Abstract
Background:
The Glasgow Prognostic Score and circulating cytokine levels are related to the prognosis of colorectal cancer and the severity of chronic inflammation. The association between the Glasgow Prognostic Score and circulating cytokines in colorectal cancer remains unclear.
Methods:
The levels of 10 circulating cytokines (TNF-α, TGF-β, IFN-γ, IL-1β, IL-4, IL-6, IL-10, IL-12, IL-13, and IL-23) were measured in 128 patients with colorectal cancer. The relationship between the Glasgow Prognostic Score, clinicopathologic variables, and cytokine levels was assessed by univariate and multivariate logistic regression analyses. The correlation among cytokines was also examined.
Results:
Patients with advanced stage colorectal cancer had lower levels of albumin (P = 0.003), higher levels of C-reactive protein (CRP; P < 0.001), carcinoembryonic antigen (CEA; P < 0.001), interferon (IFN)-γ (P < 0.001), and interleukin (IL)-10 (P = 0.006), and shorter survival outcomes (P < 0.001). Patients with a high Glasgow Prognostic Score (1 or 2) had lower 5-year progression-free survival and poor overall survival (log-rank P < 0.001). A high Glasgow Prognostic Score was significantly correlated with abnormal CEA levels (CEA > 5 ng/mL, P = 0.033), and higher levels of tumor necrosis factor (TNF)-α (TNF-α ⩾ 53.9 pg/mL, P = 0.035) and IL-10 (IL-10 ⩾ 75.95 pg/mL, P = 0.008). TNF-α, IFN-γ, IL-1β, IL-4, IL-6, IL-10, IL-13, and IL-23 were significantly correlated with each other (all P < 0.05). Only IL-10 was correlated with abnormal CEA levels (P < 0.001).
Conclusion:
The Glasgow Prognostic Score and level of circulating cytokines have an intergroup correlation, and there is a close association among cytokines in colorectal cancer.
Keywords
Introduction
Colorectal cancer (CRC) is the third most common cancer and the second most common cause of cancer death worldwide. In 2018, approximately 1.8 million people were diagnosed with CRC, with 0.86 million deaths. 1 Only 2%–5% of patients with CRC are diagnosed with inherited syndromes, such as familial adenomatous polyposis, hereditary non-polyposis CRC, and hamartomatous polyposis syndrome, 2 whereas most CRC develops from environmental and non-hereditary etiologies. Both inherited and non-inherited CRC share a common pathogenesis of cancer development: chronic inflammation. 3
During the inflammatory response, mediators such as cytokines, reactive oxygen species, and reactive nitrogen species may lead to gene mutation, affect nucleic acid repair mechanisms, block apoptosis, and alter the growth of cancer cells, all of which can facilitate cancer cell proliferation and spreading.3,4 CRC is considered the best example to delineate the relationship between chronic inflammation and cancer development. Patients with inflammatory bowel disease have an increased risk of CRC. 5 In contrast, the anti-inflammatory approaches, including the use of non-steroidal anti-inflammatory drugs, may reduce the risk of CRC. 6
The Glasgow Prognostic Score (GPS), an inflammation-based model that combines the levels of serum C-reactive protein (CRP) and albumin, reflects the systematic inflammatory and nutritional status of patients with various types of cancer. 7 The pretreatment GPS is associated with prognostic outcomes and can be used as a biomarker for cancer management in patients with CRC.4,8 A meta-analysis assessing the relationship between the GPS and survival outcomes of 9839 patients with CRC reported that an elevated GPS was associated with poor overall survival and cancer-specific survival. 8 To further clarify, CRP produced by hepatocytes is a biomarker for the inflammatory response and can be applied to predict the incidence and survival of patients with CRC.9,10 Likewise, the serum albumin level represents nutritional status, acts as an acute-phase protein that is affected by inflammation stimuli, 11 and has a positive correlation with the long-term survival of patients with CRC. 12
In addition to acute-phase proteins such as CRP and albumin, the other hallmark of chronic inflammation in cancer is an increased production of circulating cytokines. 4 Recently, accumulating evidence has shown that circulating cytokines act through a range of inflammatory pathways that potentially impact CRC pathogenesis. Indeed, some of these pathways have been identified as independent prognostic factors in CRC, including interleukin (IL)-6, tumor necrosis factor-alpha (TNF-α), and IL-1β.4,13 A meta-analysis that enrolled 1622 CRC patients who underwent surgery showed that a high expression of transforming growth factor-beta (TGF-β) had a favorable outcome on overall survival. 14 In addition, CRC patients with worse prognosis tend to have higher levels of circulating IL-10 than those with a better prognosis. 15 Moreover, IL-23, which contributes to the progression of chronic inflammation by promoting the maturation and maintenance of T helper 17 cells, 16 is gradually elevated with tumor stage progression in patients with CRC. 17 Furthermore, since multiple cytokines have been shown to actively participate in promoting the growth and metastasis of CRC, cytokine profiling has been applied to classify CRC patients into good and poor prognostic subgroups.18-20 IL-4, IL-12, IL-13, and interferon-gamma (IFN-γ) have been shown to be involved in these cytokine profiles.18-20 Taken together, measurement of circulating cytokines to predict the prognosis of patients with CRC appears to be a reasonable approach in clinical practice.
Although the analysis of acute-phase proteins and multiple cytokines may contribute to prognostic prediction, the relationship between the GPS and circulating cytokines remains unclear. The aim of this study was to investigate the association between GPS and clinicopathologic features and nutritional index in Taiwanese patients with CRC. The following clinicopathologic features were examined: age, sex, tumor node metastasis (TNM) stage, tumor location, histologic differentiation, body mass index (BMI), hemoglobin (Hb), total lymphocyte count (TLC), carcinoembryonic antigen (CEA), 10 circulating cytokines (TNF-α, TGF-β, IFN-γ, IL-1β, IL-4, IL-6, IL-10, IL-12, IL-13, IL-23), 5-year progression-free survival (PFS), and overall survival (OS). In addition, we attempted to decipher the correlations among the 10 circulating cytokines analyzed.
Material and methods
Patients
In this retrospective study, data were collected from 164 patients with CRC who underwent standard treatment at Keelung Branch of Chang Gung Memorial Hospital Taiwan, between January 2009 and December 2011. The patients’ clinical parameters and the results of clinical laboratory tests were obtained from the patients’ medical records, and included age, sex, TNM stage, tumor location, histologic differentiation, BMI, complete and differential blood count, CEA, albumin, CRP, 5-year PFS and OS. Thirty-six patients were excluded due to lack of CRP (32 patients) or albumin (5 patients) data. Finally, 128 patients were included in the analysis. The GPS was defined based on the presence of hypoalbuminemia (< 3.5 g/dL) and elevated CRP (> 10 mg/L) as follows: If both were abnormal, the score was 2; if either was abnormal, the score was 1; if neither was abnormal, the score was 0. 21 Serum cytokines, including TNF-α, TGF-β, IFN-γ, IL-1β, IL-4, IL-6, IL-10, IL-12, IL-13, and IL-23, were collected from patients before treatment. The serum samples were stored at −80°C in pyrogen-free plastic tubes until cytokine analysis by enzyme-linked immunosorbent assay. The serum cytokine concentrations were determined using a DuoSet ELISA Development Kit following the manufacturer’s instructions (R&D Systems, Minneapolis, MN, USA). The final concentrations were determined using a bioluminescencecounter (Packard Instrument Co., Inc., Downers Grove, IL, USA). All blood samples were only thawed once and were assayed in triplicate.
Tumor staging was classified retrospectively according to the 7th edition of the American Joint Committee on Cancer Staging System based on the findings of physical examination, routine laboratory tests, and computed tomography of the chest and abdomen. The diagnoses and treatment goals of all enrolled patients were reviewed and confirmed by the CRC committee at our hospital. The committee members included three colorectal surgeons, three medical oncologists, one general surgeon, one chest surgeon, one radiation oncologist, one radiologist, and one pathologist. The OS and 5-year PFS were determined as the time between the diagnosis date and mortality, and as the time between the date of diagnosis and the first evidence of cancer progression, respectively. The patients were continuously followed up until April 2019 or death, whichever occurred first. The study was approved by the Institutional Review Board (IRB) of Chang Gung Memorial Hospital in Taiwan (IRB number: 202001858B0).
Data analysis
Statistical analyses were performed using the Statistical Package for the Social Sciences version 26 (SPSS Inc., Chicago, IL, USA). The associations between categorical variables were examined using Pearson’s chi-square test. After assessment by the Kolmogorov–Smirnov normality test, the continuous variables were compared using analysis of variance (ANOVA) with Bonferroni adjustments or non-parametric statistics with the Kruskal–Wallis H test where appropriate. The optimal cutoff values were determined for continuous variables according to the values published in previous reports, including old age (> 65 years old), 22 underweight (BMI < 18.5 kg/m2), 23 anemia (Hb < 10 g/dL), 24 malnutrition status (albumin < 3.5 g/dL and TLC < 1,500 cells/mm3),25,26 elevated inflammatory status (CRP >10 mg/L), 27 and abnormal CEA level (CEA > 5 ng/mL), 28 or determined according to the Youden’s index based on receiver operating characteristic (ROC) curve analysis for serum cytokine levels. If the optimal cutoff values of the serum cytokines could not be acquired by ROC curve analysis, the median level of each cytokine was applied for stratification in the study. The associations between survival outcomes and clinicopathological characteristics (age, sex, TNM stage, tumor location, histologic differentiation), GPS, BMI, Hb, TLC, CEA, 10 circulating cytokines studied, and treatment setting were analyzed using Cox proportional hazard models. Forward stepwise selection was used in the univariate and multivariate analyses for different variables. All independent variables significantly associated with 5-year PFS and OS (P ⩽ 0.05) in the univariate analysis were included in the multivariate analysis (Supplementary Table 1). Variance inflation factors were used to test for variables collinearity. The Kaplan–Meier method and log-rank test were used to analyze 5-year PFS and OS. In Table 2, the variables with statistical significance (P < 0.05) in the univariate analysis were entered into a multivariate logistic regression to identify the independent variables associated with GPS in patients with CRC. Cytoscape, an open-source software platform for creating a two-dimensional visualization of the relationships between GPS, CEA, and serum cytokines was used, utilizing a prefuse force-directed algorithm weighted by the statistical significance of the correlations between individual variables. 29 P-values < 0.05 were considered statistically significant.
Results
The clinical characteristics of the 128 patients with CRC are shown in Table 1. The study population comprised 82 (64.1%) males and 46 (35.9%) females, with a mean age of 65.52 years (range, 18–94). Twenty-eight (21.8%) patients had stage I CRC, 34 (26.5%) had stage II, 44 (34.3%) had stage III, and 22 (17.1%) had stage IV. The tumor was located in the colon in 87 cases (67.9%) and in the rectum in 41 cases (32.1%). In all patients, the histology indicated adenocarcinoma. The histologic grade was assessed according to the criteria of the World Health Organization; 40 patients (31.2%) had well-differentiated disease, 79 (61.7%) had moderately differentiated disease, and 9 (7.0%) had poorly differentiated disease. All patients (100%) received standard surgery to remove the primary tumor and regional lymph nodes. Two (7.1%) stage I patients, 21 (61.8%) stage II patients, and 26 (56.1%) stage III patients had adjuvant chemotherapy; 18 (40.9%) stage III rectal cancer patients had adjuvant chemoradiotherapy. For stage IV patients, 13 (59.1%) patients had palliative chemotherapy, 5 (22.7%) had palliative chemoradiotherapy, and 4 (18.2%) had no postoperative treatment.
Clinicopathologic data of 128 CRC patients according to tumor stage.
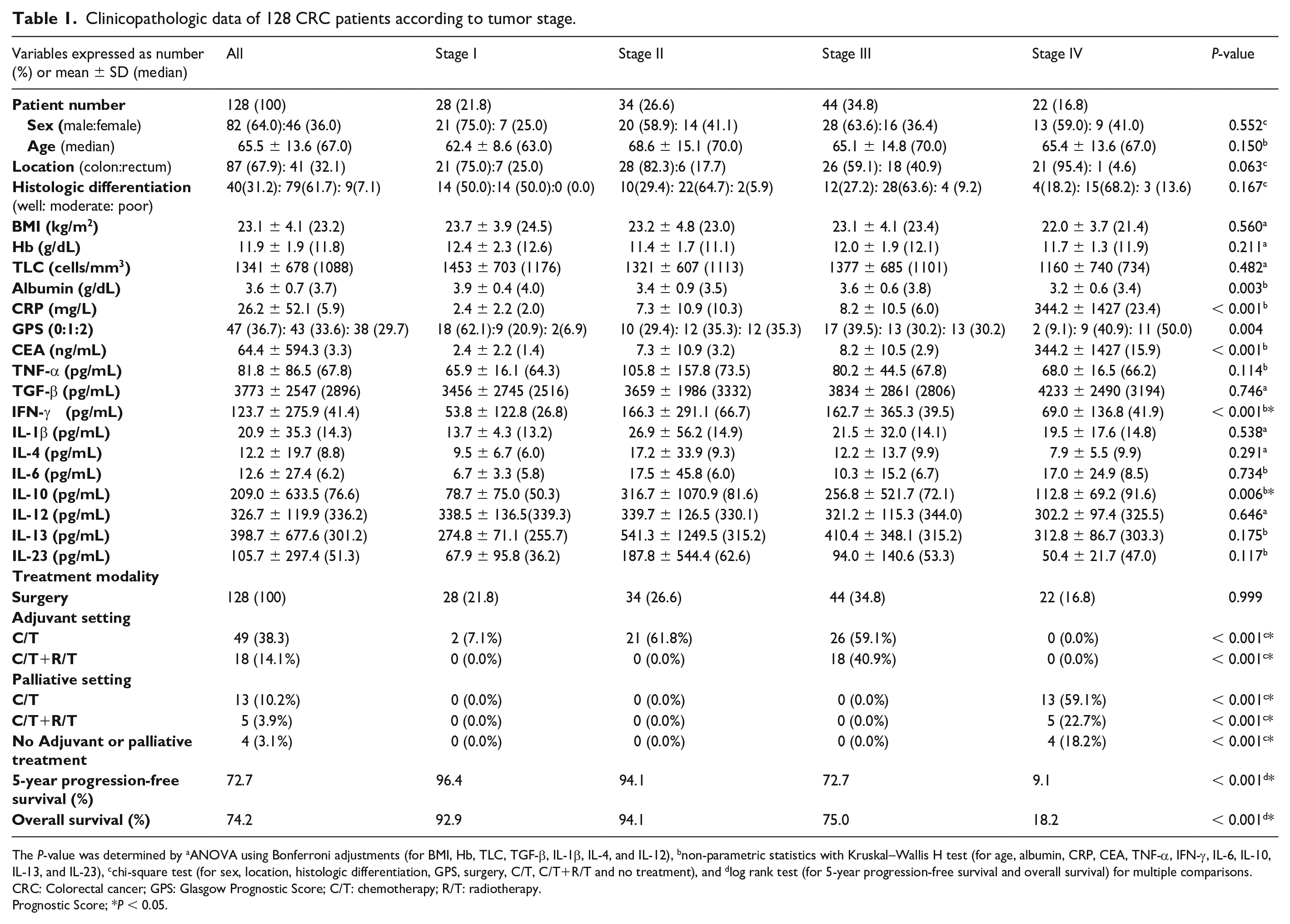
The P-value was determined by aANOVA using Bonferroni adjustments (for BMI, Hb, TLC, TGF-β, IL-1β, IL-4, and IL-12), bnon-parametric statistics with Kruskal–Wallis H test (for age, albumin, CRP, CEA, TNF-α, IFN-γ, IL-6, IL-10, IL-13, and IL-23), cchi-square test (for sex, location, histologic differentiation, GPS, surgery, C/T, C/T+R/T and no treatment), and dlog rank test (for 5-year progression-free survival and overall survival) for multiple comparisons.
CRC: Colorectal cancer; GPS: Glasgow Prognostic Score; C/T: chemotherapy; R/T: radiotherapy.
Prognostic Score; *P < 0.05.
Patients were stratified according to stage; advanced stage was associated with hypoalbuminemia (P = 0.003), higher levels of CRP (P < 0.001), CEA (P < 0.001), IFN-γ (P < 0.001), and IL-10 (P = 0.006), as well as shorter 5-year PFS (P < 0.001) and OS (P < 0.001). There was no statistical difference in sex, age, tumor location, histologic differentiation, BMI, Hb, TLC, TNF-α, TGF-β, IL-1β, IL-4, IL-6, IL-12, IL-13, and IL-23 levels among the various tumor stages (Table 1 and Figure 1).
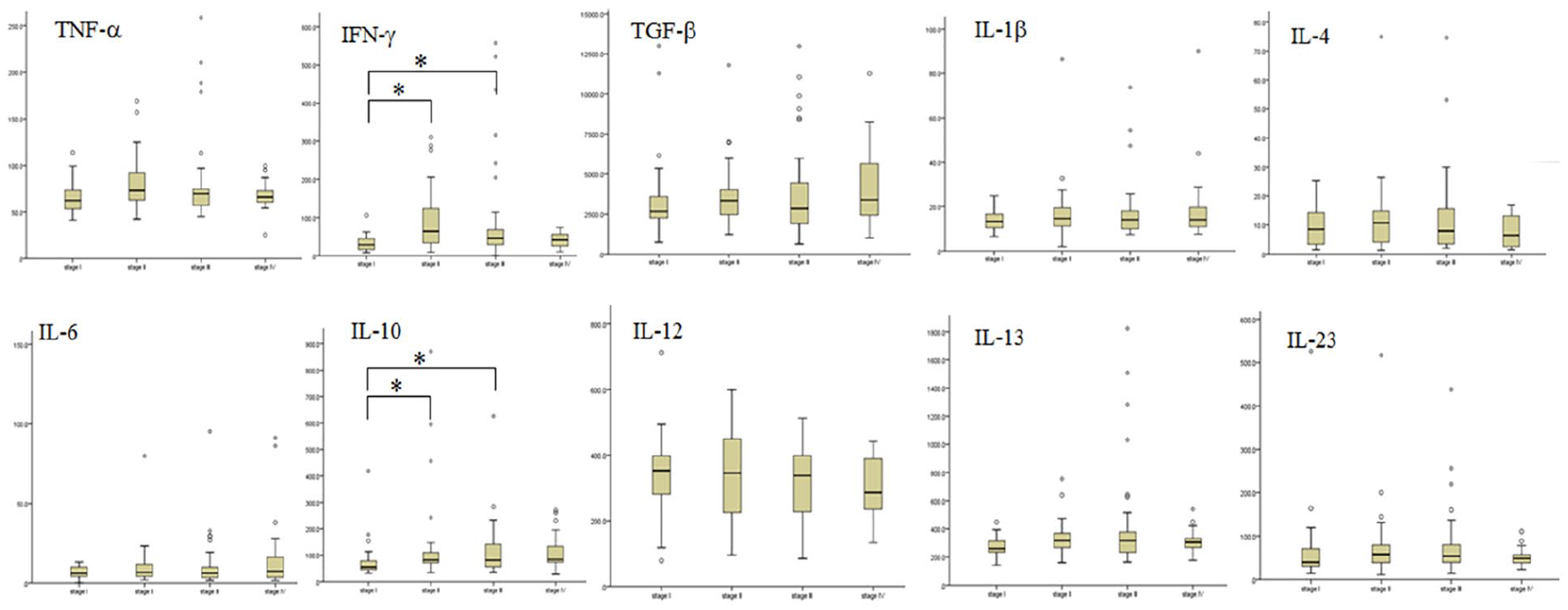
Box plots show the concentrations of 10 cytokines stratified by TNM stage. X-axis, TNM stage; Y-axis, individual cytokine concentration (pg/mL); * denotes P < 0.05, considered significant between two groups.
Patients were next classified into two groups according to GPS: 47 patients with a GPS of 0 (low GPS group; 36.71%) and 81 patients with a GPS of 1 or 2 (high GPS group; 63.29%). The high GPS group had higher levels of IFN-γ (P < 0.05), and IL-10 (P < 0.05) (Figure 2).
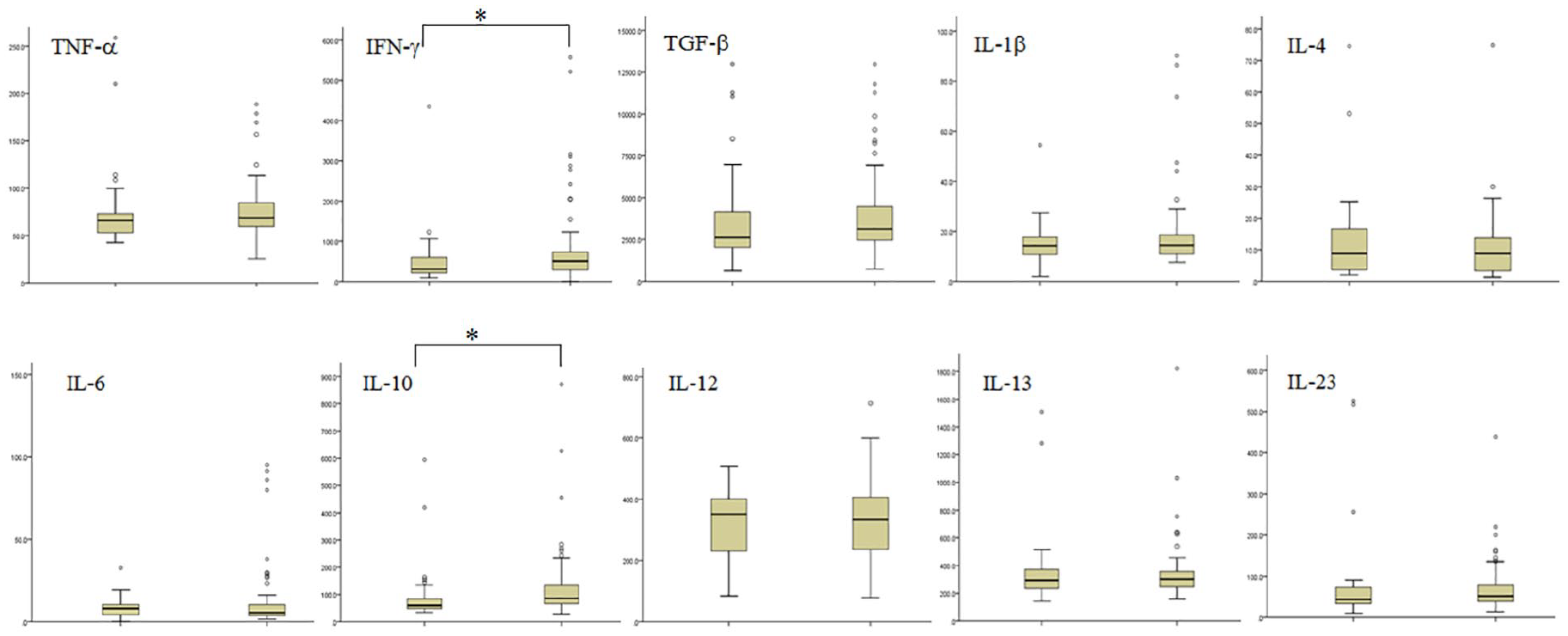
Box plots show the concentrations of ten cytokines stratified by GPS status. X-axis, GPS, 0 or GPS ⩾ 1; Y-axis, individual cytokine concentration (pg/mL); * denotes P < 0.05, considered significant between two groups.
When we investigated the cutoff values for serum cytokines using ROC curve analysis, only three cytokines achieved the optimal cutoff values: TNF-α (53.9 pg/mL; area under curve (AUC): 0.624; P = 0.046), IFN-γ (35 pg/mL; AUC: 0.643; P = 0.007) and IL-10 (75.9 pg/mL; AUC: 0.678; P = 0.001). Consequently, we arbitrarily stratified patients according to the median level as a cut-off value in other cytokines for analysis (TGF-β: median, 2896 pg/mL; IL-1β: 14.3 pg/mL; IL-4: 8.8 pg/mL; IL-6: 6.2 pg/mL; IL-12: 336.2 pg/mL; IL-13: 301.2 pg/mL; and IL-23: 51.3 pg/mL).
To assess the role of GPS in survival outcome, we examined the interactive effect among clinical variables, GPS, and cytokines studied on 5-year PFS and OS rates. On univariate analysis, tumor location, tumor stage, GPS, BMI, CEA, and treatment setting showed significance for 5-year PFS; tumor stage, GPS, BMI, TLC, CEA, TNF-α, and treatment setting showed significance for OS (Supplementary Table 1). On multivariate analysis, tumor location (P = 0.014), tumor stage (P = 0.001), GPS (P = 0.026), BMI (P = 0.002), and treatment setting (P = 0.001) were independent prognostic factors for 5-year PFS; tumor stage (P < 0.001), GPS (P = 0.007), and treatment setting (P = 0.001) were independent prognostic factors for OS. (Supplementary Table 1). Patients with a high GPS (1 or 2) had lower 5-year PFS and poor OS (log-rank P < 0.001) (Supplementary Figure 1).
The results of univariate analysis demonstrated that abnormal CEA levels and high serum levels of TNF-α, IFN-γ, IL-6, and IL-10 had a probable association with high GPS (Table 2). Moreover, we found no association between high and low GPS in terms of sex, age, tumor location, histologic differentiation, BMI, Hb, TLC, TGF-β, IL-1β, IL-4, IL-12, IL-13, and IL-23. The multivariate logistic regression model after adjustment of the covariate effects demonstrated that in addition to CEA > 5 ng/mL, higher circulating TNF-α (⩾ 53.9 pg/mL) and IL-10 levels (⩾ 75.95 pg/mL) were independently correlated with high GPS (Table 2).
Univariate and multivariate analyses of clinicopathologic features associated with a GPS ⩾ 1 in 128 patients with CRC.
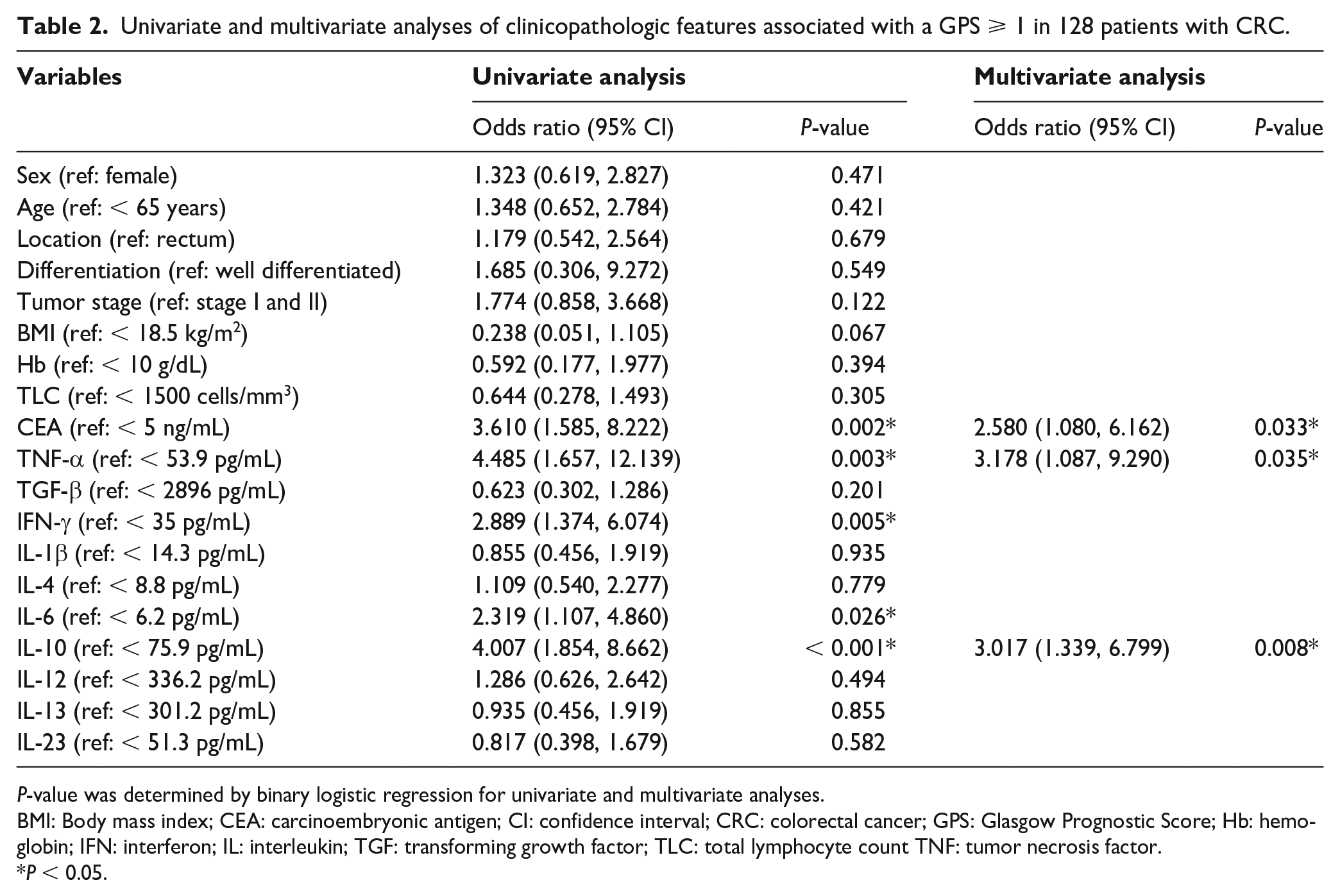
P-value was determined by binary logistic regression for univariate and multivariate analyses.
BMI: Body mass index; CEA: carcinoembryonic antigen; CI: confidence interval; CRC: colorectal cancer; GPS: Glasgow Prognostic Score; Hb: hemoglobin; IFN: interferon; IL: interleukin; TGF: transforming growth factor; TLC: total lymphocyte count TNF: tumor necrosis factor.
P < 0.05.
To facilitate the comprehension of correlation data, we utilized the Cytoscape software platform to create a two-dimensional visualization for a more detailed analysis of the interplay between cytokines, high GPS, and abnormal CEA. Figure 3 shows that multiple serum cytokines, including TNF-α, IFN-γ, IL-1β, IL-4, IL-6, IL-10, IL-13, and IL-23, were significantly correlated with each other. TGF-β was only related to IL-12 but not to other cytokines. Abnormal CEA levels were only correlated with IL-10 levels (P < 0.001).
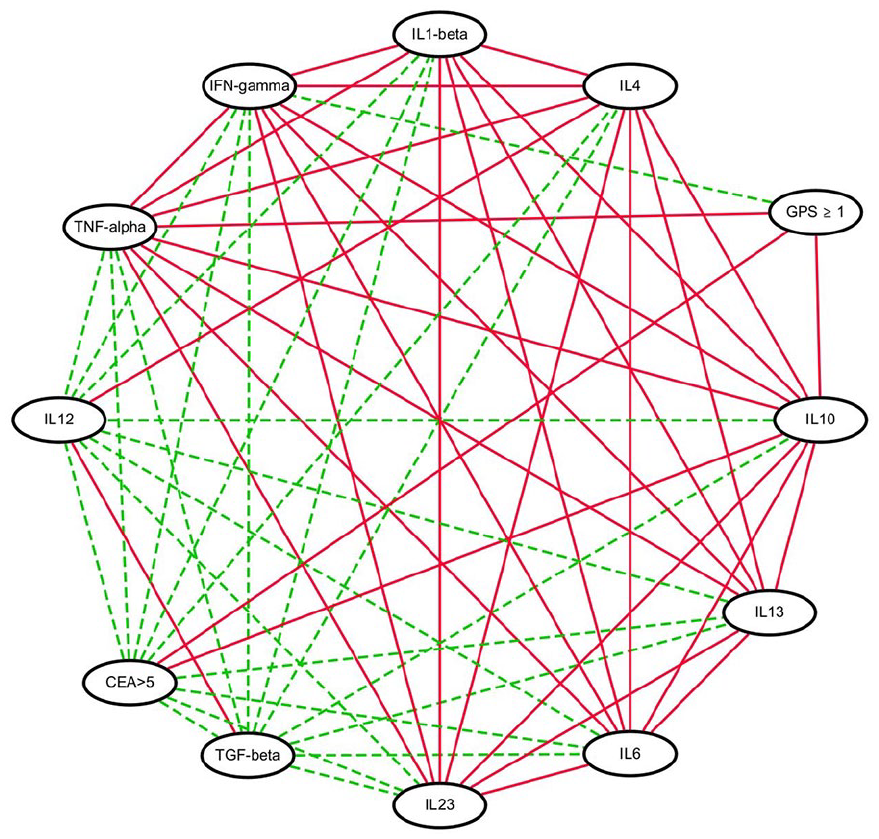
Two-dimension visualization of the interrelationships between serum cytokine levels and Glasgow Prognostic Scores (GPS). Individual variables are represented by nodes, and their associations are represented by edges (connecting lines). Red solid edges indicate significant associations (P < 0.05) between two cytokine variables or between cytokine variables and GPS or CEA. Green dash edges indicate no correlation between two cytokines.
Discussion
The present study demonstrated that abnormal CEA level (> 5 ng/mL), and higher circulating TNF-α (⩾ 53.9 pg/mL) and IL-10 (⩾ 75.95 pg/mL) levels were independently correlated with high GPS after adjustment of the clinicopathologic parameters. In addition, a high GPS (1 or 2) was associated with shorter 5-year PFS and OS. Our finding partially supports the result of a previous study in that the GPS was positively correlated with the CEA level and survival outcomes, 30 but was not associated with age, sex, tumor stage, and tumor differentiation. 31 Our results further showed that at least two circulating cytokines (TNF-α and IL-10) affect the expression of either CRP or serum albumin, GPS parameters, in patients with CRC.
CRP can be regulated by IL-1β, IL-6, and TNF-α during the inflammatory response, and the CRP level is significantly correlated with IL-1β and IL-6 expression. 32 Moreover, in animal models, high CRP concentration can enhance IL-10 synthesis.33,34 Regarding the expression of serum albumin, Brenner et al. 35 revealed that elevated circulating TNF-α levels downregulate albumin gene transcription and decrease albumin production in the liver. Higher IL-10 levels are also reported to be associated with hypoalbuminemia in critically ill patients and those with Hodgkin lymphoma.36,37 In clinical settings, cancer cachexia, which is clinically manifested by fatigue, anorexia, weight loss, hypoalbuminemia, and decreased muscle mass/strength, is a practical scenario that reflects the relationship between circulating cytokines and the GPS. It is clinically manifested by fatigue, anorexia, weight loss, hypoalbuminemia, and decreased muscle mass/strength in cancer patients. 38 Cachexia is triggered and aggravated by systemic inflammatory responses via multiple cytokine pathways, including TNF-α, IL-6, IL-1, and IFN-γ. 39 Cachexia is usually found in patients with CRC both at diagnosis and during treatment.40,41 Furthermore, the extent of increased CRP and lower serum albumin levels also reveals the severity of cachexia in a variety of cancers, including CRC. 38 Taken together, these studies, including the current one, demonstrate that the GPS is indeed correlated with the expression of circulating cytokines in patients with CRC.
The other salient feature of the present study was the intimate and intricate network of circulating cytokines, which is related to high GPS and abnormal
CEA (Figure 3). A few similar studies have been reported previously.20,42-44 Chen et al. 20 retrospectively analyzed 1180 patients with CRC using the neutrophil-to-lymphocyte ratio (NLR) derived from the routine complete blood cell count with the differential as an indicator to evaluate the systemic inflammation response. They found that in addition to a reliable prognostic factor of CRC, a high NLR is correlated with at least 20 cytokines clustered into four categories: inflammatory cytokines (IL-6), angiogenic cytokines (IL-8), epidermal growth factor ligands, and vascular epidermal growth factor. 20 Moreover, a prospective study conducted in Finland examined the expression of circulating matrix metalloproteinase-8 (MMP-8) released from neutrophils under inflammatory stimuli in 271 patients with CRC. They found that MMP-8 is associated with modified GPS (mGPS) and NLR, and is a good indicator of systemic inflammation status and survival outcomes of patients with CRC. The MMP-8 level is closely associated with several circulating cytokines including IL-1 receptor antagonist (IL-1RA), IL-6, IL-7, and IL-8. 43 Nonetheless, Sharma et al. 45 analyzed 19 circulating cytokines in 52 patients with stage IV CRC and found no association between GPS and all studied cytokines, except that a GPS of 2 was correlated with elevated IL-6 levels compared to a GPS of 0 or 1. Park et al. 28 also showed a weak association between a panel of circulating cytokines (TNF-α, IL-1β, IL-6, IL-8, IL-9, and IL-10) and systemic inflammation markers such as the mGPS and the NLR. Moreover, Chang et al. 46 measured three cytokines (TNF-α, IL-1β, and IL-6) and incorporated all three into the cytokine score, which predicted increased tumor recurrence-free survival of patients with CRP < 5 mg/L than those with CRP > 5 mg/L. Although these reports imply that the relationship between cytokine and inflammatory status in patients with CRC is negligible or limited if present, the interpretation and application of the discrepant results should made with caution depending on the study design (retrospective, prospective, or meta-analysis), enrollment patient status (cancer stage and treatment modality), panels, and tools used for cytokine measurements (multiplex bead immunoassay, real-time polymerase chain reaction, or enzyme-linked immunosorbent assay).
Some intriguing observations and potential study limitations merit further discussion. The underlying mechanism by which IL-10 outperforms other cytokines in association with abnormal CEA levels remains unclear. Some studies have proposed that, in certain circumstances, CEA may preferentially stimulate specific cytokine production.47,48 Indeed, Jessup et al. 47 observed that CEA has a tendency to stimulate secretion of IL-10, not IL6, and consequently promotes the short-term survival of weakly metastatic human CRC in an in vitro ischemic-reoxygenated liver model. Furthermore, Roselli et al. 48 examined the blood levels of TNF-α, IL-1β, IL-6, and CEA in 194 patients with CRC and found that TNF-α levels were positively correlated with the presence of distant metastasis and CEA levels, but not IL-1β or IL-6; these findings suggest that CEA plays a role in the induction of TNF-α production in CRC. Additionally, the levels of TNF-α, IFN-γ, and IL-10 were higher in patients with stage II and III tumors than those with stage IV in this study. However, statistical analysis found that—except for IFN-γ and IL-10—the levels of the other eight cytokines had no significant difference among varied stages (Figure 1). The levels of both IFN-γ and IL-10 had statistical difference between stage I, stage II, and stage III. We could not detect the difference between stage II, III, and IV, even though the levels of TNF-α, IFN-γ, and IL-10 were seemingly higher in stage II and III. Further, our institution actively promoted occult blood stool tests for the early screening of CRC from 2007. All 22 (16.8%) stage IV patients were diagnosed with no obvious clinical symptoms, such as cachexia, body weight loss, or decreased BMI, so their paraneoplastic cytokine levels or related immune reactions may not be worse than those in non-metastatic CRC patients. Although most studies favor that the serum IL-6 level is increased with CRC stage, 49 the correlation between serum levels of other cytokines and tumor stages remains equivocal.49,50 Taken together, the reasons why the levels of TNF-α, IFN-γ, and IL-10 were seemingly higher in patients with stage II and III CRC in our study could be attributed to the better medical status of stage IV patients. Another possibility, from our own perspective, is that not all cytokine levels are closely related to tumor stages. Also, we did not include RAS/RAF status, two of the major genes related to survival outcomes in CRC, into survival analysis since our data were collected and both RAS and RAF mutational tests were not standard requirements for the pathologic diagnosis of CRC in our institution during 2009–2011. Although Chen et al. 20 observed that NLR is associated with the prognosis in metastatic CRC independent of RAS/RAF status, the interactive effect between GPS and RAS/RAF status on survival outcomes of patients with CRC needs further investigation. Lastly, the retrospective design is the major limitation of this study given that—by its nature—excludes patients with incomplete data, which has the potential to affect the data analysis. As we enrolled all of the study participants from a single medical institution, had a small sample size, and examined the relationship between GPS and 10 cytokines only with no normal healthy controls, the extrapolation of the current results to patients with CRC who have been assessed using other cytokine and inflammatory indicator panels remains a concern. To understand the entire landscape of the interaction between circulating mediators and inflammatory status, a prospective study with more patients and healthy people, which includes the assessment of a comprehensive panel of cytokines, growth factors, and chemokines involved in CRC pathogenesis is warranted.
Conclusions
Inflammatory status plays an important role in the development, severity, and prognosis of CRC. GPS, an inflammation-based score, is significantly related to the 5-year PFS, OS, abnormal CEA level, and higher levels of TNF-α and IL-10 levels in Taiwanese patients with CRC. There is a close association between circulating cytokines, some of which are related to high GPS and abnormal CEA levels.
Supplemental Material
sj-pdf-1-jbm-10.1177_17246008211022769 – Supplemental material for Correlation Between the Glasgow Prognostic Score and the Serum Cytokine Profile in Taiwanese Patients with Colorectal Cancer
Supplemental material, sj-pdf-1-jbm-10.1177_17246008211022769 for Correlation Between the Glasgow Prognostic Score and the Serum Cytokine Profile in Taiwanese Patients with Colorectal Cancer by Yen-Lin Yu, Chung-Wei Fan, Wen-Ko Tseng, Pei-Hung Chang, Hsuan-Chih Kuo, Yi-Ping Pan and Kun-Yun Yeh in The International Journal of Biological Markers
Supplemental Material
sj-tif-1-jbm-10.1177_17246008211022769 – Supplemental material for Correlation Between the Glasgow Prognostic Score and the Serum Cytokine Profile in Taiwanese Patients with Colorectal Cancer
Supplemental material, sj-tif-1-jbm-10.1177_17246008211022769 for Correlation Between the Glasgow Prognostic Score and the Serum Cytokine Profile in Taiwanese Patients with Colorectal Cancer by Yen-Lin Yu, Chung-Wei Fan, Wen-Ko Tseng, Pei-Hung Chang, Hsuan-Chih Kuo, Yi-Ping Pan and Kun-Yun Yeh in The International Journal of Biological Markers
Footnotes
Authors’ contributions
Conceptualization: Kun-YunYeh; data curation: Yen-Lin Yu, Chung-Wei Fan, Wen-Ko Tseng, Pei-Hung Chang, Hsuan-Chih Kuo, and Yi-Ping Pan; formal analysis: Yen-Lin Yu, Pei-Hung Chang, and Yi-Ping Pan; investigation: Chung-Wei Fan, Wen-Ko Tseng, Pei-Hung Chang, Hsuan-Chih Kuo, and Yi-Ping Pan; methodology: Kun-YunYeh; validation: Kun-YunYeh; original draft: Kun-YunYeh and Yen-Lin Yu; writing, review, and editing: Kun-YunYeh and Yen-Lin Yu.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by grants (CMRPG2C0161, CMRPG290111, CMRPG2A0351 and CMRPG2A0352) from the Chung Gang Memorial Hospital, Keelung, Taiwan.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
