Abstract
Aim:
Due to the limits of calcitonin, other markers are warranted to better manage medullary thyroid carcinoma patients, and procalcitonin has been reported as promising. Here we aimed to evaluate procalcitonin as a marker of medullary thyroid carcinoma in the post-treatment follow-up.
Methods:
Medullary thyroid carcinoma patients previously treated by thyroidectomy were enrolled. After complete imaging work-up (i.e. ultrasonography, computed tomography, magnetic resonance and 18FDG-PET-CT) we identified patients with structural recurrent/persistent medullary thyroid carcinoma (active medullary thyroid carcinoma) and subjects with no evidence of disease. Then, both calcitonin and procalcitonin were measured and their performance analyzed.
Results:
The final series included 55 medullary thyroid carcinoma patients treated and followed-up for about five years. Of these, 43 were assessed as no evidence of disease, and 12 as active medullary thyroid carcinoma. The median value of procalcitonin was significantly higher (P < 0.0001) in active medullary thyroid carcinoma patients (3.10 ng/mL) than in those with no evidence of disease (0.10 ng/mL). Also, calcitonin levels of active medullary thyroid carcinoma (96.7 pg/mL) were significantly (P < 0.0001) higher than that of no evidence of disease (2.0 pg/mL). At the receiver operating characteristic curve analysis, the optimal cut-off of procalcitonin was ≥0.32 ng/mL with 92% sensitivity and 98% specificity, while the most accurate threshold of calcitonin was ≥12.0 pg/mL with 100% sensitivity and 91% specificity. There was no active medullary thyroid carcinoma with simultaneously negative results of procalcitonin and calcitonin.
Conclusions:
Procalcitonin is reliable in discriminating medullary thyroid carcinoma patients with active disease from those with no evidence of disease. We suggest using procalcitonin as complementary to calcitonin to follow-up medullary thyroid carcinoma patients.
Introduction
Medullary thyroid carcinoma (MTC) originates from thyroid parafollicular C cells and accounts for up to 5% of thyroid cancers (1). Four in five MTC occur sporadically and the remaining 20% are part of familial disorders (1). It is still difficult to detect this cancer due to several reasons: (a) cytology by fine needle aspiration (FNAC) has a very low detection rate (2, 3); (b) ultrasonography has low accuracy (4); and (c) the measurement of serum calcitonin (CT), even if it represents the most reliable tool in this context, has limits (4). Specifically, no fixed CT thresholds may accurately diagnose and exclude MTC, and different potential cut-off values have been reported (4). Also, some cases of MTC with negative CT exist (5). Thus, a part of MTC is diagnosed with delay and/or incidentally discovered after surgery, with consequent poor prognosis. Other tools to preoperatively diagnose MTC are needed to plan the treatment of these patients, and further markers are strongly warranted to better manage MTC patients during postoperative follow-up (1).
Recently, the measurement of procalcitonin (ProCT), a precursor of CT, has been investigated in the management of MTC (6). These studies recorded a good performance of ProCT, and when it was compared with CT. ProCT is a 116-aminoacids peptide product by thyroid C-cells, and is a very stable protein featuring a concentration-independent in vivo half-life of 20–24 hours (5). In addition, commercial ProCT assays yield similar results, and the sample does not need to be kept cool on ice during the entire process chain. These characteristics give ProCT great potential to replace CT as a marker to manage MTC patients.
Here we investigated the reliability of ProCT as a postoperative marker of MTC. The purpose of the present study was to evaluate the performance of ProCT in detecting persistent/recurrent MTC in a series of patients followed-up and assessed as active MTC or with no evidence of disease (NED).
Methods
Patient selection and study design
According to our aim, the study design included the enrollment of patients already treated for MTC, assessed as recurrent or not during follow-up. Then, we retrospectively enrolled MTC patients followed-up at our two institutes (Oncology Institute of Southern Switzerland of Bellinzona, and IFO of Rome) who had visited our thyroid centers during the period from January 2016 to June 2017. At these centers, MTC patients undergo thyroidectomy with central or lateral neck dissection, depending on risk and intra-operative findings, and the tumor features. All MTC patients were assessed as NED when we could demonstrate the complete negative results of imaging work-up, including neck ultrasonographic examination and, when indicated, computed tomography, magnetic resonance and 18fluoro-2-deoxy-d-glucose positron emission tomography-computed tomography (18FDG-PET-CT). Patients with positive imaging and/or cytology/histology (i.e. structural persistence/recurrence of disease) were considered as active MTC. Cases with uncertain imaging data (evidence of lesions that were not certainly assessed as MTC metastases) were not included. Also, patients with suspected acute infections were excluded. In all patients finally included in the study, we performed the measurement of both CT and ProCT during the last visit, and then analyzed the performance of these two markers in identifying active disease.
ProCT measurement
ProCT was measured on a Kryptor system (BRAHMS, Berlin, Germany) by a homogenous time-resolved amplified cryptate emission immunometric fluorescent assay. The assay has a functional sensitivity of 0.10 ng/mL.
CT measurement
In all patients serum CT was measured by an immunochemiluminometric assay on fully automated IMMULITE® 2000 XPi platform (Siemens Healthcare Diagnostics, Erlangen, Germany). The CT assay is standardized against the 2nd International WHO calibrator 89/620 and was performed in strict adherence to the manufacturer instructions as stated in the package insert. The limit of detection and the limit of quantification of CT assay were 2.00 and 5.00 pg/mL; upper reference limit 5.4 pg/mL and 8.2 pg/mL for females and males, respectively.
Statistical analysis
All results of ProCT were reported as median and range values. For statistical analysis all ProCT levels below 0.10 ng/mL (functional sensitivity) were considered as equal to 0.10 ng/mL; similarly, CT values below 2.0 pg/mL (functional sensitivity) were considered as 2.0 pg/mL. ProCT and CT levels of active and NED MTC were compared by unpaired Wilcoxon-Mann-Whitney test. Those cases with CT and/or ProCT values above the receiver operating characteristic (ROC)-derived cut-off and no evidence of structural recurrence were considered to be biochemical diseases, and for statistical analysis, they were included in the NED group. The performance of ProCT and CT in detecting active MTC was evaluated by ROC curve analysis and the most accurate threshold calculated with sensitivity and specificity. Statistic results were considered significant when the P value was less than 0.05. All statistical analyses were performed by StatsDirect statistical software version (StatsDirect Ltd; Altrincham, UK).
Results
The final series included 55 MTC (34 females and 21 males, mean age 57 years), four of which had hereditary MTC type (three familial MTC and one MEN2A); they had a mean follow-up of 59 (range from 12 to 360) months. Of the entire series, 12 (21.8%) patients had active MTC, which was represented by neck lymph node metastases in seven cases, a mediastinal lymph node metastasis in two cases, and unresectable masses involving the trachea in three cases. Table 1 details the main characteristics of the study series.
Patients’ characteristics
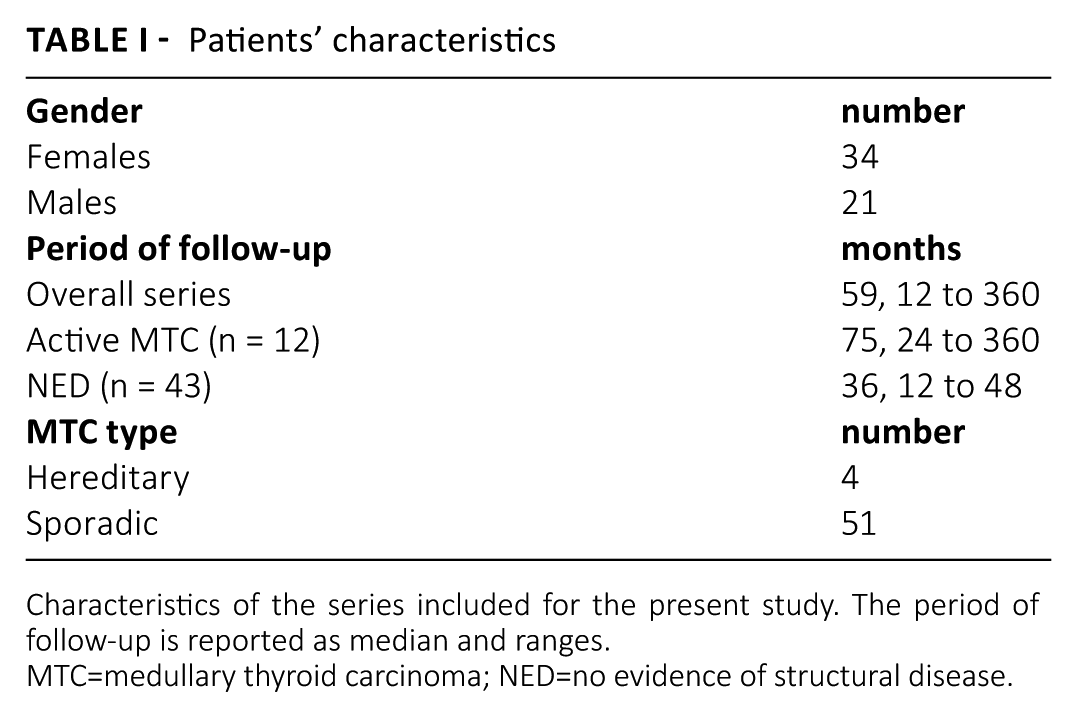
Characteristics of the series included for the present study. The period of follow-up is reported as median and ranges.
MTC=medullary thyroid carcinoma; NED=no evidence of structural disease.
Median ProCT levels of active MTC were significantly higher (P < 0.0001) than that of NED (3.10 vs. 0.10 ng/mL, respectively). Figure 1A illustrates the distribution of ProCT levels in the two groups. ProCT ranged between 0.10 and 1.04 ng/mL in NED, and between 0.10 and 12.54 ng/mL in active MTC. The ROC curve analysis showed that the most accurate cut-off of ProCT was ≥0.32 ng/mL (Figure 1B).
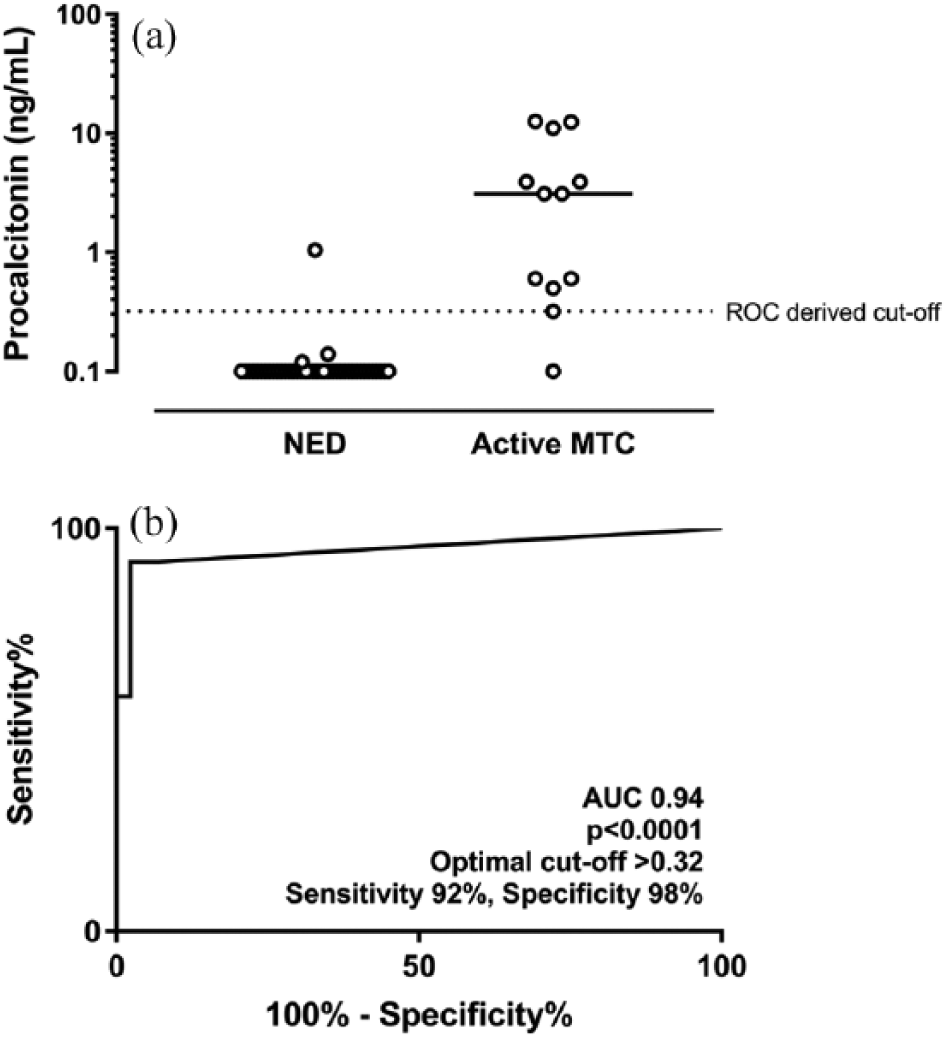
A) Distribution of ProCT values (log10) in patients assessed as active MTC or NED. B) ROC curve analysis of ProCT in detecting active MTC.
Median CT levels were significantly (P < 0.0001) higher in active MTC (96.7 pg/mL) than in NED (2.0 pg/mL). In particular, CT ranged between 2.0 and 467.8 pg/mL in NED, and between 12.0 and 1974 pg/mL in active MTC. At ROC curve analysis the most accurate threshold of CT was ≥12.0 ng/mL with 100% sensitivity and 91% specificity.
All in all, there was no active MTC with simultaneously negative results of ProCT and CT measurement. Finally, there were four biochemical diseases among NED patients: one patient had both positive ProCT and CT over a follow-up of three years, and the other three had positive CT after a follow-up of 18, 31, and 131 months.
Discussion
Traditionally, CT has been adopted as the pivotal serum tool for the identification of recurrent/persistent MTC. Then, MTC patients are generally treated by thyroidectomy and central and/or lateral neck dissection. Then, patients are periodically followed-up by clinical examination, neck ultrasound, and CT measurement. Unfortunately, some pre-analytical, analytical, and post-analytical limits can affect CT accuracy (7-12). Also, detectable CT levels may not be correlated with structural persistence of MTC; in this context, American Thyroid Association guidelines recommend imaging not be used in the presence of CT below 150 pg/mL (1). Also, the CT stimulation is not reliable enough to discriminate recurrent MTC from disease-free patients (4). Thus, other potential MTC serum markers have been recently investigated, and ProCT has appeared to be the most promising one (6).
In our series, ProCT showed a good performance in detecting 11/12 active MTC with levels equal or higher than 0.32 ng/mL, and in identifying 42/43 NED with values below this cut-off. The performance of ProCT was not significantly different from that of CT. In addition, the results of ProCT should improve the accuracy of CT; only 1/43 NED had false-positive markers, and only 1/12 with active cancer showed false-negative. A few papers (13-19) have investigated ProCT measurements in MTC after surgery, and our study corroborates those findings (Tab. II). The most relevant paper was published by Algeciras-Schimnich et al. (15) who collected a series of 42 cured and 91 active MTC, and recorded a 91.2% sensitivity and 100% specificity. The active MTC were defined as “based on clinical, imaging, or histological evidence of persistent or recurrent disease or continuous elevation of serum CT without evidence for a non-MTC cause of the elevation.” Among them, patients with stable disease had the lowest mean ProCT levels (3.6 ng/mL); patients with recurrent/metastatic disease had the highest mean value (241.7 ng/mL); and patients with newly diagnosed cancer had intermediate levels (13.8 ng/mL). An important contribution was also made by the Kratzsch et al. (19) study who compared ProCT to several CT assays (Immulite, Liaison, and IRMA Medipan) in different groups of patients with hypercalcitoninemia, including subjects affected by recurrent/persistent MTC, those with chronic kidney disease, and those with Hashimoto’s thyroiditis. The 10 MTC in that study had CT levels that ranged from 18 to 1511 pg/mL, and ProCT levels were between 0.226 and 11.6 ng/mL. The authors found the highest diagnostic sensitivity for MTC of 100% with ProCT. It is relevant that the ProCT concentrations appeared to have better stability than the CT levels. In addition, two interesting papers were reported by Polish authors who recorded 87% specificity and 100% sensitivity (16, 17) A study by Walter et al. (18) evaluated 69 MTC patients and carried out a cut-off of 0.5 ng/mL showing 84% sensitivity and specificity in discriminating MTC from controls; specifically, they showed that CT was more reliable than ProCT but the ProCT:CT ratio was accurate to discriminate the progressive MTC from the stable ones. Finally, very high sensitivity (100%) of ProCT was recorded by Bolko et al. (14). The study by Bihan et al. (13) used a chemiluminescent assay, which was not comparable with ours; therefore, we cannot discuss their data. Some other studies evaluated the role of ProCT in the preoperative diagnosis of MTC with encouraging data (6). Interestingly, ProCT could be useful in the rare cases of MTC with negative serum CT values (5). As proof, an MTC patient with undetectable CT was recently reported, and ProCT converted from detectable before surgery to undetectable after treatment (20).
Studies reporting a cut-off of ProCT to identify recurrent and cured MTC
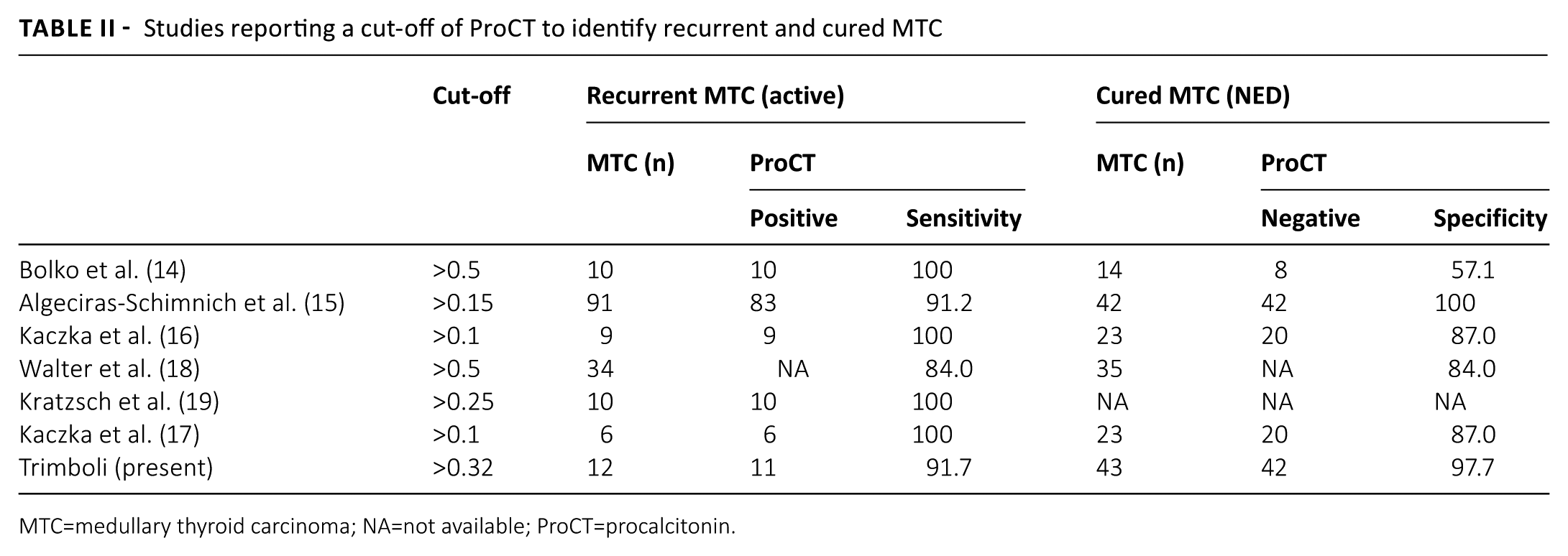
MTC=medullary thyroid carcinoma; NA=not available; ProCT=procalcitonin.
To date, only one paper has been published on the use of ProCT in the washout fluids from FNAC ( 21). In that case report, we showed that the determination of ProCT in FNAC from an MTC is technically feasible, even if this finding should be evaluated in larger series.
Calcitonin has been considered an “ideal” marker for the assessment of patients with suspected MTC and for postoperative follow-up of these carcinomas. However, CT assays have some intrinsic limitations that affect their reliability (6). ProCT data have shown promising results and, notably, ProCT is a very stable protein that does not require being kept cool on ice or frozen. In addition, all commercial ProCT assays yield similar results with the same antibodies, thus enabling cut-off standardization. Finally, it features a concentration-independent in vivo half-life of 20–24 hours (5). As a limitation for the use of ProCT in MTC patients, we have to consider that inflammatory conditions may cause elevations of ProCT by tissues that do not normally transcribe the CT gene (12, 22). These cases are represented by severe infections (i.e. sepsis). To date, we have to verify whether common bacterial infections pose difficulties in the interpretation of ProCT levels from MTC patients, while one study (23) showed that ProCT can be used for monitoring sepsis in a patient affected by MTC. This issue has to be further investigated.
Some limitations of the present study should be declared. We enrolled a small sample of patients and a few active MTC cases. Also, the time of follow-up of patients assessed as NED should be considered short for their final assessment as cured patients.
In conclusion, we found significantly higher ProCT levels in patients with active MTC compared to those with NED. This is in line with previous studies and corroborates the hypothesis that ProCT could be used in MTC management. We advise that further studies undertaken on the trend of ProCT from the preoperative assessment to long-term follow-up. According to the current knowledge, we suggest using ProCT as a complementary test to CT in patients followed-up after surgical treatment for MTC.
Footnotes
Disclosures
Financial support: No financial support.
Conflict of interest: None of the authors has financial interest related to this study to disclose.
