Abstract
Introduction
According to studies conducted in China and other countries, the incidence rate of malocclusion varies, typically falling between 20% and 100%. Patients with malocclusion, the majority of whom are youngsters and teenagers, require orthodontic treatment.1,2 The advancements in the medical field have led to a continuing improvement in orthodontic technology. A straight wire appliance is a frequently utilized fixation devices for orthodontic treatment in clinic since it is widely recognized by most practitioners. 3 Even while orthodontic treatment has advantages such as bettering facial appearance and oral health, food residues can readily accumulate due to the intrusion of equipment into periodontal tissue, which affects oral hygiene and makes it harder to remove dental plaque. 4 Inadequate dental hygiene will encourage the growth of new bacteria, which will alter the mouth’s microbiological balance and alter the oral environment. Eventually, this will cause inflammation of the soft tissue. One of the main causes of brace dislocation and unsuccessful corrections is inflammation of the soft tissues.5,6 Even while these reactions are typically reversible, if they are not treated seriously, they run the risk of becoming irreparable damage to the periodontium. Clinical monitoring of periodontal pathogenic microorganisms can effectively prevent this.
Porphyromonas gingivalis (P.g), a spore-free Gram-negative coccobacillus, is the primary pathogen responsible for the irreparable destruction to periodontal tissue. It produces immunoinflammatory periodontal disease, which is a chronic, non-infectious condition.7,8 The most active component of P.g is its extracellular cysteine protease, gingipains. It includes Lys-gingipain (Kgp) and Arg-gingipain (Rgp), the secretion of which is related to the cellular form.9,10 The former can increase vascular permeability, which in turn can cause inflammatory edema in the gingival tissues by contributing to the degradation of collagen types Ⅰ and Ⅱ in those tissues. The latter can contribute to the development of periodontal disorders by compromising host cell defense mechanisms through pathways linked to protease-activated receptors. In order to explain the positive rate of P.g and the changes in Kgp and Rgp expression with ongoing orthodontic treatment, as well as to provide a theoretical foundation for the targeted improvement of bacterial infection in the orthodontic process for adolescents, this study examined the effect of P.g expression on gingival pain and gingival inflammation during orthodontic treatment.
Materials and methods
General data
In our hospital’s orthodontic department, 105 teenage patients (35 males and 70 females, aged 12–17 with an average age of 14.34 ± 4.67) were chosen between January 2017 and June 2020. The sample size was estimated using the equivalency test, assuming that 100 patients would have the same efficacy both before and after therapy. 5 cases were added to each group for processing in order to prevent insufficient sample content caused by patient removals and sheds during the experiment, resulting in 105 samples in total. Necessary Written Informed consent of patients or the family members of patients were obtained. The Medical Ethics Committee of Shandong Medical College’s Department of Stomatology approved the treatment (2023-04-01-01).
Inclusion criteria included: (1) fixed device therapy; (2) Age 12–17 years when starting treatment; (3) no previous medical contraindications or routine medication (including antibiotic therapy); (4) Non-smokers; (5) permanent dentition; (6) mandibular arch incisor irregularity index 4∼12 mm. Exclusion criteria: (1) failure to attend follow-up visits on time; (2) Take antibiotics. 11
After the 1st, 2nd, 3rd and 6th months of orthodontic therapy, the changes in P.g positive, Kgp and Rgp levels of all patients were tallied.
Collection of specimens
Before receiving orthodontic treatment, as well as during the 1st, 2nd, 3rd, and 6th months of treatment when the device was worn, gingival crevicular fluid was obtained from the patients. To put it briefly, the patients’ dental cavities were cleansed with 0.9% NaCl before the specimen teeth were isolated. Following the blow-drying of the tooth surface, the aseptic paper tip was placed into the gingival sulcus and removed 10–30 s later. It was then plced into a sterile Eppendorf (EP) tube and refrigerated at −20°C to await additional testing.
Culture of bacteria
The samples were cultivated on blood agar or cerebroth supplemented with menaquinone (1.l g/mL) and heme (5.l g/mL) under anaerobic conditions at 37°C (10% CO2, 10% H2 and 80% N2). Add 5% defibrimmated sheep blood to pancreatic soy agar for use in blood agar plates. Agar plates have 1.5–2 mm diameter colonies grown on their surface cilia are typically lustrous but not always black. Non-Grammatical by using Gram staining, asphorocystococcus is P.g. 12
The following procedure was used to extract the specimen’s deoxyribonucleic acid (DNA): After dissolve the cultivated colony samples, centrifuge them. After removing the supernatant, the precipitate was collected and 200 μL of lysis buffer was added. Following adequate mixing, the mixture was placed in a water bath at 100°C for 10 min, and then the mixture was centrifuged. Subsequently, 150 μL of the supernatant was collected as a template and kept in a refrigerator at −20°C for further testing.
Polymerase chain reaction (PCR) amplification
On DNA samples, PCR amplification was carried out. The 16S rDNA fragments of P.g were 404 bp, primer sequences were F: 5'-AGGCAGCTTGCCATACTGCG-3' and R: 5'-ACTGTTAGCAACTACCGATGT-3'. The 16S ribosomal DNA (rDNA) fragments of Kgp were 870 bp, primer sequences were F: 5'-GAACTGACGAACATCATTG-3' and R: 5'-GCTGGCATTAGCAACACCTG-3'. The 16S rDNA fragments of Rgp were 507 bp, the primer sequences were F: 5'-GAACTGAGGACCATCATTG-3' and R: 5'-GCTGGCATTAGGAACACCTG-3'. The mixture of 10×TransStartTaq Buffer (2.5 μL), primers (1.0 μL each), TransStartTaq DNA Polymerase (0.5 μL), dNTPs (2.0 μL), and template DNA (10 μL) was supplemented with sterile double-distilled water to a total volume of 25 μL. The reaction conditions were 94°C for 5 min, 94°C for 30 s, 57°C for 30 s, 72°C for 1 min (33 cycles), and 72°C for 10 min, followed by storage at 4°C.
Polymerase chain reaction assay
The 2×EasyTaq PCR SuperMix PCR kit from Tiangen Biotech (Beijing) Co., Ltd and the gradient PCR machine (T-GradientThermoblock Biometra) were used. P.g levels are determined, and Kgp and Rgp levels are further measured in P.g-positive patients.
Methods for orthodontic treatment
The straight wire orthodontic technique was applied to orthodontic treatment and oral hygiene education was performed 1 week prior to the treatment. All patients were required to adopt the Bass brushing method and carry out post-meal tooth brushing with orthodontic toothbrushes for at least 5 min each time.
Statistical analysis
The statistical analysis of data was conducted with SPSS 23.0 software. Mean ± standard deviation (‾x±s) was used to represent continuous variables with normal distribution, which were compared by the two-independent sample t-test among groups. In terms of the measurement data with non-normal distribution, M (P 25 , P 75 ) was used as expressions and the Mann-Whitney U test was conducted for comparison among groups. Regarding the count data, frequency (%) and χ 2 test were employed, respectively. The spearman correlation coefficient test was used for correlation analysis, r is the correlation coefficient, <0.3 is uncorrelated, 0.3∼0.7 is weakly correlated, and >0.7 is strongly correlated. In addition, differences were identified to be statistically significant at p < .05.
Results
Specificity and sensitivity of 16S rDNA PCR assay
Aa ATCC29522, Fn ATCC25586 and the blank control didn’t produce the target bands in agarose gel electrophoresis following amplification, whereas the P.g ATCC33277 and P.g W83 strains produced clear, specific bands of 404 bp (Figure 1), suggesting that the primer is specific for the amplification of P.g, and that the PCR reaction system is sensitive to the detection of P.g. Specificity of P.g by 16S rDNA PCR. M: marker. 1: Aa ATCC29522. 2: Fn ATCC25586. 3: P.g ATCC33277. 4. Blank control. 5: P.g W83.
Results of PCR amplification of specimens
Porphyromonas gingivalis detection results showed that clinical specimens 2, 6, 8 and 10 positive control 1 produced target bands of 404 bp, while clinical specimens 3, 4, 5, 7 and blank control 9 as well as negative controls 11 and 12 failed to do so (Figure 2(a)). Positive controls 1 and 2, clinical specimens 4 and 5, and blank control 3 didn’t produce the target bands of 870 bp, whereas clinical specimens 6 and 7 (from P.g positive patients) and negative control 8 didn’t produce the desired bands, according to the Kgp detection results (Figure 2(b)). The findings of Rgp detection showed that, whereas negative control 3 and blank control 4 were unable to produce the target bands, positive control 1 and clinical specimens 2, 5, 6, 7 and 8 were able to produce specific bands of 507 bp (Figure 2(c)). Detection results of PCR amplification products of specimens. (a) PCR results of P.g. M: marker. 1: P.g W83. 2-8 and 10: clinical specimens. 9: blank control. 11: Aa ATCC29522. 12: Fn ATCC25586. (b) Results of Kgp detection in P.g-positive patients by PCR. M: marker. 1: P.g W83. 2: P.g ATCC33277. 3: blank control. 4-8: clinical specimens. (c) Results of Rgp detection in P.g-positive patients by PCR. M: marker. 1: P.g W83. 3: Fn ATCC25586. 4: blank control. 2 and 5-8: clinical specimens.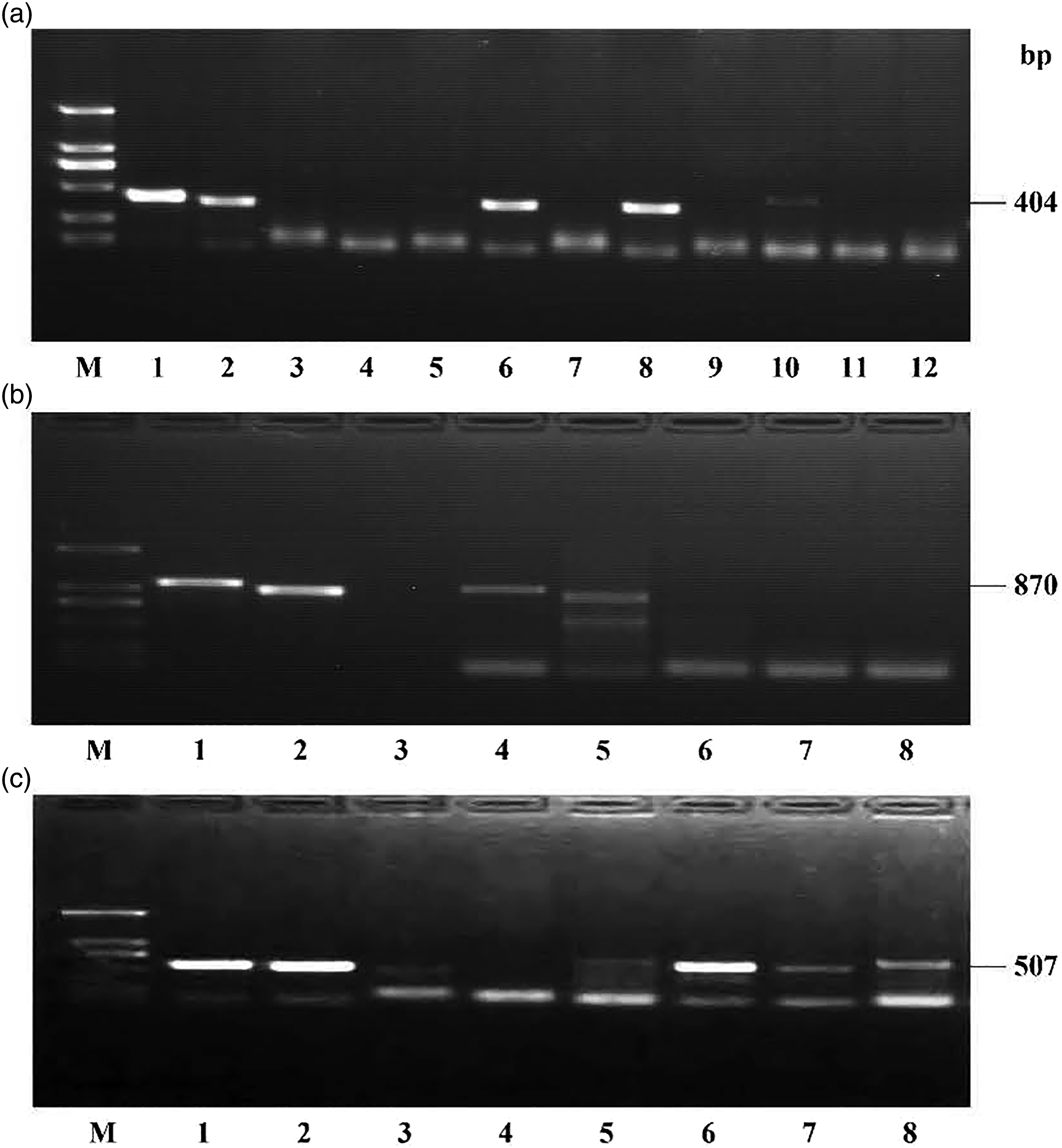
Detection rate
Detection rate.

ap < .05 vs. before orthodontic treatment.

Detection rates of P.g, Kgp and Rgp.
Detection rate of P.g and Kgp in patients with different Gingivitis indexes (GIs)
Detection rates of P.g and Kgp in patients with different GIs
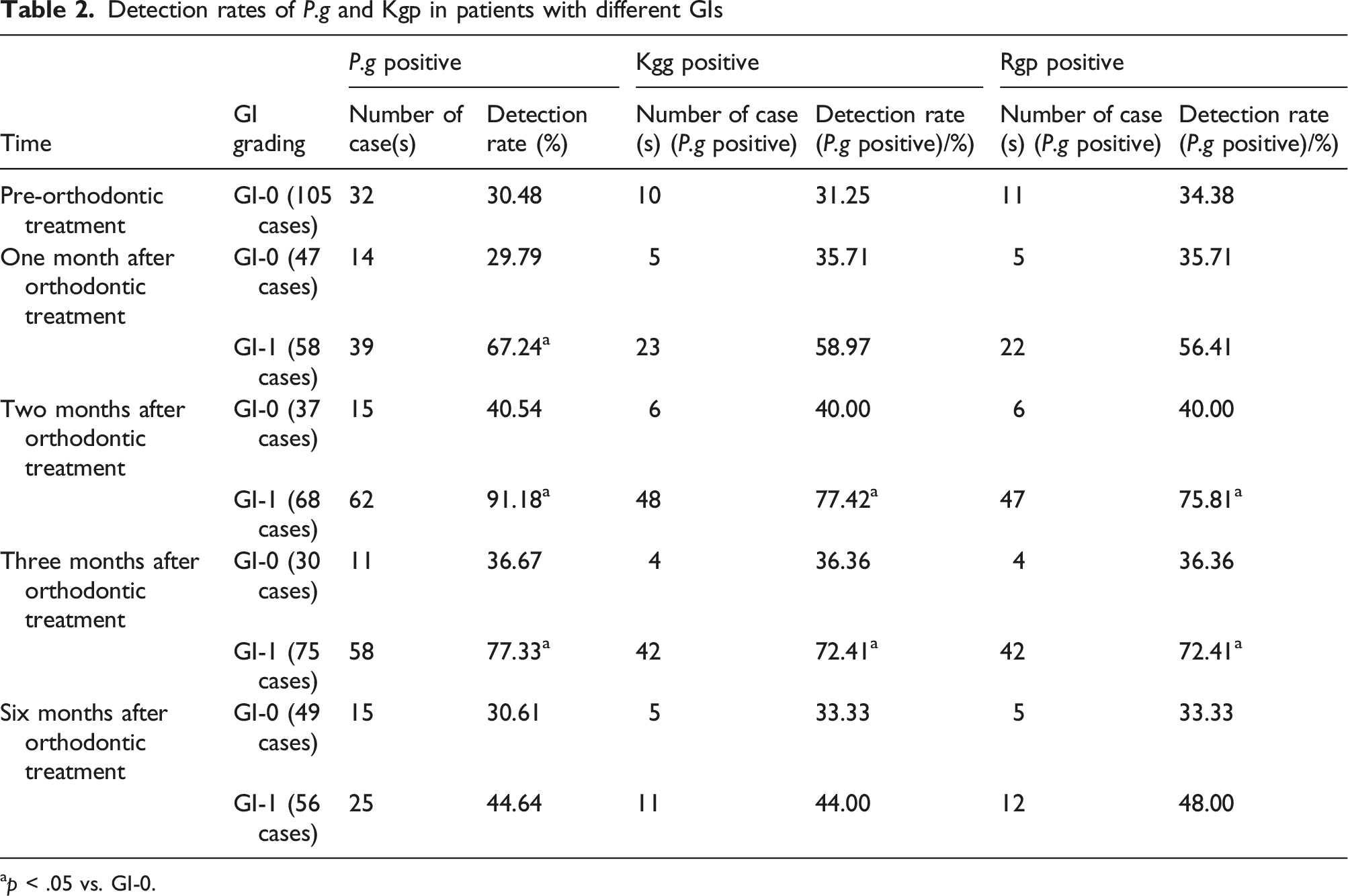
ap < .05 vs. GI-0.
Correlation analysis results
Correlation analysis results.
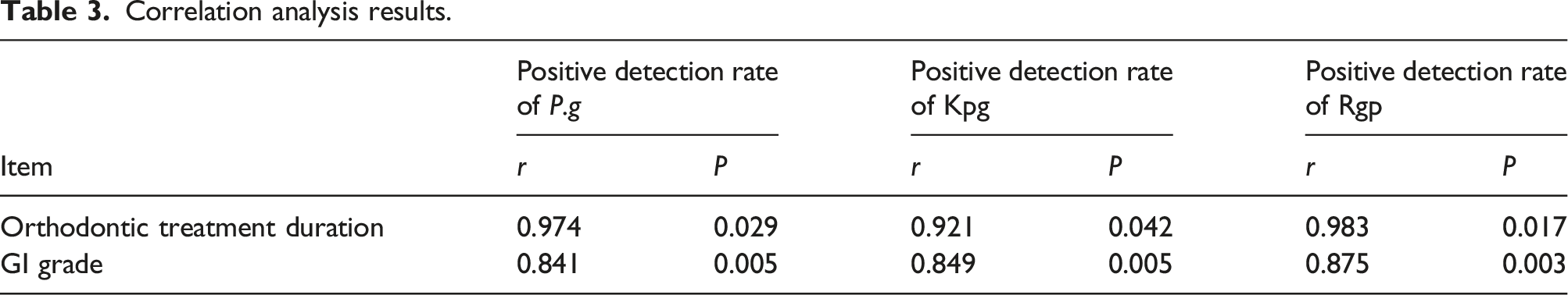
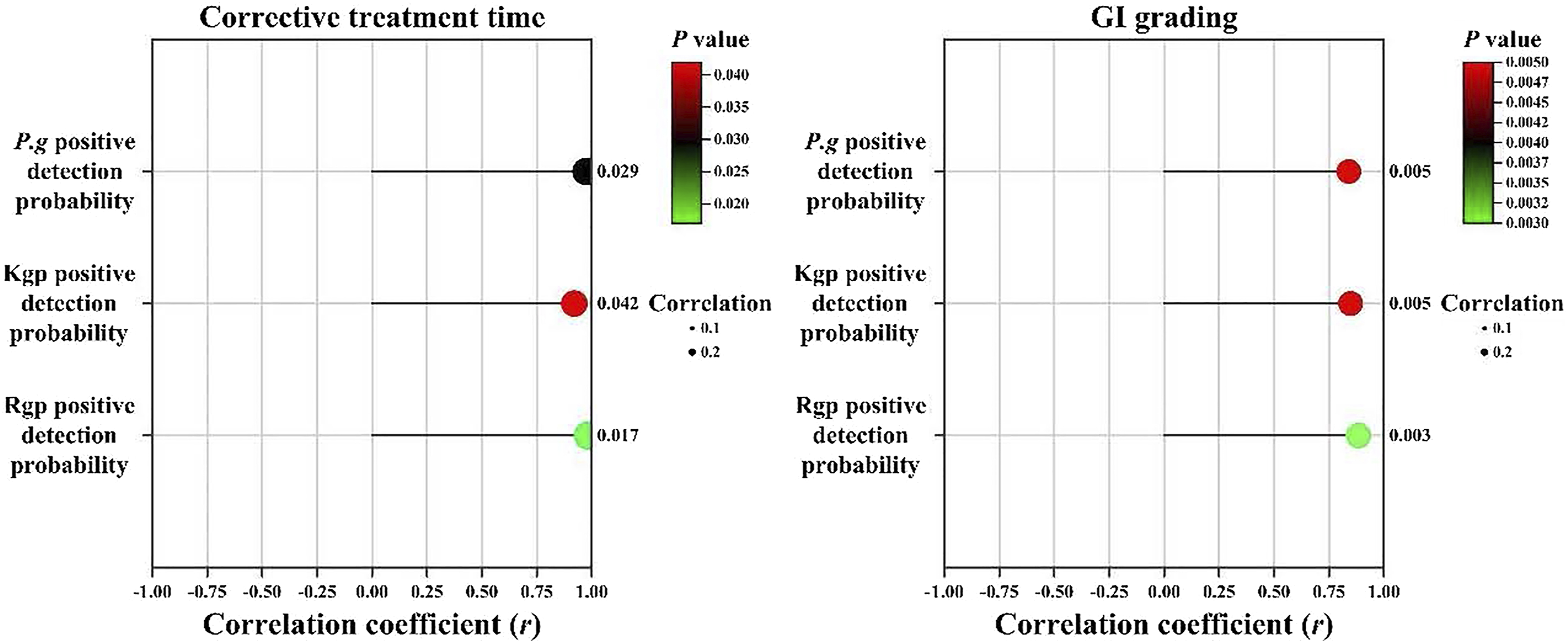
Correlation analysis results.
Discussion
With the advancement of civilization and rising living standards, people's conceptions of beauty have progressively evolved, and an increasing number of individuals are seeking to better their personal environments in order to achieve greater. In this setting, plastic surgery and medical aesthetics—including orthodontic—have become more popular. One of the most common diseases in clinical stomatology is malocclusion, which is defined as abnormal jaw size, shape and position, abnormal upper and lower arch occlusal relationships, and abnormal facial deformities. Environmental factors, such as illnesses, poor oral hygiene, disorders involving dental replacement, trauma, periodontal disease and other causes, can also sontribute to malocclusion.13,14 Patients’ oral health, facial appearance and oral function are all negatively impacted by malocclusion. As previously reported, there is a strong correlation between gingival inflammation and P.g and its gingipains. 15 In the course of orthodontic treatment, the environmental balance in the oral cavity is disrupted by the orthodontic fixing appliance, which increases plaque and converts good bacteria into harmful bacteria, causing gingival irritation.
In this study, it was confirmed that orthodontic treatment can cause abnormal expression of P.g, Kgp, and Rgp. Studies have shown that periodontal disease caused by P.g can be effectively alleviated by oral probiotics, and probiotic mouthwashes also have a significant effect on reducing P.g levels.16,17 Oral probiotics combined with probiotic gargling in pre-orthodontic patients can have the effect of preventing PG, so it is recommended to take probiotics clinically after the start of orthodontics.
The PCR assay approach can overcome challenges including P. g culture, lengthy culture periods, strict criteria for culture conditions, and challenging identification and separation procedures. These advantages include sensitivity, simplicity, specificity and rapidity.18,19 Moreover, it is specific for identification of P.g. The 16S rDNA PCR method was used in this investigation to detect P.g, and the findings showed that it was specific and sensitive for P.g detection. The percentages of patients who tested positive for P.g before and after receiving orthodontic treatment were 30.48%, 50.48%, 73.33%, 65.71% and 38.10%, respectively. When orthodontic treatment was received for 1, 2, 3, 6 months, the positive detection rate of Kgp was 31.25%, 52.83%, 70.13%, 66.67% and 40.00%, respectively. When orthodontic treatment was received for 1, 2, 3, 6 months, the positive detection rate of Rgp was 34.38%, 50.94%, 68.83%, 66.67% and 42.50%, respectively. In the first and second months following orthodontic treatment, the positive detection rate of P.g as well as that of Kgp and Rgp in P.g-positive patients gradually increased. This suggests that the orthodontic fixing appliance destroys the patients’ oral environment and microecological balance, especially during the first and second months of orthodontic treatment. The orthodontic appliance that is used to fix the braces is to blame since it makes it difficult for the patient to maintain good oral hygiene because it collects food particles, which promotes bacterial growth and microecological imbalance, which in turn leads to gingival inflammation. The positive detection rate of P.g was higher in the early stage of orthodontic treatment, according to findings of Liu 20 and Pan et al., 21 After 3 months of orthodontic treatment, the positive detection rate of P.g as well as that of Kgp and Rgp in P.g-positive patients decreased, and after 6 months, it essentially reached its pre-treatment level. In P.g-positive patients with GI-1, the positive detection rate of P.g, Kgp and Rgp were higher than those in GI-0 patients. In spite of the patients’ early-stage oral environment disorder and the excessive bacterial reproduction brought on by poor oral hygine, the patients’ bacterial reproduction was effectively controlled, and after 3 months of orthodontic treatment, gingival inflammation gradually subsided and they essentially recovered by the 6th month. This was due to the patients’ gradual adaption to the orthodontic fixing appliance and their attention to oral hygiene. The results of the correlation analysis showed a positive link between the length of orthodontic treatment and GI and the positive detection rates of P.g, Kgp and Rgp. This suggests a tight relationship between the development of gingival inflammation and P.g, Kgp and Rgp.
However, more research is necessary to address the issue of bacterial infection during the orthodontic process, as this study is still insufficient and doesn’t provide a solid treatment plan. In addition, although P.g is the primary bacteria responsible for gum inflammation during orthodontic treatment, it is still unknown whether other bacteria influence patients’ gum inflammation during orthodontic treatment. As a result, numerous clinical inflammation verification studies are still needed.
Conclusion
In conclusion, the levels of P.g, Kgp and Rgp are increased in the early stage of orthodontic treatment due to the appliance, which leads to gingival inflammation. However, they are on the decline in the 3rd month and almost close to those before orthodontic treatment by the 6th month. Therefore, aggressive oral cleaning in the early stages of orthodontic treatment is effective and necessary.
Footnotes
Author contributions
We declare that all the listed authors have participated actively in the study and all meet the requirements of the authorship. Dr. LW designed the study and wrote the paper, Dr. ZW undertook the statistical analysis, Dr. MZ managed the analyses, Dr. SX contributed to the literature searches, Dr. QG contributed to the correspondence and paper revision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Shandong University Scientific Research Program Project (No. J18KB133) from Shandong Provincial Department of Education.
Ethical statement
Data availability statement
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
