Abstract
Objectives:
This study assessed the level of appropriateness of tumor marker requests in a teaching hospital and estimated the financial cost associated with inappropriate use.
Methods:
A retrospective review of patients’ electronic records was conducted over a 3-year period (2015–2017) for tumor marker requests, including carcinoembryonic antigen, alpha-fetoprotein, cancer antigen (CA)15-3, CA125, CA19-9, and total and free prostate-specific antigen (PSA and fPSA), along with the associated clinical data that motivated the requests. Inappropriate use was defined as tumor marker requests without any relevant clinical picture. Costs due to inappropriate tumor marker requests were estimated based on the unit costs applied in the institution.
Results:
A total of 7128 patients had at least one tumor marker request between 2015 and 2017. The clinical picture that motivated tumor marker requests was absent in 71.5%, while 12.9% of the requests were associated with a malignancy. The most frequent prescribing pattern was total prostate-specific antigen alone (2128; 29.9%), followed by alpha-fetoprotein alone (1185; 16.6%), and carcinoembryonic antigen alone (506; 7.1%). Year-over-year analysis revealed an increasing tendency in requesting carcinoembryonic antigen and CA15-3. The rate of inappropriate use varied by tumor marker and ranged between 56.4% for fPSA and 86.8% for total prostate-specific antigen. The overall costs due to inappropriate tumor marker requests were estimated at $2,785,493 over the 3 years, representing an average of $0.93 million per year.
Conclusion:
Inappropriate use of tumor marker requests is a major issue regarding its high prevalence and the considerable associated costs. The role of laboratories in the management of tumor marker requests should be emphasized.
Introduction
A remarkable advancement in cancer evaluation and management is owed to the use of tumor markers (TMs), notably in the monitoring of tumor progression and early detection of recurrences and/or metastases.1, 2 In some cases, such as in breast cancer, TMs are used to predict a tumor’s response to treatment, enabling appropriate choice of the treatment, and preventing unnecessary costly interventions and their eventual side effects. 2 Additionally, TMs may be used for prognostic purposes—such as in seminomatous and nonseminomatous tumors where serum levels of alpha-fetoprotein (AFP), human chorionic gonadotrophin (hCG), and lactate dehydrogenase (LDH), which are reliable indicators of the tumor stage—besides diagnosis, prognosis, and monitoring. 3
On the other hand, lack of sensitivity and specificity of the majority of TMs hinder their use in the screening and diagnosis of many tumors. 4 Furthermore, several TMs have been associated with noncancerous disorders, notably physiological processes that modify the catabolism, leading to false positive results.5-10 Consequently, the applications and usage of TMs have been variably recommended by the international organizations concerned with cancer research. 11 For example, the American Society of Clinical Oncology (ASCO) recommends the use of CEA for breast cancer, prior to therapeutic decisions, in conjunction with other clinical and imaging data, while it rejected its usefulness in screening, diagnosis, staging, or routine surveillance of patients after primary treatment. 12 On the other hand, the European Group on TMs (EGTM) has a more generalized approach for TM usage in a range of cancers.13, 14
However, physicians’ adherence to these guidelines is questionable, and many reports show concerning rates of the misuse of TMs, which are frequently used as a screening tool for patients with suspected malignancy or an undetermined clinical picture.15-18 This exposes the patients to mismanagement, either by under- or over-treatment, in addition to the stress resulting from false positive results. At the public health level, inappropriate use of TMs impedes the decision-making process, which negatively affects care quality and patient outcomes, as well as the cost-effectiveness of related healthcare interventions. 17
The present study assessed the prevalence, and estimated the cost, of inappropriate TM requests in a teaching tertiary care hospital. The aim was to raise awareness among physicians and oncologists about the necessity to comply with evidence-based guidelines and clinical sense in requesting TMs, to avoid excessive costs associated with inappropriate use. The following objectives were explored:
To map all requests of TMs, including CEA, AFP, CA199, CA125, and PSA that were processed over 3 years at the Diagnostic Immunology Department of King Abdul-Aziz University Hospital (KAUH), and to describe the most common patterns of TM requests among single and combined requests;
To estimate the rate of inappropriate use by assessing the relevance of the clinical picture to the TMs requested for each patient;
To estimate economic costs associated with inappropriate TM use;
To assess the diagnostic value of TMs in indicating the existence of malignancy, and their usefulness as a screening tool with respect to the current request pattern.
Materials and methods
This was a retrospective review of all cases of TMs processed at the Diagnostic Immunology Department, KAUH, Jeddah, Saudi Arabia, over 3 years (January 2015 to December 2017). Requests originated from all KAUH departments and were transmitted via the hospital electronic system. It included all patients who benefitted from at least one TM request during the study period.
Patients’ electronic records were reviewed to collect the relevant demographic and clinical data: age; gender; TMs requested, including the type with level and result (positive or negative); clinical picture at the first TM request including the chief complaint that motivated the TM request, its localization, and the final diagnosis (cancer type, other disease, etc.), in addition to any significant secondary or other associated symptom or diagnosis. Patients with a previous diagnosis of cancer were excluded, and in the case of repeated requests in a given patient, the first level was considered for sensitivity analyses.
Clinical classifications
For the purpose of this study, clinical pictures at first patient presentation were summarized by localization (organ or system) of their respective chief complaint. Localizations were classified based on the classification of malignant neoplasms (C00–C97) from the 10th revision of the World Health Organization (WHO) International Statistical Classification of Diseases and Related Health Problems (ICD.10). 19 This gave rise to 14 localizations (sites) such as lip, oral cavity and pharynx, digestive organs, respiratory system and intrathoracic organs, etc. Subsequent to the significant number of patients without a clinical picture and with data missing, two categories of sites were added including (a) a non-specified site, and (b) no complaint or clinical picture reported.
The final diagnoses (available in patients’ records) were harmonized and renamed using the Medical Subject Headings (MeSH) nomenclature; then classified into seven main diagnostic categories: (a) malignant tumor, (b) benign tumor, (c) infectious disease, (d) inflammatory disease, (e) other non-tumor disease, (f) indeterminate (unspecified or unknown) diagnosis, and (g) absence of disease or diagnosis.
Characterizing appropriate and inappropriate TM requests
The patients’ records were screened to check for any relevant clinical or laboratory data demonstrating that a clinically sound diagnostic approach had been followed by the physician, leading to the TM request. In the majority cases, we found no clinically sound diagnostic approach in the patient files.
In a second step, and in the absence of a clear indication, the appropriateness of the TM request was assessed based on other clinical or paraclinical data available in the patient records, which enabled an assumption of the chief complaint and/or its localization (body site) in the given patient. For each TM, an appropriate request was assumed for complaints from relevant body sites as follows: CEA for digestive organs; AFP for digestive organs, breast, female genital organs, and male genital organs; CA15-3 for breast and female genital organs; CA12-5 for breast and female genital organs; CA19-9 for digestive organs; and both PSA and FPSA for urinary organs (male).11, 20
In a third step, a final judgment based on a case-by-case analysis attempted to determine whether the TM request would actually be motivated by the assumed complaint or symptom in respect of clinical and common sense (e.g., unreasonable chronological sequence between the complaint and the TM request, the unlikelihood of the tumor localization regarding age or gender, etc.); this final judgment was carried out by the concurrence of two physicians. Of note, complaints classified in lymphoid tissues and those in independent multiple sites were assumed to be in relation to metastasis and thus were categorized as appropriate requests.
Thus, inappropriate TM requests were defined as not being associated to any relevant clinical picture or diagnostic approach; those made for asymptomatic patients; and those for which no reasonable chief complaint could be found or assumed to be appropriate to the given TM.
Assays and cut-off values
All tumor markers were assayed with the Architect automated system (Abbott Diagnostic; USA), using a high-affinity specific monoclonal antibody. As per the manufacturer’s recommendation, the following normal range values were used to discriminate positive and negative results: CEA (0–5 ng/mL), AFP (0–8.7 ng/mL), CA 15-3 (0–25 IU/mL), CA12-5 (0–35 IU/mL), CA19-9 (0–37 IU/mL), TPSA (0–4 ng/mL), and FPSA (0–5 ng/mL).
Statistical methods
The data were entered, coded, and checked for duplicate observations in Microsoft Excel 2013 (Microsoft Corporation), then analyzed using SPSS version 21.0 for Windows (SPSS Inc., Chicago, IL, USA). The evolution over the years (2015 vs. 2016 vs. 2017) of clinical data was analyzed using the one-way analysis of variance (ANOVA) for discrete variables and the chi-square test for categorical variables. The patterns of TM requests (combined or single) were analyzed for each patient, and the most frequent patterns were presented with their respective frequencies. For each TM, the chi-square test was used to compare the percentage of positive results among appropriate versus inappropriate requests. Sensitivity analyses were performed to assess diagnostic accuracy of each TM in indicating malignancy using the presence of a malignant tumor in the final diagnosis as the golden standard: the sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy, were calculated. Costs due to inappropriate TM requests were estimated based on the unit costs applied in KAUH for each TM, including expenses related to materials (reagents, supplies, etc.), technical and medical staff, and infrastructure. A P value of <0.05 was considered to reject the null hypothesis.
Results
Participants’ demographic and clinical characteristics
The study included 7128 patients for whom at least one TM was requested during the period 2015–2017. The demographic characteristics showed 60.6% of males, mean (SD) age=57.23 (17.15) years. The most frequently requested TMs were total PSA, AFP, and CEA, which were requested for 41.9%, 39.1%, and 35.0% of the participants, respectively. Requests involved a single TM in 60.5% of the patients, two TMs in 17.7%, and three TMs in 11.7%. Analysis of the final diagnosis showed 12.9% malignancy, 6.4% benign tumors, and 9.0% non-tumoral diseases (infectious, inflammatory, or other). Analysis by year showed increasing trend in requesting CEA (P<0.001) and CA15-3 (P<0.001) (Table 1).
Participants’ demographic and clinical characteristics (N=7128).
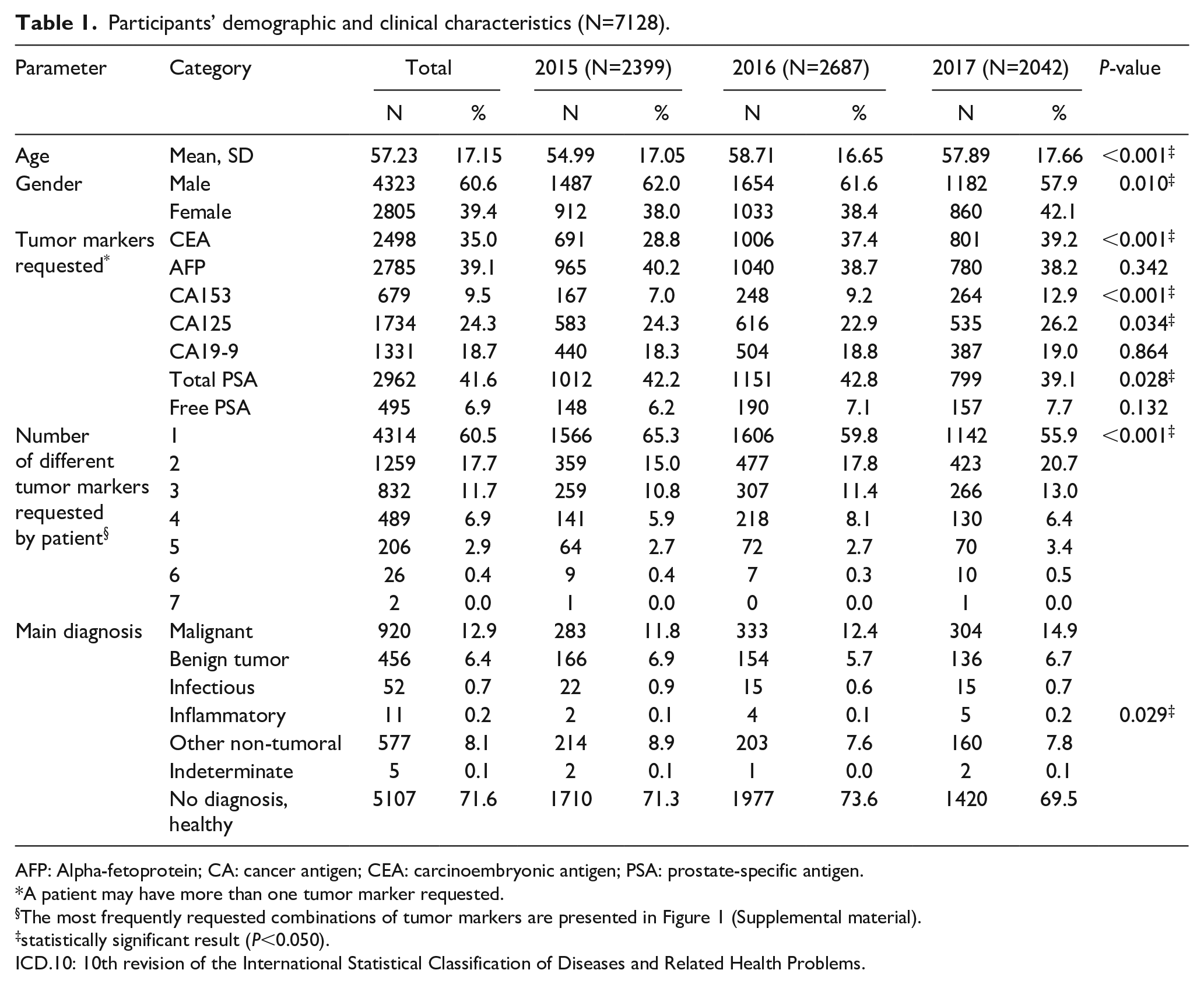
AFP: Alpha-fetoprotein; CA: cancer antigen; CEA: carcinoembryonic antigen; PSA: prostate-specific antigen.
A patient may have more than one tumor marker requested.
The most frequently requested combinations of tumor markers are presented in Figure 1 (Supplemental material).
statistically significant result (P<0.050).
ICD.10: 10th revision of the International Statistical Classification of Diseases and Related Health Problems.
Prescribing patterns of TMs
The most frequent prescribing pattern was total PSA alone (2128; 29.9%), followed by AFP alone (1185; 16.6%), and CEA alone (506; 7.1%). The most frequent combinations were total PSA+free PSA (451; 6.3%); followed by CEA+AFP+CA125 (367; 5.1%) and CEA+AFP+CA125+CA19-9 (283; 4.0%) (Supplemental Figure 1).
Correlating TM requests with the suspected tumor site and characterizing inappropriate use
By excluding cases with no clinical picture, complaints from digestive organs were the most frequent clinical pictures that motivated CEA (20.1%), AFP (21.1%), and CA19-9 (26.1%) requests. Complaints from breast and female genital organs were the most frequent clinical pictures that motivated CA153 (23.6%) and CA125 (18.2%) requests. Complaints regarding male genital organs were the most frequent motivation for total PSA (12.6%) and free PSA (42.4%). Inappropriate requesting varied by TM and ranged between 56.4% for fPSA and 86.8% for total PSA (Table 2).
Correlating tumor marker requesting with the site of the chief complaint and characterizing inappropriate requesting.
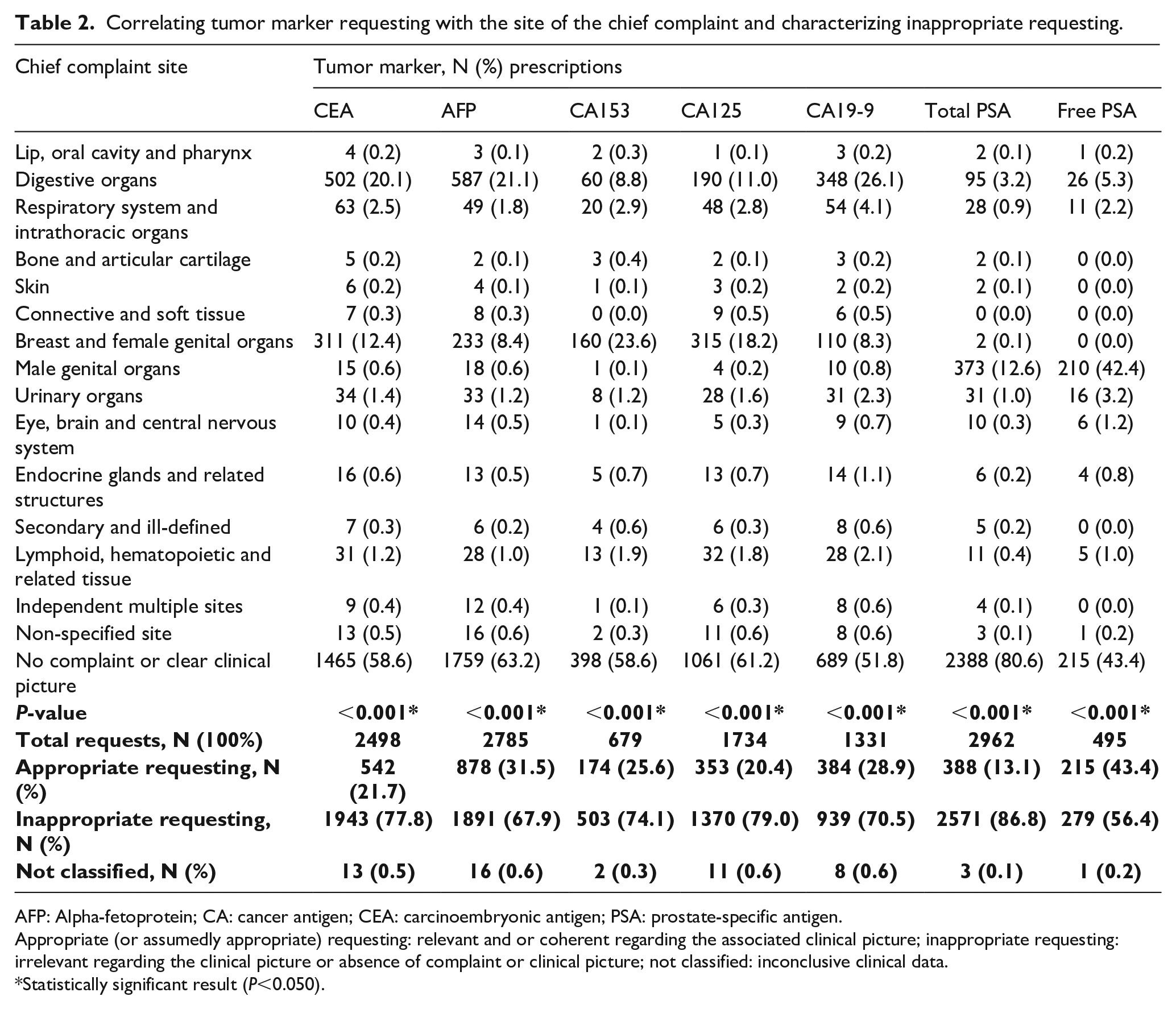
AFP: Alpha-fetoprotein; CA: cancer antigen; CEA: carcinoembryonic antigen; PSA: prostate-specific antigen.
Appropriate (or assumedly appropriate) requesting: relevant and or coherent regarding the associated clinical picture; inappropriate requesting: irrelevant regarding the clinical picture or absence of complaint or clinical picture; not classified: inconclusive clinical data.
Statistically significant result (P<0.050).
Distribution by TM, patient demographics data, and final diagnosis
Females were predominant among patients who benefited from TM requesting, accounting for 60.2% to 89.9% of the requests depending on the TM type; except for total PSA (tPSA, 0.6%) and fPSA (0.4%). Mean (SD) age of the patients ranged between 51.70 (16.45) for CA125 and 68.75 (10.89) for fPSA. The most frequently concerned age categories were >40 years for CEA, AFP, CA153, and CA125, and >60 years for CA19-9, tPSA, and fPSA. For the final diagnosis, the percentages of tumoral diseases (malignant and benign) ranged between 15.3% (5.1% and 10.2%) for tPSA to 46.5% (8.9% and 37.6%) for fPSA, respectively. The percentage of non-tumor diseases ranged from 4.0% for tPSA to 18.1% for CA19-9. TM requests were was associated with absence of disease or undiagnosed cases in 43.4% to 80.8%, depending on the TM (Table 3).
Distribution of tumor marker requesting by patient demographics data and final diagnosis.
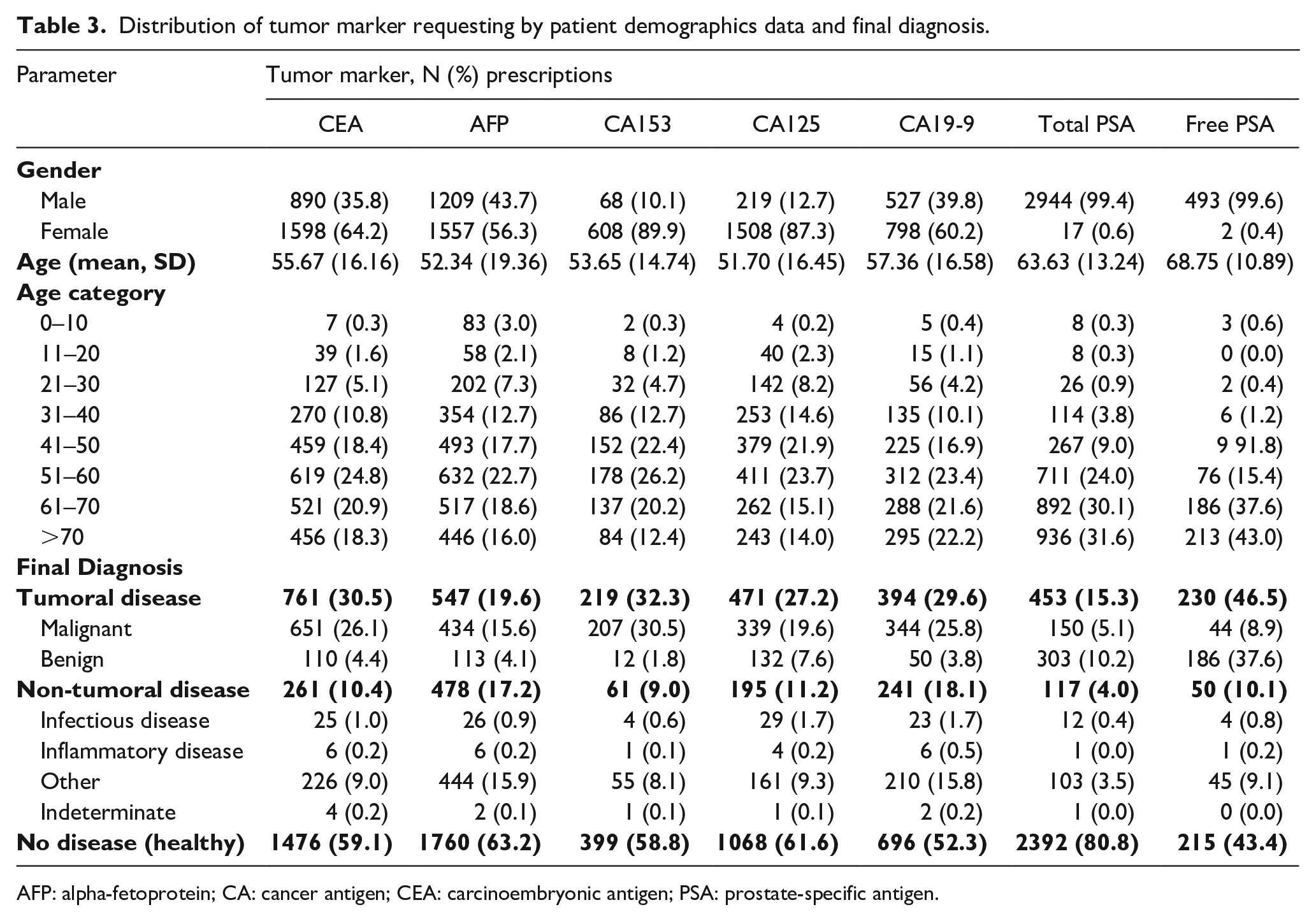
AFP: alpha-fetoprotein; CA: cancer antigen; CEA: carcinoembryonic antigen; PSA: prostate-specific antigen.
The diagnostic accuracy of TM in indicating any type of malignant tumor
In the present TM requesting pattern, TMs had low to moderate sensitivity (28.3%–75.1%) and high specificity (81.3%–93.6%) in detecting any malignancy; except for fPSA, which was highly sensitive (84.1%) but only slightly specific (24.2%) due to a high percentage of false positives (Table 4).
Diagnostic accuracy of tumor markers results in indicating any type of malignant tumor, as analyzed on retrospective data from 2015 to 2017 at KAUH.
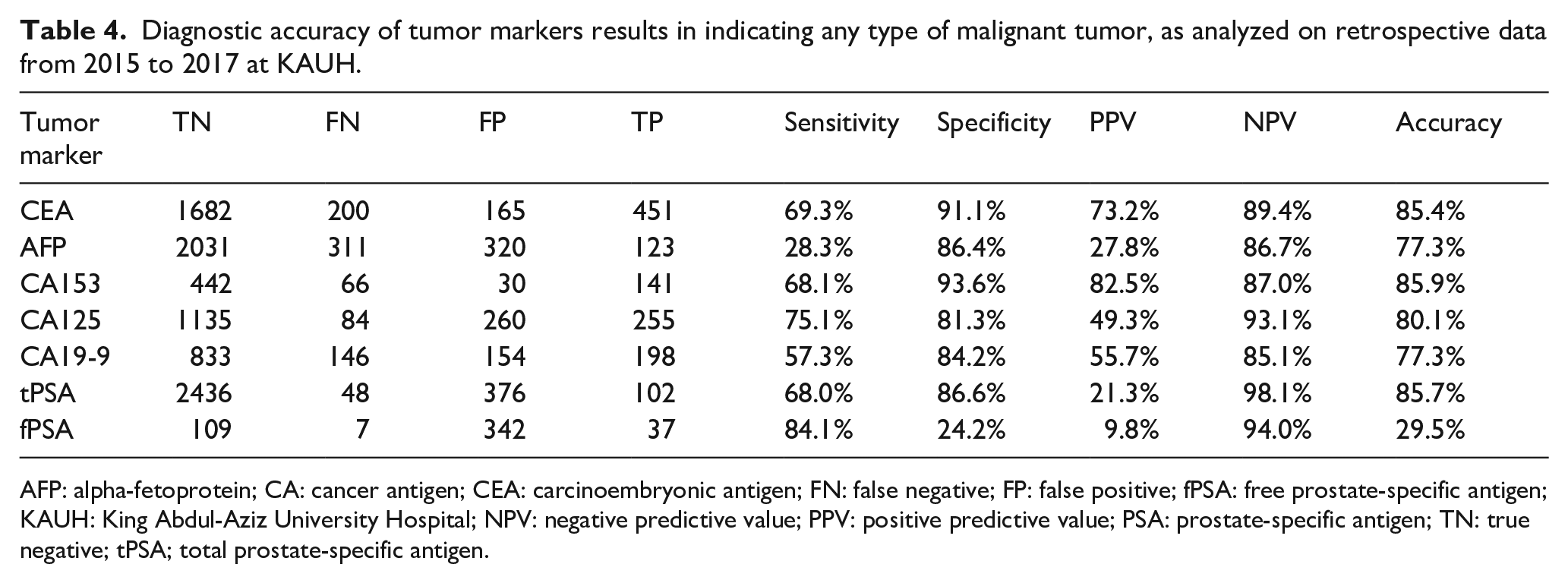
AFP: alpha-fetoprotein; CA: cancer antigen; CEA: carcinoembryonic antigen; FN: false negative; FP: false positive; fPSA: free prostate-specific antigen; KAUH: King Abdul-Aziz University Hospital; NPV: negative predictive value; PPV: positive predictive value; PSA: prostate-specific antigen; TN: true negative; tPSA; total prostate-specific antigen.
The cost of inappropriate TM use
As per the model developed in this study, the overall costs due to inappropriate TM requests were estimated as total Saudi Riyal (SAR)10,445,600 ($2,785,493) over the 3 years, representing an average of $0.93 million per year. The costliest inappropriate use was associated with tPSA (SAR2,828,100 ($754,160)), followed by CEA (SAR2,137,300 ($569,947)), and AFP (SAR2,080,100 ($554,693)) (Table 5).
Appropriate and inappropriate tumor markers requesting and related economic cost.
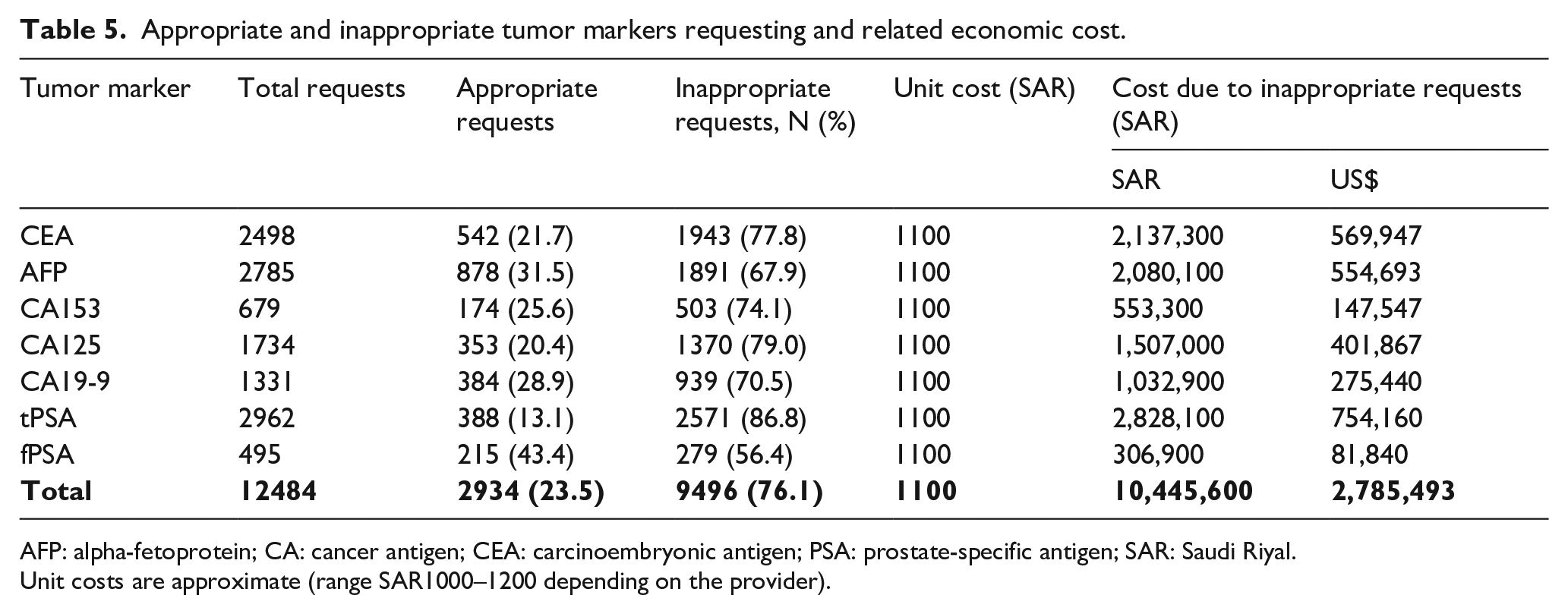
AFP: alpha-fetoprotein; CA: cancer antigen; CEA: carcinoembryonic antigen; PSA: prostate-specific antigen; SAR: Saudi Riyal.
Unit costs are approximate (range SAR1000–1200 depending on the provider).
Discussion
Since their first identification and use in the last half of the 20th century.,21, 22 TMs have been continually investigated for their feasibility and efficacy, particularly with the recent technological advances in immunology, molecular biology, and biochemistry. So far, more than 20 TMs have been identified and clinically utilized. 23 We investigated the requesting patterns of two oncofetal antigens (AFP and CEA), three tumor-associated antigens (CA15-3, CA125, and CA19-9), and the glycoprotein enzyme PSA in a large referral university hospital in Western Saudi Arabia. The findings revealed that 56.4% to 86.8% of total TM requests were inappropriate in our medical institution, and the majority were likely to be made for screening purposes in asymptomatic individuals. This was associated with considerable costs estimated at $0.93 million per year. Furthermore, in the present requesting pattern, TMs have low to moderate sensitivity (28.3%–75.1%) in detecting any malignant disease, which confirms that TMs are not appropriate as a screening tool and their utilization should comply with the clinical guidelines.
According to the year-to-year analysis, there was an increasing trend in requesting TMs, particularly CEA and CA15-3, with a significant increase in multiple TM requests for a given patient. Similarly, Walker and Crook 24 found a remarkable rise of month-on-month multiple TM requests with an average of 3.1% per month during a 36-month period. McGinley and Kilpatrick 25 have also shown a 125% increase in TM workload from 1997 to 2002, with the most reported increases in PSA and CEA requests. Both patient-related and physician-related factors may have contributed to the continuous increase in TM laboratory requests. Specifically, the need of the patient for reassurance, the fear of missing a non-predicted diagnosis, and the fear of litigation (especially for potentially serious and life-threatening conditions) can all provide a plausible explanation of such a trend. 26 Furthermore, the increasingly reported self-acquired information about cancer based on internet searches and social media in Saudi Arabia 27 and other countries 28 may provide a wealth of information about laboratory investigations and hence contribute to the increasing TM request patterns.
These factors have resulted in non-compliance with TM request guidelines and a very high rate of inappropriate TM requests estimated as 56.4% to 86.8% of the total requests. In Northern Ireland—in spite of publishing specific local guidelines for TM requests—the requests were generally inappropriate for the screening of asymptomatic individuals. 29 Similarly, Yilmaz et al. 15 conducted an analysis of TM requests over a short period (3 months), in order to go into patients’ details thoroughly; this revealed a significant rise of TM requests, indicating inappropriate request patterns for tumor screening. Franceschini et al. 30 conducted a laboratory-based survey in Italy and found no difference in the requesting attitudes of physicians even with the existence of the appropriateness guidelines. It is worthy to note that only AFP and PSA (and to a lesser extent CA125) are useful for cancer screening, whereas CEA and CA19-9 cannot be utilized for screening purpose. 31
Where CEA, CA15-3, and CA125 were reported as the most inappropriately requested TMs—possibly due to their inclusion in the same request 32 —it was quite unexpected to find that tPSA ordering yielded the highest rate of inappropriateness (86.8%) in the present study. The increased mean age of the participants (57.23±17.15 years) might provide an acceptable explanation of increased tPSA ordering. PSA is widely requested by the clinicians as a screening approach in male individuals aged 40–70 years rather than other age categories.33, 34 However, the observed disproportion between PSA ordering and definite prostatic cancer diagnosis may be related to (a) the increase in patient’s awareness regarding the disease; (b) the aforementioned physician-related factors; and (c) the low incidence of prostatic cancer among Saudi males (5.3 per 100,000 people) compared with other countries. 35
Sensitivity analysis demonstrated the poor reliability of TMs as screening approaches for malignancy, as indicated by the low to moderate sensitivity for almost all TMs except fPSA. Similarly, Arioli et al. 16 revealed a poor clinical relevance of TM requests in internal medicine units as shown by the low PPV. As such, it is important to consider sound clinical suspicion prior to ordering a TM to reduce unnecessary stress to the patient and to avoid the prospective time and financial costs.
In view of the relatively scarce knowledge concerned with the actual causes of the inappropriateness of TM requests, due to lack of linkage between electronic requests and clinical records,17, 24, 29, 36, 37 some models have been recently proposed to be effective in estimating the appropriateness of TM ordering in laboratories. Such models are usually based on incidence and prevalence data extracted on a regional scale for different malignancies. 18 For instance, the approach of Gion et al. 18 aimed at resolving the lack of clinical data and to overcome the inability to apply the established data on a more generalized scale among different laboratories in two large regions in Italy. However, the applicability of these models is endangered by distinct limitations. Actually, there are several sources of variability in the evidence of TM requests, including sample size, study setting (primary or secondary care), and study design. Furthermore, guidelines relevant to categorization, assessment, and measurement of the inappropriate TM requests may be variably demonstrated. 38 As such, the improvement of appropriateness should not only focus on the mere implementation of the pertinent guidelines, but also the integration of such guidelines into a formal way of knowledge translation, which means placing knowledge into action. 39 Furthermore, the role of laboratories in improving appropriate TM ordering has been emphasized by vetting every TM request. However, this process may bear a high cost that might go beyond the saving of a reduced volume of work. 24 Alternatively, the use of tailored request cards might be helpful to reduce the number of received tests by about 25%. 40
Some limitations in the present study may interfere with the accurate interpretation of the outcomes. Stating the appropriateness of the TM request was based on a review of the clinical records, since actual indications of TM requests—either as screening or follow-up investigations—were not available to our analysis for a substantial number of participants. This may constitute the major limitation of this study, as the absence of relevant clinical data was assumed to be an absence of motivating symptoms, which may overestimate the rate of inappropriateness. On the other hand, the exclusion of repeat requests for the same patient may relatively compensate this limitation. Furthermore, the golden standards of tumor detection, such as histopathological investigations and radiographic approaches, were not specified to confirm the final diagnosis (cancer vs. no cancer), which may downplay the power of the sensitivity analyses. Additionally, we failed to differentiate the impact of TM requests by a non-specialized clinician and/or junior physician.
Conclusion
Tumor marker ordering is likely to be inappropriately requested in a multifaceted manner due to patient-related and clinician-related factors—a matter that could be wrongfully reassuring or unduly alarming for the patient. This highlights the need of tailoring specific interventions to help reduce unnecessary stress and the financial burden of cancer care. It is imperative to consider adherence to local recommendations regarding TM requests, particularly with regard to the use of TMs for screening purposes. Relying on the scientific evidence is crucial in clinical practice by effectively eliminating the suspicion prior to requesting TMs. From another perspective, the role of laboratories in the management of inappropriate requests should be emphasized along with the introduction of cost-effective novel TMs. Finally, sound clinical knowledge should be appropriately applied and translated into efficient actions for laboratory requests in the healthcare system, notably by improving awareness of and adherence to the effective screening programs among physicians.
Supplemental Material
Supplemental_Figure_1 – Supplemental material for Inappropriate practice in tumor marker requests at a university hospital in Western Saudi Arabia: A 3-year retrospective study
Supplemental material, Supplemental_Figure_1 for Inappropriate practice in tumor marker requests at a university hospital in Western Saudi Arabia: A 3-year retrospective study by Jamil A. Al-Mughales and Mahmood Shaheen Alahwal in The International Journal of Biological Markers
Footnotes
Acknowledgements
We are grateful to Dr. Mohamed Amine Haireche and Mrs. Oolaa Saati for their contribution in the preparation of the manuscript. We would like also to thank Saudi Abbott Diagnostic Company for supporting the preparation of the manuscript.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
The SPSS data used to support the findings of this study are available from the corresponding author upon request.
Supplemental material
Supplemental material for this article is available online. Figure 1: Pattern of combined tumor marker requests.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
