Abstract
Objectives
The aim of this study was to explore the diagnostic efficiency of serum exosomal miR-451a as a novel biomarker for pancreatic cancer.
Methods
Serum samples were collected prior to treatment. First, we analyzed microRNA (miRNA) profiles in serum exosomes from eight pancreatic cancer patients and eight healthy volunteers. We then validated the usefulness of the selected exosomal miRNAs as biomarkers in another 191 pancreatic cancer patients, 95 pancreatic benign disease (PB) patients, and 90 healthy controls.
Results
The expression of miR-451a in serum-derived exosomes from pancreatic cancer patients was significantly upregulated compared with those from PB patients and healthy individuals. Serum exosomal miR-451a showed excellent diagnostic power in identifying pancreatic cancer patients. In addition, exosomal miR-451a showed a significant association with clinical stage and distant metastasis in pancreatic cancer, and the expression level of serum exosomal miR-451a was sensitive to therapy and relapse.
Conclusions
Serum exosomal miR-451a might serve as a novel diagnostic marker for pancreatic cancer.
Introduction
Pancreatic cancer (PC) is a highly lethal malignancy worldwide; it has an extremely poor prognosis, with a 5-year overall survival rate of approximately 10%.1–3 The outcomes of PC are dismal partly because patients often only receive a definite diagnosis at a late stage in the cancer's development. 4 Therefore, the key to improving patient outcomes depends on early diagnosis and constant monitoring of cancer progression. Although many potential biomarkers have been evaluated, there is still no truly reliable tumor marker for PC. 5 Carbohydrate antigen 19-9 (CA19-9), the most widely used PC biomarker, is commonly applied to assist in clinical diagnosis and managing patients. Nevertheless, the utility of CA19-9 has limitations for its underperformance in specificity.6,7 Thus, it is important to identify novel biomarkers for PC diagnosis.
Exosomes are small extracellular particles (40–150 nm in diameter) that form multivesicular bodies and carry a great variety of biological molecules implicated in cell communication, including lipids, proteins, and nucleic acids (DNA, mRNA, and microRNAs (miRNAs).8,9 miRNAs are small (18–25 nt) non-coding RNA molecules with a vital role in post-transcriptional gene silencing processes. 10 Numerous studies have indicated that exosomes contain high levels of miRNAs involved in tumor genesis and progression.11–13 In addition, miRNAs in exosomes are stable and can be monitored in a variety of bodily fluids such as ascites fluid, cerebrospinal fluid, breast milk, saliva, semen, and serum; thus, exosomal miRNAs may be candidate biomarkers for multiple cancers, including PC.14,15
In this study, a microarray-based strategy was adopted to identify the aberrant expression of exosomal miRNAs in PC. We analyzed miRNA profiles in serum exosomes from eight PC patients and eight control subjects. miR-451a was highly upregulated in serum exosomes of PC patients. We aimed to characterize its role in PC diagnosis.
Materials and methods
Serum samples
Whole blood samples from 191 patients with PC and 95 patients with pancreatic benign disease (PB) were collected before treatment in Longhua Hospital (Shanghai, China). A total of 90 individuals seeking a routine health checkup at this hospital, who showed no evidence of cancers or other diseases, were registered in the control group. Venous blood samples (2–3 mL) were collected in coagulation-promoting tubes (Becton, Dickinson and Company) and centrifuged at 5000 g for 10 min and then at 15,000 g for 10 min, at 4°C. The serum samples were stored at − 80°C until needed.
Exosome extraction
The protocol for extracting exosomes from serum using the exoEasy Maxi Kit (Qiagen GmbH, Hilden, Germany) adhered to the manufacturer's instructions.
Transmission electron microscopy
Freshly isolated exosomes were put on a copper grid for 3 min at room temperature and then moved to a 100 µL drop of distilled water. Next, the grids were placed directly in a 50 µL drop of uranyl-oxalate solution at pH 7 for 5 min, and a glass dish was covered with parafilm on ice. Then, the grids were washed five times with distilled water for 3 min each and observed by a HITACHI H7650 transmission electron microscopy (TEM) (Hitachi, Japan).
Western blot analysis
Serum exosomal protein was extracted with RIPA buffer (Sangon Biotech, China). The concentration of protein lysates was quantitated by the bicinchoninic acid assay method (Pierce, Rockford, IL, USA). Equivalent lysates were separated on sodium dodecyl sulfate-polyacrylamide (SDS-PAGE) gels and moved to polyvinylidene fluoride membranes. The membranes were blocked with 5% bovine serum albumin and then incubated at 4°C with primary antibodies (anti CD9 and anti CD63, 1:1000 dilution, Cell Signaling Technology Inc., Danvers, MA, USA) overnight. Next, the membranes were incubated with horseradish peroxidase-conjugated secondary antibody (Cell Signaling Technology Inc.) after washing three times, and were visualized using SuperSignalTM West Pico Chemiluminescent Substrate (Thermo Scientific).
Nanoparticle-tracking analysis
Nanosight NS300 analyzer (Malvern Panalytical Ltd, Malvern, UK) was used to determine the size and concentration of the isolated exosomes. The exosomes were diluted 10 times in Dulbecco's phosphate-buffered saline, and added to the NanoSight sample chamber. The data obtained were processed using the OriginLab OriginPro 8.5 software package.
miRNA microarray
Exosomes were collected from the serum of eight PC patients and eight healthy volunteers using the exoEasy Maxi Kit (Qiagen GmbH). Total RNA was extracted from serum-derived exosomes by TRIzol reagent (Life Technology) and reverse-transcribed to cDNAs by reverse transcription-polymerase chain reaction (RT-PCR) to produce smallRNA (sRNA) libraries. The libraries were sequenced using an Agilent Human miRNA microarray (Shbio Co., LTD, Shanghai, China). The database was derived from the microRNA Database (miRBase 21.0), covering 2549 human associated miRNAs. The microarray used Agilent's unique miRNA assay technology, which can specifically detect mature miRNAs and clearly distinguish highly homologous miRNA molecules. The microarray data were corrected for background and normalized by the mean intensity.
RNA isolation and quantitative RT-PCR analysis
Total RNAs were extracted from serum-derived exosomes and tissue samples using TRIzol reagent (Life Technology) in line with the manufacturer's protocol. Then, they were quantified using the NanoDrop ND-200°C spectrophotometer (Thermo Fisher). The extracted RNA samples were stored at − 80°C. Quantitative reverse transcription and real-time polymerase chain reaction (qRT-PCR) of miR-451a used the All-in-One™ miRNA qRT-PCR Detection Kit (GeneCopoeia, Inc. USA) following its detailed protocol. qRT-PCR was performed to analyze miR-451a expression levels in the serum exosomes of 376 individuals (191 PC patients, 95 PB patients, and 90 control subjects) and 23 paired PC tissues. For miR-451a expression, U6 was used as the internal standard. All experiments were performed in triplicate on a real-time PCR detection system (Qiagen GmbH). The relative expression of serum exosomal miR-451a was evaluated by 2−ΔCt (CtmiRNA−CtU6), while the relative expression of miR-451a in tissue samples was evaluated using the 2−ΔΔCt method.
Statistical analysis
IBM's SPSS Version 23 software (IBM Corporation, Armonk, NY, USA) was used to perform statistical analysis. Continuous data were compared using the Wilcoxon and Kruskal-Wallis tests. Receiver operating characteristic (ROC) curve analysis was performed to analyze the sensitivity, specificity, and accuracy of circulating biomarkers. A p-value < 0.05 was defined as statistically significant.
Results
Participants
From July 1, 2018 to January 1, 2021 a total of 519 individuals were invited to participate. Figure exhibits the flowchart of the study. Participants who were excluded or withdrew from the study were noted. In the end, 376 participants completed the study, making the completion rate 72.4% (Figure 1). The clinical characteristics of the participants are shown in Table 1.
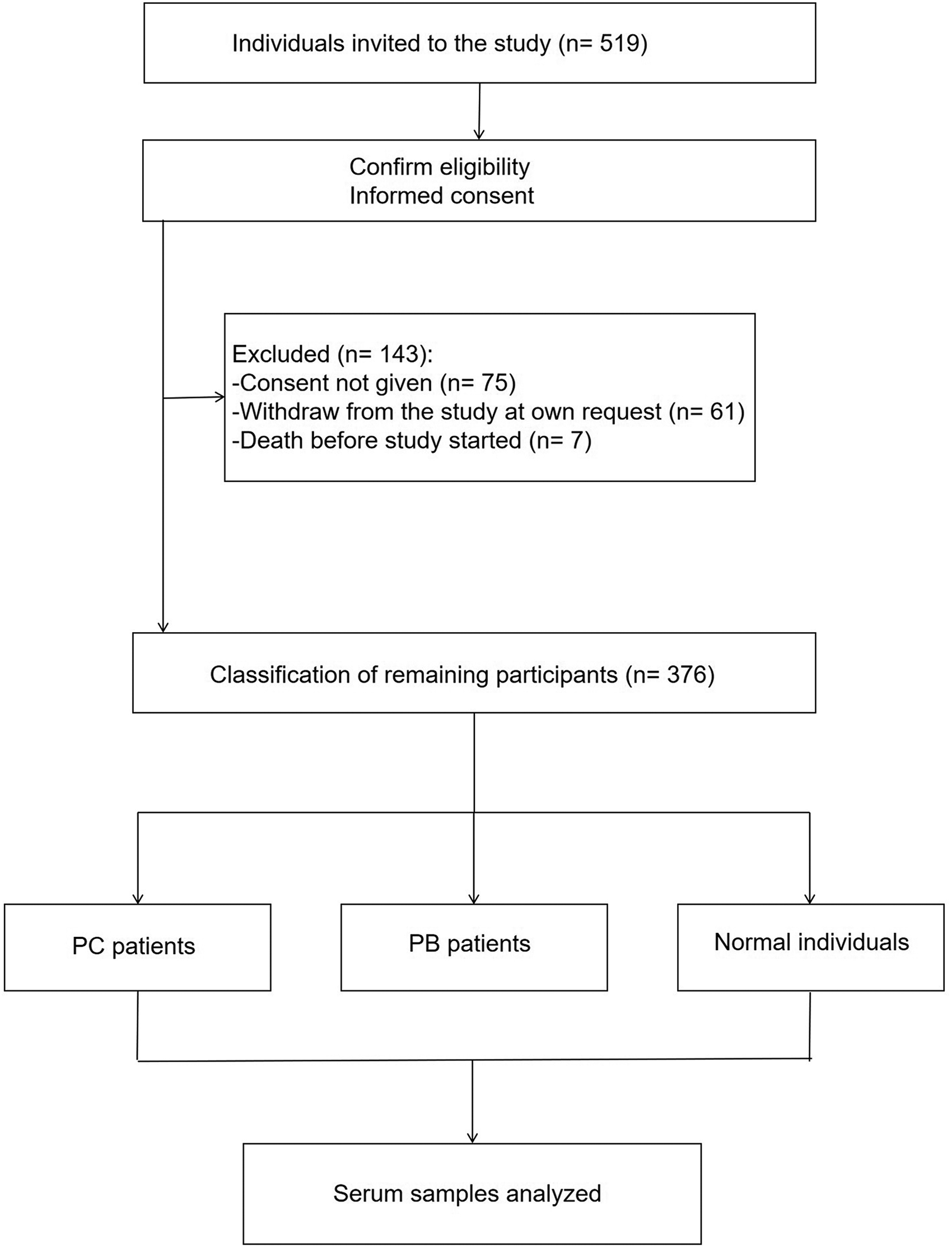
Flow diagram of participants.
Participant characteristics.
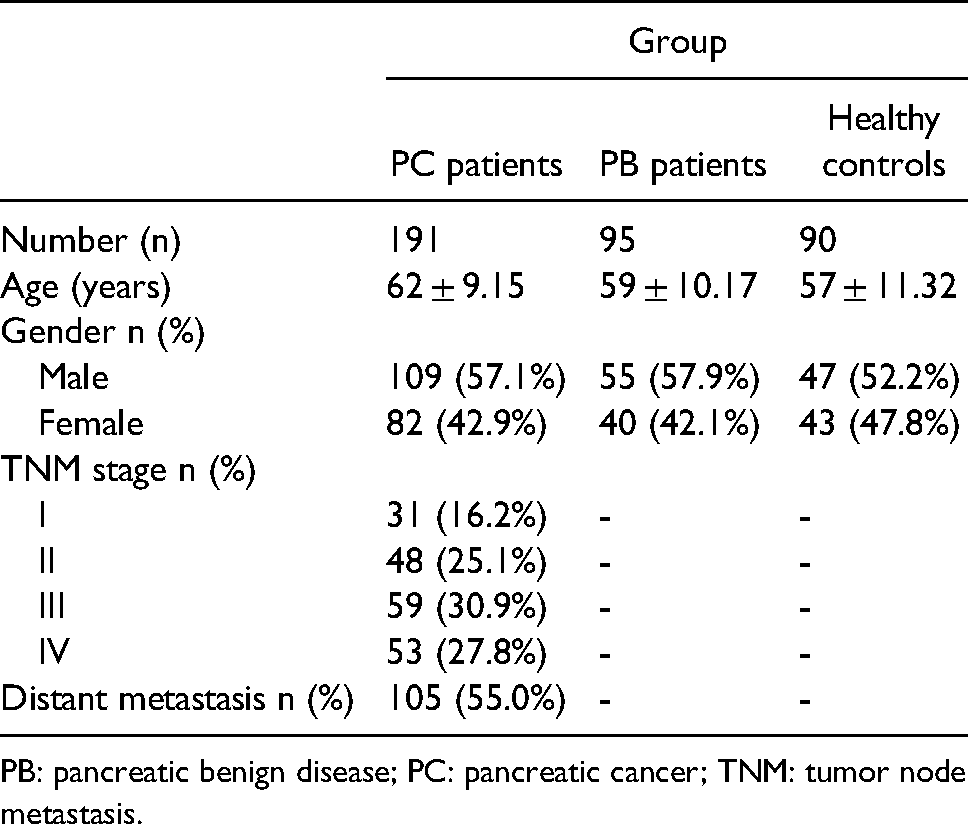
PB: pancreatic benign disease; PC: pancreatic cancer; TNM: tumor node metastasis.
Identification of exosomes
Serum exosomes were extracted using the exoEasy Maxi Kit and were validated by TEM, nanoparticle-tracking analysis (NTA), and western blot analysis. We used TEM to directly observe the morphology of exosomes; we found that exosomes had a diameter of 50–150 nm and a cup-shaped membrane (Figure 2(a)). The NTA analysis revealed the average particle size of serum exosomes and the range of the main peak. Particles with a diameter in the range from 30 nm to 200 nm accounted for 95.8% of all particles, which was in line with the size distribution of exosomal particles (Figure 2(b)). Besides, western blot analysis was used to test the markers, including CD9 and CD63, for further identifying serum exosomes (Figure 2(c)).

Characterization of serum exosomes. (a) TEM was used to observe the morphology of serum exosomes which exhibited a cup-shaped membrane. (b) NTA analysis was used to characterize the size of serum exosomes, and the data showed that the majority of exosomes were mainly between 30 nm and 200 nm in diameter. (c) Detection of CD9 and CD63 protein levels by western blot analysis in exosomes (e) and EDS.
Serum exosomal miR-451a is notably upregulated in PC patients
To screen and evaluate the serum exosome-derived dysregulated miRNAs in PC patients, miRNA expression was profiled in serum exosomes from eight PC patients and eight healthy volunteers. Cluster analysis of the miRNAs was performed (Figure 3(a)), which revealed that serum exosomal miR-451a was highly upregulated in PC patients. To further verify the microarray platform, we used qRT-PCR to compare serum exosomal miR-451a levels among PC patients, PB patients, and healthy controls. The results indicated that the expression levels of serum exosomal miR-451a were highest in PC patients (8.130 (5.180–12.080)), and progressively lower in PB patients (3.080 (1.780–4.230)) and healthy controls (2.270 (1.558–3.715)) (Figure 3(b)), which was consistent with the data generated from the microarray.

Serum exosomal miR-451a is notably upregulated in PC patients. (a) Cluster analysis of miRNAs expression in serum exosomes from PC patients and controls. (b) qRT-PCR was performed to compare serum exosomal miR-451a levels in PC patients, PB patients and healthy controls.
Diagnostic power of serum exosomal miR-451a in PC
ROC curve analysis was performed to assess the diagnostic value of serum exosomal miR-451a for PC. Serum exosomal miR-451a effectively distinguished PC patients from healthy controls (area under the curve (AUC) 0.896, 95% confidence interval (CI) 0.860–0.932, sensitivity 80.10%, specificity 86.67%, Figure 4(a)). Moreover, serum exosomal miR-451a showed excellent performance in discriminating PC patients from PB patients (AUC 0.855, 95% CI 0.812–0.899, sensitivity 71.20%, specificity 89.47%, Figure 4(b)).

Diagnostic power of serum exosomal miR-451a in PC. (a) Serum exosomal miR-451a accurately discriminated PC patients from healthy controls. (b) The performance of serum exosomal miR-451a in identifying PC patients and PB patients.
Correlation of serum exosomal miR-451a with clinical features of PC patients
Next, we analyzed the association between serum exosomal miR-451a and the clinical status of patients with PC. We used the median serum exosomal miR-451a expression level as the cutoff point to split 191 PC patients into two groups: high-expression (n = 95) and low-expression (n = 96). The results showed that serum exosomal miR-451a level closely correlated with tumor node metastasis (TNM) stage and distant metastasis in PC patients (p < 0.0001, Table 1); however, it was not associated with age, gender, tumor location, or tumor size (p > 0.05, Table 2).
Relationship of serum exosomal miR-451a level and clinical status of patients with PC.
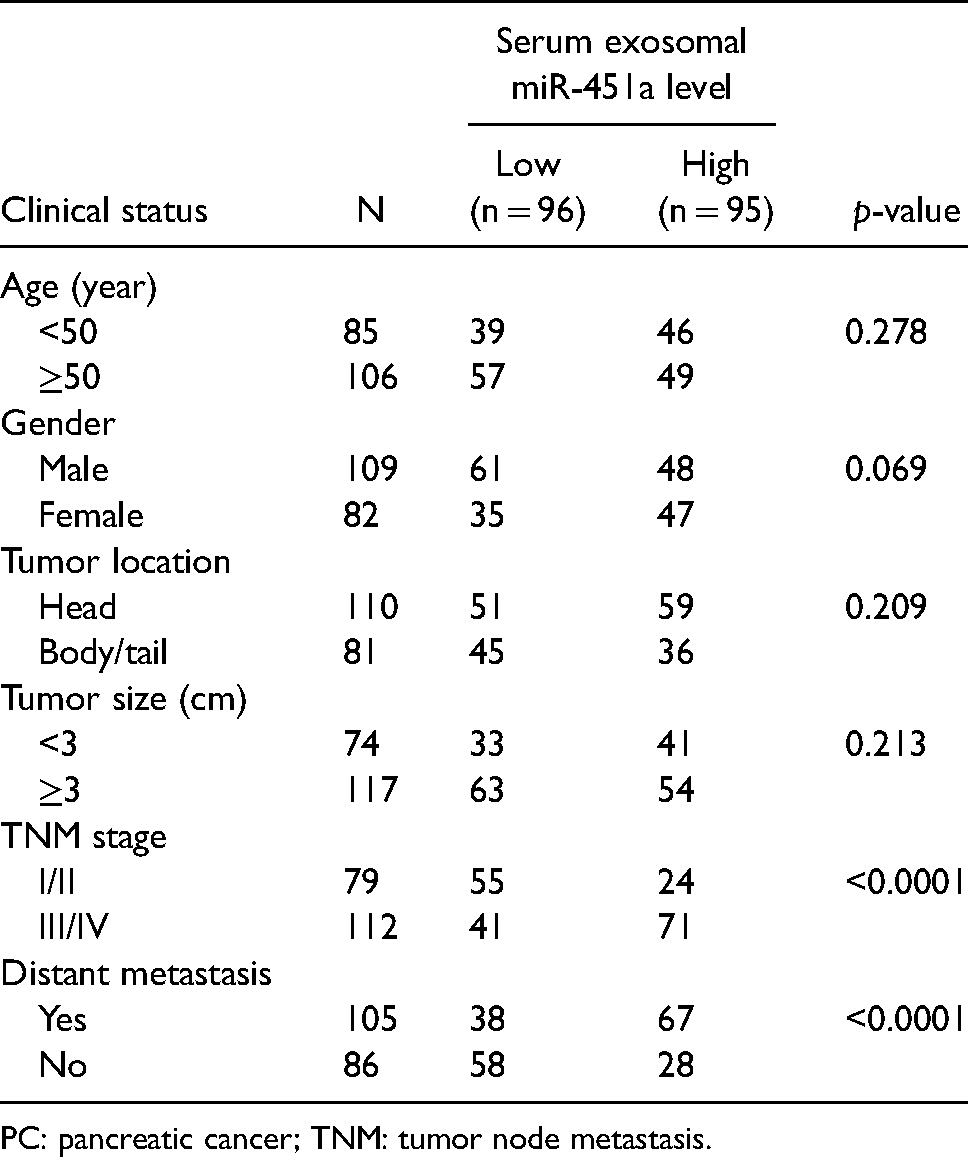
PC: pancreatic cancer; TNM: tumor node metastasis.
Serum exosomal miR-451a is sensitive to therapy and recurrence of PC
After diagnosis, of the 191 PC patients, 34 cases received operative treatment and 157 cases received chemotherapy. Two weeks after surgery or initial chemotherapy, we collected serum samples from these patients. We found that serum exosomal miR-451a levels in PC patients notably decreased following treatment (p < 0.0001, Figure 5(a)). Within half a year of the initial treatment, 69 PC patients experienced a relapse. In the patients who experienced recurrence, the expression levels of serum exosomal miR-451a were remarkably increased (p < 0.0001, Figure 5(b)). In contrast, the expression levels of serum exosomal miR-451a hardly changed in the cases without recurrence (p = 0.3619, Figure 5(c)).

Serum exosomal miR-451a is sensitive to therapy and recurrence of PC. (a) Serum exosomal miR-451a levels were significantly downregulated in PC patients following treatment. (b) Serum exosomal miR-451a levels were remarkably increased in cases with recurrence. (c) The expression levels of serum exosomal miR-451a hardly changed in the cases without recurrence.
Discussion
PC is a prevalent cause of cancer-related death worldwide. 16 While progress in therapeutic treatment strategies, including surgery and chemoradiotherapy, has ameliorated PC prognosis, the early diagnosis of PC remains very difficult.17,18 As a result, it is important to develop effective early diagnostic methods. Serum exosomes are undoubtedly novel candidate biomarkers for PC diagnosis and have gained broad attention.19–28 miR-196a and miR-1246 are remarkably upregulated in exosomes from PC, and their expression levels in plasma samples from patients with localized PC are slightly higher than those in patients without PC. 29 In addition, a previous study has shown that exosomal miR-483-3p levels in patients with intraductal papillary mucinous neoplasm (IPMN) were lower than those in patients with PC. 30 These studies have indicated that exosomal miRNAs can be candidate biomarkers for PC diagnosis.
In the present study, we first demonstrated that serum exosomal miR-451a levels in PC patients were significantly higher than those in healthy individuals using next-generation sequencing. We further identified that serum exosomal miR-451a levels were the highest in PC patients, while they were progressively lower in PB patients and healthy controls. Recent studies have shown that exosomal miR-451a is involved in a variety of cancers, but their conclusions are still controversial. For instance, Zhao et al. reported that serum exosomal miR-451a was markedly lower in hepatocellular carcinoma patients than in healthy individuals, and that exosomal miR-451a suppressed hepatocellular tumorigenesis by targeting LPIN1. 31 Kanaoka et al. found that plasma exosomal miR-451a presented the highest upregulation in non-small cell lung cancer patients with relapse and was closely related to lymphatic metastasis, vascular invasion, and clinical stage. 32 In line with our findings, Goto et al. indicated that serum exosomal miR-451a was significantly upregulated in patients with PC and IPMN compared to control subjects. Their study showed the role of exosomal miR-451a in IPMN, but the association with PC was not further explored. 33 Furthermore, exosomal miR-451a was reported to be related to recurrence and prognosis in pancreatic ductal adenocarcinoma patients.34,35 However, these studies were relatively sketchy. Our study delved into the correlation between exosomal miR-451a and PC from multiple perspectives.
ROC curve analysis revealed that the diagnostic accuracy of serum exosomal miR-451a for PC was 0.896. The overall sensitivity and specificity of exosomal miR-451a were 80.10% and 86.67%, respectively. The diagnostic accuracy of CA19-9 measured by AUC was 0.882. In addition, the sensitivity and specificity of CA19-9 were 81.15% and 83.33%, respectively (Supplementary Figure 1). These findings indicate that the sensitivity of exosomal miR-451a was slightly inferior to that of CA19-9, whereas its specificity was slightly superior, for PC diagnosis. Moreover, serum exosomal miR-451a exhibited excellent performance in discriminating PC patients from PB patients. In addition, we found that the expression level of serum exosomal miR-451a closely correlated with TNM stage and distant metastasis. PC patients with distant metastasis, or in the late stages of cancer, had relatively higher levels of serum exosomal miR-451a. Our study also revealed that serum exosomal miR-451a levels responded to therapy and relapse, suggesting that they are quite sensitive to changes in the state of PC patients.
Of note, the upregulation of miR-451a was also validated in the tissue samples of PC patients (Supplementary Figure 2). Among 23 PC patients, 16 cases showed greater miR-451a expression in tumor tissues, which was consistent with previous results.
In conclusion, the upregulation of miR-451a in serum exosomes and tumor tissues is a frequent event in PC. We demonstrated that serum exosomal miR-451a levels could effectively distinguish PC patients from PB patients and healthy controls, and that they were associated with clinical stage and the distant metastasis of PC. Moreover, serum exosomal miR-451a levels were sensitive to therapeutic response. Serum exosomal miR-451a may be a candidate biomarker for PC, which can assist in its clinical diagnosis and in monitoring changes in patients’ conditions.
Supplemental Material
sj-docx-1-jbm-10.1177_17246008211070018 - Supplemental material for Serum exosomal miR-451a acts as a candidate marker for pancreatic cancer
Supplemental material, sj-docx-1-jbm-10.1177_17246008211070018 for Serum exosomal miR-451a acts as a candidate marker for pancreatic cancer by Jia Chen, Dongting Yao, Weiqin Chen, Zhen Li, Yuanyuan Guo, Fan Zhu and Xiaobo Hu in The International Journal of Biological Markers
Supplemental Material
sj-docx-2-jbm-10.1177_17246008211070018 - Supplemental material for Serum exosomal miR-451a acts as a candidate marker for pancreatic cancer
Supplemental material, sj-docx-2-jbm-10.1177_17246008211070018 for Serum exosomal miR-451a acts as a candidate marker for pancreatic cancer by Jia Chen, Dongting Yao, Weiqin Chen, Zhen Li, Yuanyuan Guo, Fan Zhu and Xiaobo Hu in The International Journal of Biological Markers
Supplemental Material
sj-jpg-3-jbm-10.1177_17246008211070018 - Supplemental material for Serum exosomal miR-451a acts as a candidate marker for pancreatic cancer
Supplemental material, sj-jpg-3-jbm-10.1177_17246008211070018 for Serum exosomal miR-451a acts as a candidate marker for pancreatic cancer by Jia Chen, Dongting Yao, Weiqin Chen, Zhen Li, Yuanyuan Guo, Fan Zhu and Xiaobo Hu in The International Journal of Biological Markers
Supplemental Material
sj-jpg-4-jbm-10.1177_17246008211070018 - Supplemental material for Serum exosomal miR-451a acts as a candidate marker for pancreatic cancer
Supplemental material, sj-jpg-4-jbm-10.1177_17246008211070018 for Serum exosomal miR-451a acts as a candidate marker for pancreatic cancer by Jia Chen, Dongting Yao, Weiqin Chen, Zhen Li, Yuanyuan Guo, Fan Zhu and Xiaobo Hu in The International Journal of Biological Markers
Supplemental Material
sj-xlsx-5-jbm-10.1177_17246008211070018 - Supplemental material for Serum exosomal miR-451a acts as a candidate marker for pancreatic cancer
Supplemental material, sj-xlsx-5-jbm-10.1177_17246008211070018 for Serum exosomal miR-451a acts as a candidate marker for pancreatic cancer by Jia Chen, Dongting Yao, Weiqin Chen, Zhen Li, Yuanyuan Guo, Fan Zhu and Xiaobo Hu in The International Journal of Biological Markers
Footnotes
Acknowledgments
The study was supported partially by grants from The Research Foundation of the National Clinical Research Base of Traditional Chinese Medicine of Longhua Hospital (No. Seedling program LYTD-73); The Innovation Project of Longhua Hospital (No. CX202058); The Applied Basic Research Programs of Science and Technology Commission Foundation of Yunnan Provincial and Kunming Medical University (No. 2019FE001(-109)); The Open Subject of State Key Laboratory of Cardiovascular Disease of Fuwai Hospital (No. 2019kfyf-03, 2019kfyf-04); and The National Natural Science Foundation of China (No. 81803893).
Author contributions
Xiaobo Hu and Fan Zhu designed the study. Jia Chen, Dongting Yao, Weiqin Chen, Zhen Li, and Yuanyuan Guo conducted experiments, analyzed data, and wrote the manuscript. Jia Chen and Dongting Yao contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The Research Foundation of The National Clinical Research Base of Traditional Chinese Medicine of Longhua Hospital (No. Seedling program LYTD-73); The Innovation Project of Longhua Hospital (No. CX202058); The Applied Basic Research Programs of Science and Technology Commission Foundation of Yunnan Provincial and Kunming Medical University (No. 2019FE001(-109)); The Open Subject of State Key Laboratory of Cardiovascular Disease of Fuwai Hospital (No. 2019kfyf-03, 2019kfyf-04); and The National Natural Science Foundation of China (No. 81803893).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
