Abstract
Background and Aim:
Our aim was to compare the prognostic value of the American Joint Committee on Cancer (AJCC) 7th and 8th editions staging systems for patients with gastric cancer in China.
Methods:
A total of 1326 gastric cancer patients diagnosed between 2008 and 2012 were included. The discriminative ability of the AJCC 8th and 7th editions was compared using the Harrell’s concordance index (C-index).
Results:
There are two main modifications in the 8th edition. (i) pN3 staging was divided into pN3a and pN3b. The gastric cancer patients with pN3a experienced significantly better overall survival compared with those with pN3b (5-year overall survival: 34.5% vs. 15.6%, P < 0.001) (stratified by pT: pT3: 5-year overall survival: 33.9% vs. 13.2%, P < 0.001; pT4a: 32.8% vs. 16.9%, P = 0.056; pT4b: 17.0% vs. 11.1%, P = 0.048). (ii) Subgroup staging adjustments. The subgroup staging adjustments (T3N3bM0 (IIIB→IIIC), T4aN3aM0 (IIIC→IIIB), T4bN0M0 (IIIB→IIIA), and T4bN2M0 (IIIC→IIIB)) resulted in more gastric cancer patients being accurately staged. Furthermore, the C-index value of the 8th edition tumor node metastasis (TNM) staging system was significantly higher than that of the AJCC 7th TNM staging system to predict the survival of gastric cancer patients (0.701 vs. 0.685, P < 0.001).
Conclusions:
The 8th edition of the TNM staging system is superior to the 7th edition staging system for prediction of survival of gastric cancer patients in China.
Introduction
In Asian countries, the incidence and mortality of gastric cancer are the highest among malignant tumors.1, 2 Accurate and uniform staging helps physicians make informed medical decisions to determine patients’ prognoses and to assess treatment outcomes.3, 4 The gastric cancer staging system most widely used worldwide is the Union for International Cancer Control (UICC)/American Joint Committee on Cancer (AJCC) tumor-node-metastasis (TNM), which is based on tumor infiltration depth (pT), number of lymph node metastases (pN), and the presence of distant metastasis (pM). The 8th edition of TNM staging system (hereafter referred to as the “8th edition”) of gastric cancer was revised as follows: pN3 in the 7th edition was further divided into pN3a and pN3b, and the subgroup staging was adjusted. This revision is based on data from the International Gastric Cancer Association (IGCA), and the patients were mainly from Japan and South Korea (84.8%).5, 6 In contrast, the Chinese population with gastric cancer represented a relatively low percentage. People living in China have a high incidence of gastric cancer.7, 8 Therefore, it is necessary to verify the applicability of the 8th edition to patients with gastric cancer in China. The present study was designed to investigate whether the 8th edition is better than the 7th edition of the TNM staging system (hereafter referred to as the “7th edition”) for managing Chinese citizens with gastric cancer.
Materials and methods
General information
This retrospective study included patients who underwent radical gastrectomy at the PLA General Hospital from January 2005 to October 2010. Inclusion criteria were as follows: (i) pathological diagnosis of the first primary gastric cancer; (ii) absence of distant metastasis; (iii) radical gastrectomy; (iv) postoperative survival >1 month; and (v) no neoadjuvant chemotherapy. The study was approved by the Ethics Committee of the PLA General Hospital, and patients provided written informed consent before they were included in the study. Patients were followed quarterly during the first 2 years and then biannually. We included 1326 patients with a median follow-up of 71 months (range, 52–72 months).
Statistical analyses
All patients were staged according to the 7th and 8th editions. Given the influence of the number of lymph nodes on staging, stratified analysis was further conducted according to the number of lymph node biopsies. Differences between the two groups were evaluated using the chi-square test. We selected 15 lymph nodes as the cut-off value, because the National Comprehensive Cancer Network and the AJCC recommend ⩾15 lymph nodes for biopsy to avoid stage migration. The Kaplan–Meier method was used to calculate survival, and the log-rank method was used to compare the differences in survival between groups. The Cox risk-scale model was used to calculate patients’ prognostic factors, and the multivariate Cox risk-scale model was used to identify independent prognostic factors. The likelihood-ratio chi-square test was used to compare the consistency of staging between editions. Harrell’s concordance index (C-index) was used to compare the discriminative ability of the staging between editions. A C-index value closer to 1 indicates a better discriminatory model. Similarly, a smaller Akaike Information Criterion (AIC) value indicates a better model when we calculated comparative AIC values. All statistics were performed using R–3.4 software (http://www.r-project.org). P < 0.05 (two-sided) indicates a statistically significant difference.
Results
We analyzed the data for 1326 patients with gastric cancer. The median age of onset was 60 years (interquartile range, 52–68), and the median number of lymph nodes was 15 (interquartile range, 10–21). Men represented 76.32% of patients. Most tumors, which resided in the upper-third of the stomach (n = 505, 38.08%), were moderately differentiated (n = 636; 47.96%). Tumor diameters ranged from 2 to 5 cm (n = 665; 50.15%). According to the 8th edition, most tumors were divided into pT3 stage (n = 422; 31.83%) and pN0 stage (n = 503; 37.93%). Patients underwent distal gastrectomy (n = 618; 46.61%), adequate lymphadenectomy (n = 696; 52.49%), and postoperative adjuvant chemotherapy (n = 710; 53.54%). Table 1 summarizes the patients’ characteristics as well as those of their tumors.
Clinicopathological characteristics of patients with GC.
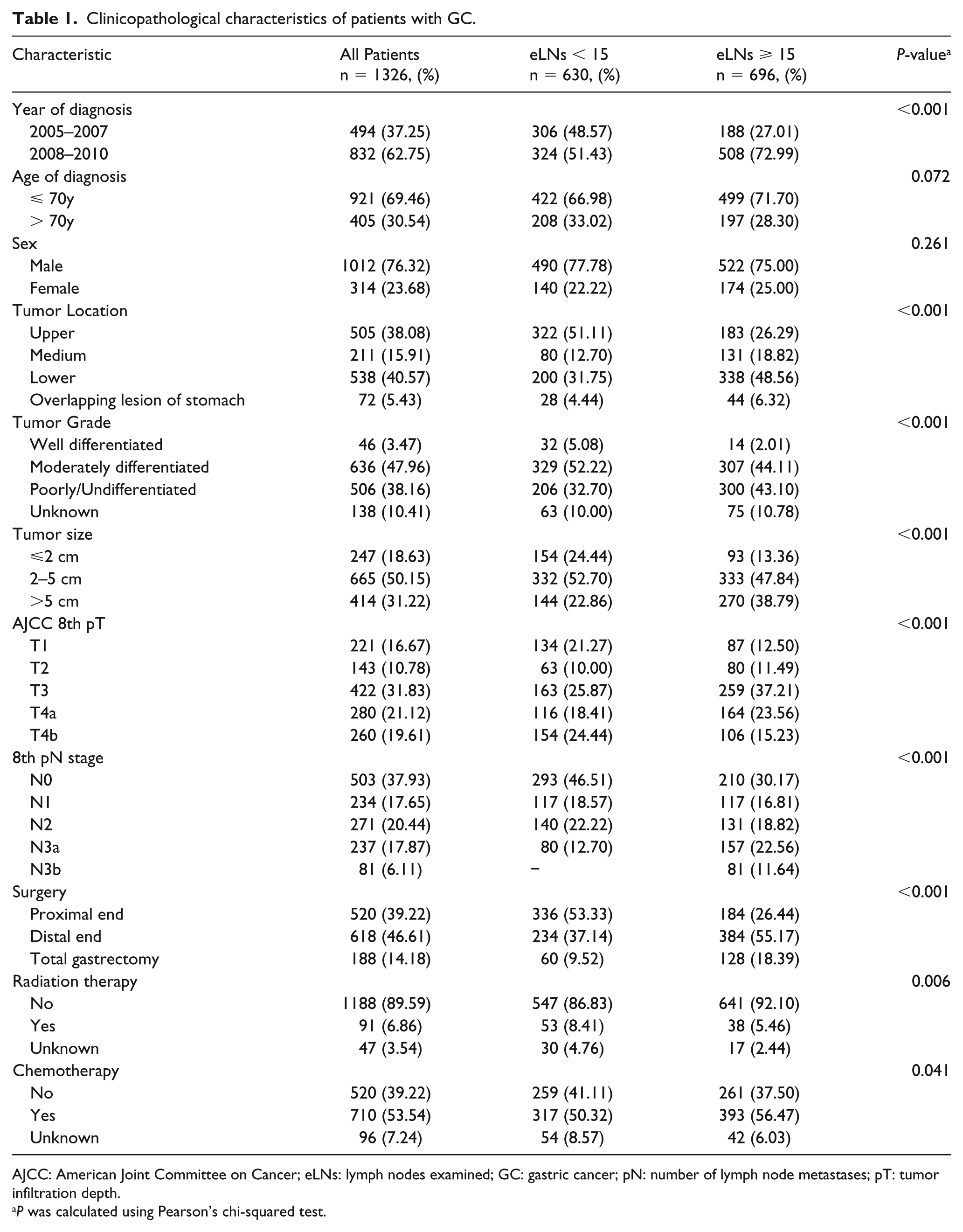
AJCC: American Joint Committee on Cancer; eLNs: lymph nodes examined; GC: gastric cancer; pN: number of lymph node metastases; pT: tumor infiltration depth.
P was calculated using Pearson’s chi-squared test.
The 5-year overall survival (OS) rate of patients with gastric cancer was 55.7% (95% confidence interval 53.0, 58.4%). The survival curves associated with staging for the 7th and 8th editions are shown in Figure 1. TNM staging subgroups and patients’ distributions of staging between editions are shown in Supplementary Material 1. There are two major changes in the 8th edition as follows: (i) The original pN3 staging was divided into pN3a and pN3b stages. Univariate analysis show that the 5-year OS of patients with pN3a was significantly longer compared with those with pN3b as follows: 5-year OS: 34.5% (28.9, 41.3%) versus 15.6% (9.3, 26.1%), P < 0.001; pT3: 5-year OS: 33.9% versus 13.2%, P < 0.001; pT4a: 32.8% versus 16.9%, P = 0.056; pT4b: 17.0% versus 11.1%, P = 0.048) (Figure 2); and (ii) subgroup staging adjustments occurred mainly in five subgroups in the present study (Supplementary Material 1). Figure 3 shows that the difference in survival after adjustment was lower compared with that before adjustment for T3N3bM0 (IIIB→IIIC), T4aN3aM0 (IIIC→IIIB), T4bN0M0 (IIIB→IIIA), and T4bN2M0 (IIIC→IIIB). Furthermore, the survival difference after adjustment was higher compared with that before adjustment only for T4aN2M0 (IIIB→IIIA).
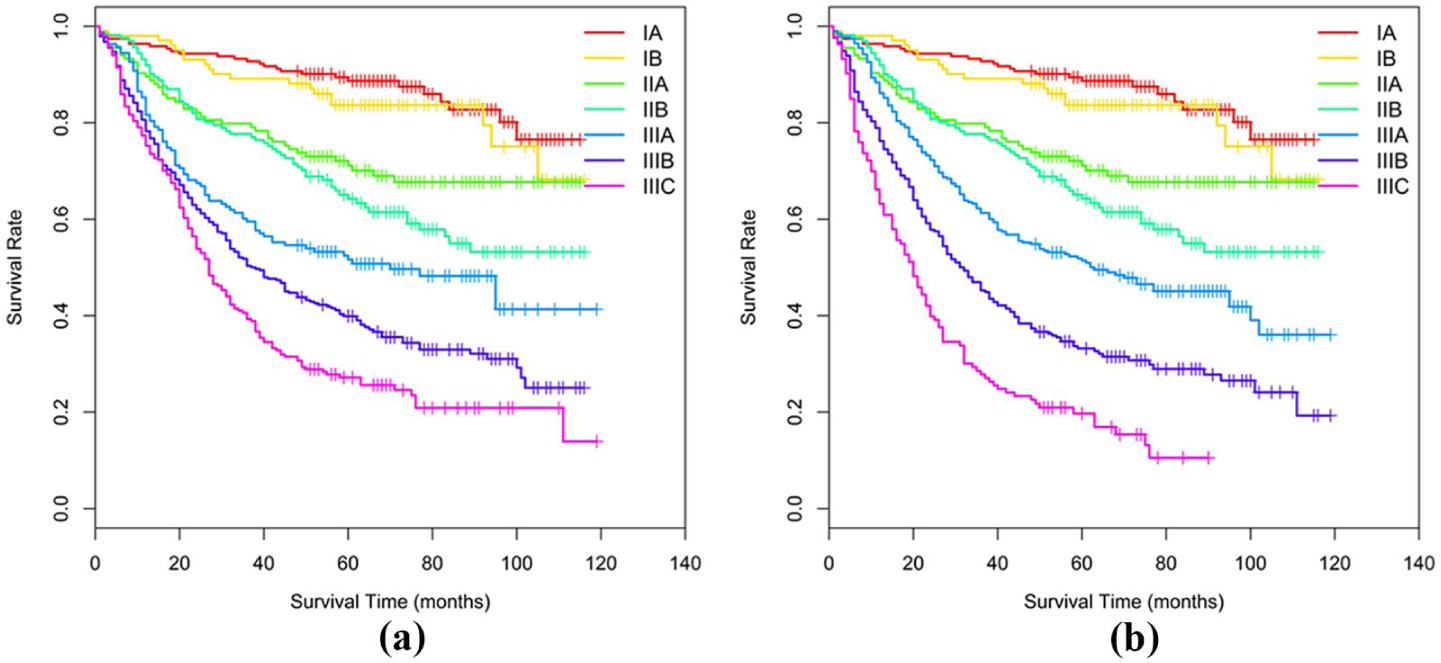
Overall survival (OS) of all patients with gastric cancer patients according to the 7th and 8th editions of the TNM staging system. (a) 7th TNM staging system. Five-year overall survival rates for stages (IA–IIIC) were 88.7%, 83.7%, 71.1%, 64.2%, 51.6%, 39.9%, and 27.2%, respectively. (b) 8th TNM staging system. Five-year overall survival for stages (IA–IIIC) were 88.7%, 83.7%, 71.1%, 64.2%, 51.4%, 33.2%, and 19.7%, respectively.
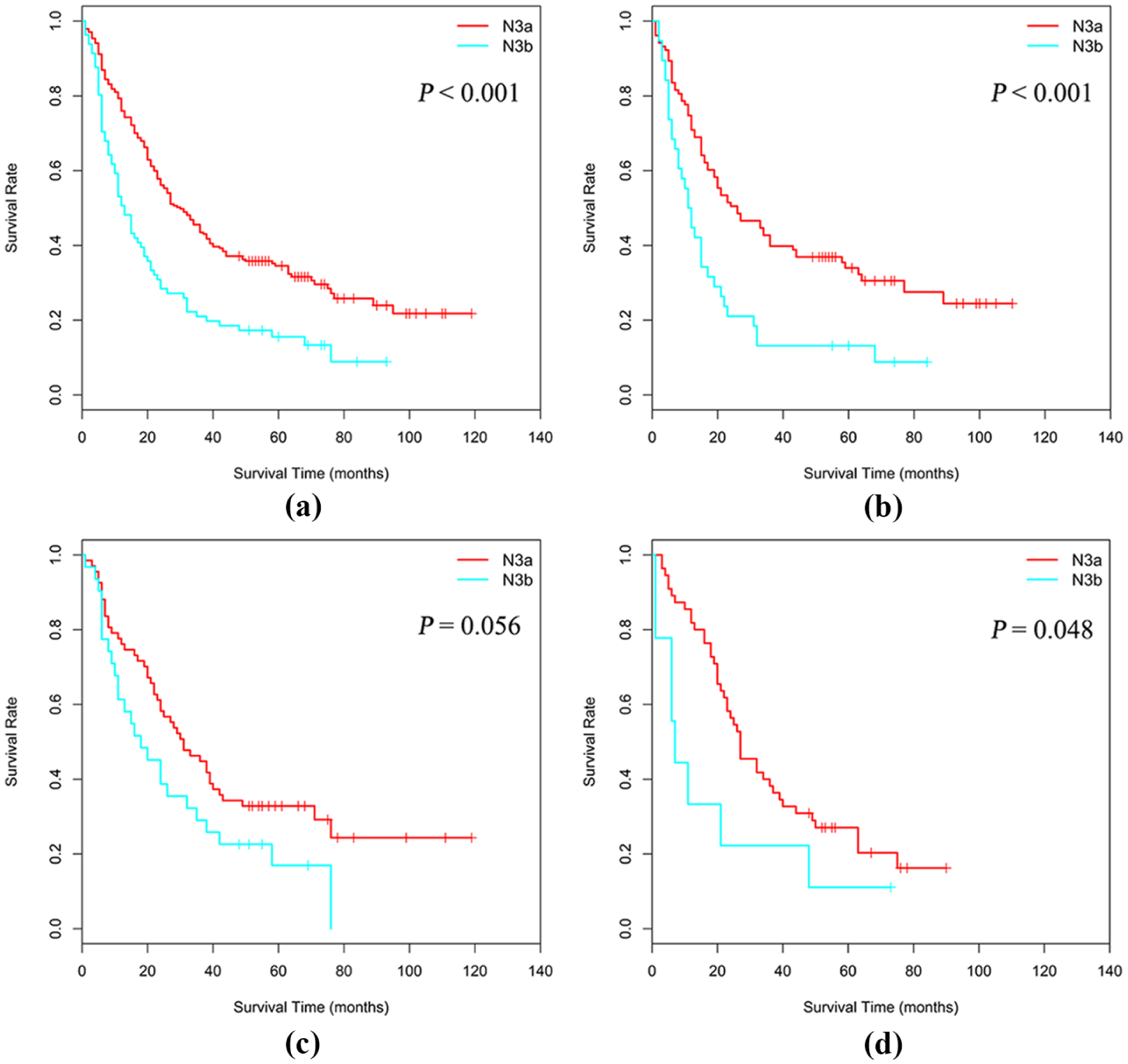
Comparison of OS between stages pN3a and pN3b stratified according to pT stage. (a) pT1-4b. (b) pT3. (c) pT4a. (d) pT4b. Patients (pT1-4b) with pN3a experienced significantly longer OS compared with those with pN3b (5-year OS: 34.5% vs. 15.6%, P < 0.001). Similarly, patients with pN3a experienced longer OS compared with those with pN3b when stratified according to pT stage (pT3: 5-year OS, 33.9% vs. 13.2%, P< 0.001; pT4a: 32.8% vs. 16.9%, P = 0.056; pT4b: 17.0% vs. 11.1%, P = 0.048). P was calculated using the log-rank test.
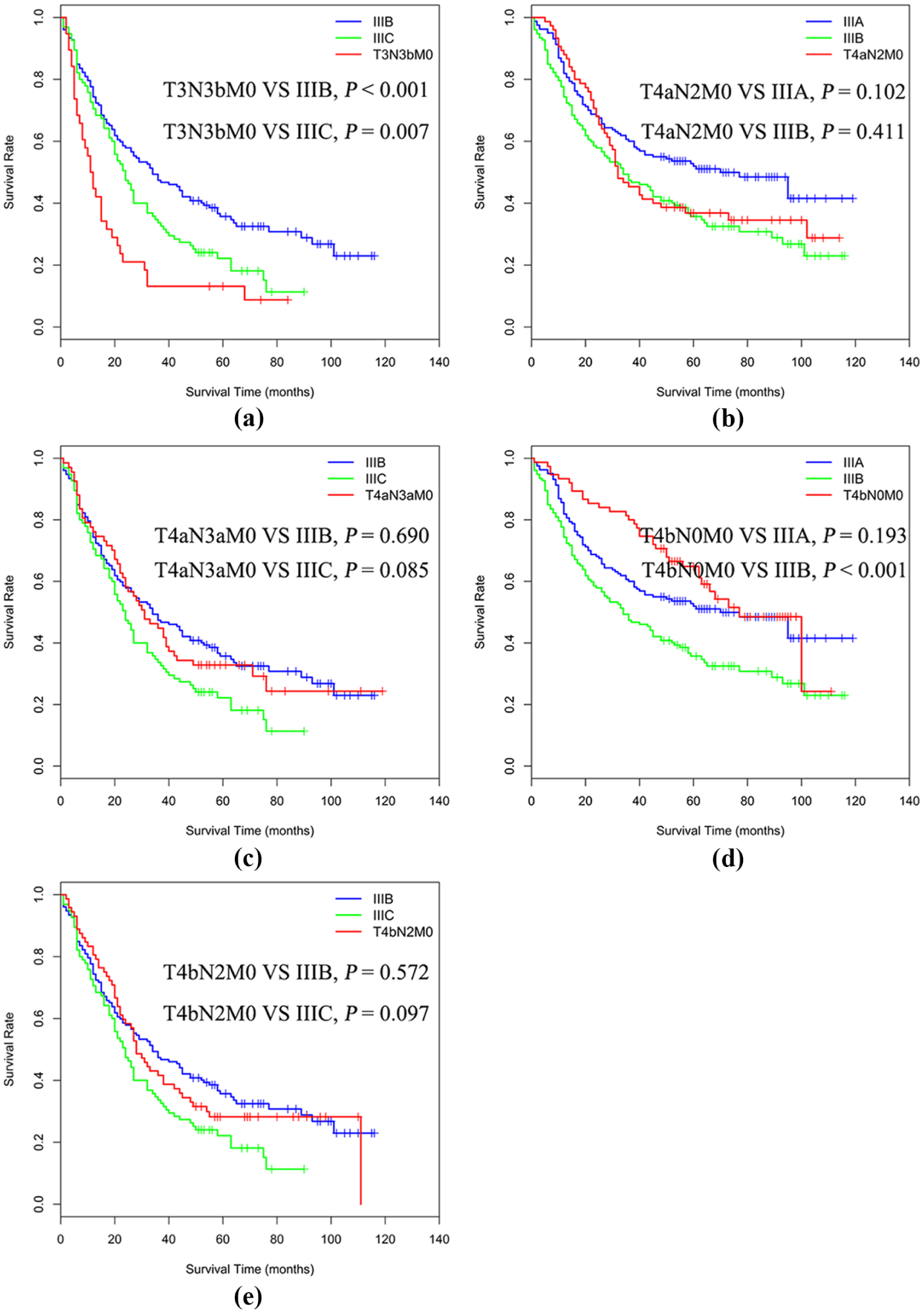
Comparison of OS among groups with stage migration according to the 8th edition of the TNM staging system. (a) T3N3bM0 shifts from IIIB to IIIC. (b) T4aN2M0 shifts from IIIB to IIIA. (c) T4aN3aM0 shifts from IIIC to IIIB. (d) T4bN0M0 shifts from IIIB to IIIA. (e) T4bN2M0 shifts from IIIC to IIIB. P was calculated using the log-rank test.
We further compared the advantages and disadvantages of the two editions for determining patients’ prognoses. The 8th edition had a higher likelihood ratio chi-squared (320.599 vs. 285.263) and C-index (0.701 vs. 0.685, P < 0.001) compared with those of the 7th edition. Similarly, the 8th edition had a smaller AIC value (8288.061 vs. 8323.398). Given the influence of the number of lymph nodes on the staging system, we further conducted a stratified analysis and found that the 8th edition had a higher likelihood ratio chi-squared value (144.139 vs. 138.383) and C-index value (0.691 vs. 0.684, P = 0.111) for patients with <15 lymph nodes. Similarly, the 8th edition had a smaller AIC value (3630.363 vs. 3636.119). The 8th edition had a higher likelihood ratio chi-squared value (196.179 vs. 161.816) and C-index value (0.718 vs. 0.689, P < 0.001) for patients with ⩾15 lymph nodes. Similarly, the 8th edition had a smaller AIC value (3783.672 vs. 3818.035) (Table 2).
Comparison of performance of 7th and 8th editions of AJCC TNM staging system for evaluating prognosis of patients with GC.
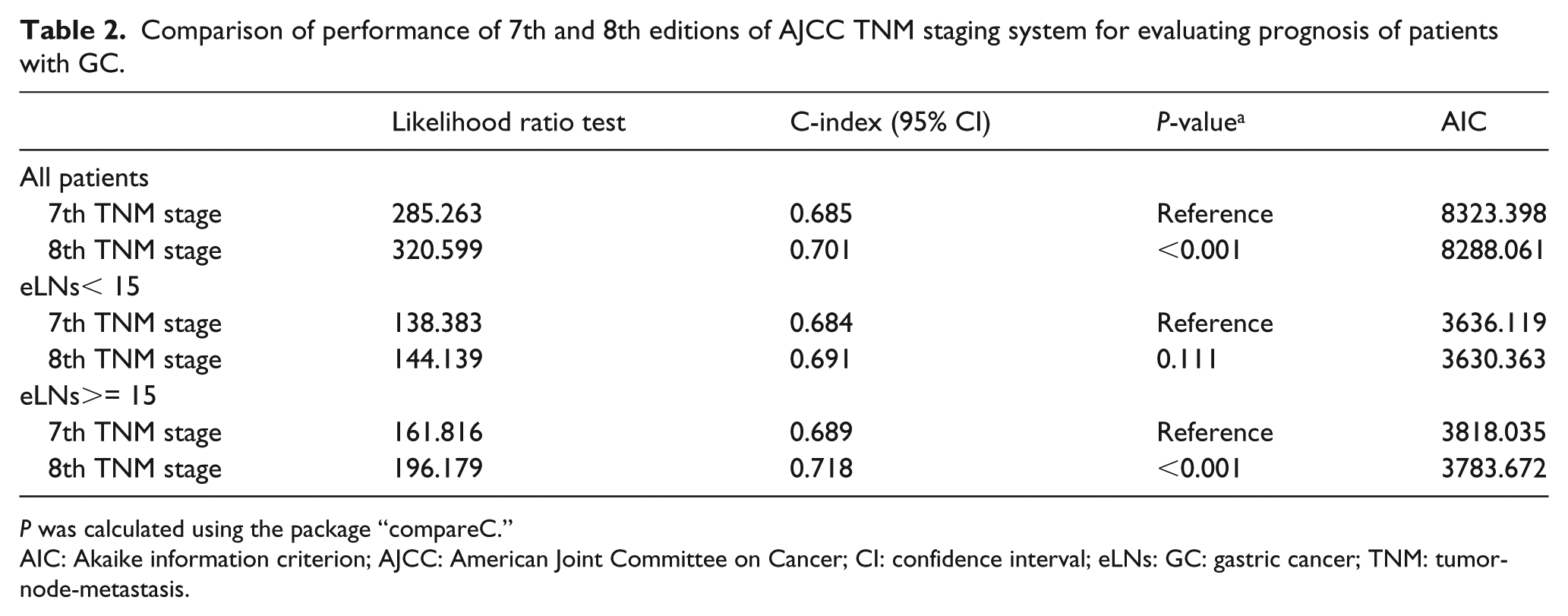
P was calculated using the package “compareC.”
AIC: Akaike information criterion; AJCC: American Joint Committee on Cancer; CI: confidence interval; eLNs: GC: gastric cancer; TNM: tumor-node-metastasis.
Discussion
Accurate staging is important for judging a patient’s prognosis and guiding clinical decision making. The TNM staging system, which was developed and maintained by the AJCC and adopted by the UICC, was first published in 1977 and has subsequently undergone significant revisions. The TNM staging system for gastric cancer is the most authoritative and widely used resource for evaluating the prognoses of patients with gastric cancer. The 8th edition was revised according to the recommendations of the IGCA. Unlike the 7th edition, the 8th edition does include modifications of the definitions of tumor infiltration (T), lymph node metastasis (N), or distant metastasis (M) stages. The 8th edition includes the major adjustments as follows: (i) distinguishes pN3 staging; and (ii) adjusts subgroups. 9 Numerous studies verify the applicability and superiority of the 8th edition.10-12
Specifically, in the 7th edition, the pN3 stages are divided into pN3a and pN3b stages, but are classified as the pN3 stage in a unified manner. For example, Sun et al. 13 retrospectively analyzed the clinical and pathological data of l998 patients undergoing radical surgery for gastric cancer and found that the 8th edition more accurately defines the N3a and N3b stages as independent components. Compared with the 7th edition, the N3a and N3b stages are independent classifications in the 8th edition. Our present study provides further support for the rationale applied to introduce this revision. For example, univariate analysis shows that patients with pN3a gastric cancer experienced significantly longer survival compared with those diagnosed with pN3b. Furthermore, our analysis stratified according to pT shows that patients diagnosed as pN3a experienced significantly longer survival compared with those diagnosed as pN3b.
Compared with the 7th edition, the 8th edition revises subgroups as indicated below. Similarly, we verified the rationale for implementing these adjustments. Specifically, we show here that applying the criteria of the 8th edition better distinguishes stage-related differences in survival. For example, differences in survival after adjustments are diminished compared with those before adjustment for T3N3bM0 (IIIB→IIIC), T4aN3aM0 (IIIC→IIIB), T4bN0M0 (IIIB→IIIA), and T4bN2M0 (IIIC→IIIB), and are greater compared with those before adjustment only for T4aN2M0 (IIIB→IIIA).
According to a theory proposed by Ueno et al., 14 the criteria for evaluating the performance of staging systems are as follows: (i) homogeneity within subgroups (small differences in survival among patients within the same stage); (ii) the ability to discriminate different groups (greater differences in survival among patients with different stages); and (iii) monotonicity of gradients as shown by the association between stages and survival rates (patients in earlier stages survive longer than those in later stages).
We show here that the 8th edition is superior to the 7th edition with respect to its consistency and discriminative ability. Our analysis stratified according to the number of lymph nodes further suggests that the 8th edition is superior to the 7th edition. Note that there was no statistically significant difference between the abilities of the two editions to distinguish differences in the survival of patients diagnosed as stage pN3a with <15 involved lymph nodes. This may be explained by the limitations of the 8th edition, which do not account for the number of biopsied lymph nodes. In contrast, patients diagnosed as pN3b required ⩾15 lymph nodes for biopsy.15-17 However, the number of patients with an adequate number of lymph node biopsies (⩾15) is increasing annually worldwide, suggesting that the value of the 8th edition will be more obvious in the future.18, 19
Here we found that the applicability of the 8th edition of the TNM staging system to Chinese patients with gastric cancer is equal to that of the previous edition. However, some limitations of our analyses should be noted. First, we were unable to determine whether it is reasonable to divide pN3 into pN3a and pN3b for patients diagnosed with pT1 and pT2 disease. This is explained by fewer patients with pT1-2 disease whose lymph node metastasis stage was pN3. Second, we conducted a retrospective study of data acquired from a single institution. Thus, the validity of our findings must be ascertained through multicenter studies.
In summary, the present study verifies the rationale of the 8th edition of the TNM for adjusting the pN3 and subgroup stages. Moreover, the 8th edition was superior to that of its predecessor with respect to its consistency and discriminative ability. Therefore, we recommend the application of the 8th edition of the TNM staging system to Chinese patients with gastric cancer.
Supplemental Material
Supplementary_Material_1 – Supplemental material for Comparing prognostic values of the 7th and 8th editions of the American Joint Committee on Cancer TNM staging system for gastric cancer
Supplemental material, Supplementary_Material_1 for Comparing prognostic values of the 7th and 8th editions of the American Joint Committee on Cancer TNM staging system for gastric cancer by Ming-hua Zhu, Ke-cheng Zhang, Ze-long Yang, Zhi Qiao and Lin Chen in The International Journal of Biological Markers
Footnotes
Author contributions
Study design and concept: MHZ and KCZ contributed equally to this work. MHZ, KCZ and ZLY. Data acquisition: MHZ and KCZ. Data analysis and interpretation: MHZ, KCZ, ZLY, ZQ and LC. Collection of clinical data and sample disposal: ZLY, ZQ and LC. Manuscript preparation: ZLY, ZQ and LC. Manuscript review: ZQ and LC. All authors read and approved the final manuscript.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
