercise regularly and have done so for longer than 6 months] corresponding to the five stages of change.
Sample size and power calculation
A sample size of 90 participants (30 ‘higher’, 30 ‘lower’ diabetes genetic risk and 30 controls) was planned to allow sufficient power to demonstrate clinically meaningful results across both behavioral and self-reported outcomes of interest. Based on the prior experience of our diabetes center in delivering 12-week diabetes prevention courses, we also accounted for a 10% loss-to-follow-up rate when calculating power. Assuming the mean number of visits among controls was 8.5 based on prior experience at our center (unpublished data) and the standard deviation was 2, the study was of sufficient sample size to detect a 20% difference (1.7 difference in number of visits between comparison groups) with 97% power with two-sided probability of 0.05 or less. The change in motivation for each patient was categorized as increased, no change, and decreased, based on responses to each of the three Stages of Change instruments (low fat diet, exercise, weight loss). The study had 90% power to show a difference between 55% positive change vs 90% positive change in motivation between arms and 80% power to show a difference between 60% positive change vs 90% positive change in motivation.
Given the relatively small study size, our analysis plan included provision to control for any differences in baseline characteristics using multivariate modeling when examining outcomes by allocation status.
Discussion
We had to address both ethical and logistic design considerations when implementing a randomized trial of diabetes genetic risk testing and counseling among patients at increased phenotypic risk for type 2 diabetes.
Although there are no published clinical trials of diabetes genetic risk testing, several prior studies have examined whether family history can motivate lifestyle change. These small studies focused on intermediate or surrogate endpoints and have shown mixed results [6,34,35]. Because individuals perceive ‘personal’ genetic risk differently to their family history [27], it was important to assess the impact of such testing using a rigorous controlled study design. Moreover, unlike family history, where the response is either ‘yes’ (with varying degrees of increased risk) or ‘no’ (perceived as ‘no increased risk’), diabetes genetic risk testing can return not only ‘increased’ but also ‘decreased’ risk results. For this reason, we determined that it was necessary to assess both the potential benefit (improved motivation) and potential risk (false reassurance or genetic fatalism) from such testing.
In designing our study, we made the following important decisions with the goal of maximizing the validity and generalizability of our results:
We randomized to genetic testing vs no genetic testing to capture the most relevant clinical question, which is: ‘Among patients willing to be tested, will testing have a clinical impact?’ We separately evaluated the impact of ‘higher’ and ‘lower’ diabetes genetic risk results relative to the untested controls to disentangle the potentially counteracting impact of these different results on the overall impact of testing. We used post-randomization allocation justified by the concept of Mendelian randomization to efficiently exclude nearly 50% of tested patients with ‘average’ results from the resource-intensive Phase 2. We created a clinically implementable diabetes relative risk score based on the most up-to-date set of genetic variants associated with type 2 diabetes as of July 2010. This was achieved by combining relative genetic risk calculated from a finely phenotyped research cohort (Framingham) with a phenotypic risk estimation calculated from the clinical population from which research patients were recruited. We created a genetic counseling intervention that in its relative brevity and explicit communication goals is responsive to the time demands and limited resources that would be expected in the real world setting. We tailored our genetic counseling intervention so as not to undermine motivation in patients at high phenotypic risk for diabetes.
Randomized clinical trials have convincingly shown that increased physical activity and dietary modifications leading to sustained weight loss significantly reduce diabetes risk among patients with pre-diabetes or MetS. However, successful clinical implementation of these lifestyle changes has been limited. Given the ongoing diabetes epidemic, interventions to increase adoption of healthy lifestyle changes among high risk patients are urgently needed. In this ‘dawn of the genomic era’ [36,37], it is imperative to establish the evidence base to guide the clinical application of genetic testing for common chronic diseases such as type 2 diabetes [38] given the rise of direct-to-consumer advertising by commercial companies [39], the eagerness with which many clinicians embrace new technology, and the increased cost of additional testing. The GC/LC Study was designed to begin establishing this evidence base for future practice.
Footnotes
Funding
This study was funded by NIDDK R21 DK84527-01. Dr Meigs received support from NIDDK K24 DK080140. ClinicalTrials. gov identifier: NCT01034319.
