Abstract

Nanotherapeutics have revealed clinical and healthcare significance in treating respiratory diseases and disorders. Implementing different types of nanoparticles for probing disease pathology and the cellular environment is needed to reconnoiter reconnoitering of inflammation-induced lung diseases. 1 Such real-world therapeutic strategies are underlined as key preference to categorize the clinical submissions of new therapeutics. The insights of nanotechnology, 2 pharmaceutical sciences and drug designings are to be scrutinized for refining the quality of state-of-the-art monitoring to have innovative drug delivery summaries for treating chronic inflammatory lung diseases. 3 Moreover, targeting the aforesaid diseases for diagnosis by applying novel theranostics will magnify pulmonary disease monitoring at the nanoscale.4,5 Inflammation is a series of chemical reactions that initiate the transmigration of leukocytes (white blood cells) at the time of tissue damage, bacterial and viral infections transpire at large. If the host cannot defuse the uncertain inflammation, it will initiate organ dysfunction and turn out to be fatal to the host.
Under typical situations, cell natural events cleared pathogens or injured tissue; then, cellular debris and inflammatory mediators transpired through inflammation resolution. 1 Unrepressed inflammation recruit pathogenesis of most diseases, including diabetes, cardiovascular diseases, 6 and tumors, which can be transported by pathogenic roots such as infection, tissue damage, or myocardial infarction. This reaction triggers the creation of cytokines caused inflammatory reactions by activating leukocyte chemotaxis. Redness, discomfort, swelling, loss of function, and heat are indicative of tissue-level inflammation brought on by local immunological, vascular, and inflammatory cell responses. Leukocyte recruitment and accumulation, inflammatory mediator release, and changes in vascular permeability are all significant microcirculatory routes that transpire throughout the inflammatory phase. Leukocytes migrate toward the location of inflammation, and this phenomenon can be used for transportation of NPs.
Biomimetic NPs, such as leukocyte-based ones, imitate the activities of cells or exosomes. They transfer innate tropism of leukocytes to the delivery platform by uniting cell membrane proteins. These strategies and therapies can eradicate noxious pathogens, cure diseases and regenerate injured tissues to reestablish homeostasis. 6 This artefact explores the genesis, mechanisms, resolution, and organ-specific inflammatory responses in different organs, focusing on the activation of nuclear factor kappa B (NF-κB), mitogen-activated protein kinase (MAPK), and Janus kinase/signal transducers and activators of transcription (JAK-STAT) pathways by inflammatory cells initiated by infectious and non-infectious stimuli and cell injury.
Numerous efforts have explored the effectiveness of nanomedicine, such as biodegradable NPs for treating inflammation at the site of tissue injury.
7
To fully fill the objective, nanocarriers based therapeutics were used for the confined and targeted delivery of drugs, nucleic acids, and imaging agents for diagnostics and treatment.
8
These targeted nanotherapeutics were applied for treating inflammatory diseases,
9
including respiratory diseases: cancer, fibrosis, and coronavirus.
10
Another type of nanotherapeutics was applied as a sustained drug delivery resource to treat pulmonary tissues suffering from acute respiratory distress syndrome
11
and for inhalation treatment of lung diseases.
12
A refined class of nanotherapeutics was acclaimed for treating pulmonary arterial hypertension.
13
New viewpoints in nanotherapeutics were described for curing chronic obstructive pulmonary disease, pulmonary arterial hypertension, asthma, silicosis, and cystic fibrosis for chronic respiratory diseases.
14
Furthermore, miRNA nanotherapeutics have the potential to complete with the continued trials in respiratory disorders (Figure 1).
16
Few efforts have been made to prepare remedies for a proper regulation of endocrine system in lung disease and inflammation.
17
The relationship between inflammation and the endocrine system has long been the subject of research. Illustrate the medication delivery methods based on nanotechnology that target pulmonary tissues, especially in acute respiratory distress syndrome. It emphasizes. Reprinted (adapted) with permission from Ref.15 Copyright (2020) Jesús Ruíz-Cabello et al. International Journal of Molecular Sciences, MDPI.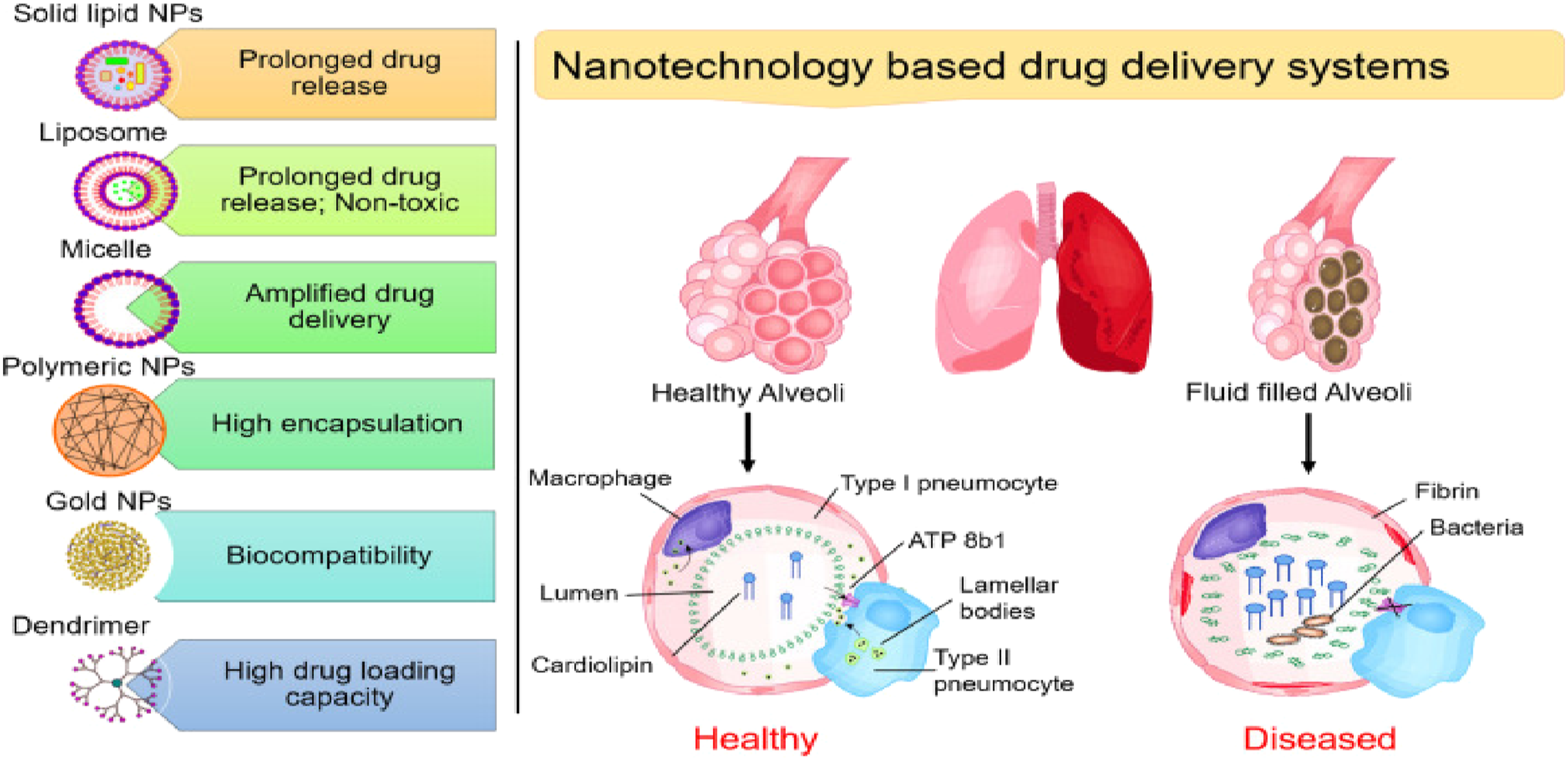
A recent hypothesis explains neuroendocrine changes in chronic inflammatory diseases (CIDs) by pointing out that inflammation-driven energy expenditure programs are beneficial for acute inflammation but not chronic systemic inflammation. Chronic use is unfavorable, and energy-rich fuel allocation is crucial for an innate immune system. Better water retention system activity is also associated with energy allocation to the immune system. Nanotherapeutics display potential in modulating micro-environment to treat lung cancer and chronic obstructive pulmonary disease and can minimize cytotoxic effects by eliminating affected constituents.
18
The limits of nanotherapeutics, clinical difficulties, and potential benefits of microRNA nanotherapeutics in lung targeting and pulmonary hypertension are clarified in a review paper.
15
Herein, the author reports the potential therapeutic claims of microRNA-based medicines as well as their restrictions. In the assessment of chemically modified microRNAs, it emphasizes assistances in stability, less side effects, effectiveness, and local administration. It puts these ideas in the context of nanovectors and also discusses expectations and existing developments in treating pulmonary hypertension (Figure 2). A design of the growth and structural changes of the pulmonary artery are depicted schematically. Excessive smooth muscle cell proliferation causes vasoconstriction, which is followed by remodeling, thrombotic lesions, and inflammation. Reprinted (adapted) with permission from Ref.15 Copyright (2020) Jesús Ruíz-Cabello et al. International Journal of Molecular Sciences, MDPI.
Nanotechnology-based inhalation was considered for treating lung tissue. More, allied routes of distribution of nanotherapeutics were detected in associated organs. 19 Once more, a mini-review was available on nanotherapeutics that highlighted the impressive and immense qualities of these remedies and their impact on the progress of the pharmaceutical industries. 20 In one more assessment, an extracellular vesicle-based nanotherapeutics engineering was emphasized as emerging front and marked as an anti-inflammatory therapy for treating interrelated disorders. 21 Urgent need exists to treat chronic respiratory disorders such as asthma, chronic obstructive pulmonary disease, tuberculosis (TB) and lung cancer by applying advanced nanotherapeutics, and that is the best option to be useful to treat aforesaid diseases and is capable of activating the airway remodeling features of the allied tissues. 22 In continuation, many other nanotechnology based therapeutics for lung disease were revealed. 23 Lung-selective 25-hydroxycholesterol nanotherapeutics are applied to suppress inflammatory nuclear factor kappa B (NF-κB) and sterol regulatory element-binding protein 2 (SREBP2)-mediated inflammasome signaling pathways. 24 These signalling routes are responsible for COVID-19 pathogenesis, inducing acute respiratory distress syndrome (ARDS) and sepsis. Discussed nanotherapeutics are effectively controlling the COVID-19-associated cytokine storm 25 leukocyte-mediated delivery of nanotherapeutics that can improve current therapies in inflammation, immune disorders and cancers at Inflammatory and tumor sites. 26
Neutrophil-mediated delivery of therapeutic NPs displayed potential in overcoming blood vessel barriers for treating inflammation and infection, as well as the ability to hijack neutrophils for the delivery of nanodrugs at the location of the diseased sites. 27 The expansion of novel targeted anti-inflammatory therapies for lung inflammation, and respiratory tract homeostasis is challenging due to the intricate administration route, biodistribution, and macrophage targeting of these NPs in the lung. 28 Designing such NPs, which can delivery resolvin D1 (RvD1) and antibiotics simultaneously, is a new strategy that is been tested in recent times. In order to treat P. aeruginosa-induced lung infections, the study suggests using RvD1 and an antibiotic (ceftazidime) in human neutrophil membrane-derived nanovesicles that target inflamed vasculature.
The current treatment of infectious disorders is made thinkable by this dynamic mechanism, which guards against host harm instigated by inflammatory reactions brought on by infections. These newly cutting-edge nanovesicles can cure infectious diseases, target host inflammatory pathways associated with existing pathogens in lungs, reduce inflammation, and clear bacteria. 29 However, molecular imaging of inflammatory lung diseases by tracking of inhaled near-infrared fluorescent NPs in the lungs that cause allergic airway Inflammation was reported. 30
Engineered cell membrane-based nanotherapeutics that hold exciting potential in modulating inflammation are effective for targeted delivery at the inflammatory tissues. These remedies cleared persistent challenges and opened newfangled overtures for targeting and manipulating the inflammatory microenvironment by directly pointing inflammation.31,32 In the meantime, antioxidant nanotherapies 33 and inflammation-responsive polymers 34 were acclaimed for treating inflammatory diseases. Bilirubin NPs displayed their character as endogenous anti-inflammatory agents and were considered anti-inflammatory therapy. 35 However, during acute lung damage brought on by sepsis, photobiomodulation plays a crucial role in supporting tissue homeostasis restoration, reducing inflammation, and modulating its resolution. 36 Evolving nanotherapeutics were also reported for antithrombotic treatment. 37 NPs were used to treat an inflammatory microenvironment with mild acidity and high reactive oxygen species and for targeted treatment of arterial restenosis. 38 Moreover, nanomedicine-based therapeutics combat acute lung injury. 39
Testified outcomes on nanotherapeutics for lung disease were high points and emphasized as new outlooks for proceeding with translational respiratory research. 40 Newly revealed claims and specified strategies in nanotechnology exhibited their potential in locating target tissues and enhancing the therapeutic effect of treating lung diseases. 41 Nanomedicine for Inflammatory disease is a book that covers progressive hallmarks in translational medicine as it unites clinicians treating inflammatory disease with scientists developing experimental nanomedicine therapeutics. 42
A review article covered the recent progress of the use of biomaterials, nanotechnology, and stem cells for detection and treatment of COVID-19, and proposed new features to deploy the aforesaid strategies in each step of the fight against SARS-CoV-2, which can provide a framework to discourse future global pandemics. 43 The creation of silica NPs as a vaccine carrier is being investigated for the SARS-CoV-2 vaccine. Using particular protective equipment, cleaning surfaces to break the transmission chain is crucial to combating the pandemic. Enrichments in cellular biology, biomaterials, and nanotechnology present prospects for more effective SARS-CoV-2 defense. Therapeutic objectives can be identified and targeted with the use of functional level NPs.
Clinical studies for the development of an effective nanovaccine can start if the viral life cycle and host response are sufficiently understood. Based on investigations carried out by experts worldwide, researchers anticipate the development of a universal NP-based vaccination with effective immunogenicity in the near future. Immunopharmacology and inflammation are the two different phenomena of lung physiology. The breathing, blood flow, and heartbeat all cause the lung to be a dynamic organ that is always in motion. Its intricate network of veins makes gas exchange easier by reducing the gap between blood and air. Red blood cells with a high hemoglobin content may move through this oxygen gradient. The lung is exposed to the world beyond breathing, which creates a vast network of immune cells that guard the blood and airways from infections. Whereas the complexity of the immune system ruled the activities allied to protection and self-damage, immunopharmacology signifies the challenge of effective and safe immunopharmacological intervention. These key investigations were applied to the advancement of personalized nanotherapeutics.
NPs impact the immune system of the respiratory tract, and create oxidative stress and genotoxicity.
44
However, the diversity of nanotechnology could revolutionize medicine, but making safety assessment a challenging task.
45
The contests persisted in developing conventional and nanotechnology based methodologies to combat chronic obstructive pulmonary disease as well as the implications of chronic airway diseases.
46
Not only are these nanotherapeutics useful for treating Inflammation-induced lung diseases, but targeted interleukin-10 nanotherapeutics were transformed into a microfluidic chip that will reduce inflammation during the treatment of atherosclerosis
The rationale and state-of-the-art nanotechnology for pulmonary drug delivery, with particular attention to new technological developments and approaches as well as the challenges associated with them, the emerging advances, and opportunities for future development in this field. 50 Overall, it can be deduced from this lookout that nanotherapeutics have the potential to cure inflammation promoted and tempted lung diseases in future.
Footnotes
Acknowledgments
One of the authors, Rajiv Kumar, gratefully acknowledges his younger brother, Bitto.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
