Abstract
Objectives
We investigated the effect and mechanism of miR-21-3p on the biological behavior of keratinocyte HaCaT cells.
Methods
HaCaT cells were transfected with miR-21-3p mimics and miR-21-3p inhibitor, respectively. Cell proliferation was detected by CCK-8, and apoptosis and cell cycle were analyzed by flow cytometry. The expression changes of Bax and Bcl-2 were measured with immunohistochemistry. The mRNA expressions of IL-17A and STAT3 were detected by real-time fluorescence quantitative PCR. Cytokeratin 17 protein was detected by Western blot. The relationship between miR-21-3p and STAT3 was verified by dual luciferase reporter gene assay.
Results
The miR-21-3p mimics significantly promoted the proliferation and cell cycle progression of HaCaT cells. However, miR-21-3p mimics significantly inhibited cell apoptosis, decreased Bax expression and increased Bcl-2 expression. Additionally, miR-21-3p mimics significantly increased the expressions of Cytokeratin 17 protein, IL-17A mRNA and STAT3 mRNA. Of note, the miR-21-3p inhibitor exhibited contrary effects to miR-21-3p mimics. Furthermore, STAT3 was a direct target of miR-21-3p.
Conclusion
miR-21-3p may promote the abnormal proliferation and inhibit apoptosis of HaCaT cells, possibly through regulating JAK/STAT pathway and the expression of Cytokeratin 17.
Keywords
Introduction
Psoriasis is one of the most common chronic inflammatory skin diseases with strong genetic susceptibility and autoimmune features. 1 Epidemiologically, the global prevalence of psoriasis is approximately 2%, but it varies by region. 2 The dermatological manifestations of psoriasis are varied. Psoriasis vulgaris, also known as plaque psoriasis, accounts for about 90% of psoriasis cases and is the most common type. 3 Its main characteristic is persistent inflammation, leading to uncontrolled proliferation and dysfunctional differentiation of keratinocytes. 4 Dysfunction of the innate and adaptive cutaneous immune responses is the reason for the development and persistence of inflammation in psoriasis vulgaris. 5 The pathogenesis of psoriasis vulgaris can be divided into an initial phase, which may be triggered by trauma, infection, or drugs, and a maintenance phase characterized by chronic clinical progression. 6 However, the pathogenesis of psoriasis vulgaris has not yet been fully elucidated.
Janus kinase (JAK) and signal transducer and activator of transcription-3 (STAT3) is related to the pathogenesis of inflammatory diseases. 7 In the pathogenesis of psoriasis, the JAK/STAT pathway is mainly related to the differentiation of T cells and the excessive proliferation of keratinocytes. Phosphorylation of STAT3 can induce abnormal proliferation of keratinocytes, and promote transcription of inflammatory factors. 8 Furthermore, the activation of STAT3 also leads to the high expression of Cytokeratin 17, which is confirmed to play an important role in the pathogenesis of psoriasis. 9
MicroRNA is a class of small non-coding RNA molecules. It can affect physiological and pathological processes such as proliferation, apoptosis and differentiation of cells by regulating the expression of targeted genes. So far, more than 250 miRNAs have been identified to be abnormally expressed in skin tissues of psoriasis vulgaris.10,11 As a promoter of the inflammatory cascade and a suppressor of apoptosis, 12 microRNA-21 (miR-21) has been found to be abnormally highly expressed in skin lesions of psoriasis vulgaris, and could regulate and participate in the occurrence of psoriasis vulgaris. 13 It has also been shown that inhibition of miR-21 could improve psoriatic lesions in experimental animals. 14 Additionally, the miR-21 promoter region has a STAT3 binding site, and miR-21 can directly regulate STAT3. 15 miR-21 can be cleaved into a guide strand miR-21-5p and a passenger strand miR-21-3p. miR-21-5p has been confirmed by many studies to promote the occurrence and development of psoriasis vulgaris. 16 The role of miR-21-3p in psoriasis is emerging 17
Herein, we investigated the effect of miR-21-3p on the proliferation, cell cycle and apoptosis of HaCaT cells and the underlying mechanism involving JAK/STAT3 pathway. Our findings may provide new targets for the diagnosis and treatment of psoriasis.
Materials and methods
Ethics approval
The study did not include any human or animal experimentation for which ethical approval was required.
Cell culture and transfection
HaCaT cells were purchased from Procell (Wuhan, China) and cultured in DMEM containing 15% FBS and 1% Penicillin- Streptomycin. Cell transfection was performed with LipofectamineTM 2000 (Invitrogen Co, Carlsbad, CA, USA). The miR-21-3p mimics and miR-21-3p inhibitor were transfected into HaCaT cells for 24 h. For control, HaCaT cells were also transfected with miR-21-3p mimics NC (negative control) and miR-21-3p inhibitor NC. The miR-21-3p mimics, inhibitor, and NC were from Wuhan Bafel Biotechnology Service Co, Ltd (China). Cells without transfection served as blank control. The transfection efficacy was verified by real-time quantitative PCR.
CCK-8 assay
Cell proliferation was detected with Cell Counting Kit-8 (MedChemExpress, Princeton, NJ, USA). In detail, HaCaT cells were seeded in 96-well plates and cell transfection was performed as above described. At 24 h after transfection, CCK8 solution was added and incubated at 37°C for 4 h. The optical density at 450 nm (OD450) was determined by a microplate reader.
Flow cytometry analysis of cell cycle
At 24 h after transfection, cells were collected and fixed with 70% pre-cooled ethanol at 4°C for more than 4 h. Then, cells were rinsed twice with pre-cooled PBS and incubated with RNase at 37°C for 30 min. Finally, cells were incubated with PI at 4°C in the dark for 30 min. Cell cycle was detected on CytoFLEX (Beckman Coulter Inc., Brea, CA, USA).
Flow cytometry analysis of cell apoptosis
Cell apoptosis was analyzed with APC/7-AAD apoptosis kit (Tianjin Sungene Biotech Co, Ltd, China). In detail, at 24 h after transfection, cells were collected and rinsed. The 7-AAD staining solution was added to the collected cells and incubated at room temperature in the dark for 5–15 min. Then, the Binding Buffer was added followed by incubation with Annexin V-APC at room temperature in the dark for 10 min. Cell apoptosis was analyzed on CytoFLEX (Beckman Coulter Inc.).
Immunohistochemistry
The cells on the coverslips were fixed with 4% paraformaldehyde for 15 min. After permeabilization with 0.5% Triton X-100 at room temperature for 20 min, 3% hydrogen peroxide was added and incubated at room temperature for 15 min. Then, the diluted goat serum was added for blocking at room temperature for 30 min. After blocking, primary antibodies against Bcl-2 and Bax (1:100; Abcam, Cambridge, MA, USA) were added and incubated overnight at 4°C. After washing, incubation with secondary antibody of goat anti-rabbit IgG H&L (HRP) (Abcam) at room temperature for 30 min was performed. DAB was used for color development. Finally, the cells were observed under a microscope after counterstaining with hematoxylin and mounting.
Real-time quantitative PCR
Primer sequences for real-time quantitative PCR.

Western blot
Proteins were extracted from cells with lysis buffer containing PMSF and phosphatase inhibitor. Protein concentrations were determined using BCA (Solarbio, Beijing, China). After SDS-PAGE separation, the proteins were transferred to PVDF membrane. The membrane was blocked with 5% BSA blocking solution at room temperature for 1 h. After that, incubation with rabbit anti-Cytokeratin 17 polyclonal antibody (Proteintech Group, Inc, Wuhan, China) was performed at 4°C overnight. Then, the membrane was incubated with HRP-conjugated goat anti-rabbit secondary antibody (Abcam; 1:50 000) for 2 h. Finally, color development was performed. The gray value of the protein bands was analyzed by GlycoBandScan 5.0 (ProZyme, Hayward, CA, USA) and the intensity ratio of the target protein to GAPDH was calculated.
Dual luciferase reporter gene assay
The wild type (control) and mutant 3′-UTR region of STAT3 gene were cloned into pYr-MirTarget vector. The constructed plasmids and miR-21-3p mimics (100 nM) were co-transfected into 293T cells. After incubation for 48 h, cells were lysed and RLU was determined using firefly luciferase as an internal reference.
Statistical analysis
SPSS 23.0 software was used for data processing. All data were expressed as mean ± standard deviation. One-way analysis of variance was used for data comparison and LDS method was used for pairwise comparison. The difference was considered to have significant statistical significance at p < 0.05.
Results
Validation of transfection effect by real-time quantitative PCR
The transfection effect was detected by real-time quantitative PCR. The results showed that miR-21-3p mimics significantly increased the expression of miR-21-3p (Figure 1) (p < 0.05). However, this increase was inhibited by miR-21-3p inhibitor. The data showed that the transfection effect was good. Real-time PCR verification of the transfection efficacy of miR-21–3p. Compared with control, *p < 0.05; Compared with mimics, #p < 0.05.
Effects of miR-21-3p on the proliferation and cell cycle of HaCat cells
CCK-8 assay and flow cytometry analysis evaluated the effects of miR-21-3p on cellular events of HaCaT cells. The results showed that compared with control groups, miR-21-3p mimics promoted the proliferative activity of HaCaT cells (Figure 2(a)), but inhibited the G1 phase of the cell cycle (Figure 2(b)). On the contrary, compared with control group, miR-21-3p inhibitor inhibited the proliferative activity of HaCaT cells (Figure 2(a)) and interfered with the cell cycle (Figure 2(b)). These data suggest that miR-21-3p may regulate the growth of HaCaT. Effect of miR-21–3p on cell proliferation and cell cycle of HaCaT cells. (a) CCK-8 assay analyzed cell proliferation. (b) Flow cytometry analysis of cell cycle. Compared with control, *p < 0.05; Compared with mimics, #p < 0.05.
Effects of miR-21-3p on the apoptosis of HaCat cells
We further assess apoptosis by flow cytometry analysis. As shown in Figure 3(a), miR-21-3p mimics induced lower level of apoptosis compared with control group. However, miR-21-3p inhibitor significantly increased apoptosis level. Additionally, the expression of apoptosis proteins in transfected HaCaT cells was detected by immunohistochemistry. As shown in Figure 3(b), compared with control, miR-21-3p mimics inhibited the expression of the pro-apoptosis protein Bax but promoted the expression of the anti-apoptosis protein Bcl-2. However, compared with control, miR-21-3p inhibitor promoted Bax while inhibited Bcl-2. This further verified the inhibitory effect of miR-21-3p on apoptosis of HaCaT cells. Effect of miR-21–3p on apoptosis of HaCaT cells. (a) Flow cytometry analysis of apoptosis. (b) Immunohistochemistry was used to detect Bax and Bcl-2 expression. Compared with control, *p < 0.05.
miR-21-3p regulates the mRNA expression of IL-17A and STAT3
The mRNA expressions of IL-17A and STAT3 were detected by real-time quantitative PCR. The data showed that HaCaT cells transfected with miR-21-3p mimics had significantly increased mRNA expressions of IL-17A and STAT3 (p < 0.05), while HaCaT cells transfected with miR-21-3p inhibitor had significantly reduced mRNA expressions of IL-17A and STAT3 (Figure 4). This data suggests that miR-21-3p may play a role by regulating the expression of IL-17A and STAT3. miR-21–3p regulates the mRNA expression of STAT3 and IL-17A. Real-time PCR measured the mRNA expression of STAT3 and IL-17A. Compared with control, *p < 0.05; Compared with mimics, #p < 0.05.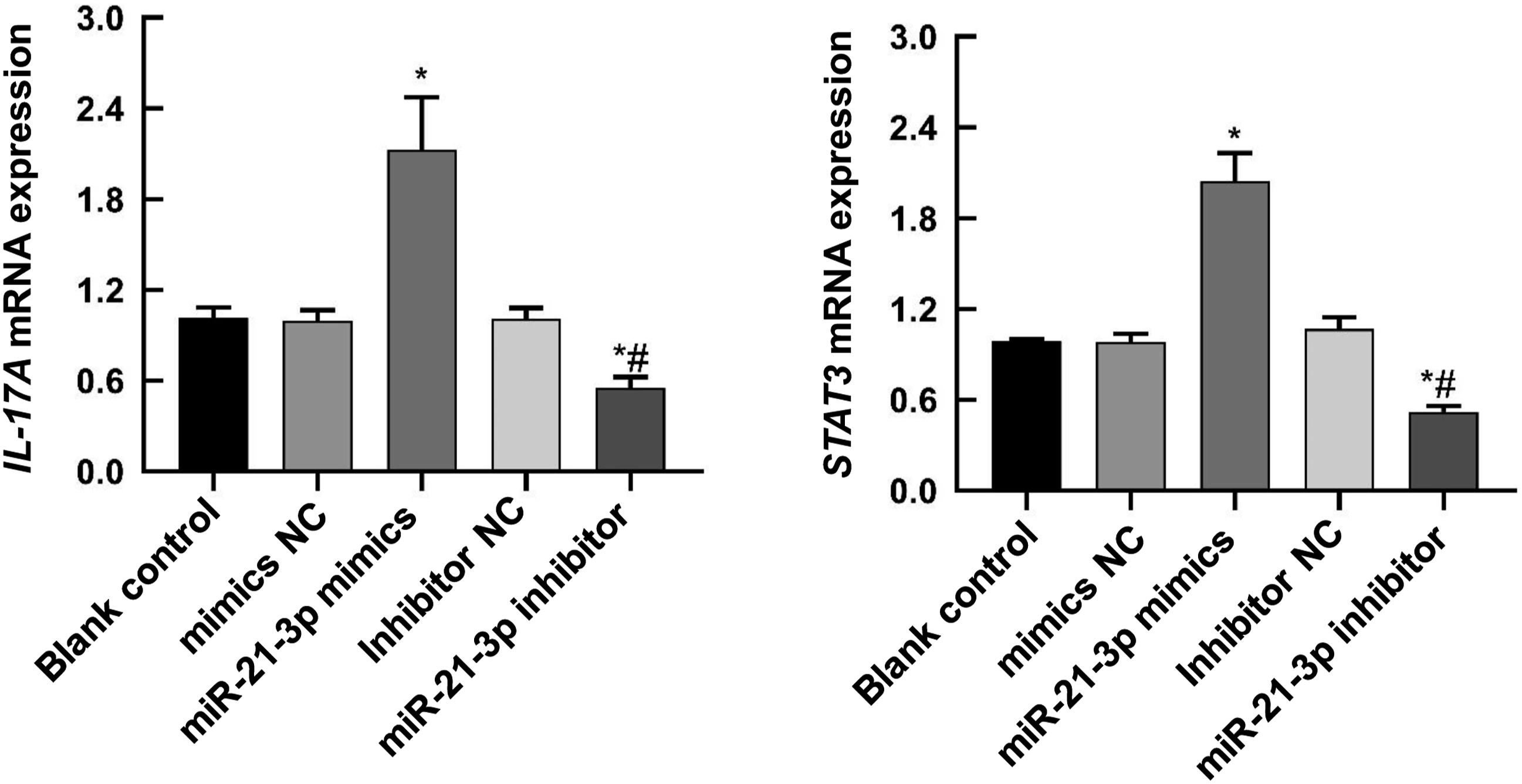
miR-21-3p regulates the protein expression of Cytokeratin 17
Western blot showed that compared with control, HaCaT cells transfected with miR-21-3p mimics had significantly increased protein expression of Cytokeratin 17 (p < 0.05) (Figure 5). Compared with control, miR-21-3p inhibitor significantly inhibited the protein expression of Cytokeratin 17 (p < 0.05) (Figure 5). This data suggests that miR-21-3p may play a role by regulating Cytokeratin 17. miR-21–3p regulates the protein expression of Cytokeratin 17. Western blot detected the protein expression of Cytokeratin 17. Compared with control, *p < 0.05; Compared with mimics, #p < 0.05.
STAT3 is a direct target of miR-21-3p
To verify the relationship between miR-21-3p and STAT3, dual-luciferase reporter gene assay was conducted. The results showed that the co-transfection of miR-21-3p-mimics + WT plasmid led to a decrease in the ratio of fluorescence intensity (p < 0.05) (Figure 6). There was no significant difference among other groups p > 0.05. This result indicates that STAT3 is a direct target of miR-21-3p. Results of dual-luciferase reporter gene assay. Compared with control, *p < 0.05.
Discussion
The pathogenesis of psoriasis is still unclear, but it is closely related to the abnormal proliferation, differentiation and apoptosis of keratinocytes. 18 The abnormal regulation of miRNA in psoriasis plays an important role in its occurrence and development. 19 miR-21-3p has been reported to be involved in traumatic brain injury. 20 Additionally, the miR-21–3p is shown to be a major driver for the pathogenesis of psoriasis. 17 However, its role in psoriasis is emerging. In this study, we found that miR-21-3p mimics promoted the proliferation and inhibited apoptosis of HaCaT cells, while miR-21-3p inhibitor showed the opposite effect. These results suggest that miR-21-3p may play a promoting role in psoriasis, consistent with previous study. 17 The role of miR-21-3p in psoriasis is consistent with the role of miR-21-5p in psoriasis.21,22 Moreover, miR-21-3p mimics could promote DNA replication and promote the G1 phase of the cell cycle, indicating that miR-21-3p may mediate abnormal changes in HaCaT cell proliferation by regulating the G1 phase of the cell cycle. Studies have confirmed that the abnormal expression of Bcl-2 is one of the main reasons for the development of psoriasis,23,24 and the apoptosis sensitivity of cells depends on the mutual antagonism between the anti-apoptotic and pro-apoptotic members of the Bcl-2 family, the imbalance of which can directly lead to the disruption of cell cycle homeostasis.25,26 The results of this study demonstrated that miR-21-3p mimics decreased Bax but increased Bcl-2, which further confirmed that miR-21-3p could inhibit apoptosis of HaCaT cells.
Psoriasis is initially characterized by hyperproliferation and abnormal differentiation of keratinocytes and later is associated with T cell dysfunction. MiRNAs have the potential to regulate keratinocyte proliferative activity, apoptosis, differentiation, cytokine production, and activation of T cell populations.27–29 Our results showed that miR-21-3p mimics promoted the mRNA expression of IL-17A, while miR-21-3p inhibitor inhibited the mRNA expression of IL-17A. This indicates that miR-21-3p may up-regulate IL-17A level, which in turn exacerbates the inflammatory response of HaCaT cells.
The JAK/STAT signaling pathway is regulated by multiple factors and plays a key role in inflammatory diseases and autoimmune diseases. Activation of STAT3 can promote the infiltration of inflammatory cells and abnormal cell proliferation. 30 It is shown 31 that STAT3 is abnormally highly expressed in the skin lesions of psoriasis patients. Inhibition of STAT3 expression can reduce abnormal cell proliferation. 32 Thus, STAT3 is involved in the proliferation, differentiation and inflammatory response of keratinocytes. Inflammatory factors can induce the expression of Cytokeratin 17 through the JAK/STAT signaling pathway,30,33 and Cytokeratin 17 can also act on T cells to promote the release of inflammatory factors such as IL-17.34,35 Cytokeratin 17 can induce inflammation and thus the occurrence and development of psoriasis. 36 The results of the present study showed that miR-21-3p mimics promoted the expression of Cytokeratin 17 and STAT3. Furthermore, STAT3 was found to be a direct target of miR-21-3p. These results indicate that miR-21-3p may activate the JAK/STAT signaling pathway and induce Cytokeratin 17 expression, thus promoting the abnormal proliferation of HaCaT cells.
This study has some limitations. For example, our results were obtained from cell experiments. Results from clinical samples or animal models are lacking. Further studies are warranted.
Conclusions
Therefore, miR-21-3p promoted the abnormal proliferation of HaCaT cells and inhibited apoptosis possibly by activating the JAK/STAT signaling pathway and inducing the expression of Cytokeratin 17. These effects are consistent to those of miR-21-5p, suggesting a possible role of miR-21-3p in psoriasis. Our findings may provide evidence for further understanding the pathogenesis of psoriasis, thereby promoting the development of therapeutic strategies for psoriasis.
Footnotes
Author contribution
Huiqin Wang and Weidong Wu designed the study. Huiqin Wang, Weidong Wu, Yuan Ding, Yang Zhao, and Xinmei Liu performed the experiments and collected the data. Huiqin Wang, Weidong Wu, and Yanjun Zhang analyzed the data. Huiqin Wang and Yanjun Zhang interpreted the data. Huiqin Wang drafted the manuscript. All authors revised the manuscript and approved the final version of the revised manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article is supported by the Natural Science Foundation of Xinjiang Uygur Autonomous Region (2019D01C130).
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Ethical approval
The study did not include any human or animal experimentation for which ethical approval was required.
