Abstract
Objective
Rheumatoid arthritis is an autoimmune disease characterized by synovial inflammation-driven cartilage and bone destruction, a process mainly mediated by osteoclasts. In recent years M2-like macrophages have been found to play an important role in the pathological process of RA by mediating pro-inflammatory effects, but their roles in the bone destruction of autoimmune arthritis have not been reported.
Methods and Results
In this study we identified that an abundant cell population of CD45+CD11b+Gr-1-F4/80+CD206+ cells, which were normally classified as M2-like macrophages, was present in synovium of collagen-induced arthritis (CIA) mice, and these cells had the potential to differentiate into osteoclasts. These M2-like macrophages sorted from CIA synovium highly expressed RANK and could be activated by RANKL and M-CSF to acquire osteoclast markers and bone resorption function both in vitro and in vivo. Furthermore, in vitro differentiated M2 macrophages from both CIA mouse bone marrow and RA patient peripheral blood mononuclear cells were also able to differentiate into osteoclasts, confirming the general osteoclastogenesis capability of M2 subtype macrophages.
Conclusion
All these results suggest that synovial F4/80+CD206+ M2-like macrophages in RA may be novel osteoclast precursors and contribute significantly to bone erosive changes seen in RA. Our studies provided new directions and targets for the diagnosis and treatment of rheumatoid arthritis.
Keywords
Introduction
Rheumatoid arthritis (RA) is a chronic, progressive inflammatory joint disease, characterized by synovitis and cartilage and bone damage.1,2 Osteoclasts are well accepted as the only cell type with bone-destroying capacity, playing key roles in steady-state bone remodeling and arthritic bone erosion.3,4 Decades of efforts on the origin of the osteoclasts have identified that monocytes/macrophages, immature DCs 5 and MDSCs 6 could differentiate into osteoclasts through classical RANK-dependent or non-classical RANK-independent 7 signaling pathway, but it still requires additional detailed characterization on the cell type as osteoclast precursor in RA pathogenesis.
Macrophages are particularly abundant among infiltrating immune cells in joints of RA patients and CIA mice, and play important roles in pathogenesis of RA.1,8,9 Macrophages exhibit phenotypical and functional plasticity and can acquire a continuum of polarization states depending on the environmental cues. Classically activated macrophages (M1) are characterized by elevated expression of CD86, release of proinflammatory cytokines (TNF-α, IL-6 and IL-1β etc.), and generation of reactive oxygen species. On the other hand, alternatively activated macrophages (M2) show more phagocytic activity, higher expression of scavenging receptor (CD163), mannose receptor (CD206), Arginase-I, IL-10 and TGF-β.10–13 During chronic inflammation when there are overlapping waves of proinflammatory and anti-inflammatory signals and cells, however, it’s hard to clearly distinguish M1 and M2 macrophage phenotypes.1,14 We previously found that after exposure to plate-coated IgG (representing insoluble IgG immunocomplex deposited in local tissues) plus bacterial lipopolysaccharides (LPS), or lactoferrin-containing immunocomplex elevated in RA sera, M2 macrophages strongly produced inflammatory cytokines, indicating that co-stimulation through FcγRs and TLRs could directly switch human macrophages from M2 into M1-like phenotype, thereby promoting excessive inflammation in autoimmune diseases such as RA.15,16 In CIA mouse synovium, up to 50% of infiltrated mononuclear cells were CD163+ M2 macrophages, and they could release pro-inflammatory cytokine TNF-α, exhibiting pro-inflammatory potentials.17,18 All these studies suggest that M2 macrophages play an important role in the pathological process of RA by mediating pro-inflammatory effects, but their role in the bone destruction of autoimmune arthritis has not been reported.
It has been reported that CD163+ CD206+ M2 macrophages composed some 60% of the RANK+ cells in Extramammary Paget’s disease, 19 and soluble RANKL released from Paget cells promotes the production of CCL17 in these macrophages RANK-dependently. 20 Our data also found that synovial F4/80+CD206+ M2-like macrophages from CIA mice strongly augmented expression of RANK compared with that from control mice. We therefore hypothesized that F4/80+CD206+ M2-like macrophages in RA synovium may be able to differentiate into osteoclasts via RANKL-RANK signaling pathway and involved in bone destruction and absorption in RA. In the present study, F4/80+CD206+ M2-like macrophages isolated from synovium of CIA mice were shown to acquire osteoclast markers and bone resorption function in vitro, and in vivo when transferred into normal male DBA/1J mice. Osteoclastogenesis capability of M2 macrophages from different sources, including F4/80+CD206+ M2 sorted from bone marrow of CIA mice and in vitro differentiated M2 macrophages from CIA mouse bone marrow and RA patient peripheral blood mononuclear cells (PBMCs), were also confirmed. Our data reveal novel osteoclastogenesis activity of M2 macrophages and its potential contribution to bone erosive changes seen in RA.
Methods
Mice
Male DBA/1J mice, 8-10 weeks old, were purchased from SLAC Laboratory Animal Center (Shanghai, China). Mice were housed under specific pathogen-free conditions. All protocols were approved by the Medical Ethical Committee of Soochow University (Ethical approval number: SYSK-(S2012-0062)).
Induction and assessment of collagen-induced arthritis
The model was generated by immunizing mouse for twice with collagen II (CII) emulsion in complete Freund’s adjuvant (CFA) and a second CII emulsion in incomplete Freund’s adjuvant (IFA) on day 21. An equal volume of CII was emulsified with CFA containing 4 mg/mL of inactivated mycobacterium tuberculosis (Sigma, USA). The emulsion was prepared and kept on ice before injection. The mice were injected intradermally at the base of the tail with 100 μL of the emulsion containing 100 μg of CII on day 0 and 21. The control mice were generated by immunizing mouse for twice with PBS in CFA and a second PBS in IFA on day 21.
Isolation of synovial cells from ankle joint
Synovial cells were isolated from ankle joints of control or CIA mice as described previously. 4 Briefly, mice were perfused with 15 mL pre-warmed 1 × PBS. After removal of the skin and biceps femoris muscle, the quadriceps femoris muscles including the vastus intermedius muscle were pinched and lifted with tweezers. The quadriceps femoris muscles and patellar ligament, including the patella, were removed from the knee joint under a stereoscopic microscope (SMZ 745T; Nikon). The hypertrophied synovium was isolated without damaging the bone. Ankle joint tendons, including the Achilles tendon, were removed, revealing the hypertrophied synovium around the talus, which allowed for isolation without damaging the bone. Synovial tissues were digested with 3 mg/mL type I collagenase in HBSS, and incubated at 37°C for 30 min. Disaggregated tissue elements were passed through a 70-µm cell strainer. The synovial cells were collected and washed twice with PBS. That centrifuging is necessary and the settings were used for 500 g 5 min.
For sorting of mouse synovial CD206+ and CD206- macrophages, the isolated synovial cells were stained with fluorescent labeled CD45, Gr-1, CD11b, F4/80, CD206 antibodies, then CD45+CD11b+Gr-1-F4/80+CD206+ or CD206- macrophages were sorted by flow cytometry.
Macrophage differentiation and analysis
Bone marrow cells from control or CIA mice were cultured for 6 days in RPMI 1640 (Hyclone) containing 10% fetal bovine serum (FBS, Biological Industries) supplemented with 20 ng/mL recombinant mouse M-CSF (Peprotech). At day 3, half of the medium was replaced by new medium containing cytokines of which M-CSF. At day 7, the medium was totally replaced in the presence of 20 ng/mL recombinant human IFN-γ for M1, and 20 ng/mL IL-4 for M2, new medium containing no cytokines for M0, respectively. We can acquire macrophage after 24 h.
PBMCs were isolated from heparinized peripheral blood from healthy donors or RA patients without treatment (N = 3) by density gradient centrifugation at 500 g for 30 min on Ficoll lymphocyte separating solution (Dakewe Biotech) at room temperature. All donors gave written informed consent to participate in the study and all protocols were approved by the Medical Ethical Committee of Soochow University (Ethical approval number: SYSK-(S2012-0062).
CD14+ blood monocytes were purified from PBMCs by magnetic cell sorting using CD14 microbeads (Miltenyi Biotec, Germany), and used for generation of M1 and M2 macrophages. Monocytes were cultured for 6 days in RPMI 1640 (Hyclone) containing 10% fetal bovine serum (FBS, Biological Industries) supplemented with 20 ng/mL recombinant human M-CSF (Peprotech). At day 3, half of the medium was replaced by new medium containing cytokines. At day 7, the medium was totally replaced in the presence of 20 ng/mL recombinant human IFN-γ for M1, and IL-4 for M2, respectively. We can acquire macrophage after 24 h.
Differentiation of osteoclast
Fifty thousand sorted macrophages or bone marrow-derived macrophages (M1, M2) were cultured with 10 ng/mL M-CSF and 100 ng/mL RANKL in 96-well flat-bottom plates containing 200 uL of osteoclast-culturing media (α-MEM with 10% serum). At day 3, half of the medium was replaced by new medium containing cytokines of which 10 ng/mL M-CSF and 100 ng/mL RANKL. After 6 days, we can see large, nucleated and tartrate-resistant acidic phosphatase (TRAP) expressing cells.
Real-time quantitative RT-PCR
Total RNA was extracted with Omega RNA Isolation Kit and reverse transcribed and real-time PCR was performed with SYBR Green (Takara, Kusatsu, Japan) on a Step One Plus real-time PCR system (Applied Biosystems, Carlsbad, CA, USA). Changes of gene expression were calculated by the 2-44 Ct method using GAPDH as internal reference gene.
Flow cytometry
Cells were harvested from tissue culture plates by treatment with 0.25% trypsin, followed by washes and staining with fluorescence-labeled Abs for 30 min on ice. For phenotyping, cells were collected, washed with phosphate-buffered saline (PBS), and the pellets were incubated for 30 min at 4°C with 50 μL AF700-conjugated CD45, APC-conjugated anti-mouse CD11b, PercP-Cy5.5-conjugated Gr-1, anti-mouse APC-Cy7-conjugated anti-mouse F4/80, FITC-conjugated anti-mouse CD206 or PE-conjugated anti-mouse CD36/IAIE/Rank, or AF700-, FITC-, PercP-Cy5.5-, APC-, APC-Cy7-, PE-conjugated isotype control Abs (Biolegend). After washe with PBS, the cells were subjected to analysis by flow cytometry Attune NxT (Life Technology).
TRAP staining
TRAP staining was performed following the procedure of a TRAP staining kit (Sigma-Aldrich). Briefly, the differentiated cells were treated with fixative solution at room temperature for 3 min, then rinsed thoroughly in 37°C deionized water for 5 min. After three washes, the fixed cells were treated with staining buffer containing diazotized fast garnet GBC, naphthol AS-BI phosphate, acetate, and tartrate solution at 37°C. The reaction was stopped by deionized water after 5 to 10 min. The cells imaging was performed with a Nikon microscope.
F-actin ring staining
After inducing to differentiate into osteoclast for 6 days, the cells were fixed with 3.7% formaldehyde solution in PBS for 10 min, permeabilized with 0.5% Triton X-100 for 5 min, and incubated with 100 nM TRITC Phalloidin (Yeasen, Shanghai, China) in 1% BSA solution for 30 min at room temperature. After washing with PBS three times, cells were stained with DAPI for 10 min to label the nuclei and were photographed under a fluorescence microscope.
Bone resorption assay
Macrophages were differentiated with M-CSF plus RANKL for 6 days, followed by seeding in Corning Osteo-Assay Surface 96-well plates (Corning Inc., Corning, NY, USA) at a density of 1 × 105 cells/well in medium, replenishing every 3 days 18 days later, cells in the plates were removed by incubating with deionized water for 2 h, and the plates were treated with 5% silver nitrate solution and placed under ultraviolet light for 30 min. The wavelength of ultraviolet light about 320–400 nm. After washing the plates with distilled water for three times, the wells were treated with 5% sodium carbonate at room temperature for 30 min and washed again with distilled water. Finally, the wells were air-dried before imaging and the resorption pits area assessed under an inverted Nikon microscope.
Osteoclast differentiation in vivo
2 × 105 synovial F4/80+CD206+ M2-like macrophages sorted by flow cytometry were transferred into the ankle joint of normal male DBA/1J mice. Prior to intra-articular injection, insulin micro injectors were used to anesthetize mice and expose the joint capsule. A 0.1 cm insert was performed along the patellar ligament, slightly inward to the needle tip, and cells were slowly injected for 1 min.2 21 An equal volume of PBS was injected into the joint as control. Three days later, mice received another injection of sorted synovial F4/80+CD206+ M2-like macrophages or PBS. Mice were sacrificed 12 days afterthe first injection. Bones were decalcified with 10% EDTA for 4 weeks and embedded in paraffin, section 4 mm in thickness were stained with H&E and TRAP.
Scores were as following. For inflammation score 0: No inflammation; score 1: Slight thickening of lining layer or some infiltrating cells in sublining layer; score 2: Slight thickening of lining layer plus some infiltrating cells in sublining layer; score 3: Thickening of lining layer, influx of cells in sublining layer and presence of cells in the synovial space and score 4: Synovium highly infiltrated with many inflammatory cells. For cartilage erosion score 0: No destruction; score 1: Minimal erosion limited to single spots; score 2: Slight to moderate erosion in a limited area; score 3: More extended erosions and score 4: General destruction.
Statistical analysis
All experiments were repeated at least 3 times and the results are expressed as mean±standard error of the mean (SEM). Comparison of the data was performed using the Student’t test using GraphPad Prism 8 software. Significance was defined as a p value of <0.05%.
Results
Phenotype of F4/80+CD206+ M2-like macrophages in CIA mice
We firstly checked the phenotypical status of M2-like macrophages in synovium of CIA mice. Synovial cells isolated from control or CIA mice (35 days after the secondary immunization with PBS or CII) were analyzed by flow cytometry, the gating strategy was shown in supplementary Figure s1(a). The number of CD45+CD11b+Gr-1-F4/80+CD206+ (F4/80+CD206+ for short) cells, normally classified as M2-like macrophages, from inflamed knee joints of mice with CIA increased significantly compared to that of control mice (data not shown). These F4/80+CD206+ M2-like macrophages displayed lower CD206 and higher IA/IE expression in CIA mice v. s. Control mice, suggesting a trend towards a pro-inflammatory phenotype. More importantly, F4/80+CD206+ M2-like macrophages from CIA mice displayed dramatically increased RANK expression (Figure 1(a)). Q-PCR analysis using sorted cells from inflamed knee joints further confirmed the pro-inflammatory change of F4/80+CD206+ M2-like macrophages from CIA mice, becaues they expressed less IL10, TGFB1 and more TNFA, IL1B and IL6 compared with same cell population from control mice (Figure 1(b)). Changes of numbers and phenotype of macrophages in the joints of control and CIA mice. Phenotypic characterization of F4/80+CD206+ or F4/80+CD206- macrophages sorted from synovium of Ctrl (n = 20) or CIA mice (n = 5) were analysed by FACS (a) and Q-PCR (b). Sorted macrophages were stimulated with Pam3, Zymosan or LPS and the culture supernatants were collected for quantification of TNF-α and IL-6 using ELISA kits. IL-1β mRNA expression of treated cells were determined by Q-PCR (c). The results are representative of at least three experiments. *p < .05, **p < .01, ***p < .001.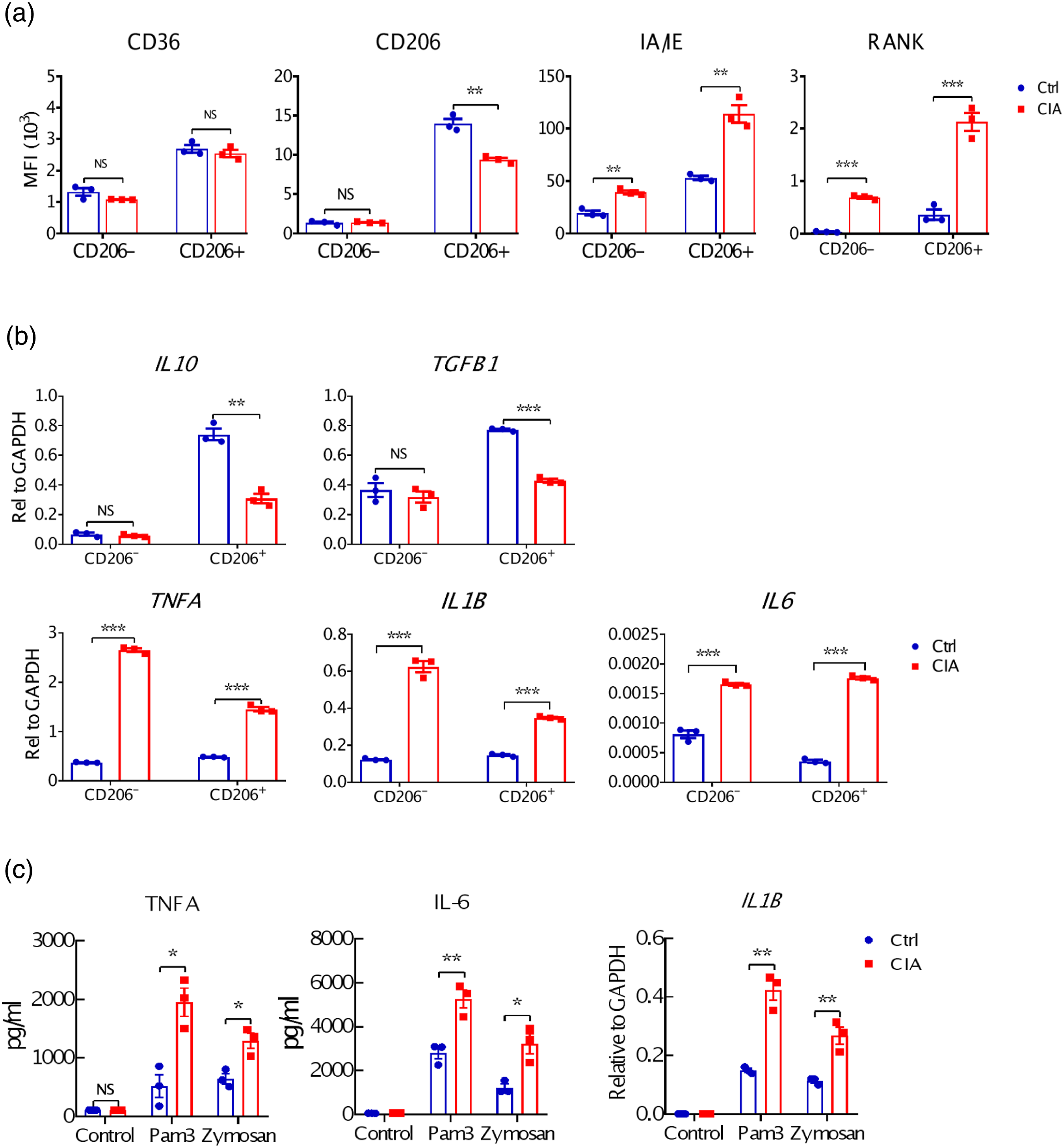
Functional analysis showed that sorted F4/80+CD206+ M2-like macrophages from the synovium of CIA mice were hyperresponsive to Pam3 or Zymosan-induced expression of TNF-α, IL-6 and IL-1β compared to those from control mice (Figure 1(c)). Interestingly, their response to LPS stimulation remained unaffected. All these data indicate that synovial F4/80+CD206+ M2-like macrophages in CIA mice adopted a pro-inflammatory phenotype in terms of both biomarkers and functionality, which might provide a positive feedback signal to promote excessive inflammation in RA. Notably, F4/80+CD206+ M2-like macrophages are much better in producing IL-10, TGF-β upon stimulation even in CIA mice while CD206- are much better in producing TNF and IL-6 upon stimulation, indicating that macrophages in CIA mice showed a chimeric M1/M2 phenotype.
Synovial F4/80+CD206+ M2-like macrophages from CIA mice had high potential to differentiate into osteoclasts
Based upon above finding that F4/80+CD206+ M2-like macrophages from inflamed knee joints of CIA mice expressed significantly higher levels of RANK, we next explored if these cells could differentiate into osteoclasts with RANKL & M-CSF cocktail stimulation. Firstly F4/80+CD206+ M2-like macrophages from CIA mice did show robust TRAP positive staining after differentiation, while M2-like macrophages from control mice, and M1-like macrophages from either mice remained TRAP negative (Figure 2(a)). Additional blocking experiment with OPG also confirmed the dependency on RANKL-RANK signaling of this differentiation (Figure 2(d)). Subsequent Q-PCR analysis further confirmed their expression of additional osteoclast-specific markers including NFATc1, MMP9, CTSK and CTR (Figure 2(b)). F4/80+ CD206+ macrophages in the inflamed synovium have high osteoclast differentiation potential. Sorted macrophages were directly stimulated with RANKL and M-CSF, followed by TRAP staining (a). Sorted macrophages stimulated with RANKL and M-CSF was used as template in Q-PCR amplification of mRNA for osteoclast markers (b). M-CSF and RANKL treated macrophages were stained with TRITC-phalloidin (red) for confocal microscopy. Microimages of the same fields observed through bright (Left panels), red channels (Middle panels) are presented. For assessment of bone resorption activity, the treated cells were incubated for 18 days in calcium phosphate-coated wells, followed by microscopic examination of the resorption pits (Right panels) (c). TRAP staining images of F4/80+CD206+ M2 macrophages cultured with M-CSF, RANKL with or without OPG at the indicated concentrations (Left panels). Box/whisker plots show the results of cellular numeration of TRAP+ MNCs with >3 nuclei (Right panels) (d). The results are representative of at least three experiments. *p < .05, **p < .01, ***p < .001.
The expression of a band of F-actin containing podosomes in osteoclasts has been shown to be involved in bone resorption, 22 we next examined this biomarker in above F4/80+CD206+ M2-like macrophage-differentiated cells. The results clearly showed that bands of F-actin containing podosomes were significantly induced in differentiated cells from F4/80+CD206+ M2-like macrophages sorted from CIA, but not control mice. Bone resorption assay also revealed that F4/80+CD206+ M2-like macrophages from CIA mice had acquire the capability of resorbing bone after differentiation (Figure 2(c)). Overall, both biomarker and functional data suggest that F4/80+CD206+ M2-like macrophages from CIA mice are readily to differentiate into osteoclast via RANKL-RANK pathway.
It has been reported that BM-derived CX3CR1hi Ly6Cint F4/80hiI-A+/I-E+ macrophages in inflamed synovium could differentiate into osteoclasts in pannus, 4 while our data showed that synovial F4/80+CD206+ M2-like macrophages of CIA mice have high osteoclast differentiation potential. By cross-examining these two cell populations through multicolor immunofluorescence staining and FACS analysis, we found that nearly 100% synovial F4/80+CD206+ M2-like macrophages expressed both LY6C and CX3CR1, while only very low proportion (0.53% in control mice and 7.24% in CIA mice) of CD45+CX3CR1hiLy6Cint macrophages are F4/80 and CD206 double-positive (supplementary Figure s2). These data suggest that synovial F4/80+CD206+ M2-like macrophages probably represent a main subpopulation of CX3CR1hi Ly6Cint F4/80hiI-A+/I-E+ macrophages (reported by Hasegawa etc in 2019) with high osteoclast differentiation potential. 4
Synovial F4/80+CD206+ M2-like macrophages from CIA mice differentiated into osteoclasts and caused bone resorption in vivo
In the in vivo experiment shown in Figure 3, we transferred synovial F4/80+CD206- M1-like or F4/80+CD206+ M2-like macrophages from CIA mice, or an equal volume of PBS as control, into the ankle joint of normal adult male DBA/1J mice that were sacrificed on day 12 for pathological analysis. Bone sections from mice that received F4/80+CD206+ M2-like macrophages displayed clear osteoclast formation in the joint as shown by TRAP staining (Figure 3(a)). Significant synovial inflammation and bone destruction in these animals were evident by H&E staining, while no obvious bone destruction was observed in mice injected with either M1-like macrophages or PBS alone (Figures 3(b) and (c)). These results confirmed that F4/80+CD206+ M2-like macrophages from CIA mice had the capability of differentiating into osteoclasts and causing bone resorption in vivo. Osteoclasts derived from F4/80+CD206+ macrophages isolated from mice with CIA are capable of bone resorption in vivo. Isolated Macrophages from mice with CIA (n = 5) were injected into the ankle joint of normal DBA/1J mice (n = 3, 5000 cells per mouse) on day 0 and day 3. Mice were sacrificed 12 days later. Joint sections were stained by TRAP (a) or H&E (b). The inflammation and cartilage erosion score in each mouse group was determined as described in Materials and Methods (c). The results are representative of at least three experiments. *p < .05, **p < .01.
Bone marrow F4/80+CD206+ M2-like macrophages from CIA mice showed high potential to differentiate into osteoclasts
It is of interest to note that, similar to synovial cells, bone marrow F4/80+CD206+ M2-like macrophages from CIA mice also expressed high level of RANK and increased IA/IE (Figure 4(a)). Their increased CD206 expression was opposite to changes seen in synovial cells. When differentiated with RANKL plus M-CSF, F4/80+CD206+ M2-like macrophages from bone marrow of CIA mice formed significantly more TRAP+ cells compared with M2-like macrophages from control mice, and M1-like macrophages from either mice (Figure 4(b)). Besides positive staining of TRAP, Q-PCR analysis confirmed their expression of additional osteoclast-specific marker NFATc1, MMP9, CTSK and CTR (Figure 4(c)), and F-actin staining with bone resorption assay also confirmed their bone resorption function (Figure 4(d)). Together these data indicated that bone marrow F4/80+CD206+ M2-like macrophages from CIA mice could also differentiated into osteoclasts. F4/80+CD206+ macrophages in bone marrow of CIA mice increased and differentiate into osteoclasts. Phenotypic characterization of cells from bone marrow of Ctrl (n = 3) or CIA mice (n = 3) were analysed by FACS staining (a). Sorted macrophages from the bone marrow of mice with CIA (n=5) or normal mice (n = 20) were cultured with M-CSF and RANKL, followed by TRAP staining. * p < .05 (b). Sorted macrophages stimulated with RANKL and M-CSF was used as template in Q-PCR amplification of mRNA for osteoclast markers (c). The RANKL and M-CSF treated F4/80+CD206+ macrophages sorted from bone marrow were stained with TRITC-phalloidin (red) (Left panels). For assessment of bone resorption activity, the treated cells were incubated for 18 days in calcium phosphate-coated wells, followed by microscopic examination of the resorption pits (Right panels) (d). The results are representative of at least three experiments. *p < .05, **p < .01, ***p < .001.
Osteoclastgenesis capability of in vitro differentiated M2 macrophage from bone marrow if CIA mice
The above results indicate that the existing M2-like cells in synovium or bone marrow of CIA mice had the potential to further differentiate into osteoclasts. We considered whether undifferentiated bone marrow cells from CIA mice, after cytokine-driven in vitro differentiation, would give rise to M2-like cells that were more likely to become osteoclasts upon RANKL signaling. Mouse M2 and M1 macrophages were differentiated from bone marrow cells of CIA and healthy control mice with M-CSF & IL-4 and M-CSF & IFN-γ, respectively, and quantified for specific marker expression (supplementary Figure s3(a) and (b)). Similar as synovial F4/80+CD206+ M2-like macrophages, in vitro differentiated M2 macrophages from CIA mice also expressed a high level of RANK (Figure 5(a)). Following treatment with RANKL and M-CSF, these cells obtained a strong TRAP staining signal (Figure 5(b)), together with other osteoclast markers including NFATc1, MMP9, CTSK and CTR (Figure 5(c)). F-actin staining with bone resorption assay also confirmed their bone resorption function (Figure 5(d)). Of note that in vitro differentiated M2 macrophages from control mice also gained some osteoclast markers, but to a lesser extent compared with that from CIA mice. Collectively these data suggested that in vitro differentiated M2 macrophages from bone marrow cells of CIA mice also have high osteoclastogenesis potential. Macrophages derived from bone marrow showed different differentiation potential into osteoclasts. Freshly differentiated M1, M2 macrophages derived from bone marrow cells of Ctrl or CIA mice were subjected to FACS analysis (a). Differentiated M1 or M2 macrophages derived from bone marrow cells of Ctrl or CIA mice were stimulated with M-CSF and RANKL, followed by TRAP staining (b). Macrophages stimulated with M-CSF and RANKL was used as template in Q-PCR amplification of mRNA for osteoclast markers (c). The treated M2 macrophages stained with TRITC-phalloidin (red) staining (Left panels). The treated M2 macrophages were incubated for 18 days in calcium phosphate-coated wells, followed by microscopic examination of the resorption pits (Right panels) (d). The results are representative of at least three experiments. *p < .05, **p < .01, ***p < .001, ****p < .0001.
In vitro differentiated M2 macrophage from RA patients had high osteoclastogenic potential
Since there are also abundant F4/80+CD206+ M2-like macrophages in the synovium of RA patients,18,23 it is of great interest to evaluate if human M2 macrophages could also function as osteoclast precursors. Human M1 and M2 macrophages, differentiated from PBMCs of RA patients or healthy volunteers using M-CSF & IFN-γ or M-CSF & IL-4, respectively, were assessed for ability differentiating into osteoclasts. As shown in Figure 6(a), in vitro differentiated M2 macrophages from RA patients also expressed higher level of RANK (Figure 6(a)). Application of M-CSF and RANKL enabled these M2 cells to acquire TRAP staining signal (Figure 6(b)) and bone absorption activity (Figure 6(c)), suggesting their successful differentiation into osteoclasts. Overall these data reveal that the osteoclast differentiation capability of M2 macrophages is conserved cross-species and could be translated into RA patients, which may play important role in disease pathogenesis. Monocytes-derived M2 macrophages from RA patients showed high osteoclastogenesis potential. M1 or M2 macrophages differentiated from monocytes of healthy donors (Ctrl, n = 3) or RA patients (RA, n = 3) were stained with RANK specific antibodies for flow cytometric analysis (a). TRAP staining images of differentiated M1 or M2 macrophages treated with M-CSF and RANKL for 6 days (b). *p < .05, **p < .01, ***p < .001. The treated macrophages stained with TRITC-phalloidin (red) (Left panels). The treated macrophages were further incubated for 18 days in calcium phosphate-coated wells to assess bone resorption activity (Right panels) (c). The results are representative of at least three experiments using cells from different donors.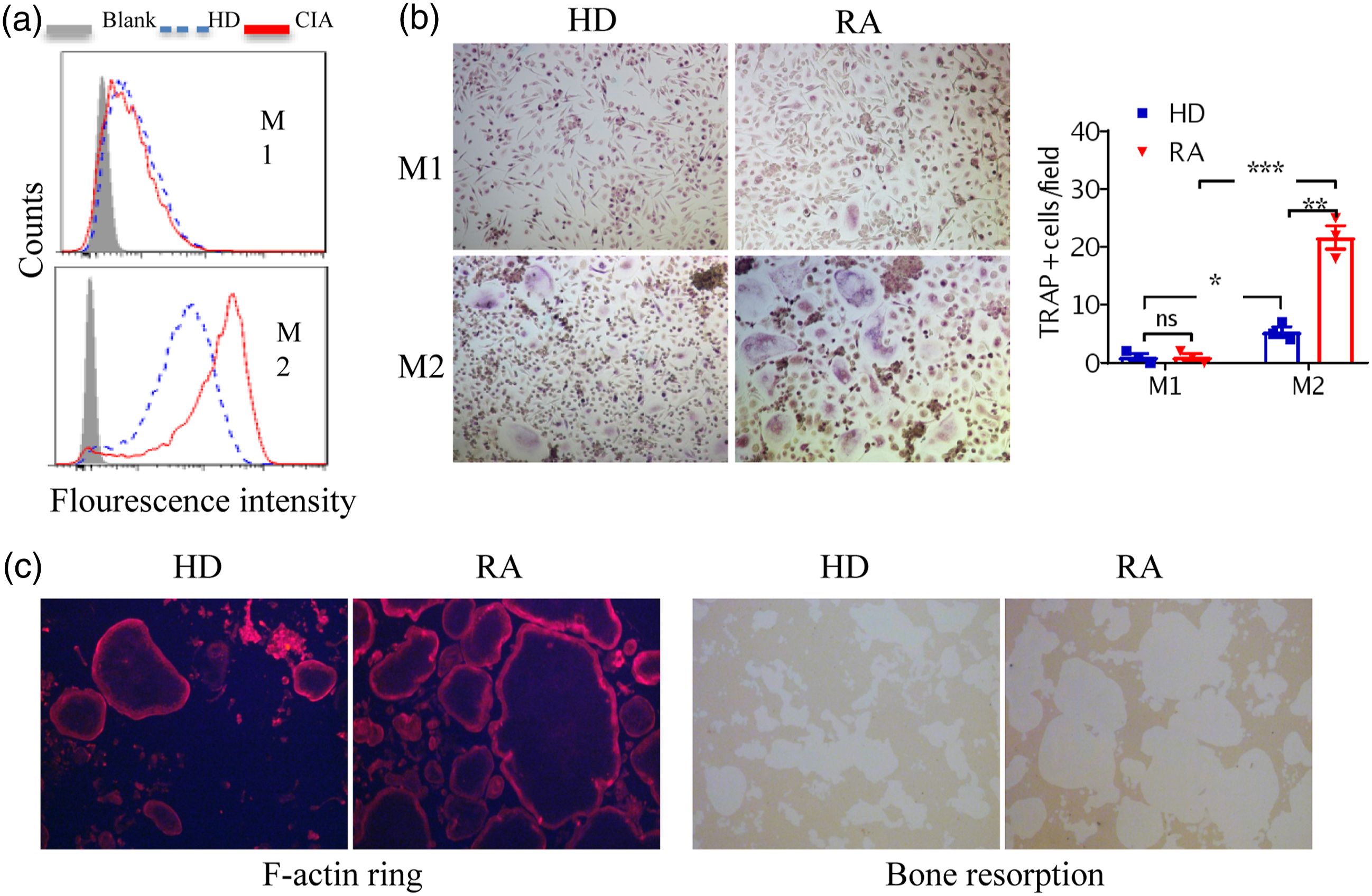
Discussion
It has long been accepted that in synovial inflammation, the production of pro-inflammatory and destructive mediators from activated M1 macrophage is of importance for RA symptoms and progression, while M2 macrophages mediate anti-inflammatory effects by producing anti-inflammatory cytokines such as IL-10 and TGF-β.24,25 However, recent studies suggest that there was no clear distinction between the proinflammatory M1 and the anti-inflammatory M2 phenotypes in RA patients15,16,26 or the arthritis animal model.3,4,17,23 Quero et al. showed that TLR2 engagement impaired the anti-inflammatory activity of M2-like macrophages derived from healthy or RA subjects, generating a chimeric M1/M2 phenotype. 26 Misharin et al. reported that both MHC II+ and MHC II− macrophages exhibited mixed M1/M2 polarization during K/BxN serum-transfer-induced arthritis, a murine model of sterile inflammatory arthritis. 23 It has also been reported that up to 50% of infiltrated mononuclear cells were CD163+ M2 macrophages in CIA synovium, and they could release pro-inflammatory cytokine TNF-α and exhibit pro-inflammatory potentials.17,18 Fc gamma receptor-TLR cross-talk elicited pro-inflammatory cytokine production by human M2 macrophages and switched human macrophages from M2 into M1-like phenotype, thereby promoting excessive inflammation in autoimmune diseases such as RA.15,16 In this study, we have demonstrated that synovial F4/80+CD206+ M2-like macrophages display a pro-inflammatory phenotype characterized by higher levels of TNFA, IL1B, and IL6, and lower levels of CD163, CD206, IL10 and TGFB1 (Figures 1(a) and (b)). Furthermore, synovial F4/80+CD206+ M2-like macrophages became functionally hyperactive to subsequent stimuli such as Pam3 and Zymosan (Figure 1(c)), but not LPS (data not shown), which was consistent with previous findings. 26
Our data demonstrate for the first time that, compared with that from normal mice and CD206- M1-like macrophages, the expression of RANK in synovial or bone marrow-derived or differentiated F4/80+CD206+ M2-like macrophages from CIA mice or M2 macrophages from RA patients is significantly elevated (Figure 1(a) and 4(a), Figure 5(a) and 6(a)). We therefore hypothesized that these M2-like macrophages might be able to differentiate into osteoclasts and contributed to RA pathogenesis. Indeed co-culture of such M2-like macrophages with M-CSF and RANKL induced expression of osteoclast markers and acquired osteoclast bone resorption function in vitro. This phenomenon was then validated using in vivo transfer model. It is therefore reasonable to suggest that F4/80+CD206+ M2-like macrophages may be involved in bone destruction and absorption in RA by differentiating into osteoclasts via RANKL-RANK signaling pathway. Our results provide additional clues for the pathological roles of M2 macrophages during RA process.
Earlier reports showed that the Ly6Chi monocytic population in the bone marrow contained osteoclast precursors (BM-OCPs) in arthritic mice.27,28 The cell population identified in our study were of synovial F4/80+CD206+ M2-like macrophages, which were distinct from above BM-OCPs, because our cells highly expressed CD11b, while their cells did not.27,28 Hasegawa et al. demonstrated that bone marrow-derived CX3CR1hi Ly6Cint F4/80hiI-A+/I-E+ macrophages displayed high osteoclast differentiation potential. 4 Notably, FACS analysis results indicated that synovial F4/80+CD206+ M2-like macrophages in our study were the main subpopulation of CX3CR1hi Ly6Cint F4/80hiI-A+/I-E+ macrophages with high osteoclast differentiation potential, and thus provided a novel precise marker for identifying an osteoclast precursor-containing population in the inflamed synovium.
Additionally, synovial F4/80+CD206+ M2-like macrophages in this study highly expressed IA/IE, and we also found similar osteoclastogenesis potential on the bone marrow derived M2 macrophages from CIA mice. As indicated in previous study that under steady-state conditions, major MHC II− macrophages represented true tissue-resident macrophages, whereas most of the MHC II+ synovial macrophages originated from bone marrow. 23 These evidences collectively suggested that these synovial F4/80+CD206+ M2-like macrophages with osteoclast differentiation capability might originate from bone marrow, which still requires further investigations.
Another interesting finding in this study was that bone marrow derived M2 macrophages from CIA mice showed higher potential to differentiate into osteoclasts in vitro compared to control mice, suggesting a “diseased” status of M2 macrophages in CIA mice. A similar phenomenon has also been reported previously. BCG 29 and β-glucan30,31 could educate haematopoietic stem and progenitor cells (HSPCs) to differentiate toward trained macrophages, which was capable of eliciting pro-inflammatory cytokine with re-stimulation and generating protective innate immunity against Tuberculosis and Candida Albicans. It has also been reported that aberrancies of immune cells in SLE32,33 and RA34,35 could be traced back to the bone marrow HSPCs, which were reprogrammed to exhibit a signature of “trained immunity”, and macrophages derived from these HSPCs also exhibited a trained phenotype with bulk production of pro-inflammatory cytokine, contributing to inflammation and the risk of flare in these autoimmune diseases. In this study we demonstrated for the first time that in vitro differentiated M2 macrophages from CIA mouse bone marrow had a higher osteoclastogenesis potential, which probably originated from the reprogrammed HSPCs in CIA mice.
Recent studies36,37 have suggested that RA is a multifactorial disease, a series of synovial cells and cytokines, like IL18, and β-nerve growth factor are involved. Our data provide evidence of M2 polarization associated cytokines such as IL-4 and IL-10 probably contribute to pathological process of RA by differentiating into functional osteoclasts.
One of the limitations of this study is that we don’t do accurate sample size calculation. We collected data only n=3 from CIA mice or RA patients limited by objective conditions in this study, but all experiments were repeated at least 3 times.
Conclusion
Our results demonstrate that synovial F4/80+CD206+ M2-like macrophages increased significantly and showed pro-inflammatory effects in CIA mice. With high level RANK expression, these cells readily differentiate into functional osteoclasts in vitro and in vivo. Similar osteoclastogenesis effect have also been validated on bone marrow F4/80+CD206+ M2-like macrophages, in vitro differentiated M2 macrophages from CIA bone marrow and RA patient PBMCs. In conclusion, F4/80+CD206+ M2-like macrophages are osteoclast precursors in synovium and contribute to bone destruction in RA pathogenesis.
Supplemental Material
Supplemental Material - F4/80+CD206+ M2-like macrophages contribute to bone erosion in collagen-induced arthritis by differentiating into osteoclasts
Supplemental Material for F4/80+CD206+ M2-like macrophages contribute to bone erosion in collagen-induced arthritis by differentiating into osteoclasts by HeHe Sun, HongMin Wang, ChenHui Gao, Li Tai, YueYao Yang, HongLiang Dong and XiaoMing Gao in European Journal of Inflammation
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the by grants from Ministry of Science and Technology (2017YFA0104502), National Foundation of Natural Science (31770942), and Priority Academic Program Development of Jiangsu Higher Education Institution (PARD) of China.
Ethics approval
All procedures involving animals were in compliance with the National Institutes of Health guide for the care and use of Laboratory animals, and ethical approval was granted by the Ethical Committee of Soochow University (Ethical approval number: SYSK-(S2012-0062). All experiments were performed in accordance with relevant guidelines and regulations.
Animal welfare
The present study followed international, national, and institutional guidelines for humane animal treatment and complied with relevant legislation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
