Abstract
Background
This study explored the relevance between rs1695 and susceptibility to the lung cancer in the Chinese Han population. Stratification analysis was conducted on the basis of age, gender, smoking status, tumor-related family history, and pathological type to observe relations between rs1695 and susceptibility to lung cancer in the subgroups.
Methods
A case-control study was performed with 974 lung cancer patients who were pathologically diagnosed and 1005 healthy cases based on physical examination to analyze the association between rs1695 and the risk of lung cancer.
Results
The frequencies of the AA, GA, and GG genotypes of rs1695 were 68.4%, 28.7%, and 2.9% in cases and 64.8%, 30.8%, and 4.2% in controls, respectively. After adjustment for age, gender, smoking status, and family history, it appears that the rs1695 G allele decreases the risk of lung cancer (OR = 0.811, 95% CI 0.684–0.961, P = 0.016). Moreover, compared with the AA genotype, the GA + GG genotype decreased lung cancer susceptibility (OR = 0.808, 95% CI 0.663–0.985, P = 0.035) and the GG genotype (OR = 0.591, 95% CI 0.347–0.988, P = 0.048). In a stratified analysis, the risk of lung cancer in the G allele carriers decreased among the males, patients without a tumor-related family history, and patients with lung adenocarcinoma, especially in smokers.
Conclusion
The polymorphism of locus rs1695 is related to the risk of lung cancer and is expected to be a target for the prediction of lung cancer.
Introduction
Lung cancer is a major cancer type, which has the highest morbidity and mortality in the world 1 and in China.2,3 A total of 2,206,771 new lung cancer cases and 1,796,144 deaths were estimated in 2020 in 185 countries. 1 Despite the remarkable improvements in diagnosis and treatment, the overall 5-year survival rate of lung cancer remains quite low. 4
The incidence of lung cancer is highly correlated with environmental factors, and cigarette smoking accounts for the major risk of lung cancer. 5 However, only 10% of long-term smokers had lung cancer, 6 which suggests that the susceptibility to lung cancer varied wildly and individually. In addition, several studies have shown that genetic variation is related to the inheritance of lung cancer.7,8 Recently, several genome-wide relation studies (GWAS) have identified a number of genetic susceptibility loci related to the risk of lung cancer.9–11 Therefore, it is essential to discover more indicators and genetic susceptibility or predisposing gene–environmental interactions for the diagnosis of lung cancer.
Single nucleotide polymorphisms (SNPs) may affect gene expression or alter protein structure–function characteristics, which may lead to differences in the cellular repairing capabilities, thereby affecting a person’s susceptibility to cancer. 12 Understanding the interaction between SNPs and cancer susceptibility may elucidate the pathogenesis of cancer. Glutathione S-transferases (GSTs) are a multiple gene family of phase II xenobiotic metabolizing enzymes, which are responsible for metabolizing carcinogenic agents by conjugation with glutathione,13,14 including GSTA, GSTM, GSTP, GSTT, GSTO. 15 The GSTP1 gene is mapped to chromosome 11q13.2, and is comprised of seven exons and six introns. 16 Two common SNPs in GSTP1, Ile105Val (rs1695), and Ala114Val (rs1138272), have been reported. GSTP1 rs1695 A > G polymorphism is an A to G transition leading to the substitution of isoleucine for valine at amino acid position 105. Since Ryberg et al. reported that GSTP1 rs1695 A > G polymorphism was related to lung cancer in 1997, 17 many studies have explored the effects of the GSTP1 rs1695 A > G polymorphisms on the risk of lung cancer.18–26 However, there are few studies on the Chinese population, and the results are inconclusive; therefore, it is not enough to draw definite conclusions about the impact of this mutation on the development of lung cancer.
To further confirm whether the GSTP1 polymorphism affects lung cancer development in the Chinese population, using a larger sample capacity we carried out a case-control study investigating the relation between GSTP1 rs1695 A > G polymorphisms and the risk of lung cancer. A subgroup analysis was stratified in age, gender, smoking status, family history, and histology types to explore the possible effects of the gene-environment interaction, and gene-tumor type interaction in GSTP1 rs1695 A > G polymorphisms was conducted to explore the relevance to the risk of lung cancer.
Methods and patients
Patients and healthy controls
A total of 974 lung cancer cases and 1005 healthy controls were recruited from multiple hospitals in China, such as Shanghai Zhongshan Hospital, Shanghai Chest Hospital, First Affiliated Hospital of Nanjing Medical University, Beijing Union Medical College Hospital, and Wuhan Union Hospital, China. All the subjects reported that they were of Chinese Han nationality. All patients were diagnosed histopathologically and were in compliance with lung cancer diagnostic criteria. Patients who did not have a previous cancer history and receive any radiotherapy or chemotherapy are included. Healthy donors were healthy individuals without any cancer disease and were adjusted for the frequency matching factors (e.g. age, gender, and residential area). After informed consent was obtained, peripheral blood samples were collected from each subject. The study was approved by the Institutional Ethical Committee of each hospital and was conducted according to the Declaration of Helsinki principles.
Genotyping
Genomic DNA was extracted from whole-blood samples using the Qiagen Blood Kit (Qiagen, Chatsworth, CA, USA) according to the manufacturer's instructions. Genotyping quality was examined by a detailed QC procedure, consisting of >95% successful call rate, duplicate calling of genotypes, internal positive control samples, and Hardy–Weinberg Equilibrium (HWE) testing. DNA samples (10 ng) were amplified by polymerase chain reaction (PCR) according to the manufacturer's recommendations. SNP genotyping was performed using a custom-by-design 2 × 48-Plex SNP scan TM Kit (cat. no. G0104; Genesky Biotechnologies Inc., Shanghai, China). This kit was developed according to patented SNP genotyping technology by Genesky Biotechnologies.
Statistical analysis
The frequencies between the cases and controls in age, gender, smoking status, and family history of cancer were calculated by χ2 test odds ratios (OR), and 95% confidence intervals (95% CI) were used to estimate the relation between the rs1965 polymorphism and the risk of lung cancer through unconditional logistic regression. Furthermore, stratified analysis was performed according to age, gender, smoking status, and family history. All statistical analysis were done by SPSS 19.0 (SPSS, Chicago, IL, USA). All P-values presented are two-sided, and a level of P < 0.05 was considered statistically significant.
Results
Characteristics of the study population
A total of 974 lung cancer cases (710 males and 264 females) and 1005 control subjects (698 males and 307 females) were included. The differences of gender and age had no statistical significance (P = 0.101 and 0.772, respectively); 69.8% of cases and 50.0% of controls had a history of smoking (P < 0.01); 34.6% of lung cancer patients had a family history of cancer; and 14.6% of healthy subjects had a family history of cancer (P < 0.01). The characteristics of the study subjects are shown in Table 1.
Frequency distribution of study subjects.
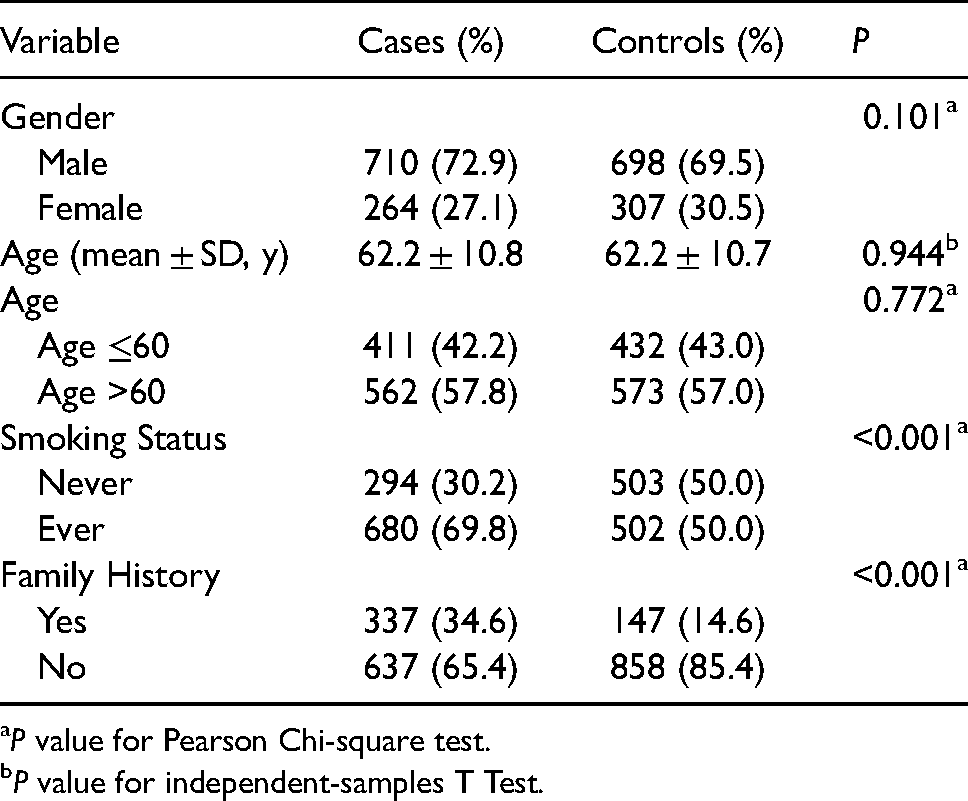
P value for Pearson Chi-square test.
P value for independent-samples T Test.
G allele of rs1695 associated with lung cancer
The genotyping call rate was 99.9% (1977/1979) and the distribution was in accordance with the HWE between cases and controls (P > 0.05). The frequencies of the AA, GA, and GG genotypes of rs1695 were 68.4%, 28.7%, 2.9% in cases and 64.8%, 30.8%, 4.2% in controls, respectively. As listed in Table 2, a significant relation was found between rs1695 and the occurrence of lung cancer. After adjusting for age, gender, smoking status, and family history by using unconditional logistic regression analysis, it appears that the rs1695 G allele decreases the risk of lung cancer (OR = 0.811, 95% CI 0.684–0.961, P = 0.016). Moreover, when the AA genotype was used as a reference, the GA + GG genotype decreased lung cancer susceptibility to 1.238-fold (OR = 0.808, 95% CI 0.663–0.985, P = 0.035) and GG genotype alone for 1.691-fold (OR = 0.591, 95% CI 0.347–0.988, P = 0.048). These results indicate that GG or GA might be a protect genotype for lung cancer.
Relation between GSP1 rs1695 and risk of lung cancer.
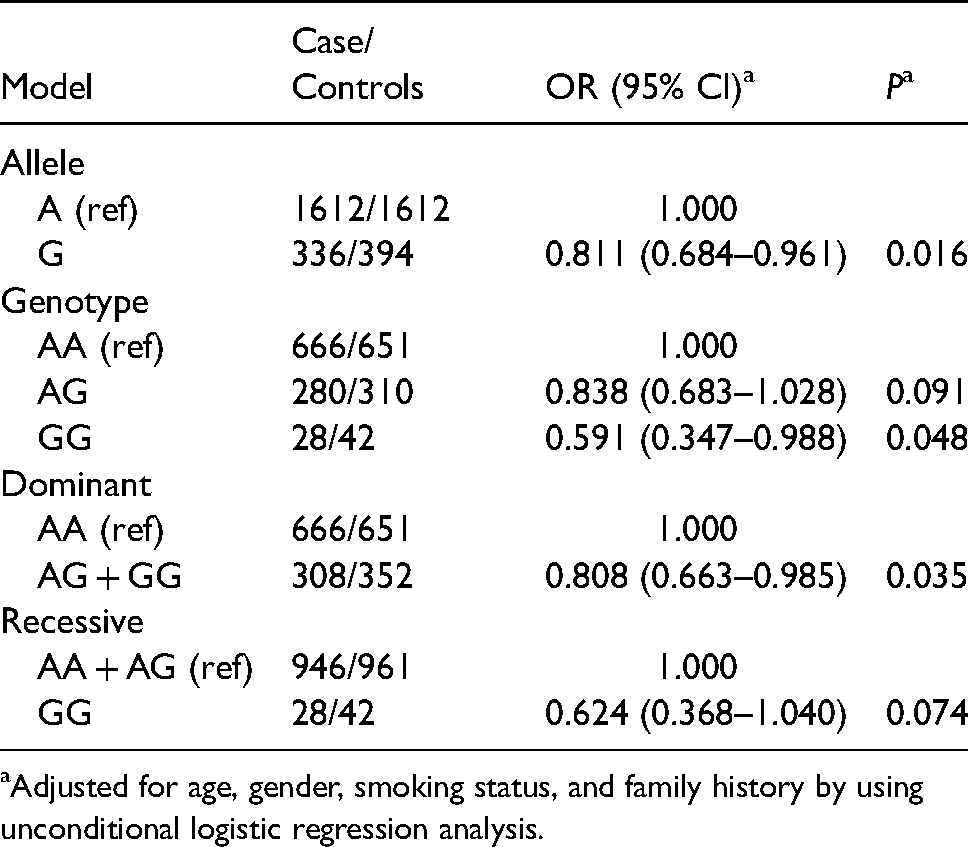
Adjusted for age, gender, smoking status, and family history by using unconditional logistic regression analysis.
Stratified analysis of relations between rs1965 genotypes and risk of lung cancer
We then performed a stratified analysis for rs1695 by epidemiological characteristics to explore the relation of rs1695 polymorphisms, gene-environment interaction, and the risk of lung cancer. As shown in Table 3, after adjustment for age, gender, smoking status, and family history, risk of lung cancer in the G allele carriers was significantly decreased in males (OR = 0.784, 95% CI 0.639–0.962, P = 0.020) and people with no cancer in their family history (OR = 0.782, 95% CI 0.643–0.950, P = 0.014) compared with the A allele carriers. Regarding smoking, significant correlation between decreased the risk of lung cancer and G allele were found in ever smokers (OR = 0.732, 95% CI 0.591–0.906, P = 0.004). In addition, when considering the histological type of tumor, lung adenocarcinoma was reduced in G allele carriers after adjustment (OR = 0.798, 95% CI 0.641–0.990, P = 0.042).
Stratified analysis of relations between rs1695 alleles and risk of lung cancer.
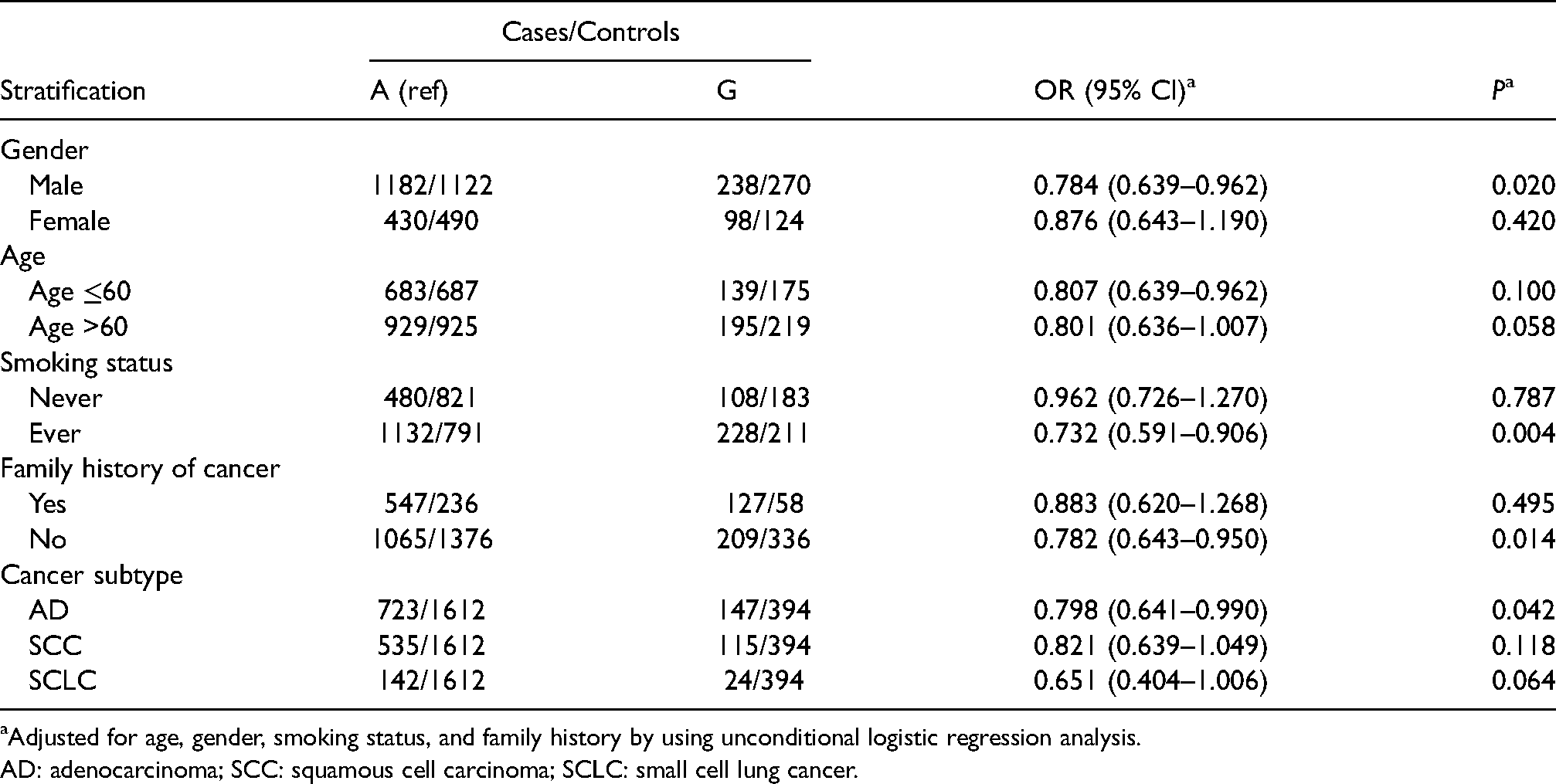
Adjusted for age, gender, smoking status, and family history by using unconditional logistic regression analysis.
AD: adenocarcinoma; SCC: squamous cell carcinoma; SCLC: small cell lung cancer.
Logistic regression under the genotype recessive and dominant models showed similar outcomes. In the genotype model after adjustment, when compared with AA carriers, GG carriers have reduced the risk of lung cancer in males (OR = 0.463, 95% CI 0.235–0.884, P = 0.022) and people with no family history of cancer (OR = 0.490, 95% CI 0.255–0.900, P = 0.026), GA carriers have reduced the risk of lung cancer in ever smokers (OR = 0.722, 95% CI 0.557–0.935, P = 0.013) were detected (Table 4). Similarly, significant risk decrease was found in the recessive model, GG versus GA + AA in males (OR = 0.488, 95% CI 0.249–0.928, P = 0.032) and people with no family history of cancer (OR = 0.518, 95% CI 0.270–0.946, P = 0.039) (Table 5). In the dominant model, GA + GG versus AA decreased the risk of lung cancer in ever smokers (OR = 0.703, 95% CI 0.547–0.902, P = 0.005) and people with no family history of cancer (OR = 0.794, 95% CI 0.633–0.994, P = 0.044) (Supplementary Table 1).
Stratified analysis of relations between rs1695 genotypes and risk of lung cancer.
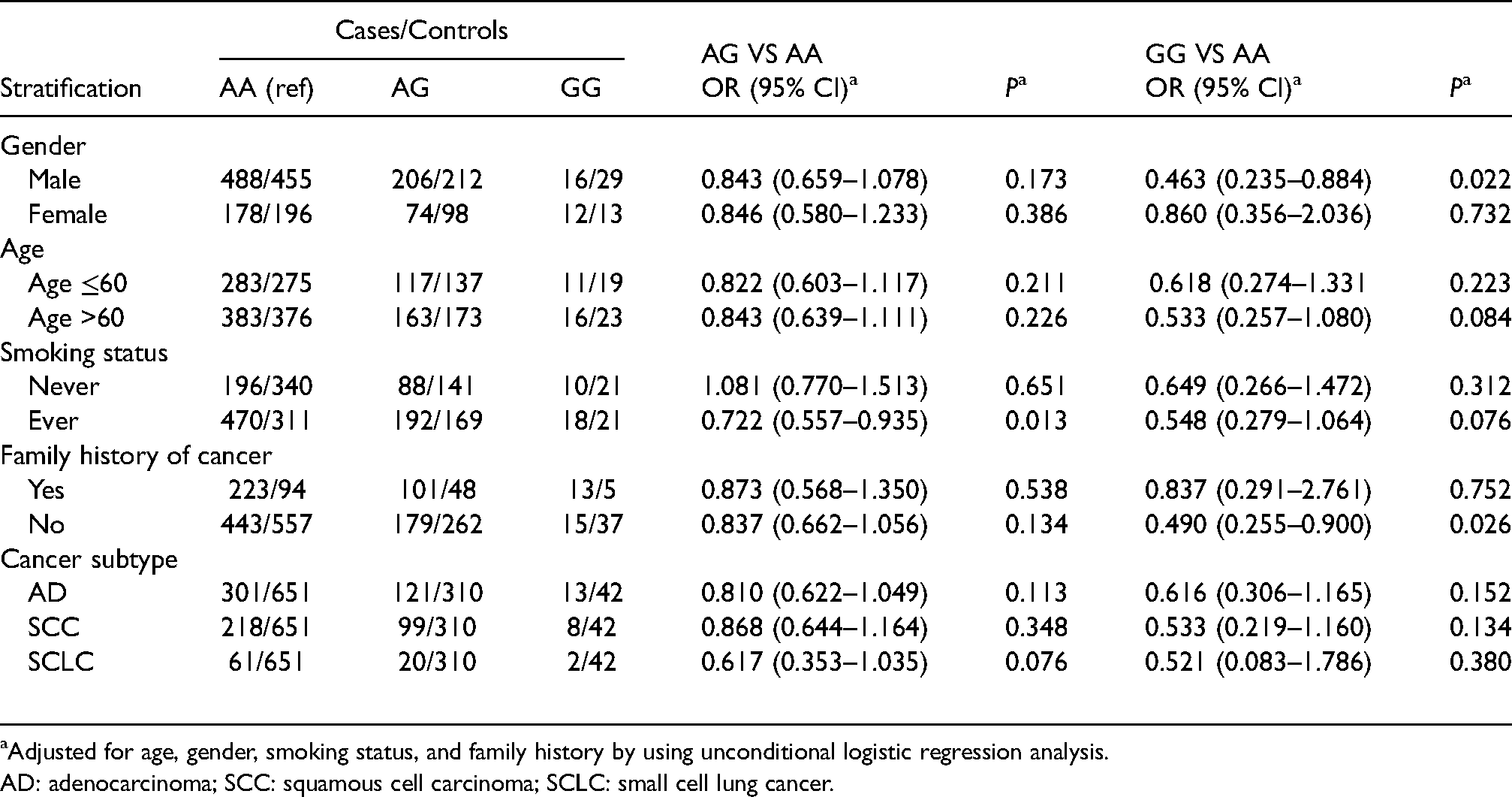
Adjusted for age, gender, smoking status, and family history by using unconditional logistic regression analysis.
AD: adenocarcinoma; SCC: squamous cell carcinoma; SCLC: small cell lung cancer.
Stratified analysis of relations between rs1695 in recessive model and risk of lung cancer.
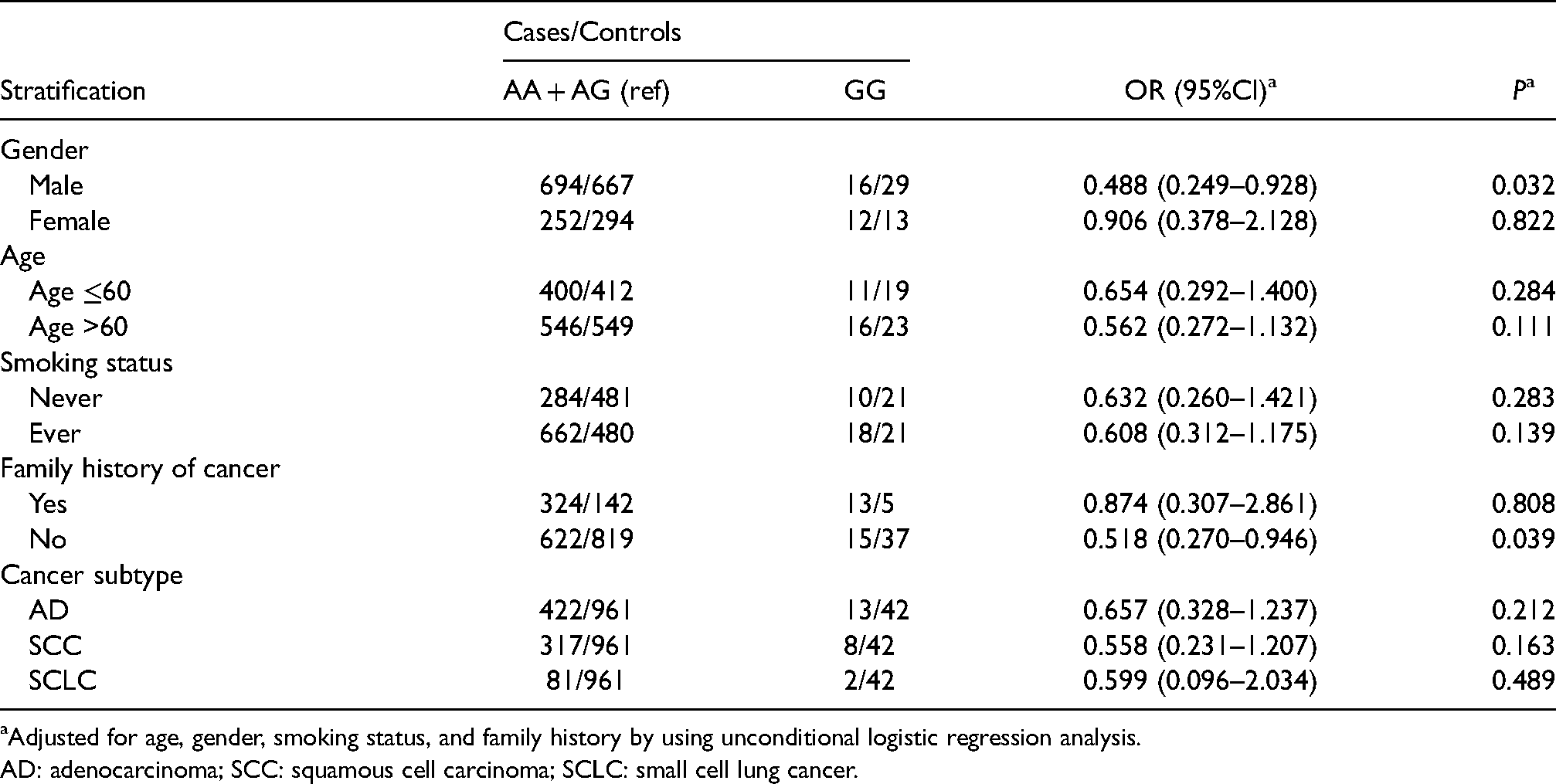
Adjusted for age, gender, smoking status, and family history by using unconditional logistic regression analysis.
AD: adenocarcinoma; SCC: squamous cell carcinoma; SCLC: small cell lung cancer.
Discussion
Our study showed that the GSTP1 rs1695 A > G polymorphism was related to a reduced risk of lung cancer in the Chinese population. Additionally, stratified analysis showed that the risk of lung cancer in G allele carriers decreased in males and in patients without family history, or with lung adenocarcinoma, and especially in smokers (P = 0.0042).
GSTs are a superfamily of enzymes that catalyze the detoxification of electrophilic compounds and therefore play a vital role in the protection of cells against oxidative stress and toxic foreign chemicals. 27 GSTP1 has the most abundant GST subtype in the lung and is involved in the metabolism of inhaled carcinogenic compounds, including benzo[a] pyrene, a tobacco carcinogen.13,28 Evidence proves that GSTP1 plays an important role in lung carcinogen metabolism and DNA damage.19,29,30 Previous studies have shown that the missense SNP rs1695 (GSTP1 Ile105Val) of the GSTP1 gene can affect the enzymatic activity of GSTP1.31–34 Since the first study reported by Ryberg et al., 17 many genetic studies have investigated the relationship between the GSTP1 rs1695 A > G polymorphism and lung cancer. The results in different races and geographic locations are inconsistent, which may be due to the fact that most studies are local studies with small sample sizes.18–26 Recently, a large-scale prospective study of a homogenous white population from 66 races showed that the GSTP1 Ile105Val (rs1695) genotype is associated with improved lung function and can prevent lung cancer and tobacco-related cancers, 19 which is consistent with the conclusion for the Caucasian population in the Cote et al. study. 20 We also assessed the relationship between the GSTP1 Ile105Val gene polymorphism and lung cancer susceptibility through our case-control study of 1979 individuals from the Chinese population, and showed that the GSTP1 rs1695 A > G polymorphism is associated with a reduced risk of lung cancer, especially among smokers.
Smoking is the main risk factor for lung cancer. In smokers, many previous studies have investigated the relationship between the GSTP1 rs1695 A > G polymorphism and lung cancer risk. Miller et al. revealed that the GSTP1 GG genotype enhances the relation between environmental tobacco smoke exposure and the risk of lung cancer. 35 Furthermore, Pliarchopoulou et al. also found the GSTP1 homozygous variant was significantly related to the increased the risk of lung cancer in a Greek smoking population. 36 However, Nørskov et al. yielded contradictory results. 19 The reason may be due to GST isoforms differing in enzyme activity and specificity, and their catalytic efficiencies depend on substrate stereochemistry. 32 In this study, we observed that the G allele of rs1695 is associated with the reduced risk of lung cancer and tobacco-related cancers in the Chinese population, which is consistent with the conclusion of the HapMap-based study on the Chinese population. 37 The explanation that GSTP1 Ile105Val (rs1695) reduces the risk of lung cancer has been shown to confer higher activity on the metabolites of polycyclic aromatic hydrocarbons (PAHs) in tobacco smoke.32,33 It is evident that PAHs can induce lung tumors. 38 In conjugation reactions in vitro, previous studies revealed that the 105Val isoform showed higher catalytic efficiency to a number of bay-region diol epoxides of PAHs when compared to the 105Ile isoform.34,39–41
The limitation of this study is that the relatively small sample size hinders its detection capabilities. The GST family contains isoenzymes, and we should also study the polymorphisms of other genes, pay attention to the interaction of other risk factors, and perform more comprehensive evaluations of the relationship between GSTP1 polymorphisms and lung cancer risk.
Conclusion
This study has shown that the presence of functional polymorphisms in GSTP1 modulate the relation to the risk of lung cancer in the Chinese population. This study is an initial step to identify environmental and genetic factors that may contribute to the high prevalence of lung cancer. Given the limited sample size, further research is warranted to confirm these observed relations.
Supplemental Material
sj-docx-1-jbm-10.1177_17246008211039236 - Supplemental material for The relevance analysis of GSTP1 rs1695 and lung cancer in the Chinese Han population
Supplemental material, sj-docx-1-jbm-10.1177_17246008211039236 for The relevance analysis of GSTP1 rs1695 and lung cancer in the Chinese Han population by Jiang Xiao, Yulu Wang, Zhimin Wang, Yao Zhang, Yutao Li, Chang Xu, Man Xiao, Haijian Wang, Shicheng Guo, Li Jin, Jiucun Wang, Yang Bao, Yan Shang and Junjie Wu in The International Journal of Biological Markers
Footnotes
Acknowledgments
We are very grateful for all the volunteers involved in this study and The First Affiliated Hospital of Naval Medical University. We thank Dr Zhenzhong Zhao (Fudan University, Shanghai, China) and Wei Feng (Changzheng Hospital, Naval Military Medical University, Shanghai, China) for their assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contributions
Jiang Xiao, Yulu Wang, and Zhimin Wang contributed equally to this work and should be regarded as co-first authors.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sponsored by National Natural Science Foundation of China (81372236, 82170033), Natural Science Foundation of Shanghai (21ZR1479200), Shanghai Postdoctoral Science Foundation (12R21411500), Shanghai Changhai Hospital Scientific Research Fund (2019SLZ002, 2019YXK018).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
