Abstract
Background:
The diagnosis and monitoring of primitive lung neuroendocrine tumors (lung pNETs) are usually performed by the measurement of serum chromogranin A (CgA) and urinary 5-hydroxyindolacetic acid (5-HIAA) levels. However, imaging techniques are necessary due to the poor diagnostic efficiency of the laboratory tests.
Methods:
A total-body computed tomography and bone scintigraphy scans showed multiple hepatic and bone metastases of a 55-year-old man affected by well-differentiated lung pNETs without severe initial symptoms. After diagnosis, he started therapy and was monitored with serum, urinary markers, and imaging techniques.
Results:
During follow-up, the urinary 5-HIAA levels did not significantly increase, while serum CgA and urinary para-hydroxyphenylacetic acid (pHPAA) levels (urinary organic acid physiologically present in the urines of healthy subjects) showed significant increases related to worsening clinical condition.
Conclusions:
The early increase in urinary pHPAA levels—usually not dosed in pNET patient monitoring—could be a promising prognostic marker.
Keywords
Introduction
Neuroendocrine tumors (NETs) are rare tumors that originate from the cells of the diffuse neuroendocrine system. The most frequent primary sites are the gastrointestinal (60%–70%) and the tracheo-bronchopulmonary (20%–30%) tract 1 ; other regions are skin, thyroid, parathyroid, and adrenal glands (10%) for example. Lung neuroendocrine tumors can be classified into neuroendocrine cell hyperplasia and tumorlets, neuroendocrine morphology tumors, non-microcytomas with neuroendocrine differentiation, and other lung tumors with neuroendocrine features. Many patients affected with primitive lung neuroendocrine tumors (lung pNETs) are asymptomatic at the time of diagnosis or have non-specific symptoms of respiratory distress such as asthma or chronic obstructive pulmonary disease, that often delay the diagnosis. 2 Although more than 90% of lung pNETs are non-functional, patients with a functional (secretory) component present hormonal symptoms (wheezing, skin redness, and diarrhea) due to the secretion of hormones and peptides. 3 Diagnostic procedures include imaging tests, such as total-body computed tomography (CT), ultrasounds, bone scintigraphy, positron emission tomography/CT (PET/CT), magnetic resonance imaging, and angiography, in order to evaluate disease location and extent. 4 In addition, the diagnosis and follow-up of NETs require serum chromogranin A (CgA) and 24-hour urinary 5-hydroxyindolacetic acid (5-HIAA) to be performed. Serum neuron-specific enolase (NSE)—even though it is not recommended in the management of patients with NETs—has been demonstrated to be elevated in 30%–50% of subjects with NETs. Preliminary data have reported urinary para-hydroxyphenylacetic acid (pHPAA, a tyrosine metabolite physiologically excreted in the urine of healthy people) as a useful marker, together with 5-HIAA, for the management of NETs patients. 5
Case description
A 55-year-old man with no relevant medical history, presented to our facility in January 2018 with cough and wheezing. A thorax CT scan showed a 19 mm maximum diameter solid nodule with irregular margins into the left upper lobe connected to an ipsilateral superior hilar lymph node agglomeration of 45 mm (maximum diameter). The total-body CT scan (Figure 1(a), right panel) revealed the presence of multiple liver and bone metastases, which were confirmed by bone scintigraphy. A liver biopsy performed in March 2018 showed a well-differentiated neuroendocrine tumor. After a multidisciplinary evaluation, 68Ga-labelled DOTA-Tyr octreotide (68Ga-DOTATOC) PET/CT was performed to confirm somatostatin receptor expression and disease staging. 68Ga-DOTATOC PET/CT functional imaging revealed an abnormal significant uptake of the tracer in the left upper lobe of the lung (SUVmax 13.6), in the ipsilateral hilar region (SUVmax 30.3), and in multiple liver (SUVmax 40.0) and bone lesions (SUVmax 48.7) (Figure 1(a), left panel). In April 2018, the patient started therapy with octreotide long-acting release (LAR) 30 mg intramuscularly every 28 days. No adverse events were reported during the treatment in accordance with Common Terminology Criteria for Adverse Events (CTCAE criteria version 4.0).
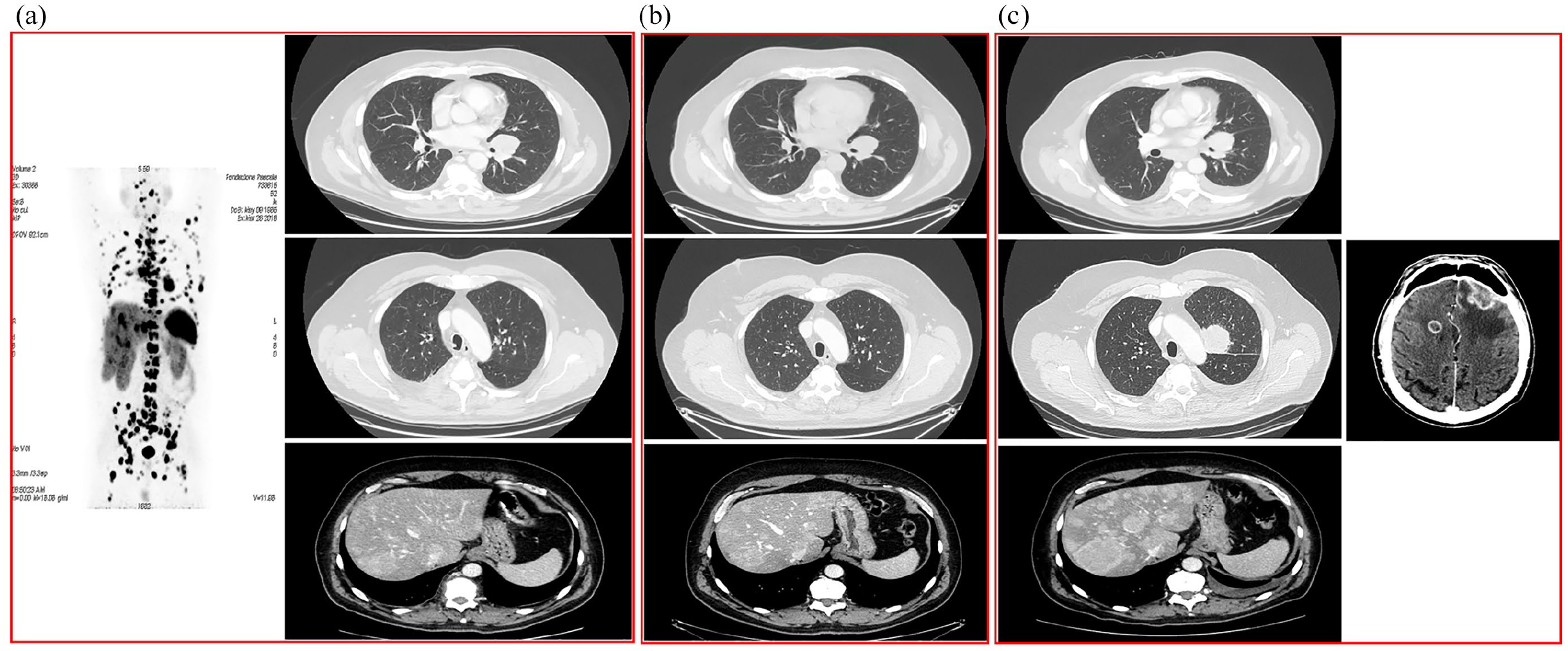
(a) (left panel): PET/CT using 68Ga-labelled DOTA-Tyr octreotide shows multiple areas of abnormal uptake into lungs, lymph nodes, liver, and bone; (right panel): total-body CT scan shows a pulmonary left hilar lesion and multiple liver lesions. (b) A total-body CT scan during octreotide long-acting release therapy shows stable disease. (c) A total-body CT scan shows progressive disease at the liver (left panel, November 2018) and the brain (right panel, January 2020) during the last follow-up.
The patient was monitored by periodic total-body CT scans, which showed a stable disease (Figure 1(b): total-body CT scan performed in November 2018). After November 2018, disease progression (Figure 1(c), left panel) occurred with a resumption of symptoms. Thereafter, the patient was included in a clinical, multicenter, double-blinded, randomized trial of axitinib versus placebo and was monitored every 12 weeks by means of total-body CT scans in accordance with Response Evaluation Criteria in Solid Tumors (RECIST criteria version 1.1) and laboratory tests. The 24-hour urinary levels of 5-HIAA and pHPAA were monitored from March 2019 until March 2020 by means of high-pressure liquid chromatography (HPLC) with electrochemical detection in 24-hour urine samples. Serum CgA levels were determined on KRYPTOR compact PLUS instrumentation and contextually serum NSE (as a marker of neuroendocrine differentiation) levels on the Roche COBAS 6000 platform. The octreotide LAR treatment allowed the achievement of stable disease until January 2020, when multiple lesions in the brain were documented (Figure 1(c), right panel). To date, the patient is continuing to perform encephalic radiotherapy, which is planned to reach a total dose of 30 Gy.
Conclusion
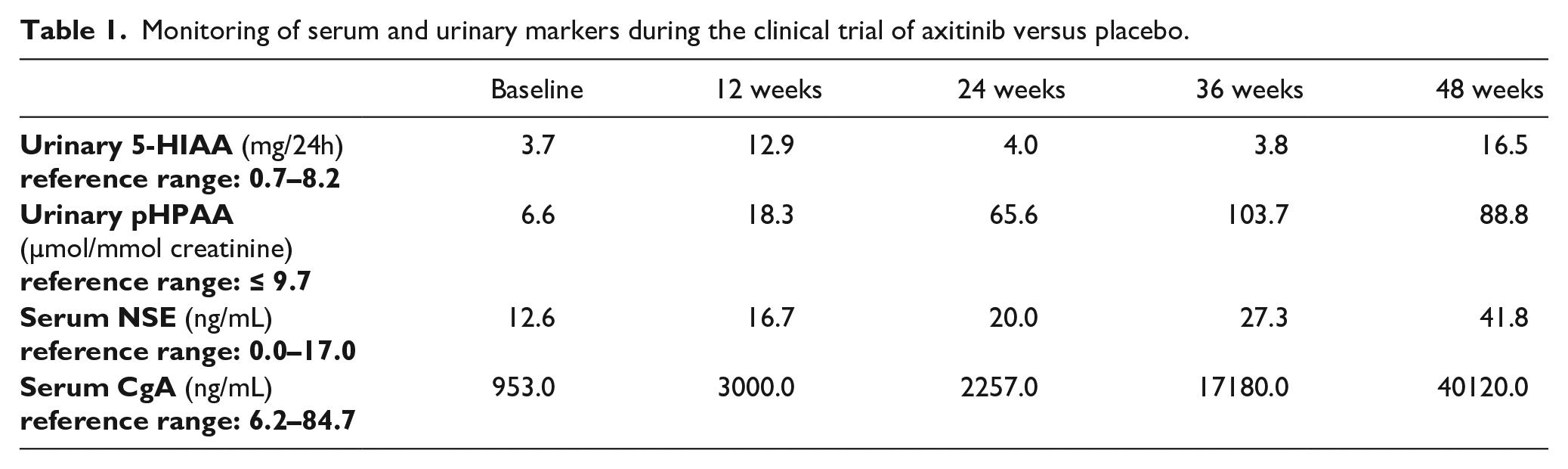
From March 2019 to March 2020, the patient was monitored by determining serum CgA and urinary 5-HIAA every 12 weeks. Urinary 5-HIAA chromatograms obtained with the HPLC method enabled the detection and measurement of pHPAA levels simultaneously (Figure 2). As shown in Figure 3 and Table 1, only serum CgA presented high levels at baseline with an important increasing trend from the first to the last follow-up. Urinary 5-HIAA levels augmented slightly by 12 and 48 weeks, while serum NSE presented a mild increase throughout the monitoring, respectively (Table 1). Unexpectedly, the urinary pHPAA levels (within the reference range at baseline) showed a great increase, which appeared earlier (at 12
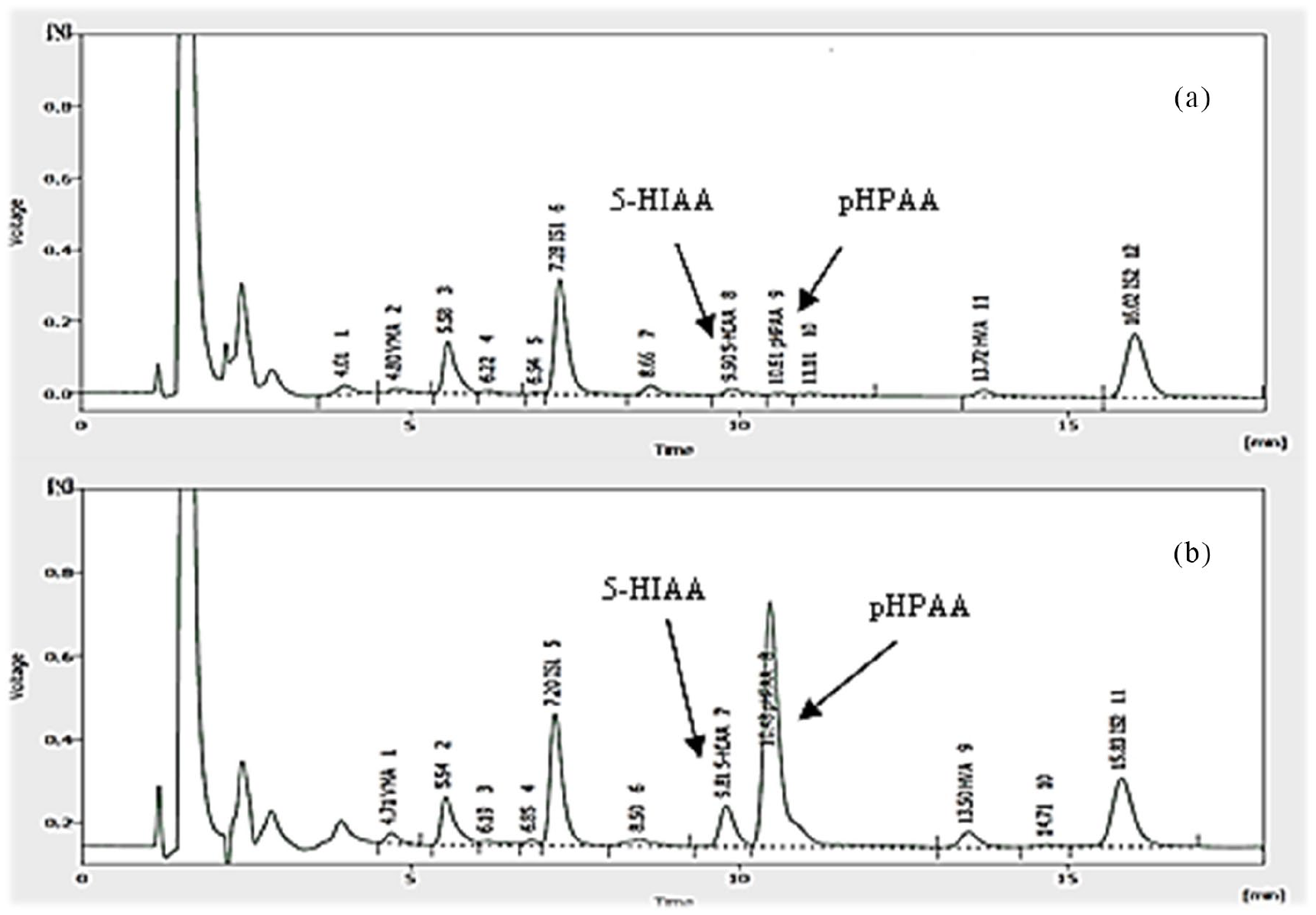
HPLC chromatograms. (a) Urinary 5-HIAA and pHPAA normal levels at first follow-up. (b) Urinary 5-HIAA and pHPAA higher levels at last follow-up.
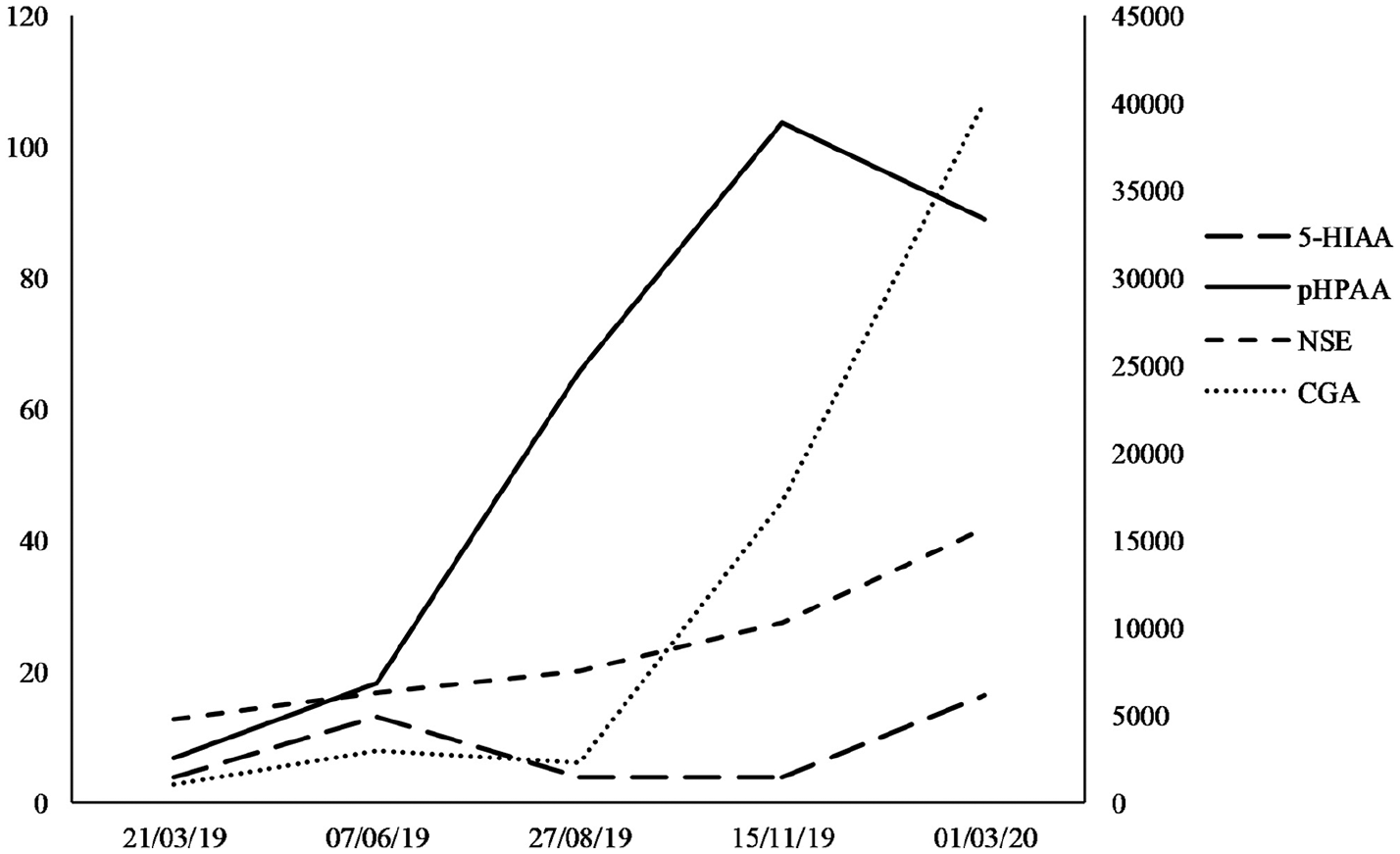
Biomarker monitoring: the scale on the left refers to serum NSE (ng/mL), urinary 5-HIAA (mg/24h), and urinary pHPAA (µmol/mmol creatinine); the scale on the right refers to serum CgA (ng/mL).
Monitoring of serum and urinary markers during the clinical trial of axitinib versus placebo.
In this case report, urinary pHPAA was evaluated during patient monitoring and was found to have increased earlier than the urinary 5-HIAA levels with an increasing trend during follow-up. This finding suggests the possibility of combining urinary pHPAA determination with serum CgA in the management of NET patients, in particular during follow-up. Moreover, HPLC elution of urinary pHPAA in the same chromatogram used for urinary 5-HIAA determination makes it technically feasible and simple to detect. In conclusion, the described case focuses on the possible utility of taking into account urinary pHPAA levels, integrating NET laboratory diagnosis with its detection on HPLC chromatograms. Nevertheless, this potential approach should be investigated in larger series of patients.
Footnotes
Author contributions
Luigi Russo and Bruna Grilli contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
