Abstract
Background:
Epidemiological and experimental literature indicates that the risk of breast cancer incidence is strongly linked to hormone-dependent factors, including reproductive history and obesity. However, the molecular mechanisms underlying the association between these factors and breast cancer risk are poorly understood. The aim of this study, therefore, was to determine whether obesity and reproductive history are associated with expression levels of two breast cancer-related long non-coding RNAs (lncRNAs), namely ZFAS1 and SRA1 in cancer-free breast tissues of women.
Methods:
In the current research, 145 healthy women were recruited, and the quantitative expression levels of the two lncRNAs were determined through qPCR assay after gathering the mammoplasty breast tissue samples.
Results:
It was found that women with body mass index (BMI)≥30 kg/m2 and BMI 25–29 kg/m2 show a low expression of ZFAS1 compared to the BMI<25 kg/m2 (P=0.031 and P=0.027, respectively). Then, the correlation analysis disclosed a negative correlation of ZFAS1 low expression with increasing BMI (r=−0.194, P=0.019). Interestingly, this analysis demonstrated a negative correlation between low expression of the ZFAS1 and high BMI in women with menarche age below 14 (r=−221; P=0.028). Lastly, it was also revealed that there was a negative association of the low expression level of ZFAS1 with increasing BMI in women through regression models (B=−0.048, P=0.019).
Conclusions:
These findings suggest interesting clues about the links between high BMI and the expression levels of ZFAS1 in non-diseased breasts that may help us better understand the underlying mechanisms through which obesity contributes to breast carcinogenesis. However, such results need more validations in future research.
Introduction
The risk of breast cancer incidence in women is increased by different factors, including obesity and reproductive factors; for example, early age at menarche, late age at the first pregnancy, nulliparity, lack of breastfeeding, and late age at menopause1,2 These risk-mediating factors are considered to affect breasts through increased exposure to estrogen hormones. 1 While their true underlying molecular mechanisms are not revealed, there is some evidence that these factors can induce gene expression signatures in the normal breast tissue at risk, as well as the prior development of breast cancer.3-6 It is thought that these early changes in breast tissue are triggers for the establishment of the breast tumors.3,6,7
Long non-coding RNAs (lncRNAs) are categorized as a type of non-coding RNA family that is described to progressively play key transcriptional or posttranscriptional regulatory roles in gene expression. 8 The lncRNAs are higher than 200 bp in length, and it has been demonstrated that they can be transcribed from the intragenic and extragenic regions of the genome, but they cannot be translated into any protein.8,9 The lncRNAs contribute to the incidence of various biological events, such as the development, growth, and differentiation of diseased breast tissues.10-12 In addition, these types of RNAs play an important role in the development of breast tumors, including initiation, progression, and metastasis.11,12 Interestingly, there are some clues that show reproductive factors can change the expression levels of cancer-related lncRNAs among normal breast women. 13 As mentioned above, lncRNA expression is dysregulated during breast cancer development; for example, ZFAS1 (zinc finger antisense 1) and SRA1 (steroid receptor RNA activator 1). 11
ZFAS1 exists as an antisense lncRNA and is positioned on the antisense strand at the promoter region of ZNFX1 (i.e. a protein-coding gene). ZFAS1 is involved in the development of breast tissue and, in this regard, experimental data have shown a high level of its expression in the alveolar structures and ducts of mammary glands.14,15 Similarly, Zfas1 shows a differential expression at diverse phases during the course of pregnancy in mice; for example, its expression decreases between pregnancy and lactation. 14 Importantly, experimental data in mice have revealed the availability of independent roles for Zfas1 lncRNA so that it can undergo splicing in the nucleus. In this way, it contributes to the generation of snoRNAs and regulation of the ribosome function, while it is transported to cytoplasm for the control of proliferation pathways.14,15 The investigation in human breasts have found some tumor suppressor activities for ZFAS1; in particular, its expression is downregulated in human ductal carcinoma. 14 Furthermore, ZFAS1 has a key role in cellular proliferation and differentiation events, and it has been shown that its reduced expression might have induced tumor progression, which suggests a role for tumor suppressor function of ZFAS1. 14
Another lncRNA with an important function in breasts is steroid receptor RNA activator 1 (SRA1) that functions as a molecular coactivator for the expression of genes encoding steroid hormone receptors, such as estrogen and progesterone receptors in the development of mammary gland.16,17 Emerging evidence indicates that SRA1 gene product is also involved in regulating the activity of non-steroid receptors and important proteins having functions in adipogenesis, myogenesis, and breast development. 18 In the same way, the results from the mouse model have revealed an activity for SRA1 towards the development of mammary glands. 19 The achieved data in this model revealed that the activation of SRA1 in mature virgin transgenic mice led to an increase in the proliferation and differentiation and, thus, early abnormalities showed up in the development of the ductal epithelium. 19 Therefore, these data represent important clues of the existence of a relationship between the roles of SRA1 in breast hemostasis and its diseases. Significantly, different studies have reported the elevated expression of SRA1 in estrogen-dependent cancers, including ovarian and breast cancers.20,21 SRA1 goes through a complicated process in the proliferation state of breasts by activating the estrogen receptor, and it can be representative of breast tumor behavior since its higher expression was found in ER-α-positive/PR-negative tumors compared to ER-α-positive/PR-positive tumors. 22 Together, the published literature proposes that SRA1 in a complex interplay with steroid hormone receptors may be an important factor of breast tumor growth and progression.14,22
In conclusion, since it has been shown that the expression of breast cancer-related lncRNAs in normal tissues may be affected by breast cancer risk factors, 13 the above-mentioned two lncRNAs, (i.e. ZFAS1 and SRA1), which play a very important role in the formation of breast tumors, may also be altered by breast-cancer-related factors. However, no study has been conducted in this regard. The identification of aberrant expression changes with regard to these lncRNAs in non-diseased breast and an understanding of their roles in breast carcinogenesis may have key implications as follows: clarification of the early and initiating changes in breast carcinogenesis and even the contribution to the introduction of preventive and detection strategies for breast cancer in women. Thus, the current study aimed at evaluating the expression levels of lncRNAs ZFAS1 and SRA1 in breast tissues of healthy women and their relationship with different patterns of obesity and reproductive factors as the risk factors for breast cancer.
Materials and methods
Sample collection and population characteristics
The subjects in this study consisted of 145 cancer-free women within the 20- to 60-year-old age range who underwent cosmetic mammoplasty surgery at Vali-e-Asr Hospital, Sohrevardi Surgery Center, and Mehr-e-Sina Surgery Clinic between 2013 and 2016. Women with a previous history of breast cancer, ovarian cancer, uterine cancer, and other cancers were excluded from the research. The breast tissues were obtained from mammoplasty samples after receiving informed consent from all participants for offering their samples not required for diagnosis purposes. The study procedure was approved by the ethics committee of Tehran University of Medical Sciences (TUMS). In addition, a clinical questionnaire has been used for data gathering on demographic features, reproductive information, family history of breast cancer and other cancers, smoking status, oral contraceptive pill use, and other medical records. The achieved samples were immediately snap frozen in liquid nitrogen and stored in −80°C for the conduct of the following analyses.
RNA isolation and cDNA synthesis
Total RNA was extracted from all breast tissues by using TRIzol reagent (Life Technology, USA) according to the standard procedures. The extracted RNA was treated with DNase I (Takara, Japan) for 70 minutes; then, the concentration and purity of all the treated RNA samples were measured by NanoDrop ND-1000 Spectrometer (Thermo Scientific, Boston, MA, USA). Moreover, RNA integrity was confirmed by agarose gel electrophoresis. A total of 0.5 µg RNA was used for the synthesis of complementary DNA using Prime Script TM RT reagent Kit (Takara, Japan) in a 10 µl reaction mixture and under the following reaction conditions: 37°C for 20 min, and 85°C for 10 s.
lncRNA expression analysis using quantitative real-time polymerase chain reaction assay
To determine the quantitative expression level of ZFAS1, SRA1 lncRNAs, and beta 2 microglobin (B2M) as a normalizer gene, quantitative real-time PCR (qPCR) was conducted using SYBR Green qPCR SYBR PremixEx TaqII (TliRNase H Plus) Master Mix (Takara Bio Inc., Japan) on Rotor Gene Q6000 real-time PCR system (Rotorgene-Q, Hilden, Germany) according to the manufacturer’s instructions. Furthermore, for normalizing the qPCR data, the B2M gene was chosen as the endogenous control. The qPCR reaction assays were done in total reaction volume of 10 µl reaction mixture containing 1 ng of cDNA, 5 pmol per primer (forward and reverse), and 5 µl of 2X SYBR Green PCR mix. Thermal cycling conditions were an initial activation for 30 s at 95°C followed by 40 cycles at 95°C for 15 s and 60°C for 20 s. The sequences of specific primers were as follows: ZFAS1; 5- AAGCCACGTGCAGACATCTAC -3 (forward), 5- CTACTTCCAACACCCGCATTCA -3 (reverse), SRA1; 5- GGGCTCGCTTCACACATCC -3 (forward), 5- TCTCTGATGCGGGGACTCTG -3 (reverse), and B2M; 5- AGATGAGTATGCCTGCCGTG -3 (forward), 5- GCGGCATCTTCAAACCTCCA -3 (reverse). The analyses of melting curve and agarose gel electrophoresis were carried out for verifying the specificity of PCR products. The samples were analyzed in duplicate and the relative expression levels of ZFAS1 and SRA1 were calculated using the comparative Livak method 2-ΔΔCT formula.
Statistical analysis
In the current research, the data were presented as mean, SD, and median. The comparison analyses of relative lncRNAs expressions were conducted for the study subgroups with two and more than two levels by the non-parametric Mann–Whitney test and the Kruskal–Wallis test, respectively. Following the Kruskal–Wallis test, pairwise comparisons were made by the Mann–Whitney test. Thereafter, the Spearman rank correlation coefficient was used to measure the correlation between the expression levels of the target lncRNAs and different variables. Furthermore, the linear multivariate regression model was run for calculating the independent effects of the factors under study on the expression level of lncRNAs. The statistical analyses were conducted in IBM SPSS version 22 (SPSS Inc., Chicago, IL, USA). The P-value less than 0.05 was defined as the statistically significant level.
Results
The detailed results of different subgroups of risk factors for breast cancer plus ZFAS1 and SRA1 expression levels in breast tissues of the subjects are shown in Table 1 and Table 1 of the supplementary data, respectively. To determine the difference in the expression level of the two lncRNAs between or among subgroups of the variables, we examined the variables categorically with quantitative measures of ZFAS1 and SRA1 expressions by the Mann–Whitney test and the Kruskal–Wallis test. The statistical results of the Kruskal–Wallis test showed that there is a significant difference in the level of ZFAS1 expression among BMI subgroups (P= 0.037) (Table 1). Then, a pairwise comparison test among BMI subgroups by Mann–Whitney analysis showed that obese women with BMI≥30 kg/m2 and overweight women with BMI 25–29 kg/m2 have a low expression of ZFAS1 compared to normal weight women with BMI<25 kg/m2 (P= 0.031 and P= 0.027, respectively) (Figure 1). These two analyses revealed no significant difference in the expression level of ZFAS1 among other subgroups of the variables (Table 1).
Expression levels of lncRNA ZFAS1 in different subgroups of breast cancer risk factors.
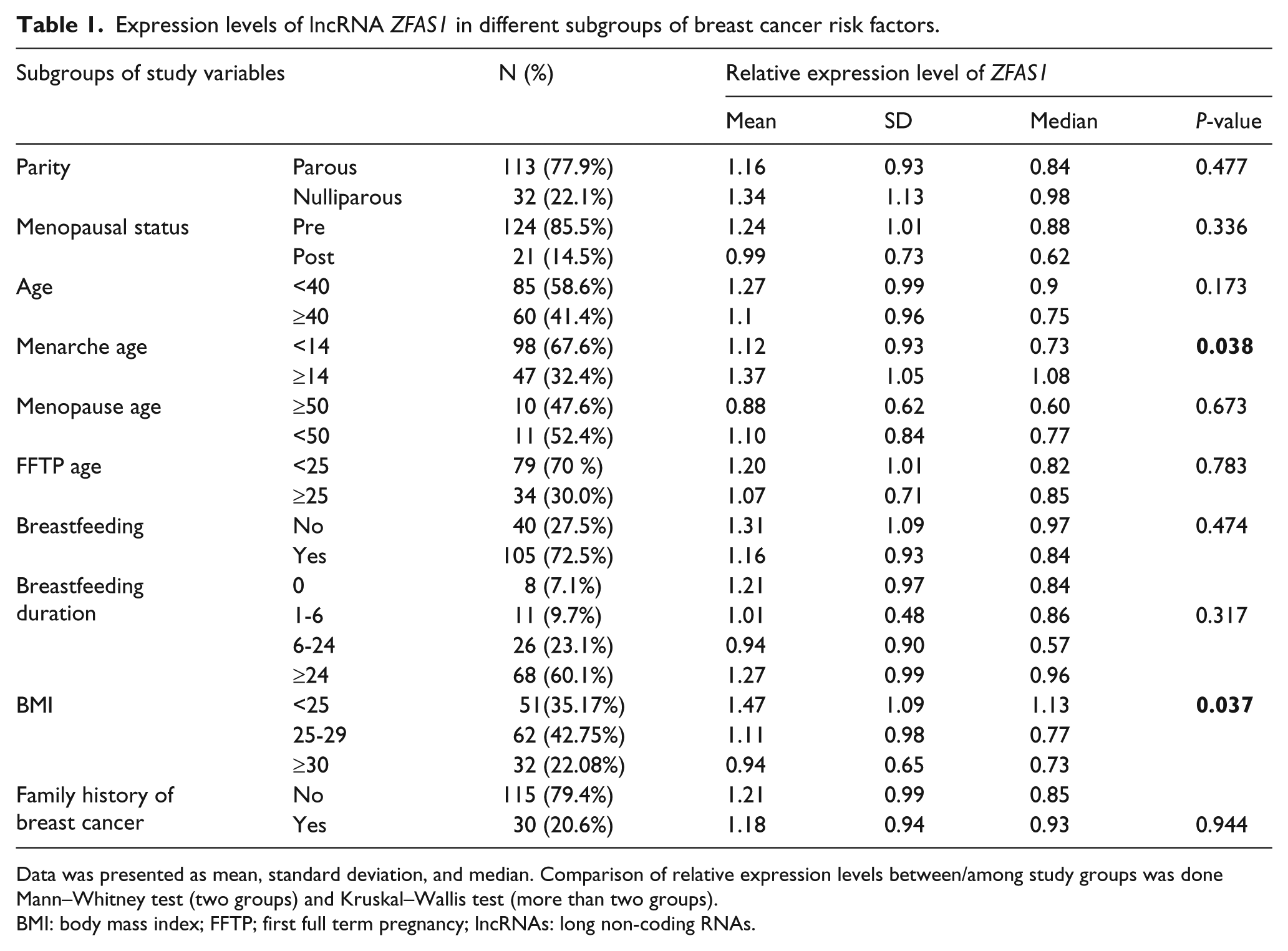
Data was presented as mean, standard deviation, and median. Comparison of relative expression levels between/among study groups was done Mann–Whitney test (two groups) and Kruskal–Wallis test (more than two groups).
BMI: body mass index; FFTP; first full term pregnancy; lncRNAs: long non-coding RNAs.
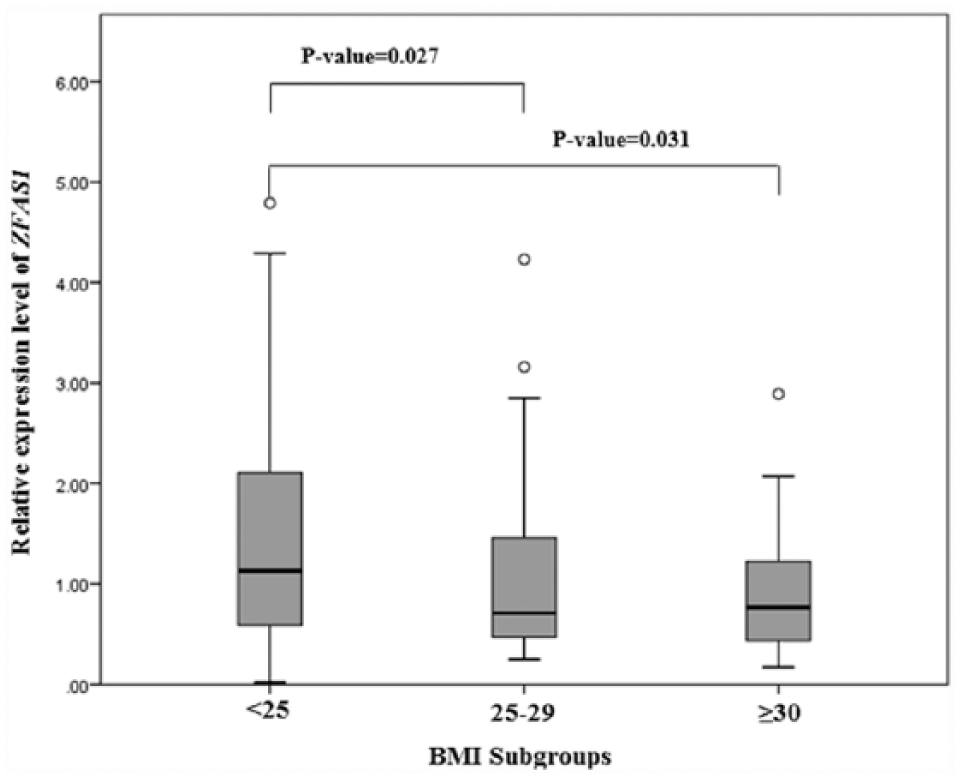
The expression level of ZFAS1 in the women with different subgroups of BMI.
In the next phase of the evaluations, the correlation between the quantitative expression level of ZFAS1 and risk factors of the study was investigated by the Spearman rank correlation coefficient test. This analysis revealed a significant negative correlation between the low expression level of ZFAS1 and the increase of BMI (r= −0.194, P= 0.019). The other factors pertaining to breast cancer were not revealed to have any important correlation with the expression level of ZFAS1 in our analyses (data not shown). In addition, the correlation of ZFAS1 expression with BMI was evaluated among postmenopausal women and among the premenopausal women. The results demonstrated a significant negative correlation between the decreased expression level of ZFAS1 and high BMI (r= −0.200; P= 0.026) in premenopausal women and a non-significant correlation r= 0.020 (P= 0.933) between ZFAS1 expression and BMI in postmenopausal women.
Finally, linear multivariate regression models were used to evaluate the independent relationship of ZFAS1 expression level with different risk factors of breast cancer. The results of this test showed that there is a negative association between the low expression level of ZFAS1 and high BMI in women of the study (multivariable B= −0.048, standard error (SE) = 0.020, P= 0.019) (Table 2). In the same way, the calculated odds ratio (OR) revealed the presence of a negative association between ZFAS1 expression levels and BMI ≥30 in women (OR= 0.537 (95% confidence interval (CI) 0.308, 0.933; P= 0.027) in comparison with BMI<25 (normal BMI) in women. This analysis also showed the odds ratio of 0.694 (95% CI 0.472, 1.020; P= 0.063) for the expression levels of ZFAS1 with BMI 25–29 in comparison with BMI<25.
Linear multivariable regression on the ZFAS1 expression and the variables under study.
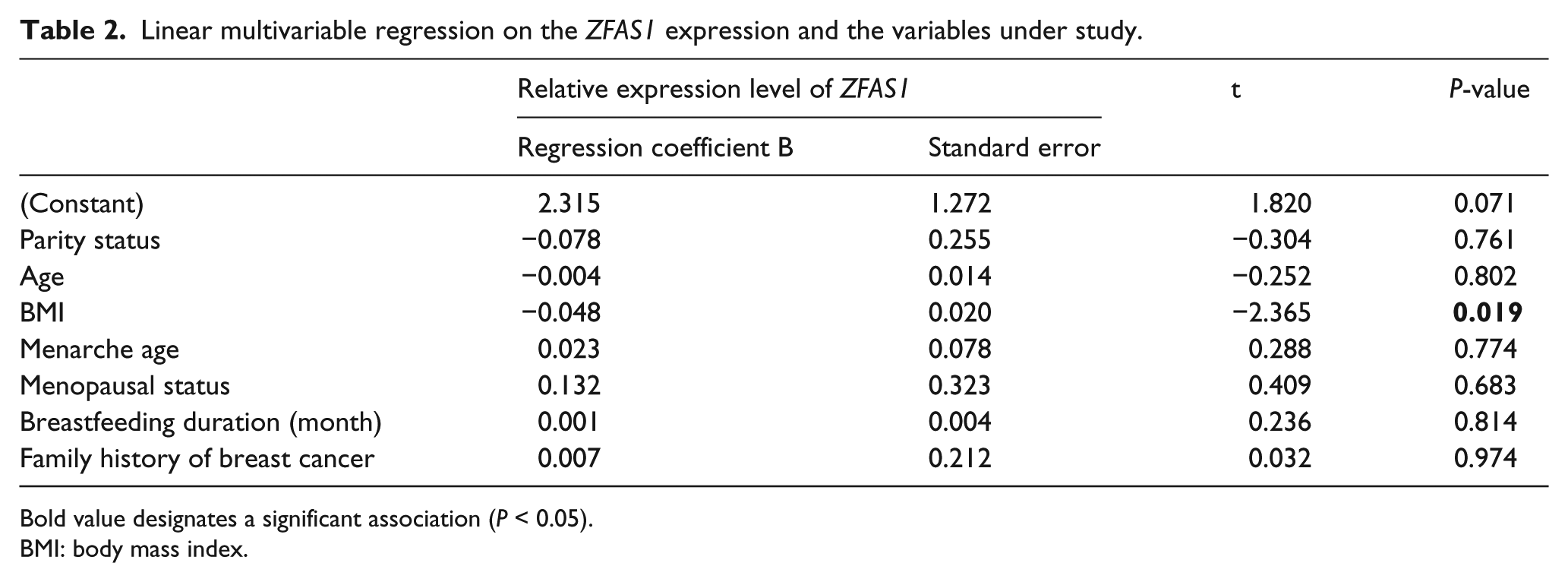
Bold value designates a significant association (P < 0.05).
BMI: body mass index.
In this study, Mann–Whitney test also indicated that women with a menarche age <14 years exhibit a lower level of ZFAS1 expression than women with menarche age at ≥14 years (P= 0.038) (Figure 2). However, the obtained correlation and association were not revealed to be significant by the Spearman rank correlation coefficient and linear multivariate regression models, respectively. Interestingly, when the correlation analysis was conducted on the expression level of ZFAS1 and BMI in relation to menarche age in women, there was a significant negative correlation between the expression level (decreased) of this gene and high BMI in women with a menarche age below 14 years (r= −221; P= 0.028).
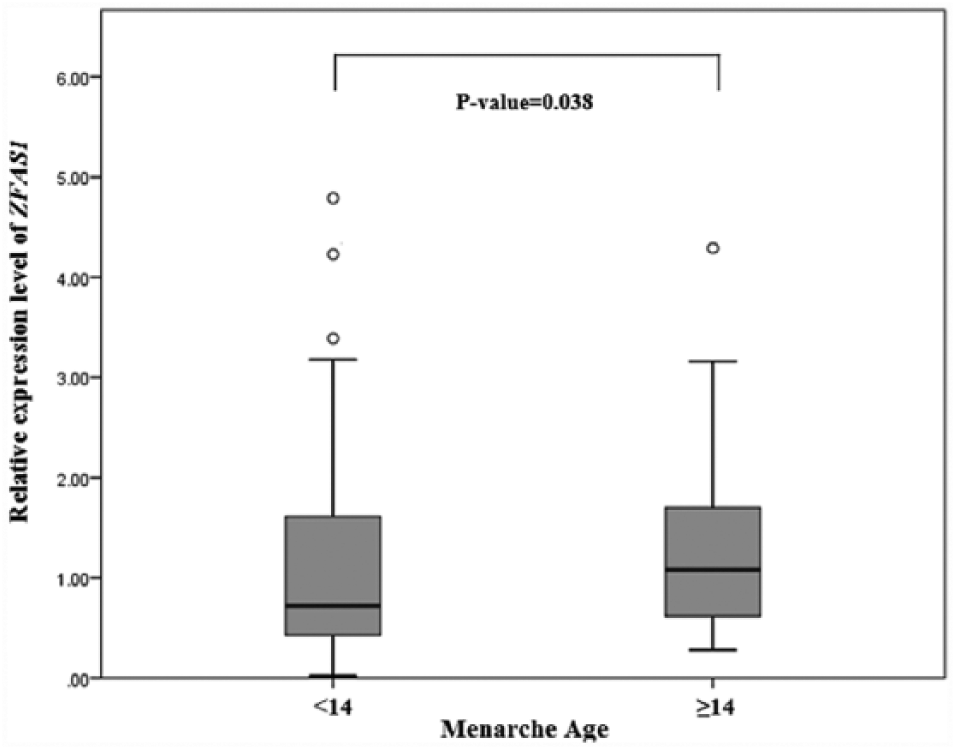
The expression level of ZFAS1 in the women with two subgroups of menarche age.
Furthermore, when the aforementioned Mann–Whitney and Kruskal–Wallis tests were applied for evaluating the possible differences in the expression levels of lncRNA SRA1 among the variables, no significant differences were observed (Table 1; supplementary data). Hereafter, no significant association or correlation was obtained between the expression level of SRA1 and the different subgroups of the breast cancer risk factors through the Spearman rank correlation coefficient test and the multivariate regression models (Table 2; supplementary data).
Discussion
Evidence has been accumulated that clinically healthy women who are at risk of breast cancer may undergo early molecular carcinogenic changes in their breast tissues.23-26 Unveiling such changes in normal breast tissues, especially at the expression levels of critical genes like those related to lncRNAs (which may be influenced by breast cancer risk factors, e.g. obesity) may contribute to the elucidation of the early and even breast carcinogenesis originating events. Today, the incidence of female obesity is on the rise and has become a health problem in communities. 27 Therefore, the revelation of the molecular basis of breast cancer risk caused by obesity in women is an important area of inquiry in breast cancer research.
The results of the current research revealed a low expression level of ZFAS1 in obese (BMI≥30 kg/m2) and overweight (BMI 25-29 kg/m2) women compared to normal weight women (BMI<25 kg/m2). This finding of our study is also supported by the correlation between the lower expression level of ZFAS1 and increased BMI in women. Although previous reports have found some connections between obesity and distinct molecular changes in genome24,26,28—especially changes in the expression of protein encoding genes in the normal breast tissue of women 24 —so far, no results have been reported on the relationship between expression changes and obesity in lncRNAs. The significance of the ZFAS1 expression signature in the present study is that previous strong evidence in both breast cell lines and breast tumors has demonstrated ZFAS1 downregulation during breast cancer development.14,15 The results of these investigations suggested some breast tumor-suppressor activities for lncRNA ZFAS1. In this regard, the study conducted by Askarian-Amiri et al. 14 demonstrated an increase in cell proliferation and metabolic activity of the breast epithelial cells by knocking down the ZFAS1, and they concluded that ZFAS1 may act as a tumor suppressor lncRNA in breast cancer. In line with these findings, the present study showed that the expression of ZFAS1 is reduced in obese and overweight women’s breasts compared to normal weight women. Thus, the current findings highlight possible key links for this important gene in relation to high BMI in women, which may influence the process of carcinogenesis in their breast and, in turn, may elevate the risk of breast cancer.
It is generally well-documented that high BMI is a risk factor of breast cancer in postmenopausal women, and it has been suggested that this occurs via elevated levels of circulating estrogens.1,29 Elevated conversion of androgens to estrogens and a decreased level of circulating sex hormone binding globulin in such situations cause a greater exposure of the breast to estrogens in obese postmenopausal women. 1 Estrogen plays an important role in elevating the risk of breast tumors through increasing breast proliferation and this has been shown to have a direct effect on the expression of the genes involved in breast cancer. 30 Although we found a decreased expression of this gene in relation to high BMI in pre-menopausal women, it was reasonably expected that postmenopausal obesity would be linked with such a decrease in the expression of this gene. This finding may be accounted for by the belief that this gene might have acted through unknown mechanisms and interactions that affect the risk of breast carcinogenesis in premenopausal women.
Therefore, we speculated that the reduced expression of ZFAS1 in the breasts of apparently healthy obese and overweight women may be indicative of the early stages of the events through which these women may be affected for breast malignancy in future. In other words, the expression and functions of ZFAS1 might have been modulated by the effects of high BMI like estrogen or other unknown mechanisms. This, in turn, could contribute to the risk of breast carcinogenesis in interaction with or without other risk factors.
In this study, it was also shown that the level of ZFAS1 expression in women with the menarche age lower than 14 years is reduced compared to the women with the menarche age above 14 years. Interestingly, this decrease in ZFAS1 expression in relation with menarche age was shown to have a significant correlation with obesity (high BMI). This finding of the present study on the link between early age at menarche and ZFAS1 expression is consistent with the results of previous epidemiologic studies where it has been shown that early age at menarche is a risk factor for breast cancer via increased exposure of the breast tissue to estrogen levels.1,2
On the other hand, as mentioned above, it appears that these two hormone-dependent risk factors may increase the risk of breast cancer through an unknown hormone mechanism and by reducing the expression of the ZFAS1 gene. It is noteworthy that such interpretations require the conduct of more studies with a higher number of subjects to examine the expression of this gene in relation to the age at the menarche and obesity. Therefore, it is conceived that the expression of the ZFAS1 gene is reduced and, in turn, the two factors of breast cancer increase the risk of breast cancer incidence when in the face of high levels of sex hormones that occur at the younger age of the menarche (with an increase in the extent of exposure time) and obesity (increased production in adipose tissue). Consequently, the outcome of our study may be mentioned as a molecular mechanism of the relationship between obesity and the age of menarche with the risk of breast cancer.
To sum up, the correlation of ZFAS1 low expression with high body mass in apparently healthy women may be a possible molecular missing link behind the role of obesity and early age at menarche in the risk of breast cancer. Such results can provide insights into how early menarche and BMI, predominantly in obese women, may be guided toward breast carcinogenesis. One of the strengths of this study is that it has evaluated the level of ZFAS1 expression in normal breasts and, thereby, they are of more genomic stability than tumor tissues. 31 Genetic signature patterns, such as lncRNAs expression, may change prior cancer initiation in the women at risk who have apparently healthy breasts, but they hold some carcinogenic-dependent molecular changes in their breast at the molecular levels.11,32 The significance of the current finding is that it may be considered as a potential predictor of the risk of breast cancer incidence in women with obesity. Nevertheless, such inferences need more validations through further investigations because the present study had some limitations, such as the number of subjects and the evaluated lncRNAs. It should also be taken into account that the female subjects in the current study may not be representative of the women in the general population because they all received cosmetic mammoplasty.
Supplemental Material
Supplymentary_data – Supplemental material for A link between expression level of long-non-coding RNA
ZFAS1
in breast tissue of healthy women and obesity
Supplemental material, Supplymentary_data for A link between expression level of long-non-coding RNA
Footnotes
Acknowledgements
This work was extracted from a part of the Ph.D. thesis by Yaser Mansoori, supervised by Dr. Javad Tavakkoly-Bazzaz.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by Tehran University of Medical Sciences (TUMS), Tehran, Iran (Grant Number: 30357).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
