Abstract
Purpose:
miR-491 functions as a tumor suppressor in several types of cancer. However, its function and mechanism in gastric cancer proliferation and metastasis have not been well defined. The aim of this study was to explore the role and regulatory mechanism of miR-491 in cell proliferation and migration in gastric cancer.
Methods:
Quantitative reverse transcriptase-polymerase chain reaction (qRT-PCR) was used to detect the expression pattern of miR-491 in gastric cancer tissues. miR-491 overexpression vector, miR-491 inhibitor, and siHMGA2 were used; and MTT, wound healing, and transwell assays were employed to examine proliferation and migration for BGC-823 cells. A dual-luciferase reporter gene was used to measure the target relationship between miR-491 and HMGA2.
Results:
Most gastric cancer patients exhibit decreased miR-491 expression. miR-491 overexpression inhibited cell proliferation and migration, whereas miR-491 inhibitor treatment produced the opposite effect. Mechanistically, HMGA2 was identified as a direct target of miR-491. Moreover, HMGA2 knockdown inhibited cell proliferation and migration, which was similar to the effect of miR-491 overexpression. HMGA2 was decreased after transfection of the miR-491 vector and increased after transfection of the miR-491 inhibitor.
Conclusion:
Our results suggest that miR-491 suppressed cell proliferation and cell motility in gastric cancer by targeting HMGA2. Silencing HMGA2 produced a similar effect to miR-491 overexpression on cell proliferation and migration. miR-491/HMGA2 signaling may be a potential therapeutic target for gastric cancer patients with decreased miR-491 expression.
Introduction
Although much progress has been made in clinical treatment, the 5-year survival rate of gastric cancer is still poor. A growing number of studies have focused on the elucidation of molecular mechanisms underlying the progression of gastric cancer, which may provide a basis for novel therapeutic targets invention. MicroRNAs are a class of small non-coding nucleotides that can negatively mediate target gene expression and act as oncogenes or tumor suppressors. These small molecules bind to 3′-untranslated regions (3′-UTRs) of messenger RNA (mRNA) due to complete or incomplete complementary sequences between miRNAs and mRNAs, and thereby prohibit translation or degrade mRNA. One miRNA, miR-491, has been reported to be deregulated in gastric cancer and to inhibit cell proliferation in vitro and in vivo. 1 miR-491 reportedly induces cell apoptosis by targeting the anti-apoptotic factor Bcl-xl in several cancers.2-4 Nonetheless, the role and mechanism of miR-491 in gastric cancer metastasis still need to be clarified. Therefore, we used several bioinformatics tools to predict potential target genes of miR-491. Considering the comprehensive information obtained regarding miR target genes, HMGA2 was selected as a potential target gene of miR-491.
HMGA2 belongs to the non-histone chromosomal high mobility group (HMG) protein family. HMG proteins play an important role by acting as architectural factors; also they are an important component of enhancer complexes. HMGA2 functions as a transcriptional regulating factor since it contains structural DNA-binding domains. HMGA2 is highly expressed in human laryngeal carcinoma, 5 renal cell carcinoma, 6 non-small cell lung cancer (NSCLC), 7 and esophageal adenocarcinoma (EAC) 8 tissues, and its overexpression has been associated with shorter survival in NSCLC and EAC patients. Meta-analysis results have shown marked associations between HMGA2 overexpression and tumor node metastasis (TNM) stage, lymph node status, vascular invasion, and shorter overall survival in gastric cancer patients. 9 HMGA2 is a promising prognostic biomarker for gastric cancer. The question of whether miR-491 plays a role in gastric cancer by targeting HMGA2 needs to be studied.
The aim of the present study was to reveal the function and regulatory mechanism of miR-491 in gastric cancer metastasis.
Materials and methods
Gastric cancer tissues and cancer-derived cell lines
In this study, 25 pairs of gastric cancer and adjacent normal tissues were obtained from patients. All specimens were collected from the First Affiliated Hospital of Xi’an Jiaotong University. Patients without cancer history, pre-operative radiotherapy, or chemotherapy were included in the study. The information of all the tissues are summarized in Table 1. All specimens were frozen at −80℃. This study was approved by the Ethics Committee of Xi’an Jiaotong University, informed consent was obtained from patients. Cells were cultured in RPMI-1640 medium (Thermo-Scientific Hyclone, USA) with 10% fetal bovine serum (FBS) added at 37℃ and 5% CO2.
Clinical data among 25 patients with gastric adenocarcinoma.
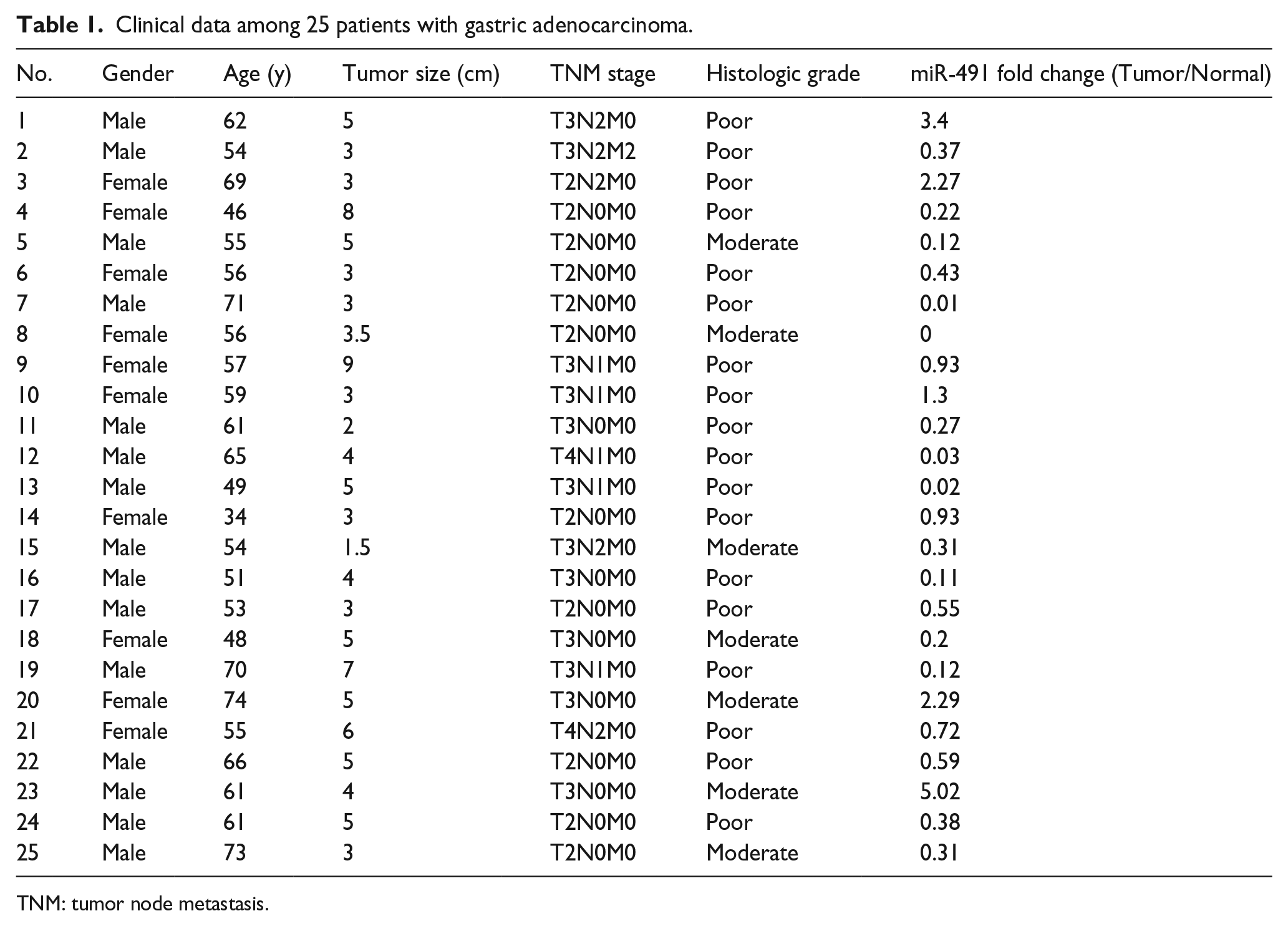
TNM: tumor node metastasis.
RNA extraction and quantitative reverse transcription-polymerase chain reaction (qRT-PCR)
Total RNA was isolated using Trizol reagents (Invitrogen, Carlsbad, CA, USA). PrimeScript RT reagent (Takara, Japan) was used to generate the cDNA. Then the quantitative reverse transcriptase-polymerase chain reaction (qRT-PCR) was conducted on the FTC-3000TM system (Funglyn Biotech Inc., Toronto, Canada) using SYBR Green Master Mix (Takara, Japan). GAPDH and U6 were treated as controls. The fold changes of mRNA and miR-491 were quantified by 2−ΔΔCT calculation. Each sample was tested the qRT-PCR in triplicate. All primer sequences are shown in Supplementary Table 1.
Expression vector construction
In short, miR-491 (pre-miR-491) overexpression vectors were constructed by cloning into pcDNA6.2-GW/EmGFP vector (Invitrogen, USA) as previously mentioned.10,11 After analyzing the targeting information from public databases, such as miRbase, Targetscan, and miRTarBase, HMGA2 was considered as the candidate target of miR-491. Several potential miR-491 binding sites on HMGA2 3′-UTR were predicted. The binding sequences in 3′-UTR of human HMGA2 were constructed via cloning into the pmiRGLO Dual-Luciferase miRNA Target Expression Vector (Promega) as previously mentioned.10,11 miR-491 inhibitor and related control were chemically synthesized (GenePharma, China). The vectors were validated by sequencing. The sequence information is provided in Supplementary Table 1.
3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay
As mentioned previously, MTT was used to detect cell proliferation. 12 MTT analysis data reflect cell viability and quantity by detecting the activity of metabolic enzymes in mitochondria of living cells. In short, cells (4000 cells/wells) were inoculated into 96-well plate and cultured for 24 h, 48 h, and 72 h respectively after transfection. Before the prescribed time, MTT reagent (Sigma) was added, after 4 h, the medium was taken out, replaced by dimethyl sulfoxide (Sigma), and then shaken vigorously at room temperature for 10 min. The optical absorbance at 492 nm (OD492) was measured by FLUO star OPTIMA (BMG). Since the absorbance value was positively correlated with the number of living cells, the absorbance value in different groups was compared with the Student T-test in the indicated time. Each test was repeated three times.
Wound healing assay
BGC-823 cells were cultured and scratches were made in wells with sterile plastic head. The plates were washed with phosphate buffer saline (PBS) and cultured in 1% FBS fresh medium, then transfection was conducted. Scratch images were taken with microscope at 0 h, 24 h, and 48 h after transfection. Each well image was photographed in three different areas, and the same area at each time point was taken. The relative wound area was compared in different groups with the Student T-test. Each test was repeated three times.
Transwell migration assay
Migration analysis was carried out by using transwell chambers (8.0 um, Millipore, Billerica, MA, USA). In short, after transfection, 200 μL cells (5 × 105 cells) were added into serum-free medium and 600 μL complete FBS medium was added into the lower chamber. Cell plates were cultured for 24 h, then the cells that stayed in the upper chamber were wiped away with a cotton swab. Cells migrating and remaining on the surface of the membrane were stained with 0.5% crystal violet. The mobility was quantitatively analyzed by solubilization of crystal violet and observing the optical absorbance at 590 nm. The absorbance value in different groups was compared with the Student T-test. Each experiment was conducted three times.
Oligonucleotide and plasmid transfection
According to the manufacturer’s instructions, JetSI-ENDO transfection reagent (Polyplus transfection, French) was used for cell transfection. The transfection efficiency was evaluated by observing distribution and brightness of fluorescence in cells and validating by qRT-PCR (Figure 1(c) and (d)). All the transfection was done in triplicate.
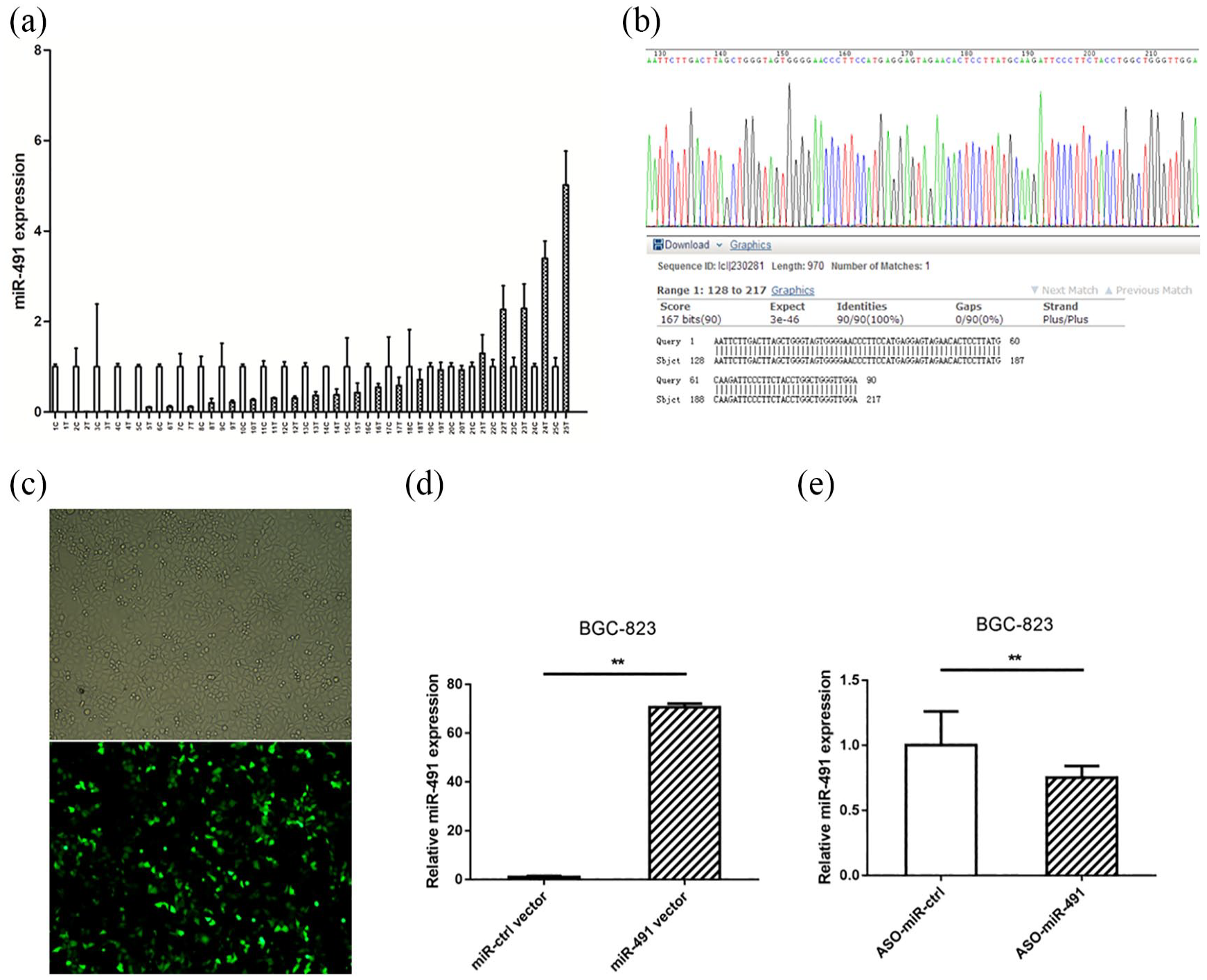
miR-491 was mostly downregulated in gastric cancer and miR-491 overexpression vector was constructed. (a) Most of the gastric patients showed decreased expression of miR-491. (b) The miR-491 overexpression vector was validated by direct sequencing. (c) The transfection efficiency was evaluated by observing GFP fluorescence. (d) miR-491 was highly increased after transfection of miR-491 overexpression vector compare to the cells transfected with microRNA negative controls. (e) miR-491 was slightly decreased after transfection of ASO-miR-491 compared to the cells transfected with scramble nucleotide. (*P < 0.05, **P < 0.01).
Western blot analysis
In short, radioimmunoprecipitation assay cell lysis buffer (Wolsen) was used to extract protein 48 hours after transfection, and the protein concentration was detected by BCA protein detection kit (Thermo Scientific Pierce, China). An equal amount of protein solubilization was added to the band and separated by 10% sodium dodecyl sulfate (SDS) polyacrylamide gel. Then the separated protein was transferred to polyvinylidene fluoride (PVDF) membrane (Millipore Corporation). After incubation in 5% skimmed milk containing 0.1% TBST for 1 h, primary antibodies were added, then the membrane was incubated overnight at 4°C: HMGA2 (bios, China), MMP-2 (cell signaling technology), β-catenin (cell signaling technology). After three rinses, the secondary antibodies (Santa Cruz Biotechnology) were added and the membrane was incubated at room temperature for 1 h. In each sample, the target protein expression level was normalized to GAPDH. Western Lightning chemiluminescence reagent PLUS (Perkinelemer, USA) was used to observe protein bands. The blots’ images were obtained, and image J software was used to quantify the band density.
Dual luciferase assay
BGC-823 cells were incubated and seeded into 96-well plates (12,000 cells per well). Then 100 ng pmiRGLO-HMGA2-3′-UTR vectors, which included wild-type or mutant miR-491 binding sites, and 100 ng pre-miR-491 expression vectors were co-transfected into BGC-823 cells. The pmiRGLO vector was defined as the control. The dual-luciferase analysis system (Promega) was used to detect the activity of luciferase 24 h after transfection. The relative firefly luciferase activity was calculated by normalized to Renilla luciferase activity. Each experiment was conducted three times.
Statistical analysis
All data were shown as mean ± SD of at least three independent experiments. The Student T-test or one-way ANOVA were used to analyze the differences among varied groups. When the p value was less than 0.05, the result was defined as having statistical significance. All data were analyzed by SPSS 17.0 software.
Results
miR-491 was downregulated in gastric cancer and miR-491 overexpression vector was constructed
We used qRT-PCR to assess the expression of miR-491 in 25 pairs of gastric cancer tissue and matched adjacent non-cancerous tissue samples. Compared with the matched normal tissue samples, 20 (80%) gastric cancer samples exhibited decreased miR-491, and 5 (20%) gastric cancer samples exhibited increased miR-491 (Figure 1(a)); these findings were consistent with those of other studies.13,14 With respect to functional analysis, gain- and loss-of-function analyses were performed. The miR-491 overexpression vector was constructed and verified by sequencing (Figure 1(b)). BGC-823 cells were selected for further analysis, and transfection efficiency was validated by observing green fluorescent protein (GFP) in fluorescence microscopy images. As expected, transfection efficiency reached approximately 40% (Figure 1(c)). miR-491 expression was markedly upregulated after transfection of the miR-491 overexpression vector compared with transfection of a control vector (Figure 1(d)), whereas miR-491 expression was slightly downregulated after transfection of anti-sense oligodeoxynucleotide (ASO)-miR-491 compared with transfection of scrambled nucleotide (Figure 1(e)). Notably, up to 80-fold overexpression of miR-491 was observed in vitro.
miR-491 inhibits cell proliferation and migration of gastric cancer cells
To examine the role of miR-491, gain- and loss-of-function experiments were performed. MTT, wound healing, and transwell assays were utilized, and the results showed that miR-491 overexpression in BGC-823 cells produced an inhibitory effect on cell proliferation and migration after transfection (Figure 2(a) to (c)), whereas the miR-491 inhibitor produced the opposite effect on these gastric cancer cells (Figure 2(d) to (f)). These data indicated that miR-491 played a tumor-suppressive role in proliferation and motility in gastric cancer cells.
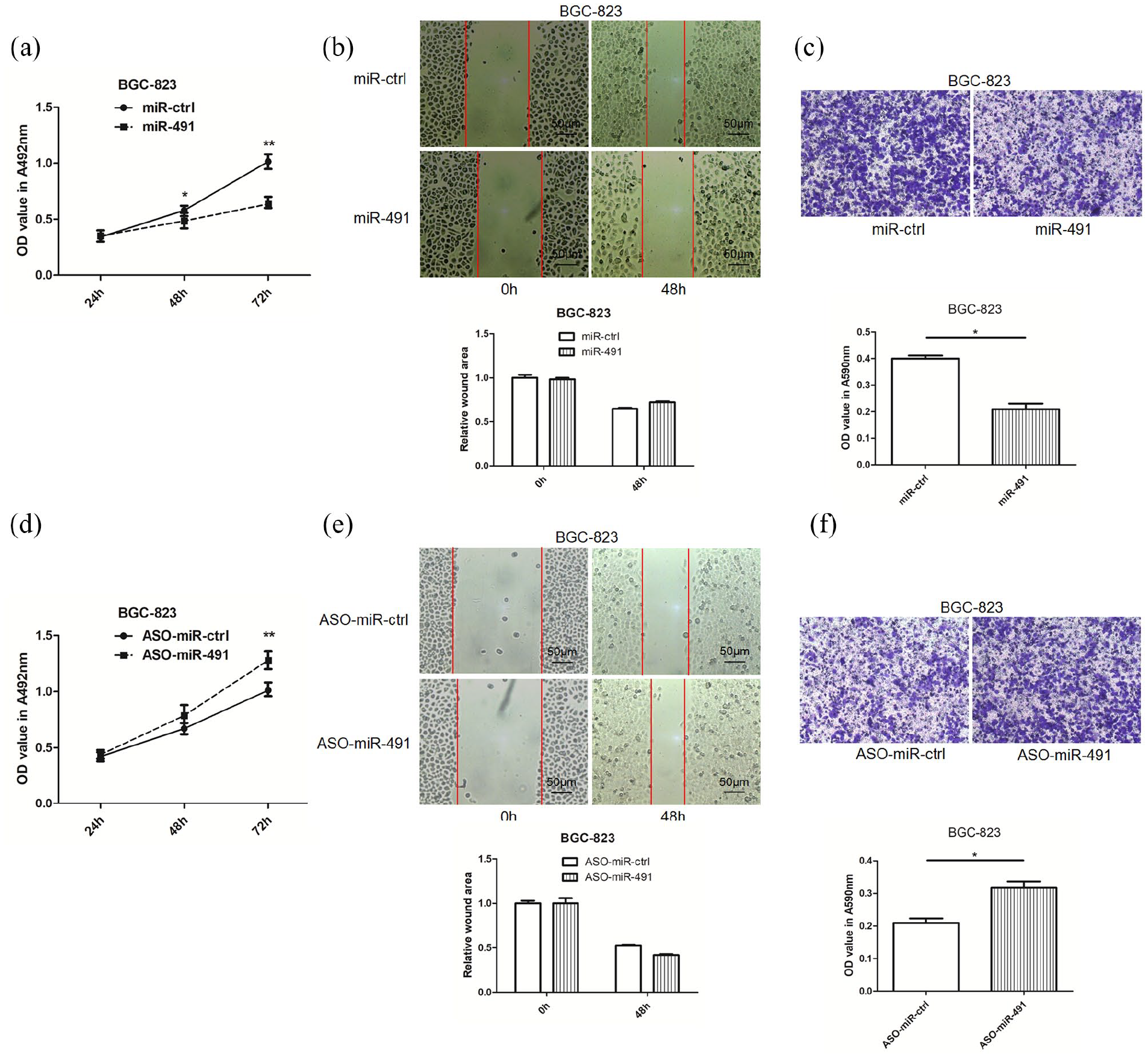
Overexpression of miR-491 inhibits and knockdown of miR-491 promotes cell proliferation and migration in gastric cancer. (a) The effects of miR-491 on gastric cancer cell proliferation were determined by MTT assay after transfection of miR-491 or control vector. (b) The effects of miR-491 on gastric cancer cell migration were determined by wound healing after transfection of miR-491 or control vector. (c) The effects of miR-491 on gastric cancer cell migration were determined by transwell assay after transfection of miR-491 or control vector at 24 h, the migrated cells were stained using crystal, 33% acetic acid was used to dissolve the crystal, then the Student T-test was used to evaluate the OD value at 590 nm obtained by the lysate. (d) The cell proliferation effects of miR-491 inhibitor on gastric cancer was determined by MTT assay after transfection of miR-491 inhibitor or negative control vector. (e) The cell migration effects of miR-491 inhibitor on gastric cancer were determined by wound healing after transfection of miR-491 inhibitor or negative control vector. (f) The cell migration effects miR-491 inhibitor on gastric cancer was determined by transwell assay after transfection of miR-491 inhibitor or negative control vector; the migrated cells were stained using crystal; 33% acetic acid was used to dissolve the crystal; then the Student T-test was used to evaluate the OD value at 590 nm obtained by the lysate (*P < 0.05, **P < 0.01).
HMGA2 is a direct functional target of miR-491 in gastric cancer
We searched for potential miR-491 target genes by using miRNA target prediction programs, software, and websites. HMGA2 was treated as a candidate since there is a potential binding site between miR-491 and the 3′-UTR of HMGA2. Furthermore, HMGA2 has been reported to be increased in human cancers and involved in tumor growth and metastasis.7,9
To determine whether miR-491 directly targets HMGA2, we subcloned 3′-UTR HMGA2 fragments that included wild-type (HMGA2-WT) and mutant (HMGA2-MUT) miR-491-binding sites into the pmiRGLO dual-luciferase reporter vector (Figure 3(a)). The resulting vectors were verified by sequencing. HMGA2-WT or HMGA2-MUT pmiRGLO-3′-UTR vector and pre-miR-491 were co-transfected into BGC-823 cells. As expected, miR-491 had no effect on the luciferase activity of the HMGA2-MUT pmiRGLO-3′-UTR vector, whereas the relative luciferase activity of the HMGA2-WT pmiRGLO-3′-UTR vector was markedly decreased in miR-491-overexpressing BGC-823 cells (Figure 3(b)), suggesting that miR-491 binds directly to the 3′-UTR of HMGA2. In addition, when pre-miR-491—the miR-491 inhibitor—and related controls were transfected into gastric cancer cells, HMGA2 was upregulated after pre-miR-491 transfection and downregulated after miR-491 inhibitor transfection (Figure 3(c) and (d)).
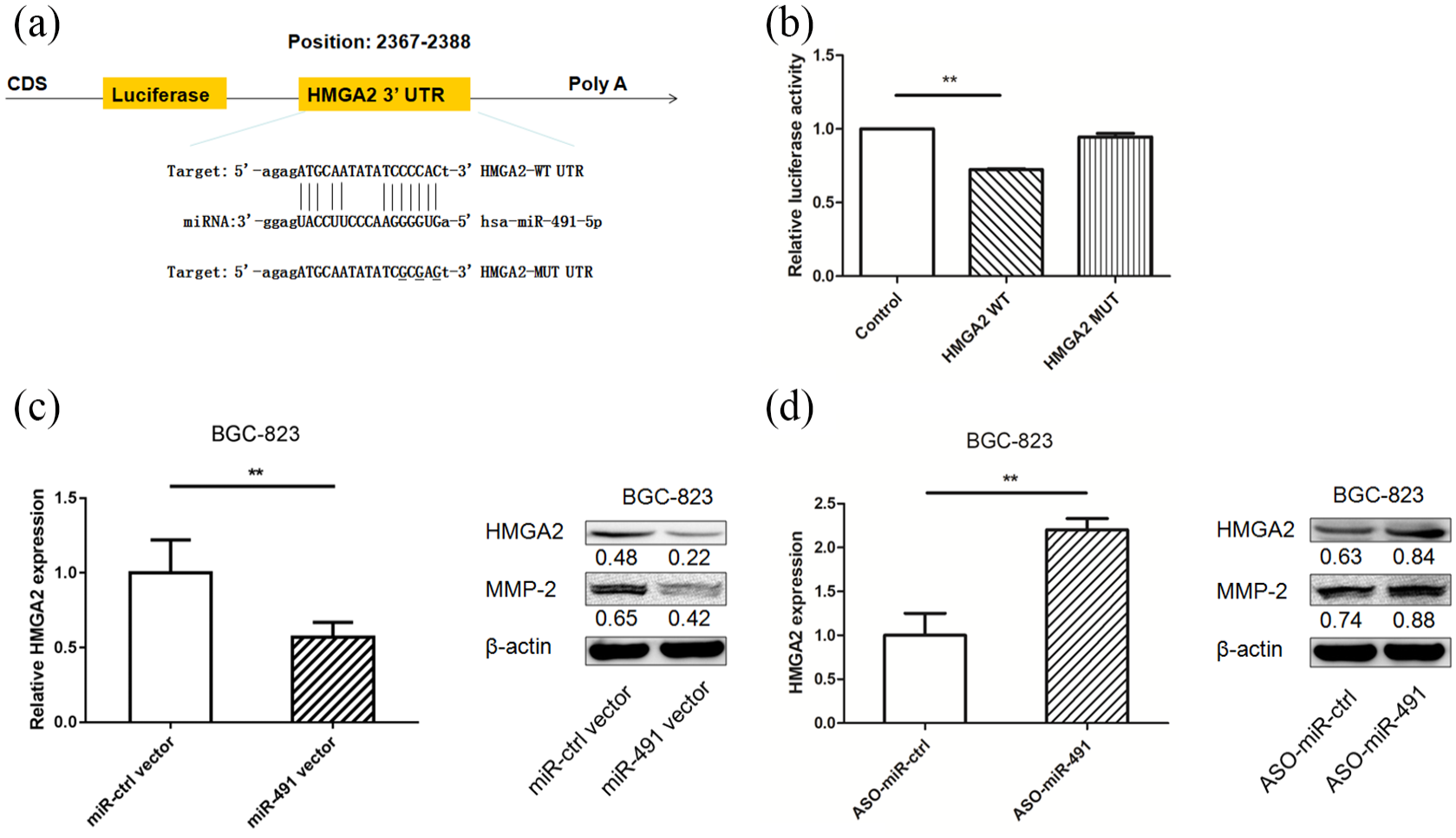
HMGA2 is a direct target of miR-491 in gastric cancer cell lines. (a) miR-491 is highly conserved among different species, and has a binding site between miR-491 and 3′-UTR of human HMGA2. (b) miR-491 was co-transfected with pGLO-HMGA2 wild type or pGLO-HMGA2 mutant vector into BGC-823 cells, the relative luciferase activity was assayed. (c) The mRNA level of HMGA2 was tested after transfection of miR-491 (left panel); HMGA2 and MMP-2 expression levels were detected in BGC-823 cells after miR-491 transfection by western blotting (right panel). (d) The mRNA level of HMGA2 was tested after transfection of miR-491 inhibitor (left panel); HMGA2 and MMP-2 expression levels were detected in BGC-823 cells after miR-491 inhibitor transfection by western blotting (right panel) (*P < 0.05, **P < 0.01).
Knockdown HMGA2 inhibits BGC-823 cell proliferation and migration
siHMGA2 was used to knock down HMGA2 expression, and inhibitive efficiency was validated at the mRNA and protein levels (Figure 4(a) and (d)). The results showed that in BGC-823 cells, HMGA2 knockdown had an inhibitory effect on cell proliferation and migration after transfection (Figure 4(b) to (d)); this effect was similar to that of miR-491 overexpression. Western blotting indicated that MMP-2 was downregulated after siHMGA2 transfection, suggesting that miR-491 produces an inhibitory effect on cell motility by targeting HMGA2 in gastric cancer cells (Figure 4(d)).
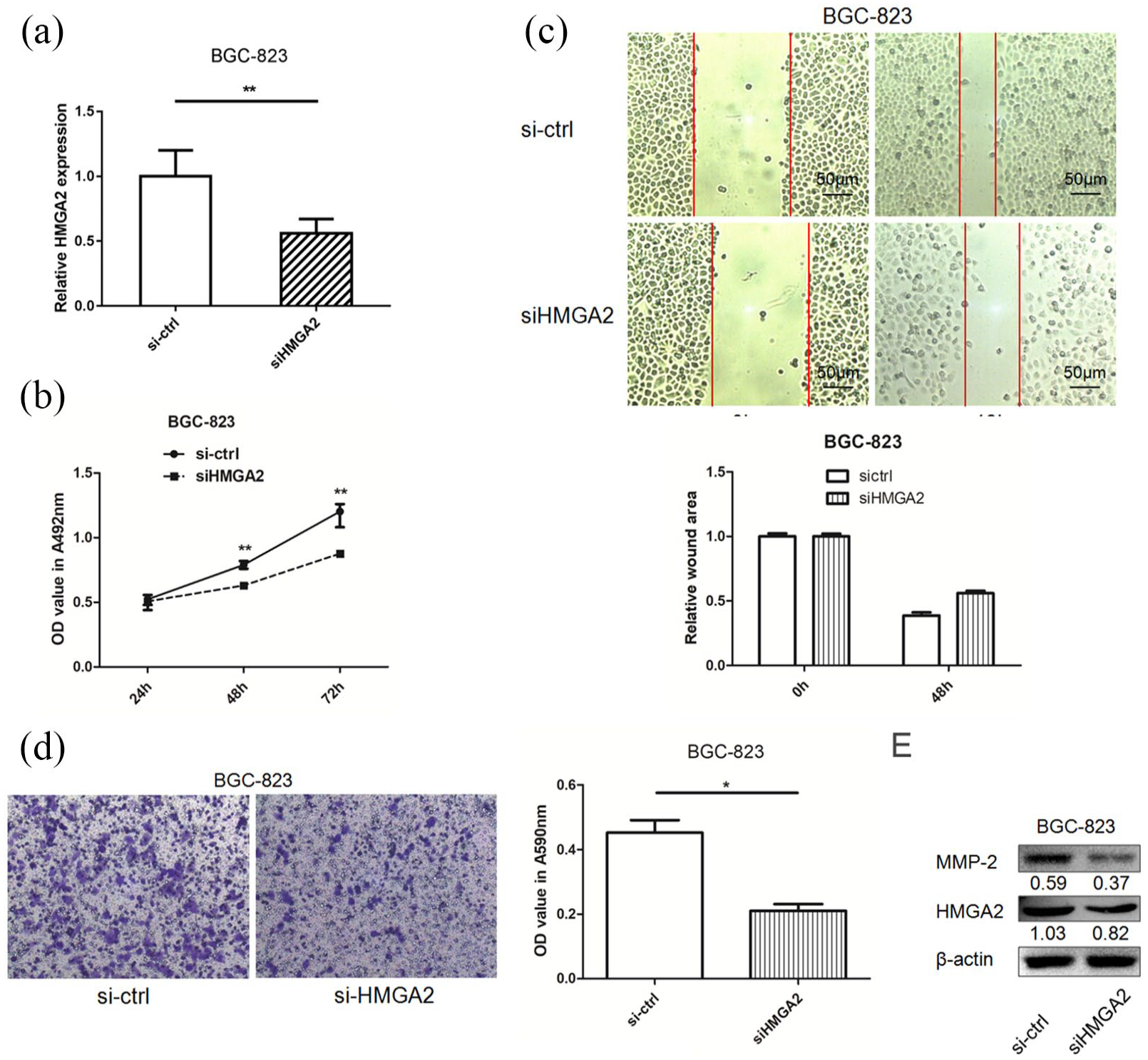
Knockdown of HMGA2 suppresses cell migration in gastric cancer cells. (a) HMGA2 mRNA expression level was decreased after transfection of siHMGA2. (b) The effects of HMGA2 on gastric cancer cell proliferation were determined by MTT assay after transfection of siHMGA2 or control. (c) The effects of HMGA2 on gastric cancer cell migration were determined by wound healing after transfection of siHMGA2 or control. (d) The effects HMGA2 on gastric cancer cell migration was determined by transwell assay after transfection of siHMGA2 or control at 24 h, the migrated cells were stained using crystal; 33% acetic acid was used to dissolve the crystal; then the Student T-test was used to evaluate the OD value at 590 nm obtained by the lysate. (e) HMGA2 and MMP-2 expression levels were detected in BGC-823 cells after siHMGA2 transfection by western blotting (*P < 0.05, **P < 0.01).
Discussion
Gastric cancer is a major cause of cancer-related mortality. Since early-stage gastric cancer has few typical symptoms and unique biomarkers, this cancer is often diagnosed at a late stage, and the survival rate is extremely low. There is an urgent need to search for better predictive biomarkers. Increasing numbers of studies have reported that miRNAs play roles as cancer suppressors or oncogenes. miRNAs may be increased or decreased in cancer tissues relative to adjacent normal tissues. miRNAs may be new molecular markers and therapeutic targets for gastric cancer.
In this study, our data showed that miR-491 inhibits the motility of gastric cancer cells. In addition, we validated the role of miR-491 by targeting HMGA2 signaling. To our knowledge, ours is the first report to disclose that miR-491 can mediate reversible HMGA2 expression in gastric cancer cells.
It has been reported that miR-491 is downregulated in certain human cancers and serves as an important tumor suppressor.15-17 miR-491 acts as a tumor suppressor via targeting JMJD2B in ERalpha-positive breast cancer. 15 In addition, miR-491 inhibits cell migration and invasion by targeting TPX2 17 in esophageal cancer and PDGFRA 16 in prostate cancer. Few studies have focused on the role of miR-491 in gastric cancer. Our data showed that miR-491 was frequently decreased in gastric cancer tissues and that miR-491 overexpression inhibited BGC-823 cell motility. miRNA-target information in public databases and a literature review drew our attention to HMGA2, which we chose as a candidate target of miR-491. In this study, the function and mediating role of miR-491 with respect to HMGA2 was assayed.
HMGA2 is a nuclear binding protein that plays key roles in cell proliferation, differentiation, and migration. HMGA2 overexpression has been found in certain cancers,18,19 including oral squamous cell carcinoma20,21 and breast cancer. 22 A meta-analysis showed that higher levels of HMGA2 were significantly correlated with TNM stage, lymph node status, and poor survival in gastric cancer patients; thus, HMGA2 is a promising biomarker in gastric cancer. 9 The underlying mechanism of HMGA2 is to regulate gene transcription, including IL11, FN1, 23 Twist, 24 and TGF-β/Smad signaling. 25 Pre-miR-491 was transfected into BGC-823 cells, and HMGA2 was found to be decreased by miR-491 at the mRNA and protein levels. We constructed vectors containing binding sites with a wild-type or mutated sequence. As expected, the relative luciferase activity test indicated that HMGA2 was a direct target of miR-491. Moreover, HMGA2 knockdown inhibited cell migration, a result consistent with data from transfection of the miR-491 overexpression vector. These findings suggested that miR-491 played a cell motility-inhibiting role in gastric cancer cells by targeting HMGA2.
In conclusion, our data showed that miR-491 suppresses gastric cancer cell motility by targeting HMGA2. Silencing HMGA2 produced a similar effect to miR-491 expression on cell mobility. Further in vivo mouse studies are needed to confirm these results. miR-491/HMGA2 signaling may be a potential therapeutic target for gastric cancer patients with decreased miR-491 expression.
Supplemental Material
Supplementary_Table_1 – Supplemental material for miR-491 inhibits BGC-823 cell migration via targeting HMGA2
Supplemental material, Supplementary_Table_1 for miR-491 inhibits BGC-823 cell migration via targeting HMGA2 by Zhigang Liu, Yun Lü, Qiuyu Jiang, Yang Yang, Chengxue Dang and Ruifang Sun in The International Journal of Biological Markers
Footnotes
Author contributions
RF Sun, ZG Liu, and CX Dang designed the research; Y Yang, K He, L Han, and QY Jiang performed the research; RF Sun, ZG Liu, and Y Yang analyzed the data; and RF Sun and ZG Liu wrote the paper. ZG Liu, Yun Lü, and QY Jiang contributed equally to this work.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by National Natural Science Foundation of China (81802788), Shaanxi Provincial Natural Science Foundation of China (2018JM7028), Postdoctoral Science Foundation of China (2019T120918, 2017M620458), Postdoctoral Science Foundation of Shaanxi Province, and the Fundamental Research Funds for the Central Universities (xjj2017192).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
