Abstract
Objective:
To investigate the role of programmed death-1 (PD-1), programmed death-ligand 1 (PD-L1), and P16 in patients with head and neck squamous cell carcinoma (HNSCC).
Methods:
A total of 95 paraffin-embedded samples of tumorous tissue of HNSCC were collected. Expression levels of PD-1, PD-L1, and P16 were determined by immunohistochemistry.
Results:
A significantly higher proportion of PD-1 among patients infected with the human papillomavirus was found. PD-L1 expression is closely associated with the primary site of the tumor, postoperative recurrence, survival, PD-1 expression and P16 expression. Univariable analysis indicated that T stage, N stage, tumor node metastasis stage, tumor differentiation, and PD-L1 expression were all shown to be prognostic variables for overall survival in patients with HNSCC. In the multivariate analysis, only N stage (P = 0.010) and PD-L1 expression (P = 0.001) were found to be independent prognostic variables for overall survival. In addition, for disease recurrence, multivariate analysis showed that only PD-L1 expression was the associated independent risk factor. For the patients with negative PD-L1 expression, Kaplan-Meier analysis revealed that they had significantly worse outcomes in terms of overall survival (P = 0.001). Similarly, compared with the patients with positive PD-L1 expression, those with negative PD-L1 expression had a higher probability of recurrence (P = 0.026).
Conclusions:
The expression of PD-L1, PD-1, and P16 in HNSCC is significantly correlated. Human papillomavirus infection (P16 positive) is negatively related to postoperative recurrence. HNSCC patients with positive PD-L1/PD-1 expression tend to have better overall survival outcomes and lower probability of recurrence, providing more evidence for the PD-l-targeted immunotherapy of HNSCC.
Keywords
Introduction
Worldwide, head and neck cancers remain the sixth leading cause of cancer incidence and mortality, of which squamous cell carcinoma accounts for more than 90%. 1 Head and neck squamous cell carcinoma (HNSCC) includes nasopharyngeal carcinoma, nasal sinus, oropharynx, hypopharynx, larynx, and oral cancer. The 5-year survival rate of the patients with HNSCC is about 40% to 60%, but the median survival time after palliative chemotherapy is only 6 to 9 months for patients with local recurrence and distant metastasis, and the survival time for patients with chemotherapy tolerance has reduced to 3 to 6 months. 2
In recent years, with the continuous development of immunology, tumor immunotherapy has gradually become one of the most promising anti-tumor therapies. Immunosuppressive agents, represented by programmed death-1 (PD-1) inhibitors, have obtained a certain therapeutic effect in various malignant tumors. PD-1 is mainly expressed in activated T lymphocytes, B lymphocytes and macrophages. Programmed death-ligand 1 and 2 (PD-L1 and PD-L2) are the two ligands of PD-1—the main one being PD-L1. Although many targeting drugs have been approved for the treatment of many malignant tumors, the progress of immunotherapy for HNSCC is still relatively slow. 3 Further research on the expression level and the function role of PD-1/PD-L1 in HNSCC may provide more evidence for the clinical therapy of HNSCC.
The mechanism of the PD-1/PD-L1 pathway is not yet fully defined, and the mechanism of action in the HNSCC is also not clear. As an important immunomodulator in immune regulation, PD-1 is expressed in activated monocytes, dendritic cells, natural killer cells, B lymphocytes, and tumor-infiltrating CD8+T cells and CD4+T cells.4,5 PD-L1 is expressed in antigen-presenting cells, monocytes, macrophages, activated T lymphocytes and B lymphocytes, as well as a variety of tumor cells. 6 According to the existing studies, PD-L1 are mainly expressed in two ways 7 : in the persistent way, it is regulated by oncogenes of the tumor cells, which are induced by EGFR, ALK/STAT3, PI3K-AKT, and other signaling pathways, and in this way, the expression of PD-L1 does not relate to the inflammatory response8-10; in the non-persistent way, PD-L1 is regulated by T lymphocytes in the immune system, and the expression of PD-L1 this way is induced by the inflammatory signal produced during the immune response process, which prevents the body from immune damage caused by infection-induced inflammation. 11 A large number of studies have shown that the upregulation of PD-L1 expression on the cell surface is not only related to tumor cells, but also to chronic infection.12,13
At present, a large number of studies have proved that the human papillomavirus (HPV) infection is closely related to the occurrence of head and neck cancer,14-16 of which people are gradually becoming aware. According to the HPV status, patients with HNSCC can be divided into two groups: HPV positive and HPV negative. 17 In HNSCC, the HPV infection is closely related to the expression of P16, and P16 protein can be used as a substitute biomarker for HPV infection. 18 The HPV integrates its own DNA into the DNA of the above cells by infecting the skin and the squamous epithelial cells of the mucous membrane. The DNA of HPV is used to synthesize eight viral proteins to regulate the life cycle of HPV, among which E6 and E7 play an important role in the process of HPV-induced tumorigenesis. The E6 protein mainly induces the degradation of P53 by convening E6-related proteins, and the degradation of P53 causes the cells to lose control of the G1/S and G2/M cell cycle, which further promotes the DNA damage of infected cells. E7 is combined with the retinoblastoma protein (pRb protein) to form a complex, and pRb protein is combined to lose its function and is degraded by a ubiquitin protease. pRb protein has the function of inhibiting the E2 F transcriptional factor, and the degradation of pRb protein causes the E2 F transcription factor to lose effective inhibition, which also causes the cell to enter the S phase earlier. In addition, the downregulation of pRb protein can induce the expression of P16 gene, so in the case of HPV infection, the expression of P16 protein is upregulated by a series of the HPV virus protein.19-21
In this study, we investigated the role of PD-1/PD-L1 in HNSCC. We also investigated the relationship between PD-1/PD-L1 and P16 expression.
Subjects and methods
Subjects
From January 2010 to December 2015, a total of 95 cases of paraffin-embedded tissue specimens of HNSCC were collected in the Sun Yat-sen University Cancer Center; all cases were confirmed by pathological diagnosis. Radiotherapy, chemotherapy, or immunotherapy were not conducted before the operation and sample acquisition. The follow-up period was defined as the interval between the date of operation and the date of death or the last follow-up. This study was approved by the Medical Ethics Committee of Sun Yat-sen University Cancer Center. Because all the specimens used were anonymous, the Medical Ethics Committee of Sun Yat-sen University Cancer Center waived the need for informed patient consent.
Immunohistochemistry
All the specimens were fixed with 4% paraformaldehyde in 0.1 M phosphate buffer for 24 h and embedded in paraffin and then sectioned into 4-μm sections and mounted on glass slides. After dewaxing, the slides were treated with 3% hydrogen peroxide in methanol and blocked with a biotin blocking kit (Dark, Germany). After blocking, the slides were incubated overnight with PD-1 antibody (1:1000; Proteintech, USA) or PD-L1 (1:1000; Proteintech, USA) or P16 (1:1000; Santa Cruz, USA) in a humid chamber at 4°C, washed three times with phosphate buffered saline (PBS), incubated for 1 h with biotinylated goat anti-mouse antibody, and then stained with 3,3′-diaminobenzidine tetrahydrochloride. Finally, these sections were stained with hematoxylin and observed under a microscope. Semi-quantitative immunohistochemistry (IHC) was used to detect the protein expression levels of epidermal fatty acid binding protein, according to the following standard scores: “0” (negative staining), “1” (weak staining), “2” (moderate staining), and “3” (strong staining). The final score was calculated as the percentage of positive expression multiplied by the intensity score, which was independently determined by two pathologists. The median IHC score was used as the cut-off to define high and low expression levels.
Statistical analysis
All statistical data were analyzed by SPSS software (IBM, Standard Version 21). The relationship between the expression of PD-1, PD-L1, and P16 in HNSCC with the clinicopathological features of the patients was estimated by the chi-square test. The differences in survival curves were compared using the Kaplan-Meier and log-rank tests, and the independent prognostic factors of the patients with HNSCC were analyzed by the Cox proportional risk regression model. A P value < 0.05 (bilateral test) was considered statistically significant in all the examinations.
Results
Expression of PD-1/PD- L1 and P16 in HNSCC
We performed IHC of PD-1/PD-L1 and P16 on all HNSCC specimens (Figure 1). PD-L1 is mainly expressed on the cell membrane of tumor cells, while PD-1 is expressed in the cytoplasm and is closely related to the expression of P16. Of these 31 PD-L1-negative-expressed patients, 77.42% were PD-1-negative, and among the 64 PD-L1-positive expressed patients, 68.75% were PD-1-positive (P < 0.001), indicating an obviously positive correlation between PD-L1 and PD-1. HPV infection (P16 positive) also showed a positive relationship with PD-1/PD-L1. We found that 63.49% of PD-1-positive tissues were positive for P16-positive tissues (P = 0.007) and 74.60% for PD-L1 expression (P = 0.035; Figure 1).
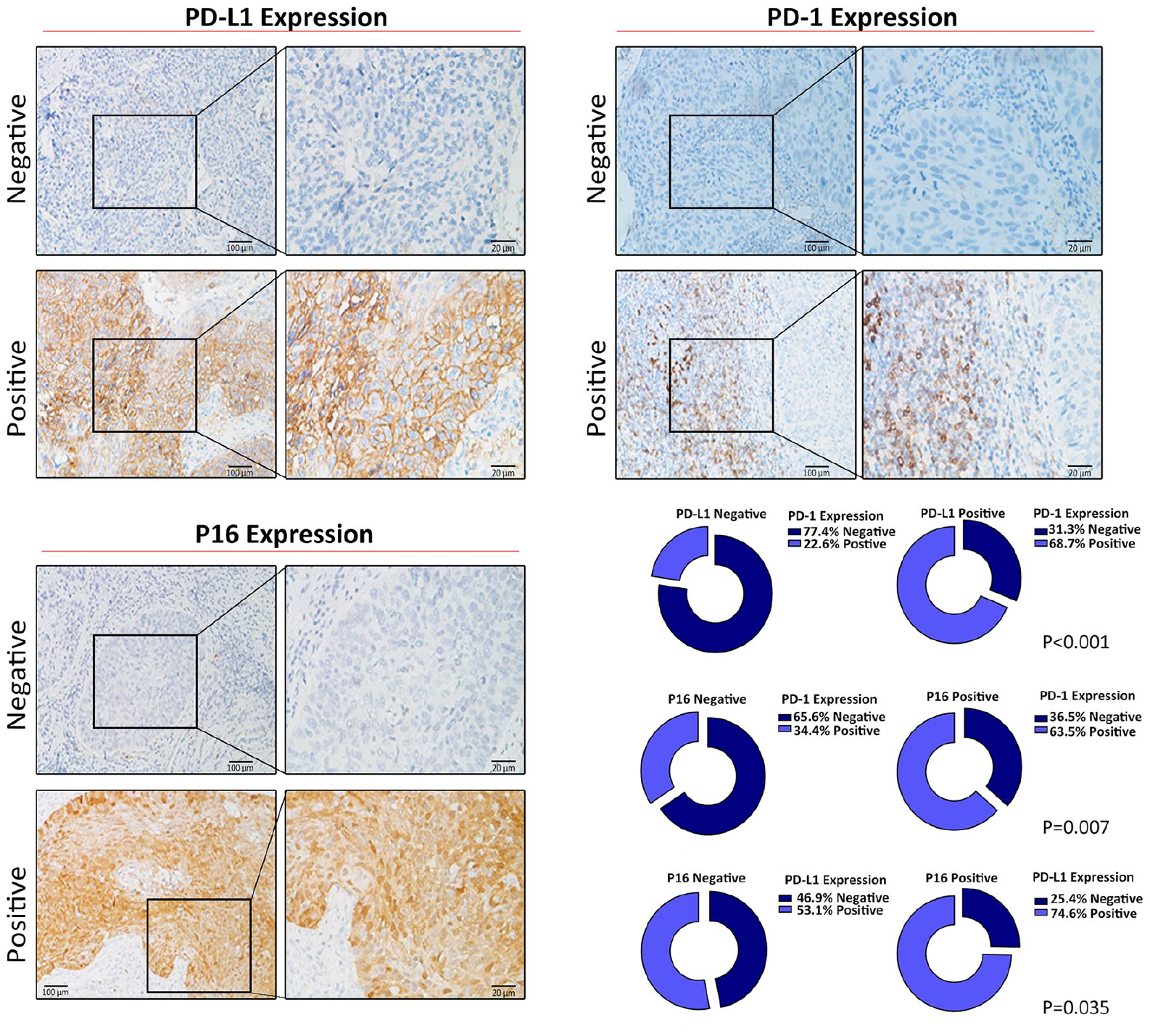
Representative images of HNSCC tissues showing negative and positive PD-1, PD-L1 and P16 expression. (Left panel: magnification 100X; right panel: magnification 400X). In 31 cases of PD-L1 negative tissues, 77.42% of PD-1 expression was negative (P < 0.001). 63.49% of the PD-1-positve tissues were P16-positive (P = 0.007), and 74.6% of them were PD-L1-positive (P = 0.035).
Association of PD-1/PD- L1 and P16 expression with the clinical features of HNSCC
The expression levels of PD-1, PD-L1, and P16, and the demographic and clinicopathological features were analyzed. We found a higher proportion of positive PD-1 expression among the patients infected with HPV, as shown in Table 1. We further compared the patients with positive PD-L1 expression and negative PD-L1 expression. The results indicated that the PD-L1 expression was significantly related to the primary site of the tumor, postoperative recurrence, survival, PD-1 expression, and P16 expression (P < 0.05; Table 2). Then we compared the HPV-infected group with the HPV-uninfected group; the results showed that HPV infection (P16 positive) was negatively related to postoperative recurrence (P < 0.05; Table 3).
Association between PD-1 expression and clinical features of HNSCC.
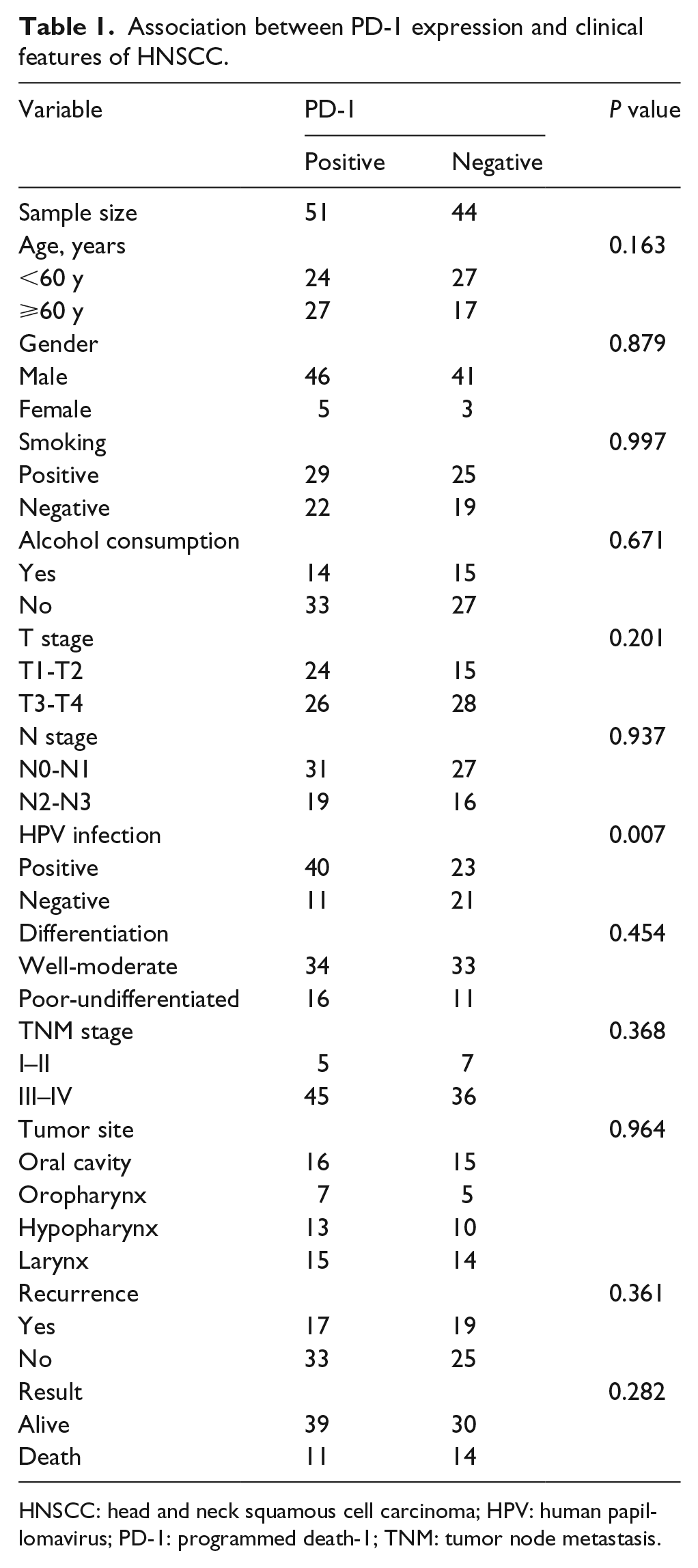
HNSCC: head and neck squamous cell carcinoma; HPV: human papillomavirus; PD-1: programmed death-1; TNM: tumor node metastasis.
Association between PD-L1 expression and clinical features of HNSCC.
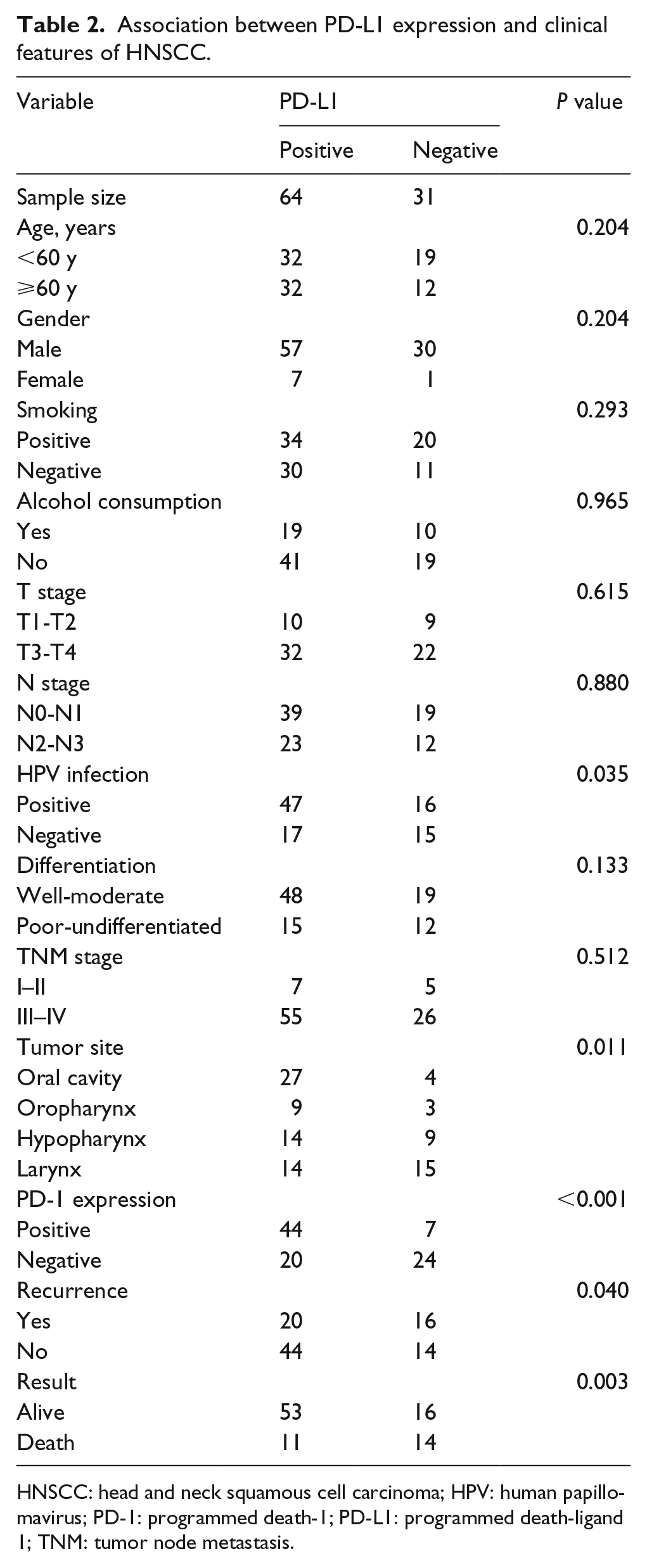
HNSCC: head and neck squamous cell carcinoma; HPV: human papillomavirus; PD-1: programmed death-1; PD-L1: programmed death-ligand 1; TNM: tumor node metastasis.
Association between the HPV infection and the clinical features of HNSCC.
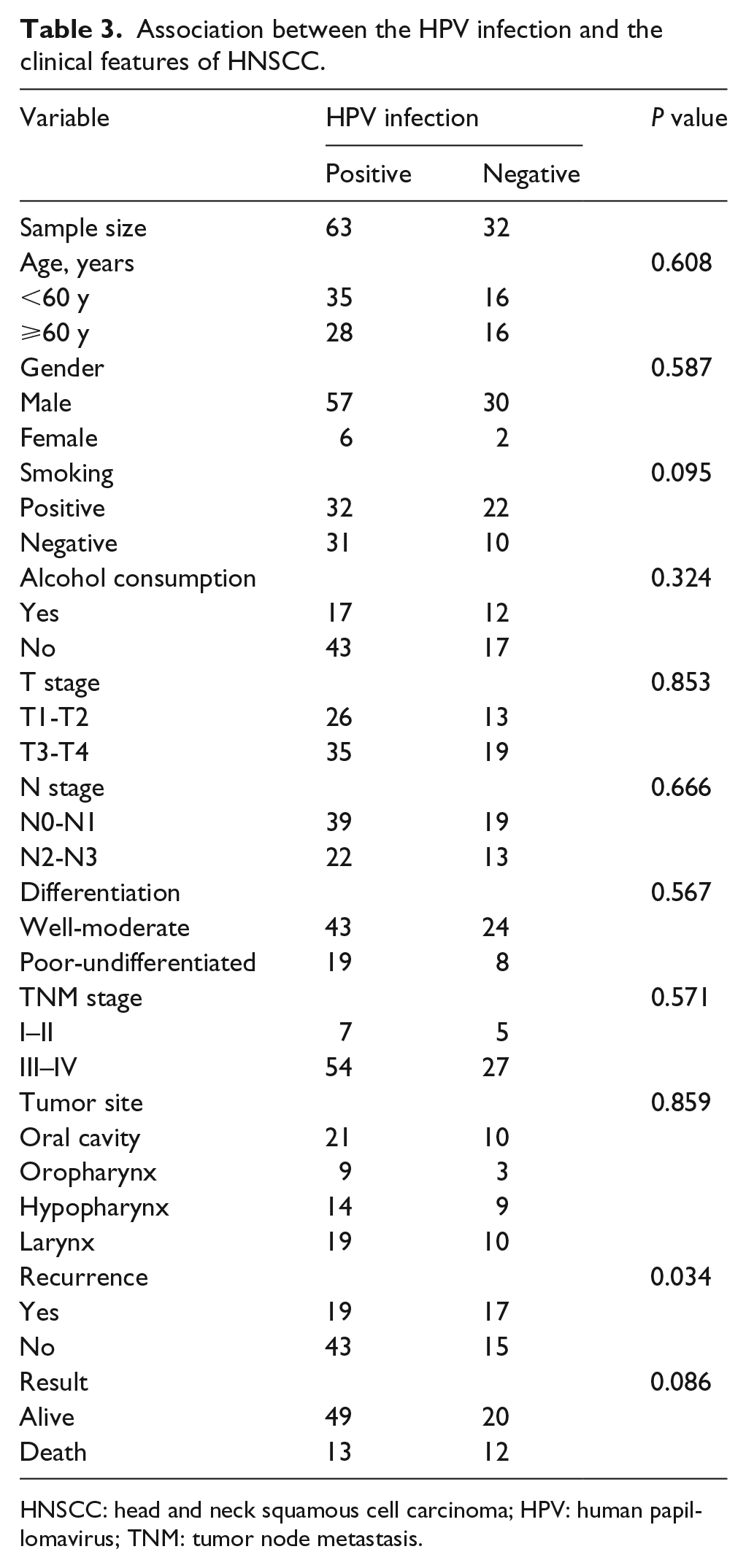
HNSCC: head and neck squamous cell carcinoma; HPV: human papillomavirus; TNM: tumor node metastasis.
Univariate and multivariate analyses of the prognostic variables in HNSCC
To evaluate whether PD-L1, PD-1, or P16 expression was an independent risk factor for survival in HNSCC, both univariate and multivariate analyses were conducted. The T stage, N stage, tumor node metastasis stage, tumor differentiation, and PD-L1 expression were all found to be prognostic variables for the overall survival (OS) in patients with HNSCC. In the multivariate analysis, only N stage (P = 0.010) and PD-L1 expression (P = 0.001; Table 4) were found to be independent prognostic variables for OS.
Univariate and multivariate analyses of prognostic variables for overall survival.
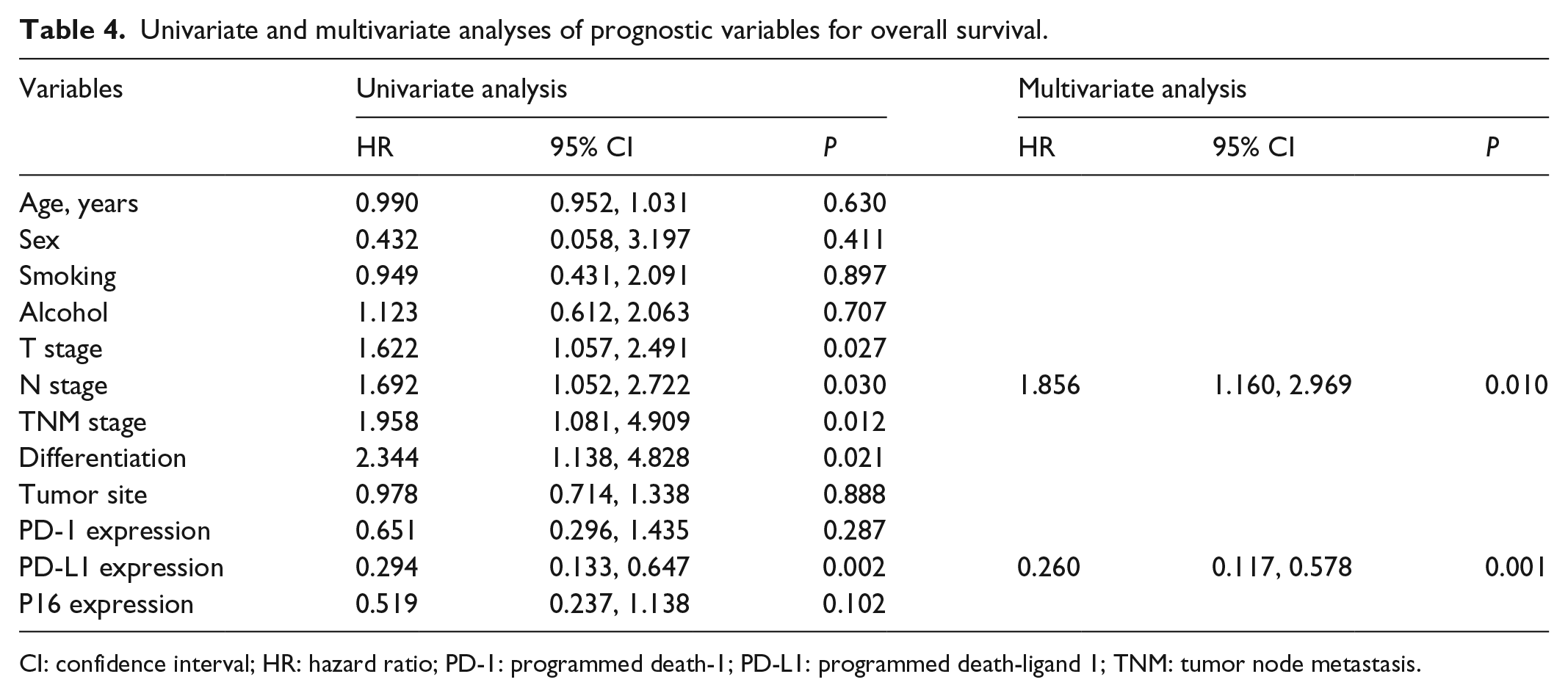
CI: confidence interval; HR: hazard ratio; PD-1: programmed death-1; PD-L1: programmed death-ligand 1; TNM: tumor node metastasis.
We further explored the risk factors associated with recurrence (Table 5). Univariate analysis showed that PD-L1 expression and P16 expression were negatively associated with recurrence. In the multivariate analysis, only PD-L1 expression was revealed to be an associated independent risk factor.
Univariate and multivariate analyses of the prognostic variables for recurrence.
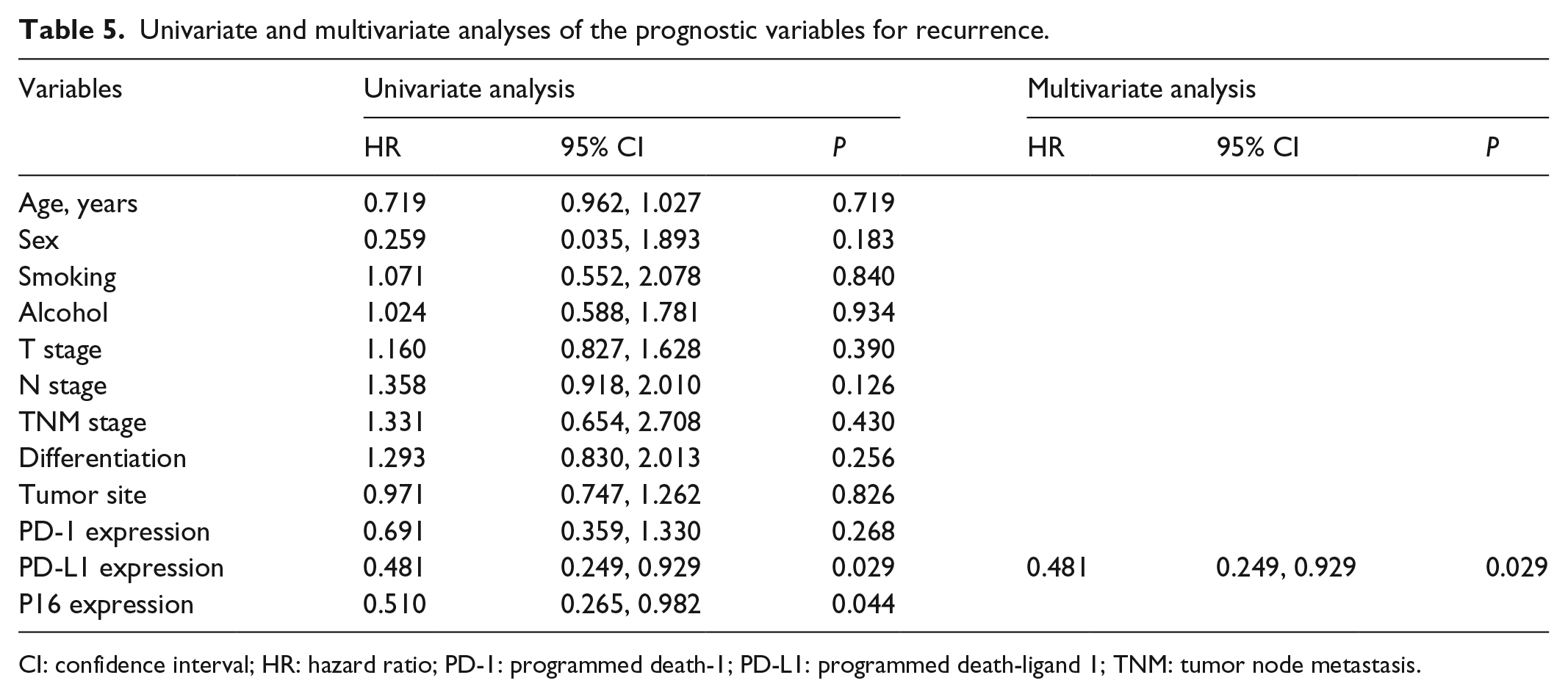
CI: confidence interval; HR: hazard ratio; PD-1: programmed death-1; PD-L1: programmed death-ligand 1; TNM: tumor node metastasis.
Association of PD-L1 and P16 expression with the clinical outcomes in patients with HNSCC
Multivariate analysis indicated that PD-L1 was positively associated with the prognosis of HNSCC. To further determine the prognostic effect of PD-L1 expression on patients with HNSCC, we conducted a Kaplan-Meier survival analysis. For the patients with negative PD-L1 expression, Kaplan-Meier analysis revealed that they had significantly worse outcomes in terms of OS (P = 0.001). Furthermore, the patients with positive PD-L1 expression tended to have a lower probability of recurrence (P = 0.026; Figure 2).
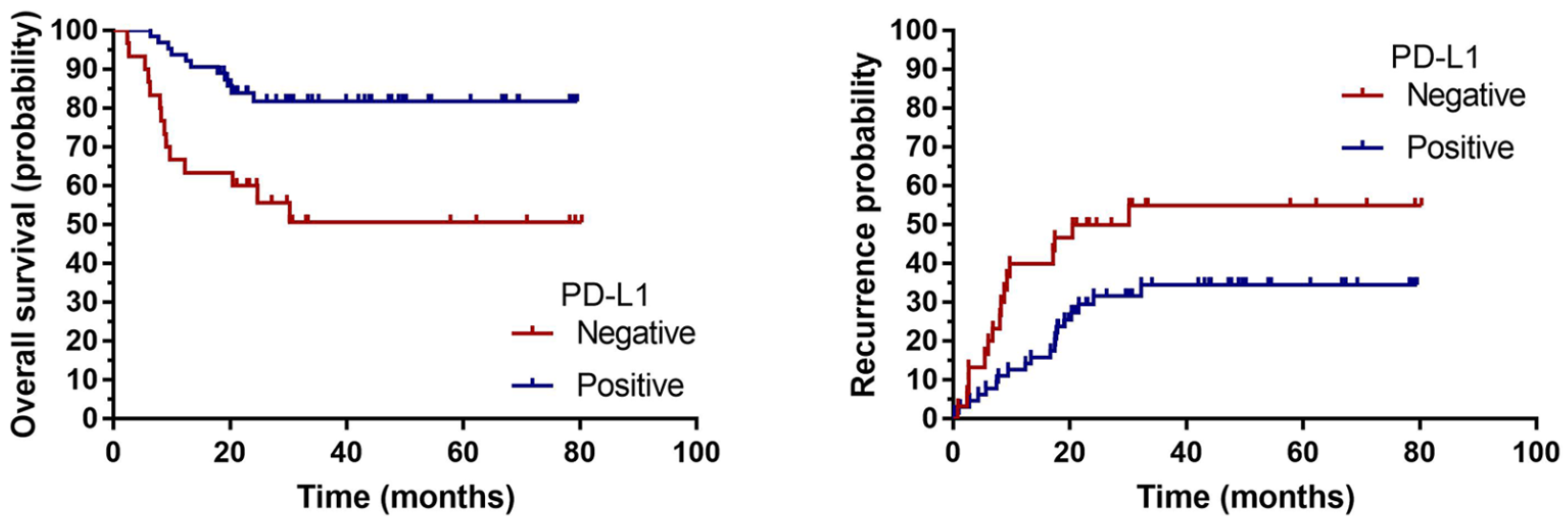
Kaplan-Meier analysis revealed that the patients with negative PD-L1 had significantly worse outcomes in terms of overall survival (P = 0.001). Compared with the patients with positive PD-L1 expression, those with negative PD-L1 expression had a higher probability of recurrence (P = 0.026).
Finally, we compared the HPV-infected group with the HPV-uninfected group, showing that HPV infection (P16 positive) is apparently related to postoperative recurrence (P < 0.05; Table 3).
Discussion
The pathological tissue of 95 cases of HNSCC was detected by IHC staining. The result of the statistical analysis showed that there was obvious correlations between PD-1, PD-L1, and P16 (PD-L1 and P16: P = 0.035 < 0.05; PD-L1 and PD-1: P = 0.000 < 0.05; PD-1 and P16: P = 0.007 < 0.05, all of which were statistically significant), which is in accordance with the results of some existing literature reports. 22 In the P16 positive HNSCC, the positive rate of PD-1 and PD-L1 was significantly higher than that of the P16-negative group. We speculate that the increase of PD-L1 expression on the cell surface may be influenced by the double effects of tumor cell induction and inflammatory mediating, leading to the high expression of PD-L1 in the HPV-infected group, and is also due to the dual effects of tumor and inflammation.8-11 This enables the immune system to mobilize the activated immune cells to infiltrate around the tumor, which also promotes the expression of PD-1 in the immune cells infiltrating around the tumor.
The survival analysis indicated that PD-L1 expression was positively associated with the OS time (OS rate: 82.8% vs. 53.5%; OS: 36.66 months vs. 30.54 months, P = 0.001 < 0.05; Figure 1). PD-1 was mainly expressed on the surface of activated T lymphocytes, B lymphocytes, and macrophages, while stationary phase T lymphocytes did not express PD-1.4,5 In addition to the expression of the tumor cells, endothelial cells, and epithelial cells, PD-L1 can also be expressed in the immune cells—such as dendritic cells, macrophages, B lymphocytes, and T lymphocytes—and its expression is associated with the activation of these immune cells. 6 As a molecule that inhibits immunization and promotes tumor immune escape, PD-L1 turned out to be contrary to its own role in this study. The immune cells at the stationary phase do not express PD-1 and PD-L1; however, once the immune system is activated, the activated immune cells can induce an immune escape for the tumor cells and protect themselves from being attacked and removed by PD-L1. Therefore, a high level of PD-1 and PD-L1 may suggest an activated immune system, in other words, PD-L1-positive HNSCC patients are in a state of immune activation and have a better immune level, indicating a better prognosis.
At present, many studies have found that there are different levels of PD-L1 positive expression in a variety of malignant tumors, and the level of PD-L1 expression is positively related to the efficacy of the drugs. In the American Ib phase clinical trial of PD-1 inhibitors of HNSCC (KEYNOTE-012) and the III phase clinical trial Checkmate-141, the positive expression of PD-L1 was 65% and 57.3%, respectively. In this study, the positive expression rate of PD-L1 was 67.4% in HNSCC, and was 87.1% in oral squamous cell carcinoma, 75% in oropharyngeal squamous cell carcinoma, 60.9% in hypopharyngeal squamous cell carcinoma, and 48.3% in laryngeal squamous cell carcinoma, indicating a high expression in HNSCC and approximately corresponding with other related researches. In the KEYNOTE-012 study, the expression of PD-L1 in tumor cells—which was treated with the PD-1 inhibitor pembrolizumab—was associated with progression-free survival (PFS) and OS. In the extended cohort study, the expression of PD-L1 in tumor cells and immune cells was positively correlated with objective response rate (ORR), PFS, and OS. 23 Checkmate-141 studies showed that, compared with the traditional chemotherapeutic drug, nivolumab contributed no significant improvement in PFS but contributed to an increase in the ORR, duration of overall response (DOR), and OS in PD-L1-positive patients. 24 A phase I/II clinical study of durvalumab also suggested that ORR was higher in PD-L1-positive group than in the PD-L1-negative group. 24 These clinical studies show that the expression level of PD-L1 in HNSCC is positively correlated with the efficacy of the target drugs. Therefore, the quality and efficiency of the clinical treatment can be improved by screening the HNSCC patients on their expression level of PD-1 and PD-L1, which significantly benefits patients.
Conclusion
This study was designed to clarify the correlation between PD-1, PD-L1, and P16 expression (as estimated by IHC) and the impact of their expression on clinicopathological parameters—including prognosis—in HNSCC. Although these current results need further validation, they may help us achieve a deeper understanding of the roles that PD-1, PD-L1, and P16 play in HNSCC, providing more evidence for clinical treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Natural Science Foundation of China (No. 81802713) and the Science and Technology Program Fund of Guangdong Province (No. 2016A020215090).
