Abstract
Excessive exercise can lead to decreased immune function of phagocytes. This study used 50 male Kunming mice aged 40 days to evaluate the effects of lentinan (LNT) on the number of functional blood cells, the phagocytosis, peripheral blood neutrophils, and peritoneal macrophages of long-term heavy-duty exercising mice. The mice were subjected to 45 min of 5% load weight swimming training for 4 weeks, 6 times per week. Different doses of LNT were administered in the HL + T group (400 mg/kg/day), ML + T group (200 mg/kg/day), LL + T group (100 mg/kg/day), and S + T group (saline only) during the 4 weeks of excessive exercise. The phagocytic function of mice in each group was measured after long-term heavy-load exercise. The results showed that the number of white blood cells decreased significantly; the number of neutrophils and the phagocytic index of blood neutrophils in mice were significantly decreased at the end of the exercise. The macrophage phagocytosis index also decreased significantly on the fourth day after the end of the exercise. The difference was significant, and the difference in the phagocytosis rate was not obvious. The numbers of white blood cells in ML + T and HL + T groups were significantly higher than that in S + T and LL + T groups. The number of neutrophils in the ML + T and HL + T groups was significantly higher than that in the S + T and LL + T groups. The phagocytic rate and phagocytic index of neutrophils in the HL + T group were also significantly higher than those in the S + T group. The macrophage phagocytosis rate of ML + T and LL + T mice was significantly higher than that of S + T, and the phagocytic index difference was not significant. Four weeks of heavy-load exercise resulted in a decrease in phagocytic immune function in mice. Excessive exercise while feeding on LNT can improve the immune function of mice phagocytic cells. There was a positive correlation between the effect of LNT on the function of phagocytes and the LNT dose.
Introduction
Lentinus edodes contains a variety of effective medicinal components, especially L. edodes polysaccharides, that is, lentinan (LNT), which is an important biological response modifier. 1 Through stimulating the immune cell maturation, differentiation, and proliferation, it improves the host body balance, restores, and improves the reactivity of host cells to lymph, hormones, and other physical activity factors. 2
For athletes (especially endurance athletes) in late high-intensity exercise training, immune function is reduced, making them prone to infectious diseases. This affects the ability to improve and play sports. Neutrophils and macrophages are the main effector cells of the innate immune system with the role of phagocytosis and digestion of pathogens and foreign substances. However, the effects of LNT on immune function of the body have not been reported. This study was to investigate the effects of LNT on the function and activity of phagocytic cells of heavy-load, long-term exercising mice and to explore its immunoregulatory mechanism.
There is no report on how excessive exercise changes the function of neutrophils, and whether LNT can modulate the change of phagocyte function of the body after excessive exercise. In this study, we divided the mice into several comparing groups and investigated the impact of LNT on the immune function in exercise-induced immunosuppressed mice. Excessively exercising mice were fed with LNT in order to observe LNT’s modulating effect on phagocyte function. The means of using nutritional science to prevent phagocyte immune function decline caused by excessive exercise are evaluated and assessed.
Materials and methods
Experimental animals and grouping
Fifty male specific pathogen-free (SPF) grade Kun Ming (KM) mice, weighing 18–22 g, were used for this experiment. The KM mice were provided by the animal laboratory of Green Leaf Pharmaceutical Co., Ltd. with the animal certification number: SYXK (Lu) 20030020. LNT with authorization # Z20080579 was acquired from Hubei Chuangli Pharmaceutical Company. The mice were divided into cages with five per cage and were fed a diet that was in accordance with the national standard for routine feeding of the rodents. The investigation was conducted in an animal husbandry environment of 23°C ± 2°C with humidity kept at 40%–60%. The rodents followed the natural circadian rhythm. After the 50 mice underwent adaptive feeding for 3 days, they were randomly divided into a saline group (S + C), a saline + exercise group (S + T), a high-dose LNT + exercise group (HL + T), a medium-dose LNT + exercise group (ML + T), and a low-dose GFP + exercise group (LL + T) (n = 10 for each group).
Exercise conditions and training designs
The mice were placed in a plastic swimming pool with dimensions of 100 × 60 × 50 cm3. Water depth was set at 40 cm. The water depth was more than twice the mouse body length and water temperature was 27°C–30°C. The water temperature was similar to that used in other studies. 3
Mice in the S group without exercise underwent normal physiological activities and did not swim. Mice in the S + T, HL + T, ML + T, and LL + T groups swam with loads of 5% of their body weight (a fine copper wire equivalent to 5% of the mouse weight was tied to the root of the mouse tail). Flowing water forced the mice to swim. LNT administration was conducted between 9 a.m. and 10 a.m., and exercise training started at 3 p.m. The swimming training lasted for 4 weeks, 6 times per week with no training on Sundays. From the first week, the loaded swimming training started at 20 min with daily increases of 5 min until it reached 45 min per day. Exhausted mice that exercised less than 45 min were quickly picked up, dried off, and allowed to continue swimming. Exhaustion is accompanied by increased serum-free radical levels and decreased hypoxia tolerance time. The cumulative 45 min/day training time was required. At the end of the training, mice were quickly dried off with a hair dryer.
A total of four mice died due to improper gavage, untimely judgment of mice exhaustion, and failure to pick the mice up before they drowned. Two died in the S + T group, one died in the HL + T group, and one died in the LL + T group.
Drugs and methods of administration
LNT was administered by gavage. The standard human dosage of LNT is 3 g/day. Surface coefficient is a comprehensive parameter for comparing the conversion of the LNT between humans and animals according to body surface area. This coefficient considers a variety of factors, including metabolism, and body surface areas of humans and mice. The equivalent dose in the mice was calculated as follows: Animal dose (per kg) = the known daily animal dose × body surface coefficient/unknown animal body weight.
The body surface coefficient was assigned to be 0.00264. Thus, the dose for each mouse (per kg) = 3000 mg × 0.0026/0.02 kg = 390 mg/kg. Gavage dose: the HL + T group was dosed at 800 mg/kg/day; the ML + T group was dosed at 400 mg/kg/day; and the LL + T group was dosed at 200 mg/kg/day.
LNT solutions were prepared with normal saline to concentrations of 800 mg/10 mL, 400 mg/10 mL, and 200 mg/10 mL, respectively. LNT aqueous solutions were intragastrically administered at 0.2 mL/day, and the S + C and S + T groups were given the same volumes of saline. Intragastric administration was performed daily between 8 a.m. and 9 a.m. for 4 weeks.
Experiment measurement
Measurement of functional cell numbers and number of neutrophils
Two to three drops of EDTANa2 were transferred into a 1.5 mL centrifuge tube and dry tubes at 50°C for 2 days to use later. Four days before the end of the training, tail blood was collected from mice in subgroup A. The mouse was placed in the restraint holder with tail exposed. The tail was immersed in 45°C water for a few minutes, so that the tail vein became vascular filling. The tail was sterilized and dried with a sterile gauze. A length of 5 mm was snipped off the tip of the tail. The tail was pushed from the root to the tip, and the blood sample was collected at the tip of the tail with a pipette. The blood was pipetted into the centrifuge tubes containing EDTANa2, and the bottom of the tube was continuously tapped to give the blood full access to EDTANa2. About 0.1 mL of blood was collected from each mouse. 4 Hemogram and blood analysis were performed using an automatic three classification hemocytometer M-2100 manufactured by BlueBridge Medical Technology Co., Ltd.
Following the procedures in Wu and Zhang, 5 the Staphylococcus aureus was inoculated in 5 mL of broth, cultured for 12 h in a 37°C incubator, and then 0.1 mL was taken out for bacterial count. We put it in 100°C bath water for 10 min to kill bacteria and calculated the number of bacteria per milliliter. We adjusted saline solution to 6 × 108 bacteria/mL and set it at 4°C for further use. We used a hemoglobin pipette to take 40 μL of blood and immediately filled it into the concave recess hole of a clean slide containing 20 μL heparin (concentration of 20 U/mL). We gently stirred the mix, added 20 μL prepared broth, and mixed well. We placed it in a 37°C preheated container covered with wet gauze, put the container in the 37°C incubator for 30 min, and shook it at 10 min intervals. We put a drop of the mixture onto a clean glass slide and pushed it into thin slices. After it dried, we used methanol to fix for 4–5 min and then dyed the staining for 2–3 min with alkaline methylene blue dye. We examined it under an oil microscope, counted 100 neutrophils randomly, and wrote down the number of neutrophils for which phagocytosis occurred and did not occur, respectively. For the occurrence of neutrophil phagocytosis, we recorded the number of bacteria it swallowed
Determination of macrophage phagocytosis
Six grams of soluble starch was dissolved into 100 mL of broth culture fluid. It was mixed and boiled to sterilize for later usage. Four days before the end of training and after tail blood collection and hemogram analysis, each mouse from each subgroup was intraperitoneally injected with 6% soluble starch, 1 mL per mouse, and then continued with gavage and training. Three days later, the mouse was intraperitoneally injected with 2 mL of 5% chicken red blood cell (RBC) suspension, with their abdomens rubbed softly after injection. Thirty minutes after the injection, 1 mL of normal saline was injected intraperitoneally. To collect ascites, the mice were placed their natural standing position. At the leading edge of the pubis, both sides along the abdominal midline were sheared. After sterilization and anesthesia, the skin was tightened around the puncture and then pierced vertically. The ascites were withdrawn slowly with a syringe. A droplet was placed onto a clean glass slide and pushed into thin slices. Once the drop air-dried, it was fixed with methanol for 4–5 min. Swiss-Giemsa dye was added and allowed to stain for 3 min. The slide was washed with running water, air-dried, and observed using a microscope.
One hundred macrophages were counted randomly. The percentage of macrophages that engulfed the chicken RBCs was the phagocytosis rate. The number of macrophages that engulfed chicken erythrocytes divided by 100 was the phagocytic index 3
Data processing
The data were presented as mean ± standard deviation (X ± SD). SPSS11.5 statistical software was used for data processing. S, S + T, HL + T, ML + T, and L + T group data were processed with one-way analysis of variance (ANOVA) first. Considering the sample size of this experiment was not large, least significant difference (LSD) method was used for multiple comparisons. The criterion of significance level (P) was defined to be 0.05, while a very significant level was defined to be 0.01.
Experimental results
Effects of high-load training and LNT on the mouse blood functional cells
Table 1 shows the functional cell counts in the blood in different groups. After 4 weeks of heavy exercise training, the total numbers of white blood cells (WBCs) and platelets (PLT) in S + T group were significantly lower than those in S + C group. No significant change was observed in the total number of RBCs.
Functional cell count in the blood..

RBC: red blood cell; WBC: white blood cell; PLT: platelet.
and ** denote P < 0.05 and P < 0.01 in comparison with the S + C group, resp ectively. # and ## denote P < 0.05 and P < 0.01 in comparison with the S + T group, respectively.
In the LL + T group, no significant change was observed in the total numbers of RBC and PLT. The total numbers of WBC indeed increased from 6.97 ± 1.27 103/mL to 7.93 ± 1.37 103/mL. The most significant change was found in the ML + T. All the three numbers for the WBC, RBC, and PLT were back to or even above the counts in the reference S + C group.
Effect of high-load exercise training and LNT on the function of mouse neutrophil phagocytosis
The number of neutrophils, phagocytic rate, and phagocytic index in the S + T group were significantly lower than those in the S + C group after 4 weeks of heavy-load training (Figure 1(a) and (b)).
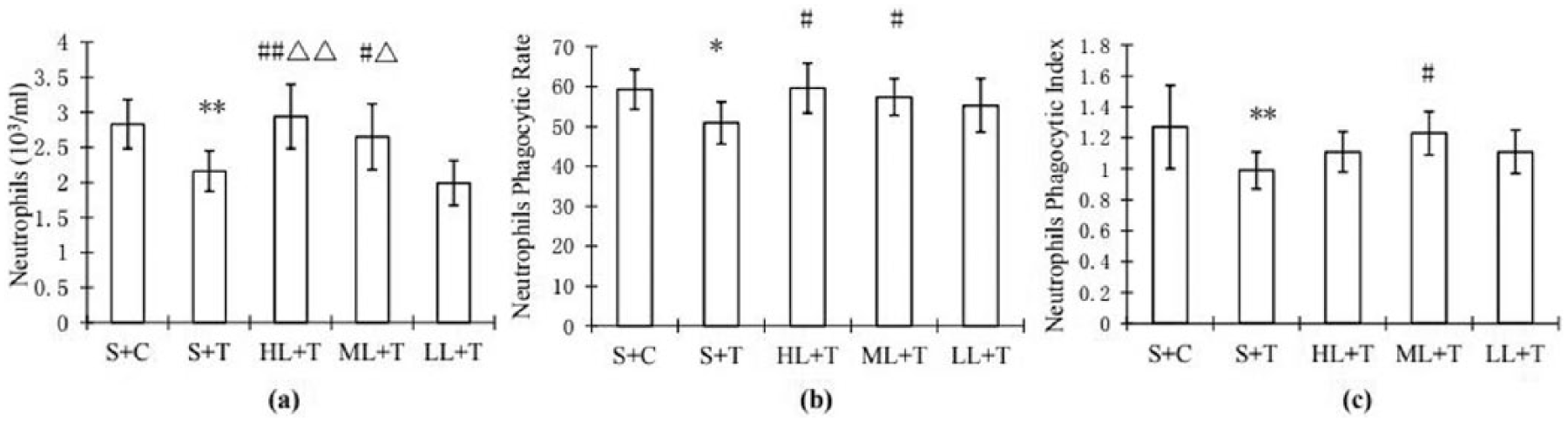
Comparisons between groups S + C, S + T, LL + T, ML + T, and HL + T for (a) number of neutrophils, (b) neutrophil phagocytic rate, and (c) neutrophil phagocytic rate. Each value is represented with mean ± standard deviation.* and ** denote P < 0.05 and P < 0.01 in comparison with the S + C group, respectively. # and ## denote P < 0.05 and P < 0.01 in comparison with the S + T group, respectively. Δ and ΔΔ denote P < 0.05 and P < 0.01 in comparison with the LL + T group, respectively.
In the ML + T and HL + T groups, the numbers of blood neutrophils were significantly higher than in the S + T and L + T groups. The difference between them was significant. The mean values of blood neutrophils for S + C, S + T, HL + T, ML + T, and LL + T are 2.83, 2.16, 2.94, 2.65, and 1.99 × 109 cells/L with standard deviations of 0.35, 0.29, 0.46, 0.47, and 0.32 109 × cells/L, respectively (Figure 1(a)). Similar patterns can be found for the phagocytic rates with 59.3, 50.9, 59.6, 57.4, and 55.3 for S + C, S + T, HL + T, ML + T, and LL + T (Figure 1(b)). Figure 1(c) shows the phagocytic index for S + C, S + T, HL + T, ML + T, and LL + T. The phagocytic indexes for S + C, S + T, HL + T, ML + T, and LL + T are 1.27, 0.99, 1.11, 1.23, and 1.11, respectively (Figure 1(c)).
Effects of high-load exercise training and LNT on phagocytosis of mouse peritoneal macrophages
After 4 weeks of intensive exercise training, the macrophage phagocytosis rate for S + T decreased significantly. The difference in macrophage phagocytosis rate between the S + T and S + C groups was significant. The macrophage phagocytosis rates of HL + T, ML + T, and LL + T mice were significantly higher than that of S + T. The mean macrophage phagocytic rates for S + C, S + T, HL + T, ML + T, and LL + T are 33.6, 26.3, 35.6, 38.2, and 35.3 with standard deviations of 3.83, 3.16, 3.99, 2.94, and 5.72, respectively (Figure 2(a)). The macrophage phagocytic index was not significantly different. Figure 2(b) shows the macrophage phagocytic index for S + C, S + T, HL + T, ML + T, and LL + T. The macrophage phagocytic indexes for S + C, S + T, HL + T, ML + T, and LL + T are 4.13, 3.69, 4.87, 3.67, and 3.76, respectively.

Comparisons between groups S + C, S + T, LG + T, MG + T, and HG + T for (a) macrophage phagocytic rate and (b) macrophage phagocytic index. The legends and symbols are the same as Figure 1.
Discussion and summary
A number of studies have confirmed that long-term intensive training leads to a decline in the numbers of WBCs and neutrophils.
Our study shows that long-term heavy-load exercising mice were in exercise-induced immunosuppression. The results of this study showed that the number of the WBCs and neutrophils in mice with long-term heavy-load exercise decreased significantly. This is consistent with previous study. 10 The number and phagocytic index of neutrophils in the blood of 4-week high-load exercising mice also decreased significantly, reaching an extremely significant level. The mouse macrophage phagocytosis rate also decreased significantly, but no significant decline in the macrophage phagocytic index was observed. It is also noted that the difference in macrophage phagocytosis index between the S + C and S + T groups was not obvious.
There are many other studies on the effects of fungus such as Ganoderma lucidum polysaccharides (GLP) on immune function.11–13 Similar to GLP, LNT is an immune activator that activates macrophages and lymphocytes, increases the chemotaxis of macrophages and the toxic response of lymphocytes to Yac-1 cells and P-815 cells, and antagonizes the carcinogenicity of BBN on mice. 14 Lentinus is a fungus that induces α-interferon. LNT can activate CD4+ lymphocytes and improve the CD4+ proliferation and IL-2R expression. 15 LNT can boost the thymus, spleen index significantly, and improve bone marrow hematopoietic function. LNT also induces the increase of iNOS activity of the mouse peritoneal macrophages and causes the decrease of the GSH in the cell. 16
This study provides further evidence that LNT can also improve the phagocytic rate and phagocytic index of neutrophils in the ML + T and HL + T groups. Feeding LNT can also improve the immune function of mouse phagocytic cells. The number of WBC in ML + T and HL + T was significantly higher than that in S + T. High and medium doses of LNT can both significantly increase blood neutrophils in long-term heavy-load exercising mice to the same level as the S group. The macrophage phagocytosis rates in long-term heavy-load exercising mice treated with high doses and medium doses of LNT were also higher, and at the same level as measured for the S group while the phagocytic index was not significantly different.
In conclusion, 4 weeks of heavy-load exercise can lead to exercise-induced immunosuppression in mice. A supplement of LNT enables improvement of immune responses among long-term heavy-load exercising mice. Effects of high and medium doses of LNT supplements were significant.
Footnotes
Acknowledgements
The authors thank the reviewers for their useful comments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by discipline construction funds from Ludong University, People Republic of China.




