Abstract
Rheumatoid arthritis (RA) is associated with premature atherosclerosis and increased risk of cardiovascular disease (CVD). The purpose of this study was to assess the relationship between disease activity and arterial stiffness indexes for CVD in RA patients. Two hundred twenty-two female RA patients were retrospectively recruited. Non-invasive arterial stiffness was assessed by brachial-ankle PWV (baPWV) and ankle-brachial index (ABI) using pulse wave analyses and the augmentation index (AIx). The AIx was classified into two groups of normal (grade 0) and abnormal (grade 1, 2, and 3). The baPWV was associated with only age and rheumatoid factor titer, but not disease activity index, DAS28-ESR. Univariate analysis indicated that AIx was related to body mass index, disease duration, and DAS28-ESR. In addition, the baPWV was associated with DAS28-ESR and disease duration after adjusting for confounding factors (β = 0.147, p = 0.032 and β = 0.183, p = 0.034, respectively). Multivariate logistic regression analysis showed that the tendency for abnormal AIx increased as DAS28-ESR increased, and the result was significant (p for trend = 0.026). The results of this study indicated that the relationship between AIx and DAS28-ESR suggests that disease activity could affect arterial stiffness in RA.
Introduction
Rheumatoid arthritis (RA) is a chronic inflammatory autoimmune disease characterized by synovial inflammation with infiltration of inflammatory and immune cells, neovascularization, and bone erosion by osteoclast activation that results in disability and poor quality of life. 1 Research has established that patients with RA have increased risk of cardiovascular disease (CVD) caused by premature atherosclerosis compared to the general population.2,3 This increase in CVD is considered the primary cause for the decrease in overall life expectancy and higher mortality rate in RA patients. 4
Previous prospective cohort studies have confirmed that traditional CV risk factors such as hypertension, diabetes, hyperlipidemia, family history of CVD, and body mass index are important in development of CVD in RA patients.5,6 However, the causal relationship for development of CVD in RA patients is not sufficiently explained by traditional risk factors alone. 7 Systemic inflammation, therapeutic agents for RA, and an RA-related specific genetic background also have been considered as other CV risk factors in RA.8,9 This suggests that additional mechanisms are related to the increased risk of CVD in RA. Some clinical studies have reported that the risk of CVD could be regulated by management of disease activity in RA.10,11 Recently, the European League Against Rheumatism (EULAR), which is focused on the causal relationship between RA and CVD, presented guidelines for preventing and treating CVD in patients with RA by lowering disease activity through appropriate treatment for RA and controlling traditional CV risk factors. 12
Atherosclerosis is an important pathological mechanism in CVD, and early detection is important to minimize damage to vascular endothelial cells in the preclinical stage. Arterial stiffness assessment is an important index for predicting CVD events. 13 Non-invasive tests are used commonly in clinical practice to evaluate arterial stiffness, which include pulse wave velocity (PWV), augmentation index (AIx), and ankle-brachial index (ABI).14,15 Several studies have demonstrated that patients with RA showed higher PWV and AIx compared to healthy controls.16,17
In Korea, RA patients also have a higher prevalence of CVD than the general population. 18 In a recent study for the awareness and recognition of CVD in RA patients, only 39.5% of patients accurately recognized the risk of CVD. 19 The relationship between systemic inflammation and risk of CVD in RA remains under debate. The purpose of this study was to evaluate the correlation between disease activity and non-invasive arterial stiffness markers for CVD in Korean RA patients.
Subjects and Methods
Study population
Patients with RA who were admitted to the Department of Rheumatology at Daegu Catholic University Medical Center from November 2020 to March 2021 and who met the 2010 American College of Rheumatology (ACR) classification criteria were enrolled in this study. 20 Patients with underlying different autoimmune or inflammatory rheumatic diseases such as systemic lupus erythematosus, Sjogren syndrome, ankylosing spondylitis, mixed connective tissue disease, and systemic sclerosis were excluded.
A total of 222 female patients who underwent an arterial stiffness test retrospectively enrolled in this study. The arterial stiffness test was performed to evaluate the risk of CVD regardless of preexisting CV symptoms or signs. This study was approved by the Institutional Review Board (IRB No. CR 21–032) of Daegu Catholic University Medical Center and the written informed consent was waived by IRB.
Assessment of clinical information
Sex, age (years), and disease duration (years) were determined through medical records. Information about the use of therapeutic drugs was investigated based on the drugs prescribed at the time of study enrollment. Treatment drugs were nonsteroidal anti-inflammatory drugs (NSAIDs), glucocorticoids, conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), targeted synthetic DMARDs (tsDMARDs), and biological DMARDs (bDMARDs). Conventional synthetic antirheumatic drugs included methotrexate, hydroxychloroquine, sulfasalazine, azathioprine, leflunomide, and tacrolimus, and the tsDMARDs included tofacitinib and baricitinib. The bDMARDs were classified into tumor necrosis factor inhibitors (TNF inhibitors) and non-TNF inhibitors. TNF inhibitors included infliximab, adalimumab, etanercept, and golimumab, and non-TNF inhibitors included tocilizumab and abatacept.
In addition, evaluation of acute phase reactants was performed using the erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) measured at the time of the arterial stiffness test. Rheumatoid factor (RF) and anti-cyclic citrullinated peptide (anti-CCP) antibody also were identified.
Height (cm) and weight (kg) were measured using a body composition analyzer. Body mass index (BMI) was measured automatically by the formula BMI = weight (kg)/height (m2).
Pulse wave velocity measurement
PWV was measured using a VP-1000 (Colin Co. Ltd, Komaki, Japan) machine after allowing sufficient stability for about 10 min in a lying position, since the value is affected by heart rate and blood pressure. 21 The machine was a device that records blood pressure, electrocardiogram (ECG), and heart sounds simultaneously. The cuffs were wrapped around both upper forearms and both ankles to measure the blood pressure of the limbs, ECG electrodes were attached to both wrists to record the heart rhythm, and a microphone to record heart sounds was placed on the left edge of the sternum.
In addition, the cuff was connected to a plethymographic sensor to determine the volume pulse form, and blood pressure was measured with an oscillometric pressure sensor. The PWV (cm/s) was defined as the value obtained by dividing the distance (cm) between two points by the pulse wave transmission time (seconds). This study used the brachial-ankle PWV (baPWV) measured from the brachial artery and ankle.
The ankle-brachial index (ABI) was defined as the ratio of systolic blood pressure measured at the ankle and systolic blood pressure measured at the brachial artery. 22 The test results were analyzed by classifying the subjects into two groups of normal (grade 0) and abnormal (grade 1, 2, 3) using the automatically calculated result values for analysis.
Augmentation index measurement
The AIx was determined by measuring radial arterial pressure by placing a probe in the position where the pulse was best palpated in the radial artery of the left hand in a seated position after 10 min of rest using an HEM-9000AI (Omron, Japan). 23 At the same time, blood pressure was measured in the right upper arm. This method is used to calculate late systolic blood pressure of the radial artery pulse wave by substituting brachial artery blood pressure. The method measures aortic blood pressure by substituting the blood pressure of the brachial artery into the formula for the generalized transfer function from the pulse wave detected in the radial artery.
AIx is expressed as a percentage by dividing the difference between the height of the pulse changed by the reflected wave of the aortic waveform and the height of the pulse caused by central arterial pressure cardiac contraction. AIx@75 is an index normalized for AIx based on a heart rate of 75 bpm. The results were classified into two groups of normal (grade 0) and abnormal AIx (grade 1, 2, and 3).
Assessment of disease activity in RA
Disease activity in RA was evaluated using a Disease Activity Score of 28 joints (DAS28). DAS28-ESR used the tender joint count (TJC) and swollen joint count (SJC) for the 28 joints, the visual analogue scale (VAS) that was evaluated by the patient, and the ESR. The result is calculated based on the following formula: DAS28-ESR = 0.56x√TJC + 0.28x√SJC + 0.70xln (ESR) + 0.014xVAS.
The disease activity status of RA patients at the time of this study was classified into remission (<2.6), low (≥2.6 and ≤3.2), moderate (>3.2 and ≤5.1), and high (>5.1) based on DAS28-ESR. 24
Statistical analysis
Variable values were expressed as mean value ± standard deviation (SD) or frequency (percentage, %). The normality of the data was verified using the Kolmogorov–Smirnov test, showing a normal distribution. Student’s t-test was used to identify difference between the two groups, and chi-square test was performed for frequency distribution analysis. The correlation analysis between variables was verified using the Pearson correlation coefficient (r). Multivariate regression analysis was used to identify variables related to the AIx@75. Odd ratio (OR) was evaluated using multivariable logistic regression analysis to identify variables related to abnormal AIx. In this study, the csDMARDs and ts/bDMARDs groups were compared according to therapeutic agent characteristics. The statistical significance level was set to p < 0.05. The software used for statistical analysis of this study was SPSS/WIN version 25.0 (IBM Corp, Armink, NY, USA).
Results
Baseline characteristics of the study population
Baseline characteristics of enrolled rheumatoid arthritis patients.

Data were described as mean ± standard deviation or number (%).
Abbreviation: DAS: disease activity score; ESR: erythrocyte sedimentation rate; CRP: C-reactive protein; VAS: visual analogue scale; csDMARDs: conventional synthetic DMARDs; bDMARDs: biological DMARDs; tsDMARDs: targeted synthetic DMARDs; TNF: tumor necrosis factor.
All patients used at least one csDMARD, and 85.6% (n = 190) of patients received glucocorticoids. bDMARDs and tsDMARDs were used by 79 patients and 20 patients, respectively. Of the patients receiving bDMARDs, 60.8% (n = 48) were taking TNF inhibitors.
Analysis of arterial stiffness indexes
Values of arterial stiffness in rheumatoid arthritis patients.

Abbreviation: baPWV: brachial-ankle pulse wave velocity; ABI: ankle-brachial index; AIx: augmentation index.
Correlations between arterial stiffness indexes and clinical variables
Correlation of arterial stiffness markers and other variables in rheumatoid arthritis patients (n = 222).

Data were described as correlation coefficients (r).
Abbreviation: BMI: body mass index; DAS: disease activity score; ESR: erythrocyte sedimentation rate; RF: Rheumatoid factor; Anti-CCP: Anti-Cyclic citrullinated peptide baPWV: brachial-ankle pulse wave velocity; ABI: ankle-brachial index; AIx: augmentation index.
Comparison for differences in clinical variables according to AIx
The difference in clinical variables was measured between the normal (n = 187) and abnormal (n = 31) AIx groups (Figure 1). In this analysis, patients with abnormal AIx values showed statistically significantly lower BMIs than those with normal AIx values (23.5 ± 3.2 vs 22.1 ± 2.9, p = 0.025). There was also a statistical difference for DAS28-ESR and ESR between the two groups, and the values of these variables were higher in patients with abnormal AIx compared to those with normal AIx (3.4 ± 1.2 vs 3.9 ± 1.4 for DAS28-ESR and 26.9 ± 19.6 vs 35.1 ± 26.2 for ESR). Age, RF titer, anti-CCP antibody titer, patient VAS score, CRP, baPWV, and ABI were not statistically different between the groups. Comparison of variables between normal and abnormal augmentation index in rheumatoid arthritis (n = 222).
Analysis for clinical variables related with AIx
Determination of correlation factors for AIx@75 in rheumatoid arthritis patients (n = 222).

Linear regression models were used: Model 1, adjusted for age; Model 2, adjusted for age, hypertension, diabetes mellitus, and cardiovascular disease; Model 3, additionally adjusted for steroid and ts/bDMARDs.
Abbreviation: AIx: augmentation index; BMI: body mass index; DAS: disease activity score; ESR, erythrocyte sedimentation rate.
Analysis of clinical variables related with abnormal AIx
The correlation analysis of abnormal AIx and RA-related variables included RF, anti-CCP antibody, DAS28-ESR, disease duration, comorbid diseases, glucocorticoid, and use of ts/bDMARD. The RF, anti-CCP antibodies, DAS28-ESR, disease duration, glucocorticoid use, and comorbid diseases were not significantly different based on AIx.
Determination of clinical variables for abnormal AIx in rheumatoid arthritis patients (n = 222).
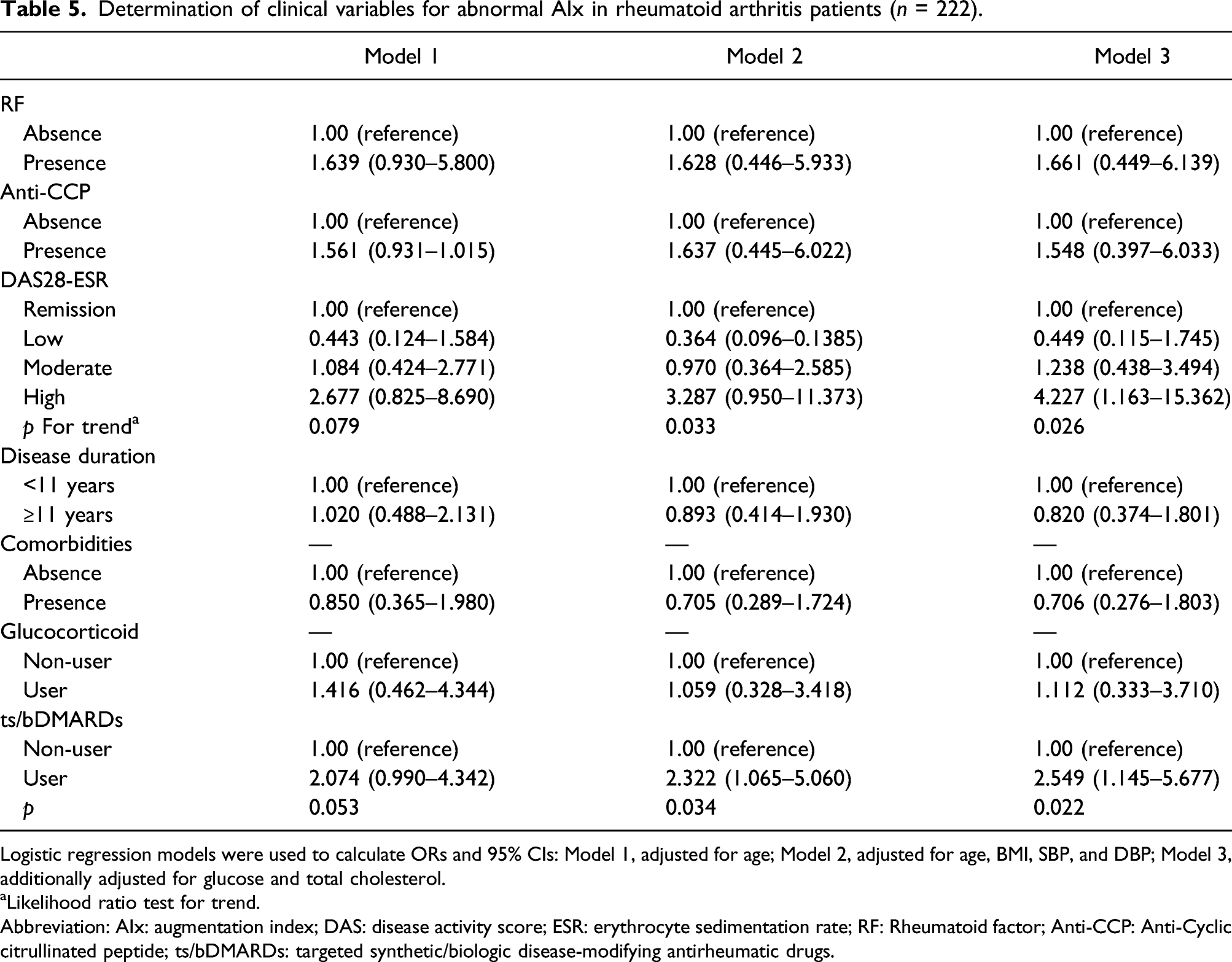
Logistic regression models were used to calculate ORs and 95% CIs: Model 1, adjusted for age; Model 2, adjusted for age, BMI, SBP, and DBP; Model 3, additionally adjusted for glucose and total cholesterol.
aLikelihood ratio test for trend.
Abbreviation: AIx: augmentation index; DAS: disease activity score; ESR: erythrocyte sedimentation rate; RF: Rheumatoid factor; Anti-CCP: Anti-Cyclic citrullinated peptide; ts/bDMARDs: targeted synthetic/biologic disease-modifying antirheumatic drugs.
Discussion
It is well known that RA has a higher prevalence and mortality rate in cases with CVD compared with the general population. Previous studies have identified traditional risk factors for CV events such as hyperlipidemia, diabetes mellitus, and BMI for patients with RA,5,6 similar to the general population. However, the increased incidence of myocardial infarction in patients without traditional CV risk factors suggests that the increase in CVD in RA patients contributes to other mechanisms associated with inflammatory responses.7,25 Disease-specific factors have been investigated to determine the impact on development of the development of CVD in RA, including the duration of disease, RA-related autoantibodies, degree of inflammatory response, and therapeutic drugs such as glucocorticoid. 26 The objective of this study was to analyze the relationships between disease activity and arterial stiffness indexes in Korean RA patients, and the main findings revealed that AIx is correlated with DAS28-ESR and disease duration, and abnormal AIx is related to DAS28-ESR and use of ts/bDMARDs.
The inflammatory response is a critical mechanism for progression of atherosclerosis. Chronic inflammatory reactions lead to endothelial cell activation and dysfunction, resulting in atherosclerotic changes in the endothelium of the artery. 27 RA and atherosclerosis share inflammatory and immunological pathogenic mechanisms. 28 Several studies have suggested that RA patients with higher inflammation had increased risk of more severe and frequent CV events. One study followed 24,989 patients for about 2.7 years to assess the association between disease activity and risk of CVD and found that lower CDAI disease activity resulted in a lower incidence of CVD. 10 A study that analyzed Nijmegen in an early RA inception cohort demonstrated that low DAS28 (≤3.2) was associated with a lower risk of CVD compared to DAS28 > 3.2 (HR 0.65, 95% CI 0.43 to 0.99). 11 The incidence of atherosclerotic plaques increased in patients with high swollen joint number and CRP in RA patients who underwent carotid ultrasound follow-up visits for about 3 years. 29 Additionally, results of an analysis with the Framingham Risk Score confirmed that the risk of CVD was increased. These findings provide evidence to suggest a close relationship between the inflammatory response of RA and CVD caused by progressive atherosclerosis.
Multiple studies have evaluated the degree of atherosclerosis in the preclinical stage using non-invasive arterial stiffness indexes such as PWV and AIx to assess the risk of CVD of RA.16,17,29,30 The PWV and AIx were statistically higher in RA patients than in the healthy control group, which suggests that these markers could be used as predictors for CVD.16,17,30 However, data on clinical variables related to these arterial stiffness indexes in RA patients are lacking. Taverner et al. reported that PWV and AIx were not related to the inflammatory variables and disease activity of RA. 31 However, the present study showed that DAS28-ESR was positively correlated with AIx in linear regression analysis. However, it is necessary to consider the moderate disease activity of the patients participating in this study, with an average DAS28-ESR of 3.5. Based on the DAS28-ESR, it cannot be excluded that the bias of the study population’s disease activity affects AIx. And DAS28-ESR also was confirmed as a determinant of abnormal AIx in logistic regression analysis. Furthermore, our results indicated that baPWV was not related to disease activity index in RA, although it was significantly associated with age and RF titer even in linear regression analysis using confounding factors (supplementary Table 1). These results suggest that disease activity indicators such as DAS28-ESR can be an alternative marker for predicting the degree of arterial stiffness.
Several studies have shown that risk of CVD in patients with RA increases with increasing disease duration.32,33 In an analysis of 185 female RA patients with disease duration of at least 2 years, Kao et al. reported an increased risk of coronary artery calcification in patients with disease duration of 8 to 13 years (OR 3.03, 95% CI 1.06–8.66). 32 In addition, the incidence of CVD increased as the disease duration increased in early RA patients. 34 Therefore, research indicates that disease duration is a major risk factor for CVD. In comparison, a recent study of RA patients in Spain demonstrated that baPWV and Aix, which are indicators of arterial stiffness, were not related to disease duration. 31 In contrast, our study found a positive correlation between disease duration and AIx, but not baPWV.
Among the current therapeutic agents for RA, NSAIDs and glucocorticoids can increase the risk of CVD development. 26 Recently, TNF inhibitors, which are one of the bDMARDs that have a strong anti-inflammatory effect for RA, showed favorable effects not only on inflammation, but also lipid metabolism, which suggested the possibility of reducing the risk of CVD. 35
Additionally, analysis of 983 patients treated with TNF inhibitors indicated that the incidence of CVD was significantly lower than for patients that did not receive TNF inhibitor treatment (95% CI 0.25–0.85, p = 0.013). 36 PWV was statistically significantly reduced before and after 12 weeks of TNF inhibitor administration in RA patients, and PWV measured at 12 weeks was improved to a level similar to that of the healthy controls. 17 These results suggest that bDMARDs, including TNF inhibitors, have a beneficial effect on arterial stiffness as biomarkers for CV events. However, in this study, administration of ts/bDMARDs was a determinant of abnormal AIx. This is a paradox since such treatments are expected to beneficially affect parameters of vascular function following the reduction of the inflammatory burden. Nevertheless, patients on DMARDs are more likely those with the more active disease and probably more extensive atherosclerotic disease. Therefore, it could be related to higher disease activity in patients that receive biological therapeutic agents, compared with patients that did not use ts/bDMARDs.
Subclinical atherosclerosis can be detected using ABI as a non-invasive test to evaluate arterial stiffness, in addition to PWV and AIx. The clinical utility of ABI for evaluating arterial stiffness or subclinical atherosclerosis is still debated. ABI was considered to be beneficial for detecting atherosclerosis or decreased peripheral arterial blood flow in RA patients.37,38 In contrast, the usefulness of the ABI test for evaluating CVD development or progression has been questioned. 39 Similar to the results of the latter study, our study demonstrated that ABI was not correlated with traditional risk factors or RA-specific factors in patients with RA. Therefore, further research is needed to determine the relationships between ABI and RA-related indicators.
BMI-based on obesity has been well established to be closely associated with increased CVD risk in general population and more firmly in patients with RA.5,6 In this study, patients with abnormal AIx showed lower BMI than patients with normal AIx, but statistical significance was lost in the analysis using various confounding variables. The findings of low BMI in patients with abnormal ABI may be due to the following several reasons. First, mechanical techniques such as ultrasound, computed tomography, and dual-energy X-ray absorptiometry may be more accurate than BMI in order to accurately measure the body composition of RA patients. 40 Second, because the number of patients with abnormal ABI is too small, the results may be lack of consistency.
There are several limitations for interpreting the results of this study. First, this study was conducted as a cross-sectional study in which subjects were retrospectively recruited within a specific period. The correlation between disease activity and arterial stiffness or atherosclerosis in RA could be determined, but there was a limitation for clearly clarifying the causal relationship. A prospective cohort study in a large study population is needed to confirm the trend of changes in arterial stiffness markers based on changes in disease activity. Second, there were no healthy control subjects to be compared with RA patients. Therefore, it was not possible to determine whether the arterial stiffness of RA patients was increased compared to healthy controls. Additional case–control studies that include a control group should be conducted to address this limitation. Third, there might be a selection bias for study subject enrollment. It is hypothesized that there is a higher likelihood of undergoing the arterial stiffness tests required for this study for patients with more stable disease activity. Fourth, due to the lack of clinical information on the traditional CV risk factors, such as smoking or a family history of CVD, these variables were not considered sufficiently in the analysis for disease activity of RA. Fifth, a total of 222 patients with rheumatoid arthritis participated in this study, and it was not evaluated whether this was a sufficient sample size. In order to prove this hypothesis, a study with a more appropriate number of patients is needed in the future.
Conclusion
Increased risk of CVD development in RA was not clearly explained by traditional CV risk factors. This study demonstrated that higher disease activity DAS28-ESR values had abnormal arterial stiffness, as demonstrated by AIx, even considering some traditional risk factors. This finding suggests that RA-related inflammatory response contributes to increased arterial stiffness or premature atherosclerosis.
Supplemental Material
sj-pdf-1-eji-10.1177_1721727X221090158 – Supplemental Material for Augmentation index, but not pulse wave velocity, is associated with disease activity in rheumatoid arthritisds
Supplemental Material, sj-pdf-1-eji-10.1177_1721727X221090158 for Augmentation index, but not pulse wave velocity, is associated with disease activity in rheumatoid arthritis by Jung-Yoon Choe, Seo-Hyeon Park and Seong-Kyu Kim in European Journal of Inflammation
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
